Cellular Respiration Subtopic 2 8 The essential Idea

Cellular Respiration Subtopic 2. 8

The essential Idea: Cell respiration supplies energy for the functions of life. All living organisms need energy to stay alive, which is why we eat. The nutrients in foods, such as carbohydrates, are broken down into glucose, which is absorbed into the blood and taken up by cells. And that is where the actual energy-generating process takes place.
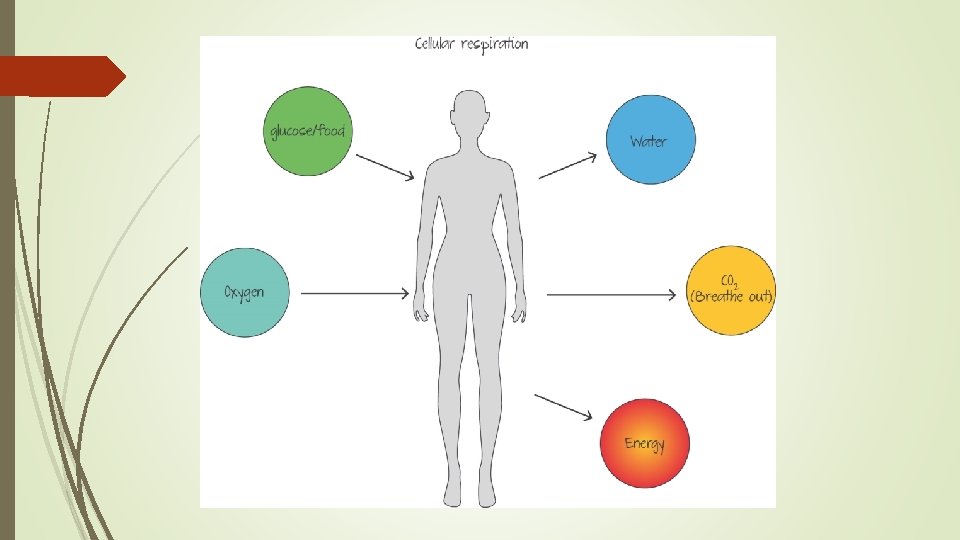
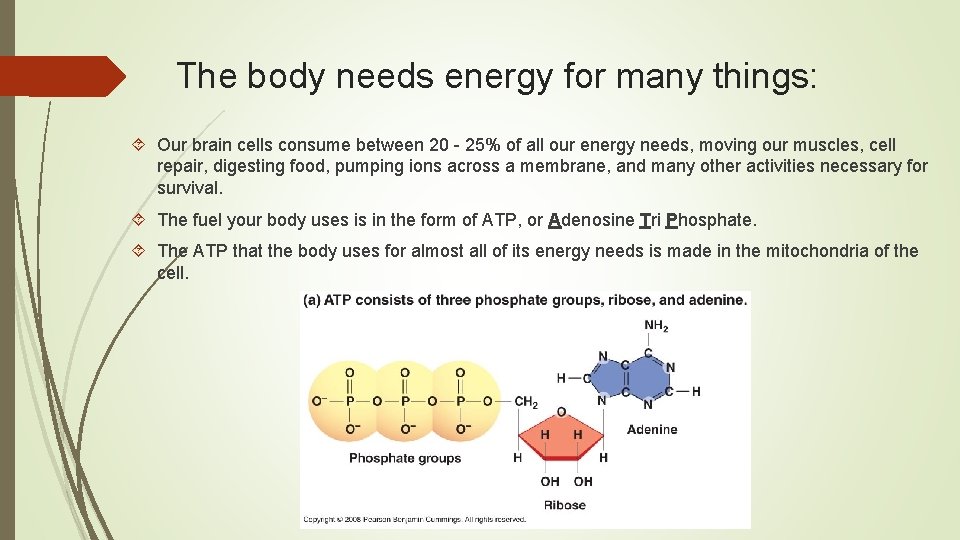
The body needs energy for many things: Our brain cells consume between 20 - 25% of all our energy needs, moving our muscles, cell repair, digesting food, pumping ions across a membrane, and many other activities necessary for survival. The fuel your body uses is in the form of ATP, or Adenosine Tri Phosphate. The ATP that the body uses for almost all of its energy needs is made in the mitochondria of the cell.

