Cellular Respiration Making ATP from food crashcoursebio ATP

Cellular Respiration (Making ATP from food) crash-course-bio ATP

RESPIRATION 1

3 All living cells are made up of chemical substances The processes of living involve reactions between the substances A reaction is an event which produces a change in a substance For example, a reaction between carbon and oxygen (such as burning coal in air) changes the carbon in the coal, and oxygen in the air into This reaction can be represented by the equation carbon dioxide C carbon + O 2 CO 2 oxygen carbon dioxide
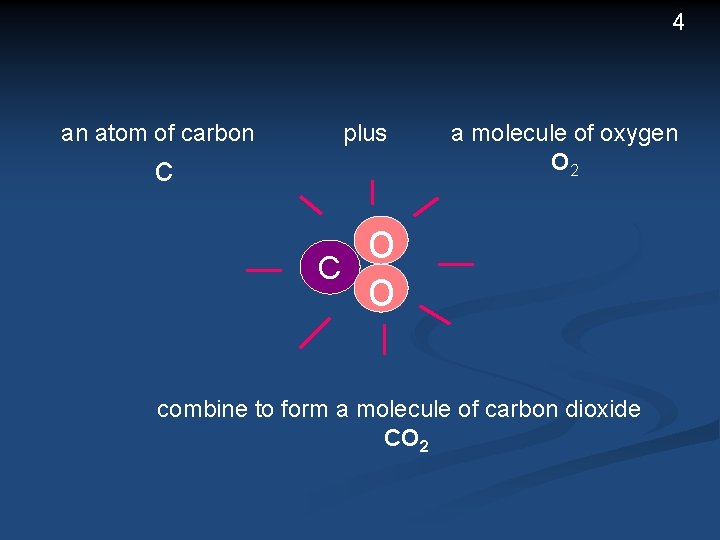
4 plus an atom of carbon c C a molecule of oxygen O 2 o o combine to form a molecule of carbon dioxide CO 2

Metabolizing Molecules Metabolism – all the chemical reactions occuring in your cells at one time

n Two types of chemical reactions can occur as an organism metabolizes molecules: n Anabolic reactions - this type of reaction builds molecules (specifically, small molecules are combined into large molecules for repair, growth or storage). n Catabolic reactions – this type of reaction breaks down molecules to

What do you use to get yourself out of bed in the morning? n ENERGY n

Where do our cells get energy? 6 -C sugars are the MAJOR source of energy for cell n What type of macromolecule are 6 -C sugars? n n n Carbohydrates Cells break down glucose a 6 -C sugar to make ATP “energy”

n. What is a molecule of energy? n ATP n. So, did you eat ATP last night? n Or did you eat pizza? n. Food you eat must be

Setting the stage… n n n Eat! Amylase breaks down starch glucose Digestion Circulation Into cells (facilitated diffusion) Phytosynthesis is done by autotrophs to convert solar energy into chemical energy. Cellular respiration is the process of converting chemical energy to ATP. C 6 H 12 O 6 + 6 O 2 ATP + 6 CO 2 + 6 H 2 O At first glance it may appear as if phytosynthesis and cellular respiration are the reve But they occur in different organelles (chloroplasts and mitochondria) and the chemic reactions are very different.

Transferring Energy with ATP Cells transfer energy between anabolic and catabolic reactions by using an energy middleman -ATP (adenosine triphosphate) n When ATP supplies energy to a process, one of its phosphates gets transferred to another molecule, changing it into ADP (adenosine diphosphate). n Cells can recreate ATP by using a catabolic reaction to reacttach a n
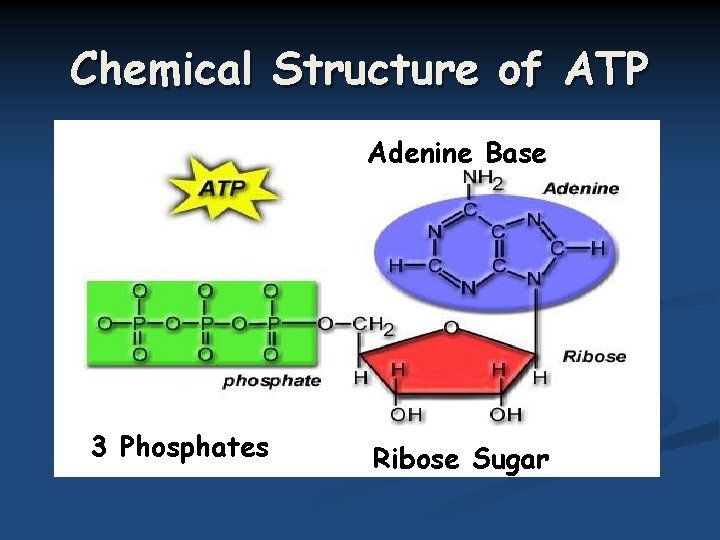
Chemical Structure of ATP Adenine Base 3 Phosphates Ribose Sugar

