Celsius Kelvin Temperature Scales Kelvin Celsius Boiling point
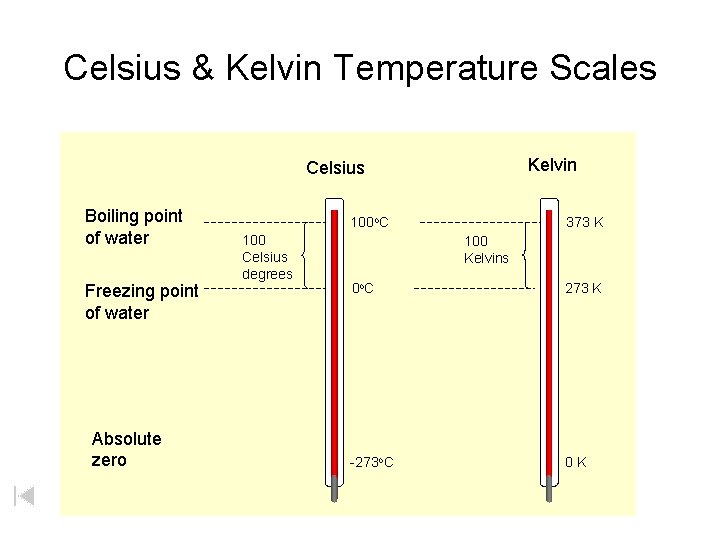
Celsius & Kelvin Temperature Scales Kelvin Celsius Boiling point of water Freezing point of water Absolute zero 100 o. C 100 Celsius degrees 373 K 100 Kelvins 0 o. C 273 K -273 o. C 0 K
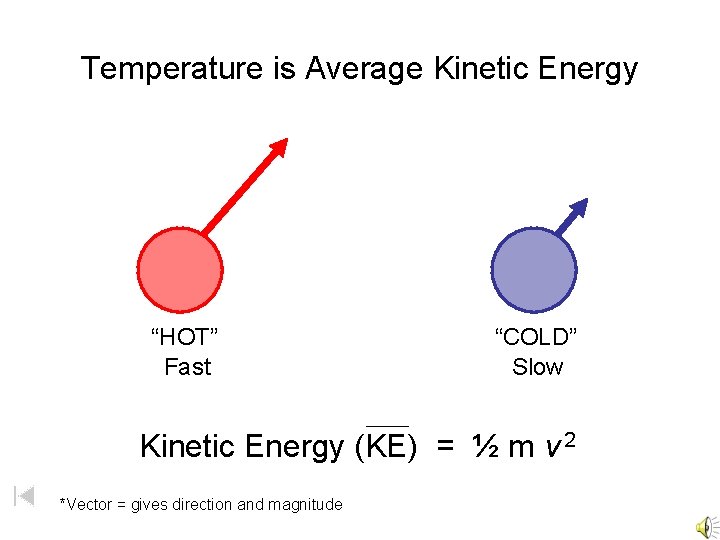
Temperature is Average Kinetic Energy “HOT” Fast “COLD” Slow Kinetic Energy (KE) = ½ m v 2 *Vector = gives direction and magnitude

Temperature Scales Boiling point of water Fahrenheit Celcius Kelvin 212 o. F 100 o. C 373 K 180 o. F Freezing point of water 32 o. F 100 o. C 100 K 273 K Notice that 1 kelvin degree = 1 degree Celcius

Kelvin Scale blue 5250 K Sunlight 5000 K PIAA Plasma Blue 4300 K PIAA HID Bulb 4150 K PIAA Xtreme White 3800 K PIAA Super White 3200 K Halogen Bulb white yellow 2600 K Incandescent Bulb