2. 8. 1 Cell respiration and ATP Glucose is the preferred energy source for brain cells. Glucose is broken down in muscle cells for energy. Glucose is the most widely used source of energy in living organisms. Cellular respiration is the gradual and controlled release of energy by breaking down organic compounds to produce ATP (Adenosine Tri Phosphate). Cellular respiration involves enzymes and hence the process is fully controlled to ensure that energy is produced when it is required.
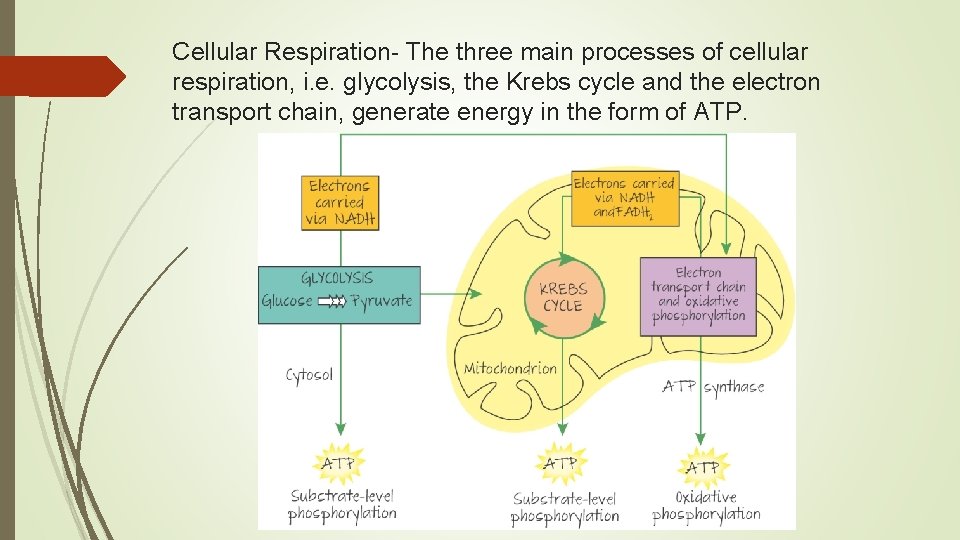
Cellular Respiration- The three main processes of cellular respiration, i. e. glycolysis, the Krebs cycle and the electron transport chain, generate energy in the form of ATP.

Energy stored in the organic molecules of food ultimately comes from the sun. Energy flows into an ecosystem as sunlight and exits as heat; while chemical elements essential for life are recycled. 3 pathways of respiration: Glycolysis Citric Acid cycle Oxidative phosphorylation (Electron Transport Chain)

The production of ATP Organic compounds possess potential energy. Compounds that can participate in exergonic reactions can act as fuels. The cell breaks down complex organic molecules with the help of enzymes.

Cellular Respitation Fermentation Partial degradation of sugars and other organic fuels that occurs without oxygen. Aerobic respiration Oxygen is consumed as a reactant along with the organic fuel. Carried out by most eukaryotes and many prokaryotes. Anaerobic respiration Use substances other than oxygen as reactants in order to obtain energy. Technically cellular respiration includes both aerobic and anaerobic processes. However, cellular respiration is often used to refer to the aerobic process.

Which molecules can be used in Cell. Resp? Carbohydrates Lipids Proteins Glucose is the fuel that cells most often use.

Why Glucose? Breakdown of glucose is exergonic. ∆G (Gibbs free energy) = -686 kcal per mole of glucose. Negative ∆G means the products of the chemical process store less energy than the reactants and that the reaction can happen spontaneously. Cellular respiration is a series of catabolic reactions! Catabolic pathways do not directly do work such as moving flagella. Instead, catabolism is linked to work by ATP. To keep working a cell must regenerate its ATP supply form ADP + P

Redox Reactions: Oxidation and Reduction Reactions. Redox reactions are based on the transfer of electrons during chemical reactions. The relocation of electrons releases energy stored in organic molecules, and this energy is used to synthesize ATP. Oxidation-reduction reactions take place when electrons are transferred from one reactant to another. If the molecule loses electrons, its been said to be oxidized. If the molecule gains electrons, its been said to be reduced. We say it is being reduced because you are reducing the amount of positive charge of that atom.