How Do We Get Energy From ATP? By breaking the high- energy bonds between the last two phosphates in ATP


Define Cellular Respiration Cellular respiration is a process where oxygen is used to break down organic compounds (sugars) to produce ATP (energy). n

7 One of the energy-producing reactions is called respiration (Respiration is not the same thing as breathing) The chemical reactions of respiration take place in all living cells The reaction takes place between oxygen and a substance which contains carbon. The reaction produces carbon dioxide and water, and releases energy

8 The carbon-containing substances come from FOOD The oxygen comes from the AIR (or water) The energy is used to drive other chemical reactions taking place in cells One example of this is the release of energy in muscle cells to make them contract and produce movement
10 Some examples of the use of energy in organisms muscle contraction Respiration supplies the energy for germination chemical changes in cells cell division
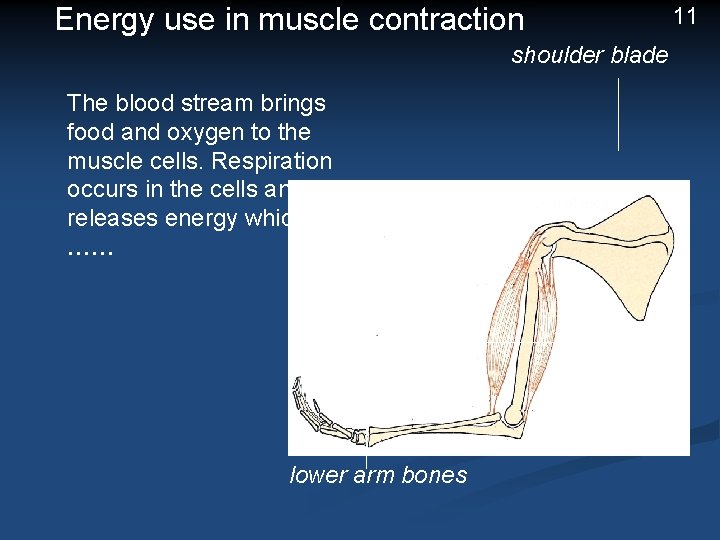
Energy use in muscle contraction 11 shoulder blade The blood stream brings food and oxygen to the muscle cells. Respiration occurs in the cells and releases energy which …… upper arm bone lower arm bones
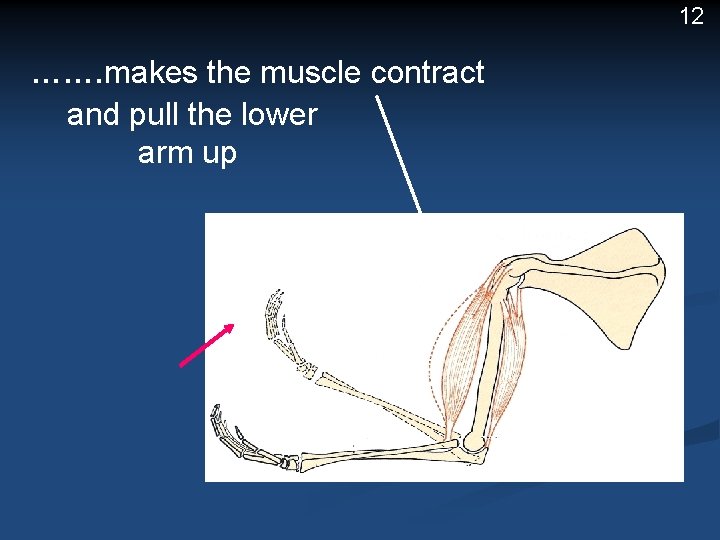
12 ……. makes the muscle contract and pull the lower arm up
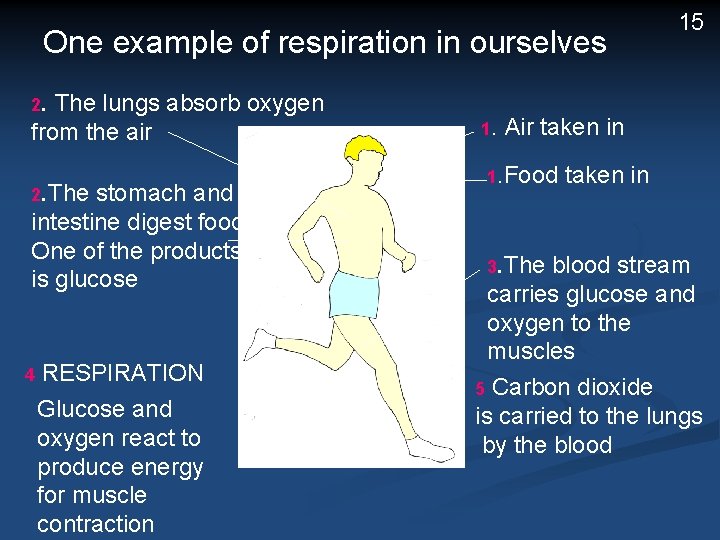
One example of respiration in ourselves 2. The lungs absorb oxygen from the air 2. The stomach and intestine digest food. One of the products is glucose 4 RESPIRATION Glucose and oxygen react to produce energy for muscle contraction 1. 15 Air taken in 1. Food 3. The taken in blood stream carries glucose and oxygen to the muscles 5 Carbon dioxide is carried to the lungs by the blood
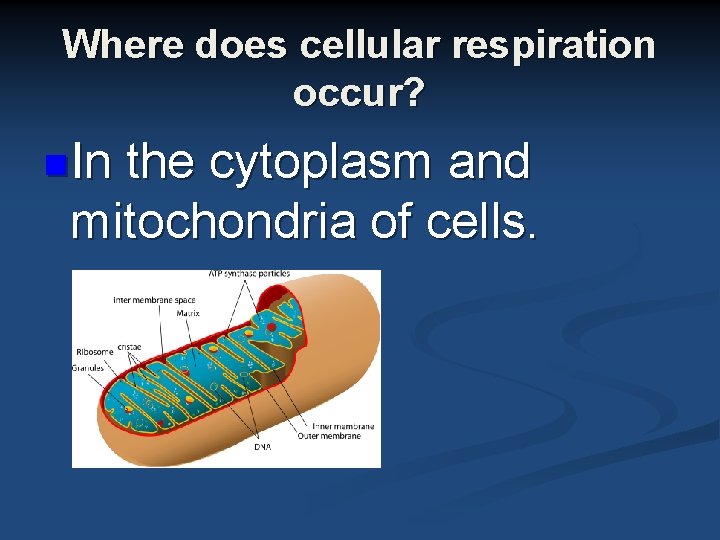
Where does cellular respiration occur? n. In the cytoplasm and mitochondria of cells.

