CELLULOSE ETHANOL Module 3 BIOFUEL PROCESS BIO DIESEL

CELLULOSE ETHANOL

Module 3 BIOFUEL PROCESS

BIO DIESEL PROCESS Converting fat or oil into bio diesel involves the reaction of the oil, methanol (or ethanol), and a catalyst. Glycerin and fatty acids must be extracted from the product to yield bio diesel. The main reaction for converting oil to biodiesel is called transesterification. The transesterification process reacts an alcohol (like methanol) with the triglyceride oils contained in vegetable oils, animal fats, or recycled greases, forming fatty acid alkyl esters (biodiesel) and glycerin. The reaction requires heat and a strong base catalyst, such as sodium hydroxide or potassium hydroxide. The simplified transesterification reaction is shown below. Triglycerides + Free Fatty Acids (<4%) + Alcohol ——> Alkyl esters + glycerin
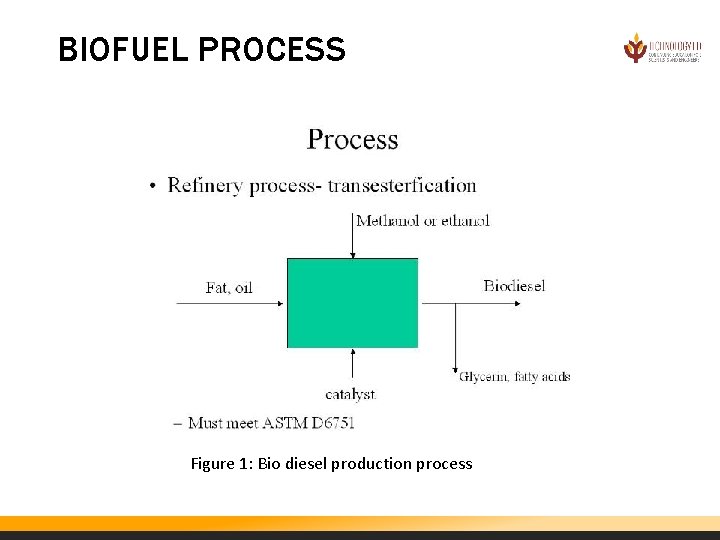
BIOFUEL PROCESS Figure 1: Bio diesel production process

Pretreatment Reaction Some feedstocks must be pretreated before they can go through the transesterification process. Feedstocks with less than 4% free fatty acids, which include vegetable oils and some food-grade animal fats, do not require pretreatment. Feedstocks with more than 4% free fatty acids, which include inedible animal fats and recycled greases, must be pretreated in an acid esterification process. In this step, the feedstock is reacted with an alcohol (like methanol) in the presence of a strong acid catalyst (sulfuric acid), converting the free fatty acids into biodiesel. The remaining triglycerides are converted to biodiesel in the transesterification reaction. Triglycerides + Free Fatty Acids (>4%) + Alcohol ——> Alkyl esters + triglycerides

Acid Esterification. Oil feedstocks containing more than 4% free fatty acids go through an acid esterification process to increase the yield of biodiesel. These feedstocks are filtered and preprocessed to remove water and contaminants, and then fed to the acid esterification process. The catalyst, sulfuric acid, is dissolved in methanol and then mixed with the pretreated oil. The mixture is heated and stirred, and the free fatty acids are converted to biodiesel. Once the reaction is complete, it is dewatered and then fed to the transesterification process. Transesterification. Oil feedstocks containing less than 4% free fatty acids are filtered and preprocessed to remove water and contaminants and then fed directly to the transesterification process along with any products of the acid esterification process. The catalyst, potassium hydroxide, is dissolved in methanol and then mixed with and the pretreated oil. If an acid esterification process is used, then extra base catalyst must be added to neutralize the acid added in that step. Once the reaction is complete, the major co-products, biodiesel and glycerin, are separated into two layers. Methanol recovery. The methanol is typically removed after the biodiesel and glycerin have been separated, to prevent the reaction from reversing itself. The methanol is cleaned and recycled back to the beginning of the process. Biodiesel refining. Once separated from the glycerin, the biodiesel goes through a clean-up or purification process to remove excess alcohol, residual catalyst and soaps. This consists of one or more washings with clean water. It is then dried and sent to storage. Sometimes the biodiesel goes through an additional distillation step to produce a colorless, odorless, zero-sulfur biodiesel. Glycerin refining. The glycerin by-product contains unreacted catalyst and soaps that are neutralized with an acid. Water and alcohol are removed to produce 50%-80% crude glycerin. The remaining contaminants include unreacted fats and oils. In large biodiesel plants, the glycerin can be further purified, to 99% or higher purity, for sale to the pharmaceutical and cosmetic industries.