Redox reactions cont. The electron donor is called the reducing agent The electron acceptor is called the oxidizing agent Oxidation and reduction ALWAYS go together. Not all redox reactions involve complete transfer of electrons, some change the degree of sharing of electrons in covalent bonds.

The role of oxygen in redox reactions Oxygen is one of the most potent of all oxidizing agents. This is because oxygen is very electronegative. Think of how much energy would be required to pull an electron from an electronegative atom!!! It is going to require some energy. The more electronegative the atom (the stronger its pull on electrons), the more energy is required to pull that electron away. An electron loses potential energy when it shifts from a less electronegative atom toward a more electronegative one. Think of a ball losing potential energy as it rolls down a hill.

Oxidation of Organic Molecules during Cell Respiration is the oxidation of glucose and other molecules in food. Organic molecule with a lot of hydrogens available are excellent fuels because their bonds are a source of “hilltop” electrons, whose energy may be released as these electrons “fall” down the energy gradient when they are transferred to oxygen.

Cont. Hydrogen is transferred from glucose to oxygen. The energy state of the electron changes as the hydrogen (with its electron) is transferred to oxygen. Oxidation of glucose transfers electrons to a lower energy state, liberating energy that becomes available for the synthesis of ATP. Energy-yielding foods are reservoirs of electrons associated with hydrogen. Only the barrier of activation energy holds back the flood of electrons to a lower energy state.

The role of enzymes Body temperature is not high enough to initiate fuel burning. Instead, enzymes in your cells lower the activation energy, allowing the sugar to be oxidized in a series of steps.

Harvesting electrons for the production of ATP Energy must be released in steps in order to be more efficient (less heat loss). Glucose is broken down in a series of steps, each step catalyzed by an enzyme. Electrons are stripped from glucose. In redox reactions electrons often travel with a proton – thus a hydrogen atom.
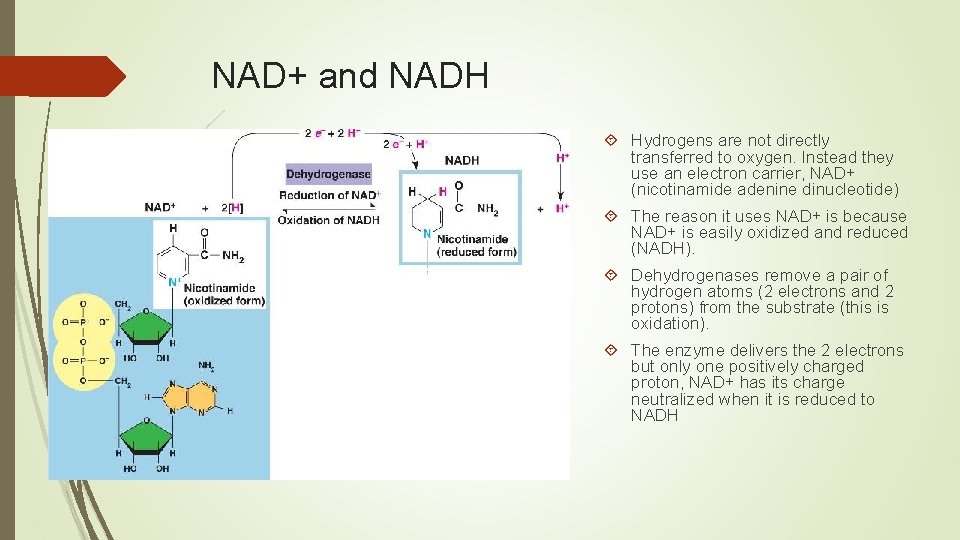
NAD+ and NADH Hydrogens are not directly transferred to oxygen. Instead they use an electron carrier, NAD+ (nicotinamide adenine dinucleotide) The reason it uses NAD+ is because NAD+ is easily oxidized and reduced (NADH). Dehydrogenases remove a pair of hydrogen atoms (2 electrons and 2 protons) from the substrate (this is oxidation). The enzyme delivers the 2 electrons but only one positively charged proton, NAD+ has its charge neutralized when it is reduced to NADH

Using the energy released to make ATP Electrons lose very little of their potential energy when they are transferred from glucose to NAD+. Each NAD+ molecule formed during respiration represents stored energy stored that can be tapped to make ATP as electrons complete their “fall” down an energy gradient from NADH to oxygen.