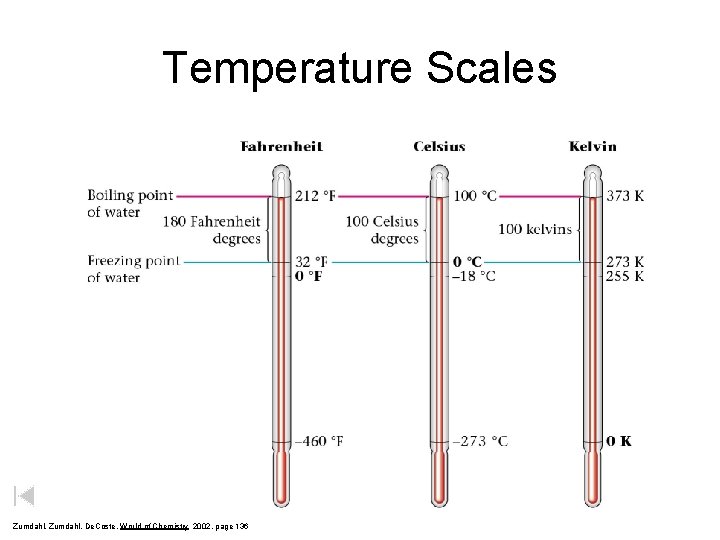
Temperature Scales Zumdahl, De. Coste, World of Chemistry 2002, page 136
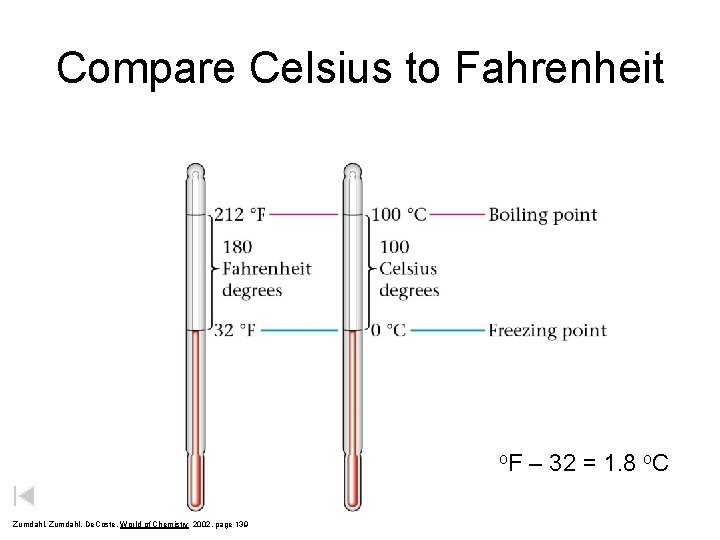
Compare Celsius to Fahrenheit o. F Zumdahl, De. Coste, World of Chemistry 2002, page 139 – 32 = 1. 8 o. C
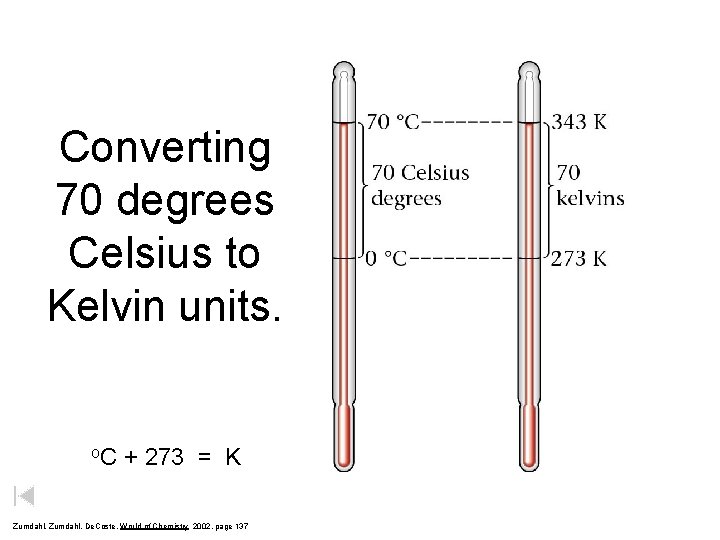
Converting 70 degrees Celsius to Kelvin units. o. C + 273 = K Zumdahl, De. Coste, World of Chemistry 2002, page 137

Temperature Scales • Temperature can be subjective and so fixed scales had to be introduced. • The boiling point and freezing point of water are two such points. • Celsius scale (o. C) – The Celsius scale divides the range from freezing to boiling into 100 divisions. – Original scale had freezing as 100 and boiling as 0. – Today freezing is 0 o. C and boiling is 100 o. C. • Fahrenheit scale (o. F) • Mercury and alcohol thermometers rely on thermal expansion