Ethanol Process Converting biomass to ethanol process consist of two primary processing steps for starch based biomass (i. e. corn, sugar cane) and four primary processing steps for lignocellulosic biomass (i. e. corn stover, switchgrass): Ethanol from starch biomass • · Yeast fermentation • · Distillation Ethanol from cellulosic biomass • · Pretreatment • · Enzymatic hydrolysis • · Yeast fermentation • · Distillation
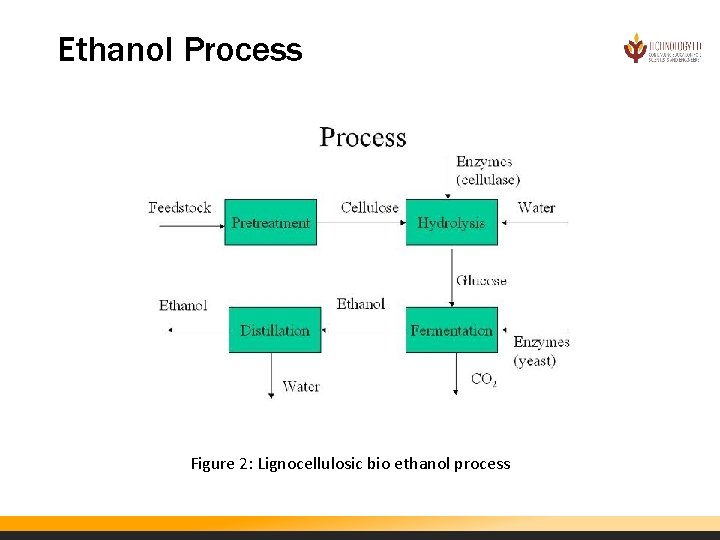
Ethanol Process Figure 2: Lignocellulosic bio ethanol process
PRETREATMENT A primary technological challenge in converting cellulosic biomass feedstock into fuel is overcoming the recalcitrance of it's lignocellulosic matrix to microbial conversion. Naturally occurring lignocellulosic materials, as shown in Figure 1, have carbohydrate-rich cellulose and hemicellulose fibers that are surrounded by a lignin seal. This forms a complex structural matrix that is resistant to enzymatic hydrolysis. Here, lignin acts like a glue that fills the voids between and around cellulose and hemicellulose fibers, providing the rigid characteristics. This structure reduces accessibility to the polysaccharide molecules. Hence, removal of the lignin will largely enhance polysaccharide accessibility. In addition to the lignin seal, cellulose chains are held together laterally by intermolecular hydrogen bonds. These intramolecular hydrogen bonds form between repeating glucose units. The combined effect of the bonding energies of the hydrogen bonds increases the rigidity of cellulose, causing further insolubility to hydrolysis.

PRETREATMENT A primary technological challenge in converting cellulosic biomass feedstock into fuel is overcoming the recalcitrance of it's lignocellulosic matrix to microbial conversion. Naturally occurring lignocellulosic materials, as shown in Figure 1, have carbohydrate-rich cellulose and hemicellulose fibers that are surrounded by a lignin seal. This forms a complex structural matrix that is resistant to enzymatic hydrolysis. Here, lignin acts like a glue that fills the voids between and around cellulose and hemicellulose fibers, providing the rigid characteristics. This structure reduces accessibility to the polysaccharide molecules. Hence, removal of the lignin will largely enhance polysaccharide accessibility. In addition to the lignin seal, cellulose chains are held together laterally by intermolecular hydrogen bonds. These intramolecular hydrogen bonds form between repeating glucose units. The combined effect of the bonding energies of the hydrogen bonds increases the rigidity of cellulose, causing further insolubility to hydrolysis. To qualify as effective, the pretreatment must meet the following criteria: 1) maximize fermentable sugar yields, 2) preserve utility of the hemicellulose fraction, 3) avoid, or minimize degradation of carbohydrates, 4) avoid, or minimize the formation of microbial growth-inhibiting byproducts, and 5) be economically efficient. Principal substrate factors that have been correlated with pretreatment effectiveness include cellulose pore volume, hemicellulose and lignin removal, and cellulose crystallinity
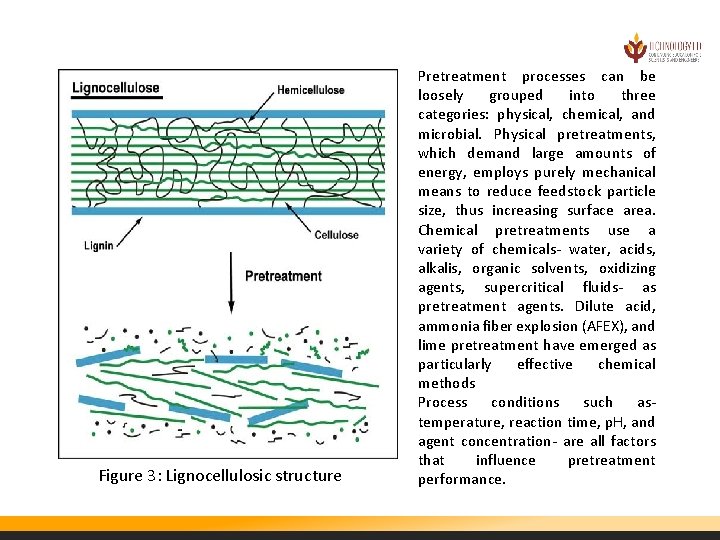
Figure 3: Lignocellulosic structure Pretreatment processes can be loosely grouped into three categories: physical, chemical, and microbial. Physical pretreatments, which demand large amounts of energy, employs purely mechanical means to reduce feedstock particle size, thus increasing surface area. Chemical pretreatments use a variety of chemicals- water, acids, alkalis, organic solvents, oxidizing agents, supercritical fluids- as pretreatment agents. Dilute acid, ammonia fiber explosion (AFEX), and lime pretreatment have emerged as particularly effective chemical methods Process conditions such astemperature, reaction time, p. H, and agent concentration- are all factors that influence pretreatment performance.