Electron Transport chain Electron transport chain breaks the fall of electrons to oxygen in several energyreleasing steps. ETS happens in the inner membrane of the mitochondria of eukaryotic cells and the plasma membrane of aerobically respiring prokaryotes. Electrons removed from the glucose are shuttled by NADH to the “top”, higherenergy end of the chain. At the “bottom”, lower-energy end, O 2 captures these electrons along with hydrogen nuclei (H+), forming water.

Oxygen the final electron acceptor Electron transfer from NADH to oxygen is an exergonic reaction. Electrons cascade down the chain from one carrier molecule to the next in a series of redox reactions, losing a small amount of energy with each step until they reach oxygen, the terminal electron acceptor. Electrons removed from glucose by NAD+ fall down an energy gradient in the electron transport chain to a far more stable location in the electronegative oxygen atom.

Location of Cellular Respiration within the cell. Glycolysis takes place in the cytoplasm, while the Kreb's cycle and oxidative phosphorylation (and electron transport chain) occur in the mitochondria. Within the mitochondrion, Kreb's cycle occurs in the matrix, while the electron transport chain is located on the inner membrane of the mitochondrial envelope.

ATP fuels most of our body's energy needs. ATP from cell respiration is immediately available as a source of energy in the cell. Without it we would not be able to move, think, digest food, conduct nerve signals or breathe. Energy is produced when ATP is hydrolyzed. It should be noted that this same energy was generated during respiration and stored in the energy rich phosphate bond of ATP.

Do NOT confuse respiration with breathing!!! In some languages, e. g. French, 'respire' means breathing. In biology, respiration always refers to the breakdown of organic compounds and the production of ATP in the cell.

2. 8. 2 Anaerobic cell respiration Cell respiration can take place with or without the presence of oxygen. When it takes place without oxygen it is called anaerobic respiration, and it occurs only in the cell cytoplasm as it involves only glycolysis (to be able to complete the other steps, oxygen is required). Humans have utilized the ability of certain organisms to carry out cell respiration without oxygen, including yeast and certain bacteria. The former is far more important if you like to drink beer, wine or eat bread for breakfast.

Alcoholic Fermentation Yeast respires anaerobically to produce ethanol and CO 2 when fermenting beer, wine or bread. The CO 2 produced by yeast is used to make bread rise while the ethanol is evaporated away during baking.

Lactic Acid Fermentation During strenuous exercise, our muscle cells may use up all the available oxygen. Nonetheless, the muscle cells still need ATP to continue the exercise. Without oxygen, the muscle cells start to respire anaerobically and are able to produce a small amount of ATP which allows the power of muscle contractions to be maximized. During anaerobic respiration, animals produce lactate (also known as lactic acid), which causes the soreness in muscles after strenuous exercise.
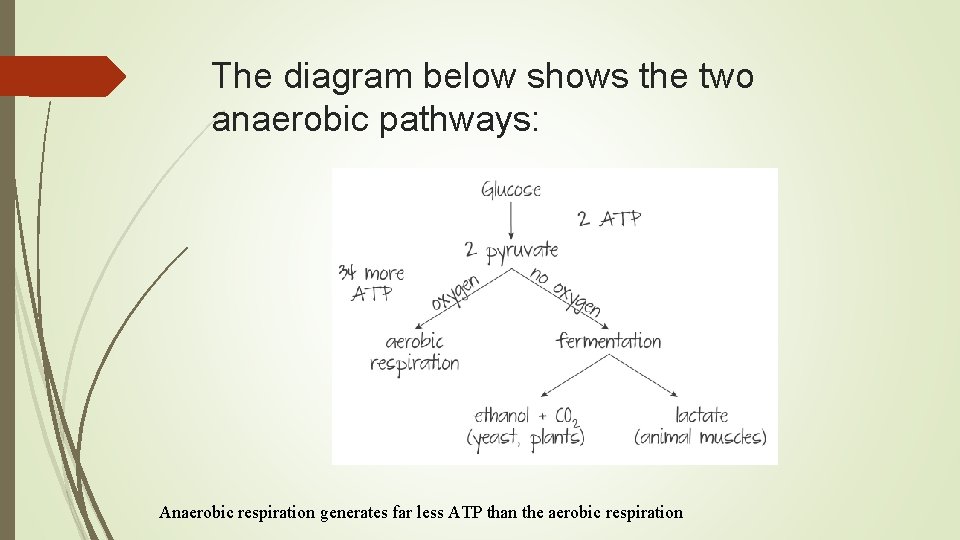
The diagram below shows the two anaerobic pathways: Anaerobic respiration generates far less ATP than the aerobic respiration