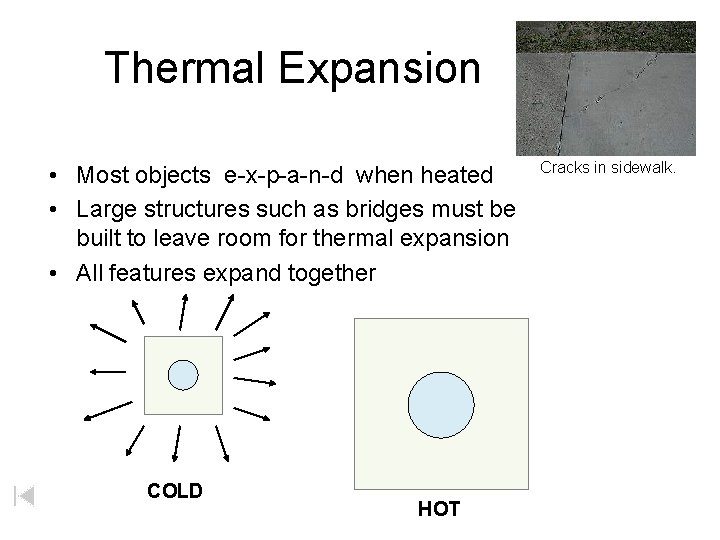
Thermal Expansion • Most objects e-x-p-a-n-d when heated • Large structures such as bridges must be built to leave room for thermal expansion • All features expand together COLD HOT Cracks in sidewalk.
Equal Masses of Hot and Cold Water Thin metal wall Insulated box Zumdahl, De. Coste, World of Chemistry 2002, page 291
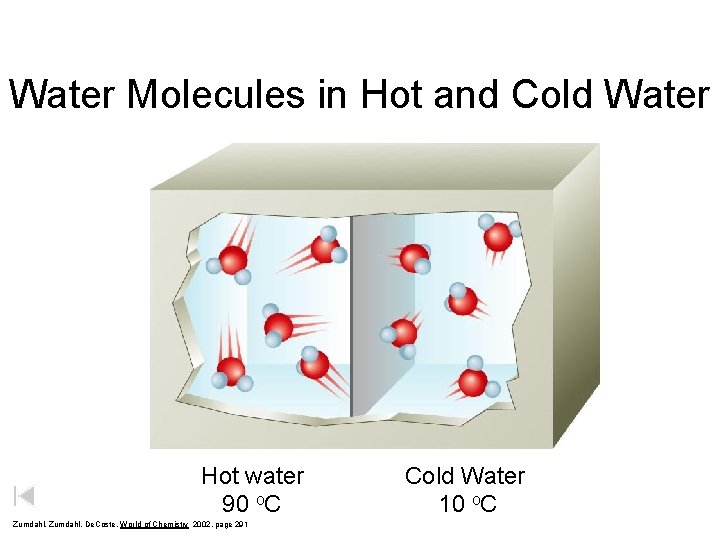
Water Molecules in Hot and Cold Water Hot water 90 o. C Zumdahl, De. Coste, World of Chemistry 2002, page 291 Cold Water 10 o. C
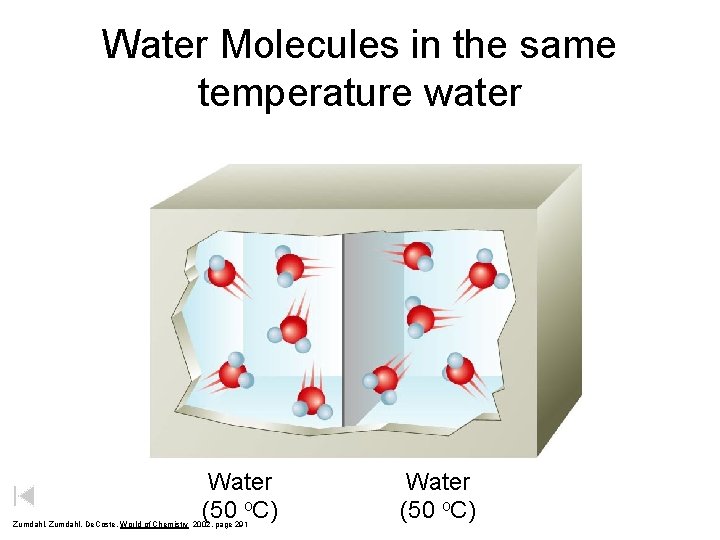
Water Molecules in the same temperature water Water (50 o. C) Zumdahl, De. Coste, World of Chemistry 2002, page 291 Water (50 o. C)

Heat versus Temperature lower temperature Fractions of particles higher temperature TOTAL = Heat Kinetic ENERGY Kinetic energy