Pretreatment Example 1: Steam Explosion Steam explosion involves exposing lignocellulosic biomass to high pressure steam. The steam penetrates the biomass by diffusion under the high pressure. The moisture content of the material rises through condensation under the high pressure. The water initiates hydrolysis of the acetyl groups of the hemicellulose, resulting in organic acids (i. e. acetic and uronic acid). These acids catalyze hemicellulose depolymerization, yielding xylan and small quantities of glucan. An “explosion” is believed to occur when the reactor is depressurized. The condensed moisture within the biomass structure evaporates immediately due to the sudden depressurization in the reactor. Here, the water vapor expands and exerts a shear force on the structure. High shear forces are believed to cause physical breakdown of the biomass structure. T. he optimized factors for this process are retention time and temperature. The steam pressure within the reactor is managed by the temperature (i. e. temperature and pressure are directly proportional). The pressure corresponds to the differential between the reactor and atmospheric pressure. This differential pressure is also directly related to the shear force of the evaporating moisture. Hemicellulose hydrolysis is a direct function of the residence within the reactor. This hydrolysis plays a key role in the downstream enzymatic processes

Pretreatment Example 2: Chemical Pretreatment Chemical pretreatment has been a widely explored approach to overcoming the recalcitrance of natural biomass. Many acids, bases, and other chemicals promote hydrolysis and improve fermentable sugar yield through the removal of hemicellulose and/or lignin. An extensive array of chemical pretreatment such as oxidizing agents, acids, bases, and other solvents have been investigated. Oxidizing agents tested include alkaline peroxide, sodium and calcium hydroxide, ozone, dioxane, and organosolv. Acids evaluated include sulfuric acid, hydrochloric acid, phosphoric acid, and nitric acid. Chemical solvents such as ammonia, aprotic solvents (i. e. DMSO), and metal complexes have been explored. These chemicals have shown varying degrees of effectiveness reducing cellulose crystallinity, disrupting the lignin matrix, and dissolving cellulose.
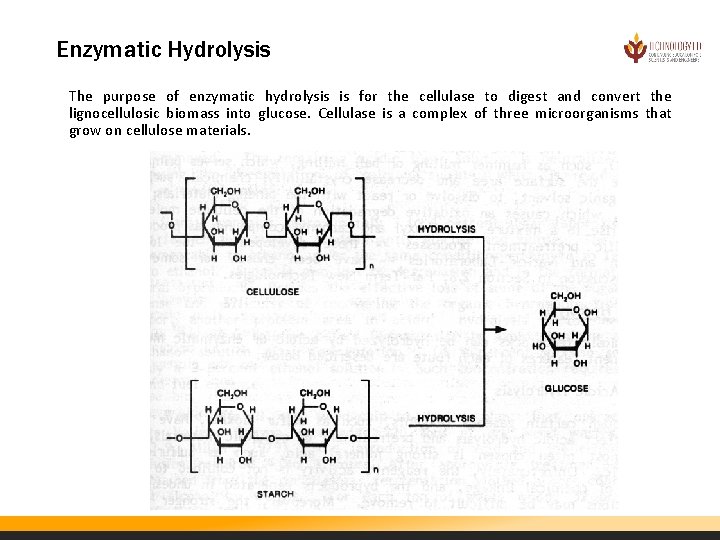
Enzymatic Hydrolysis The purpose of enzymatic hydrolysis is for the cellulase to digest and convert the lignocellulosic biomass into glucose. Cellulase is a complex of three microorganisms that grow on cellulose materials.