Mitochondria n n The matrix where 3 carbon pieces that came from carbohydrates are broken down to (CO 2 and water) The cristae is where ATP is made

Mini Lab 1. Open and close your hand as many times as possible for 30 seconds. Have your partner time and record the results…QUICKLY! After only a 5 second break repeat this step for a total of 5 trials. n 2. After you have completed the 5 trials. Switch jobs with your partner, and record your partner’s 5 trials. Trial 1 Trial 2 Trial 3 Trial 4 Trial 5 Name n

Observations: n How did your hand feel after the 1 st trial? n 3 rd trial? n 5 th trial? n How did the amount of times you could open and close your hand change from the 1 st trial to the 5 th trial?

Analysis: n Can you think of a time where your body got tired from working hard? Give an example. n Why do you think your hand began feeling this way? n Why do you think your muscles get sore after a hard work out?

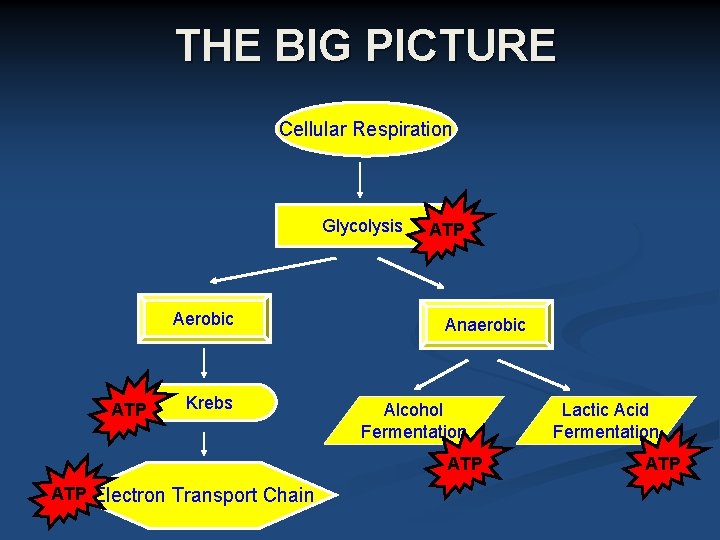
Cellular Respiration Cellular Energy • The Stages of Cellular Respiration- Cellular respiration has two main stages: • Glycolysis The first stage of cellular respiration is called glycolysis. • Aerobic and Anaerobic Respiration The second stage of cellular respiration is either aerobic respiration (in the presence of oxygen) or anaerobic respiration (in the absence of oxygen). A large amount of ATP is made during aerobic respiration.
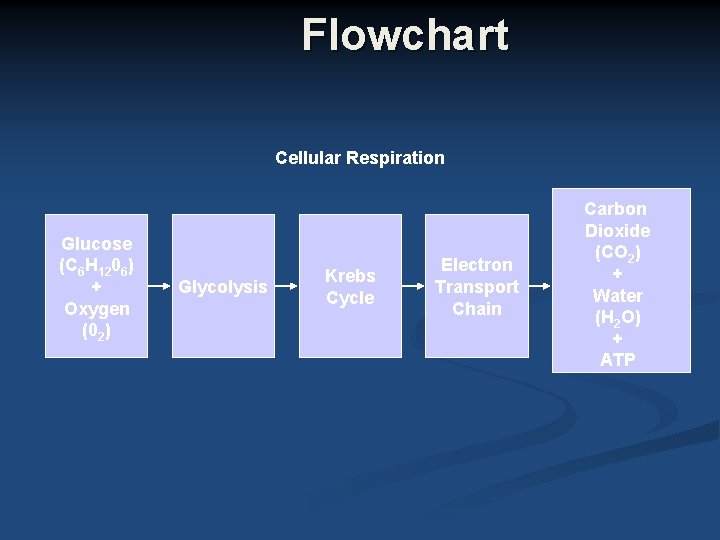
Flowchart Cellular Respiration Glucose (C 6 H 1206) + Oxygen (02) Glycolysis Krebs Cycle Electron Transport Chain Carbon Dioxide (CO 2) + Water (H 2 O) + ATP
THE BIG PICTURE Cellular Respiration Glycolysis Aerobic ATP Krebs ATP Anaerobic Alcohol Fermentation ATP Electron Transport Chain Lactic Acid Fermentation ATP

Key Terms: n. Aerobic – occurs with oxygen n. Anaerobic – occurs without oxygen
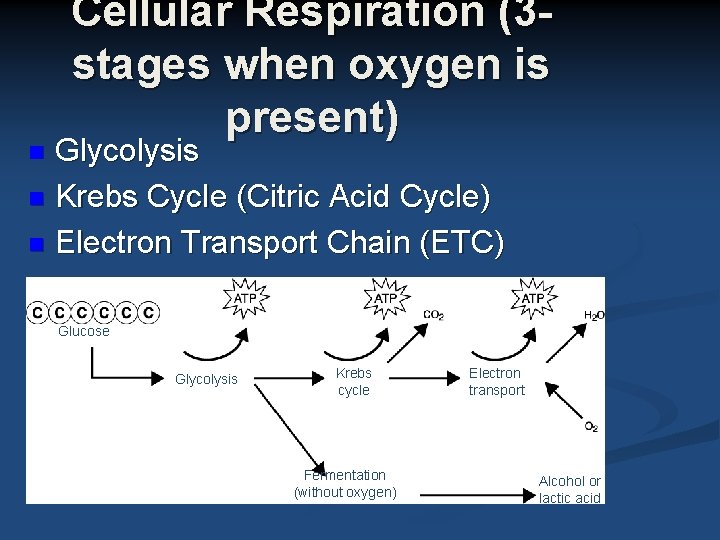
Cellular Respiration (3 stages when oxygen is present) Glycolysis n Krebs Cycle (Citric Acid Cycle) n Electron Transport Chain (ETC) n Glucose Glycolysis Krebs cycle Fermentation (without oxygen) Electron transport Alcohol or lactic acid