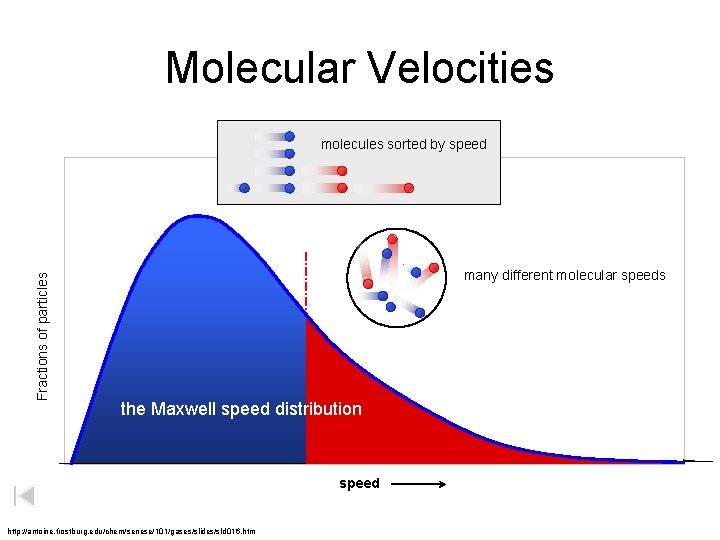
Molecular Velocities Fractions of particles molecules sorted by speed many different molecular speeds the Maxwell speed distribution speed http: //antoine. frostburg. edu/chem/senese/101/gases/slides/sld 016. htm
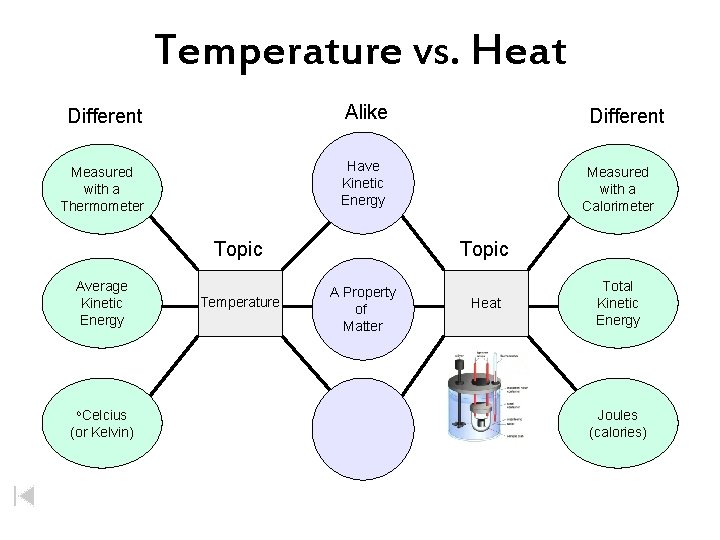
Temperature vs. Heat Different Alike Measured with a Thermometer Have Kinetic Energy Topic Average Kinetic Energy o. Celcius (or Kelvin) Temperature Different Measured with a Calorimeter Topic A Property of Matter Heat Total Kinetic Energy Joules (calories)

Conservation of Matter Reactants yield Products

Heavy Metal Poisoning TREATMENT: Chelation therapy EDTA (ethylenediamine tetra acetic acid) Exposure to mercury made the Hatter “mad”. Eating chips of lead paint causes brain damage. Arsenic treated lumber. ‘Green-treated’ wood will not rot outdoors for 50 years.
- Slides: 17