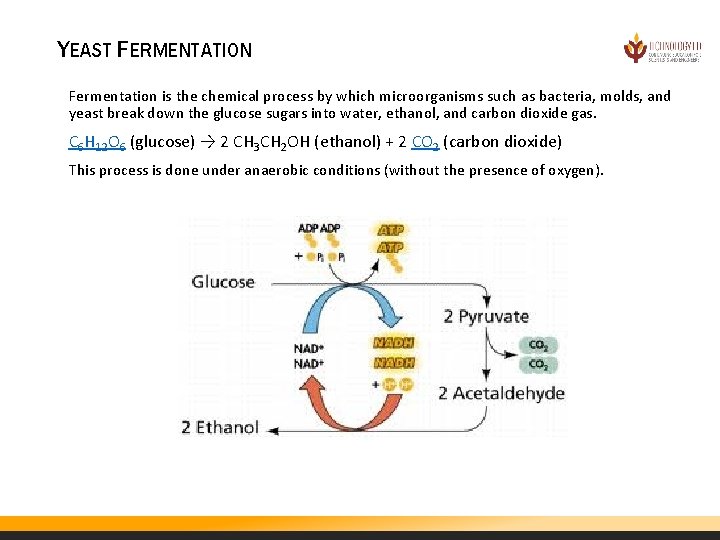
YEAST FERMENTATION Fermentation is the chemical process by which microorganisms such as bacteria, molds, and yeast break down the glucose sugars into water, ethanol, and carbon dioxide gas. C 6 H 12 O 6 (glucose) → 2 CH 3 CH 2 OH (ethanol) + 2 CO 2 (carbon dioxide) This process is done under anaerobic conditions (without the presence of oxygen).
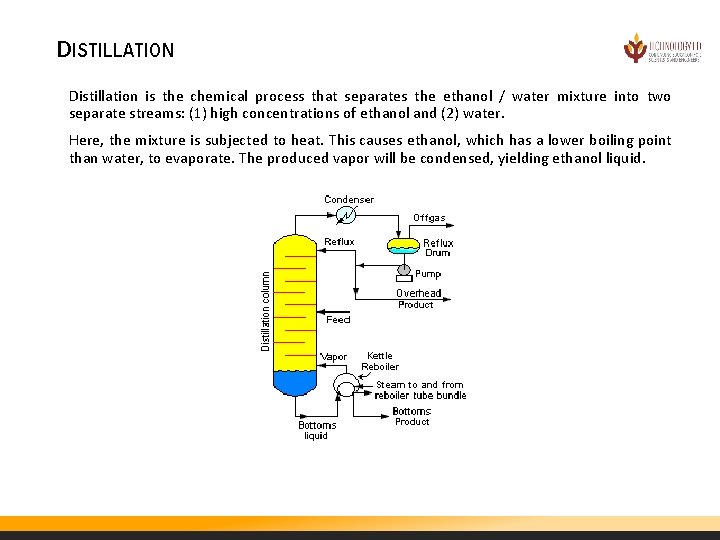
DISTILLATION Distillation is the chemical process that separates the ethanol / water mixture into two separate streams: (1) high concentrations of ethanol and (2) water. Here, the mixture is subjected to heat. This causes ethanol, which has a lower boiling point than water, to evaporate. The produced vapor will be condensed, yielding ethanol liquid.

BUTANOL PROCESS Butanol is an alcohol that can be used as a transport fuel. It is a higher member of the series of straight chain alcohols with each molecule of butanol (C 4 H 10 O) containing four carbon atoms rather than two as in ethanol. The butanol production process is different from the ethanol production process. The butanol process technology is very embryonic, with no production at the present. Butanol can be produced by traditional ABE fermentation - the anaerobic conversion of carbohydrates by strains of Clostridium into acetone, butanol and ethanol. This process yields a 6: 3: 1 ratio of butanol, acetone, and ethanol. Figure 4 illustrates the ABE ferementation process. Environmental Energy, Inc. (EEI) has patented a two step process (Figure 5): · step 1: biomass -> butyric acid (BA) · step 2: BA fed to C -> butanol (42% yield)

Butanol Process However, cost issues, the relatively low-yield and sluggish fermentations, as well as problems caused by end product inhibition and phage infections, meant that ABE butanol could not compete on a commercial scale with butanol produced synthetically and almost all production ceased as the petrochemical industry evolved. There is now increasing interest in use of biobutanol as a transport fuel. 85% Butanol/gasoline blends can be used in unmodified petrol engines. It can be transported in existing gasoline pipelines and produces more power than ethanol. Biobutanol can be produced from cereal crops, sugar cane and sugar beet, etc, but can also be produced from cellulosic raw materials.
- Slides: 18