Anaerobic Respiration Overview Takes place in the cytoplasm. Takes place without the presence of oxygen. Generates a smaller amount of ATP (only 2 ATP) than aerobic respiration. In yeast it produces alcohol and CO 2. In human muscle cells it produces lactate.

2. 8. 3 Aerobic cell respiration We breathe in oxygen to support aerobic respiration. When oxygen (O 2) is present, aerobic respiration can yield far more ATP than under anaerobic conditions as all the energy contained in a glucose molecule can be harvested and converted into ATP. The main reason for this increased ATP production is that during aerobic respiration, all the four steps of cellular respiration are completed (compared to only glycolysis being completed during anaerobic respiration). The reason for this increased yield is that 6 -carbon glucose is systematically and gradually broken down to 6 CO 2 molecules. Each step yields potential energy, which can be converted into ATP in the electron transport chain.

Aerobic respiration: Requires oxygen. Gives a large yield of ATP from oxidation of glucose.

Aerobic Vs. Anaerobic Respiration It is much more efficient to respire aerobically than anaerobically. That is why long distance runners try to maintain an aerobic speed. If they use up the available oxygen, their muscles will start anaerobic respiration, producing lactate which will make their muscles cramp up.

2. 8. 4 The use of a respirometer A respirometer is a simple apparatus that can measure the rate of respiration. Cellular respiration uses oxygen and produces CO 2 and H 2 O. We can measure the consumption of O 2 as an indication of the respiration rate.

Using a Respirometer In Tube A, the organism to be tested (or germinating seed) is positioned, and the tap is closed. The organism starts respiring, consuming O 2 and producing CO 2 and H 2 O. The alkali solution at the bottom of Tube A will absorb the CO 2. Tube B is the control where no O 2 is used or CO 2 produced as no living organism is present. The capillary connecting the two tubes is a manometer. The reduction in oxygen in Tube A will reduce the pressure in Tube A and will move the colored liquid in the manometer in the direction of tube A, providing an indirect measurement of the oxygen consumed, allowing the rate (amount of oxygen consumed per time unit) to be calculated. Setting up the respirometer in a water bath will ensure that temperature does not affect the pressure and hence volume of air in the system.

Respirometer and Respiration Rates

Ethical Issues in science Why would we wish to do such experiments which are potentially unethical? Even if scientists use insects for such a lab test, you should question if such experiments will harm the living organisms you are investigating before performing the experiment. Remember, you should respect the IB animal experimentation policy. A respirometer can give us a good idea of the respiration rate of germinating seeds, a resting animal or a moving animal, and it allows us to compare different species. It can measure the influence of temperature or, for example, sleep, on respiration. The whole range of tests can be performed with such a simple setup.
You should be able to analyze results from experiments involving measurement of respiration rates in germinating seeds or invertebrates using a respirometer. The results could be in the form of data tables or graphs.

Nature of Science There are ethical implications in the use of animals and humans in research. The use of animals in scientific experimentation has ethical implications. It is, therefore necessary to consider four issues when using animals in school experiments: Is it absolutely necessary to use an animal in the experiment? Will the animal possibly be harmed during the course of the experiment? Will the animal be exposed to risk or hurt during the course of the experiment? Will the animal be able to be returned to its natural habitat at the end of the experiment? In the case of the respirometer: Germinating seeds can demonstrate most aspects of respirometer use rather than an animal. The placing and confinement of an animal in a respirometer tube could harm or damage the animal. Exposure to the alkali (that absorbs carbon dioxide) must be completely avoided. The habitat and place the animal was taken from should be noted and then it can be returned to the natural habitat. The IB has strict guidelines which must be adhered to in all laboratory procedures involving experimentation with animals (including humans). Local and national regulations must also be respected.
- Slides: 39