A little help with some important terms we will need: Enzymes are proteins BUT many need a nonprotein partner in order to do their job. n Inorganic partners such as iron, potassium, magnesium and zinc are called cofactors. n Organic partners are called coenzymes. They are small molecules that can separate from the protein and participate directly in the chemical reaction. Examples of coenzymes include many derivatives of n

Oxidation is a chemical process that removes electrons from molecules. n Reduction is the process that gives electrons to molecules. n During cellular respiration enzymes remove electrons from food molecules and transfer the electrons to coenzymes. n

Glycolysis (Splitting Sugar) n Begins with a 6 -carbon molecule, usually glucose, ends with 2, 3 carbon molecules of pyruvate. n Produces 2 ATP and occurs in the cytoplasm of prokaryotic and eukaryotic cells. C C C Glucos en Will occur with or without oxygen. Pyruvat e C C 2 ATP C C

Glycolysis n n Glucose (and phosphate from 2 ATP molecules are required to get it started) is broken down into two - 3 carbon and phosphate groups, which is then broken down into pyruvic acid and 4 ATP. Net gain of 2 ATP.

n During cellular respiration, enzymes remove electrons from food molecules and then transfer the electrons to the coenzymes nicotinamide adenine dinucleotide (NAD+) and flavin adenine NAD+ and FAD receive the electrons as part of hydrogen (H) atoms, which change them into their reduced forms: NADH and FADH 2 n

n NAD+ and FAD act like electron shuttle buses for the cell. n The empty buses NAD+ and FAD collect electron passengers. The “H” sign goes up to show that the bus is full – NADH and FADH 2 The bus drives over to the reactions that need the electrons, drop off the passengers and go back to the oxidation reaction to collect new passengers.

Glycolysis Summary Where? Cytoplasm n Oxygen required? No n n In Glucose (6 -C) n Out 2 pyruvate (3 -C) 2 NADH 2 ATP (net)
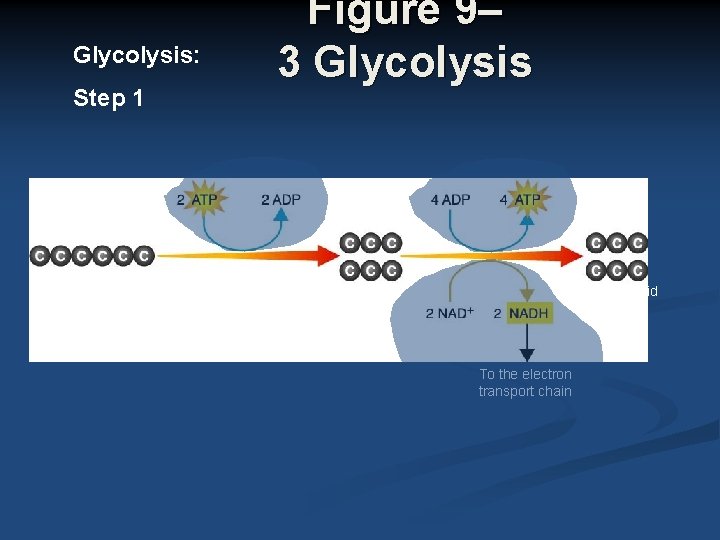
Glycolysis: Step 1 Glucose Figure 9– 3 Glycolysis 2 Pyruvic acid To the electron transport chain

A Little Krebs Cycle History n n n Discovered by Hans Krebs in 1937 He received the Nobel Prize in physiology or medicine in 1953 for his discovery Forced to leave Germany prior to WWII because he was Jewish

Krebs Cycle (Citric Acid Cycle) n Begins with two, 3 carbon molecules of pyruvate which go from the cytoplasm into the mitochondrial matrix. Together two pyruvate molecules produce: 2 ATP, 2 CO 2 and then two types of electron carriers: NADH and FADH 2 Pyruvat e C C C Electron Carrier - NADH ATP + + Electron Carrier – FADH 2 O C O

Krebs Cycle Summary Where? Mitochondrial matrix n Oxygen required? Yes n Out n In 2 CO 2 (as waste) 2 Pyruvate NADH FADH 2 2 ATP n Pyruvate is broken into a two carbon molecule called acetyl-co. A. The Krebs cycle b down acetyl-co. A into carbon dioxide. Cells use the Krebs cycle for breaking down fatty acids and amino acids.
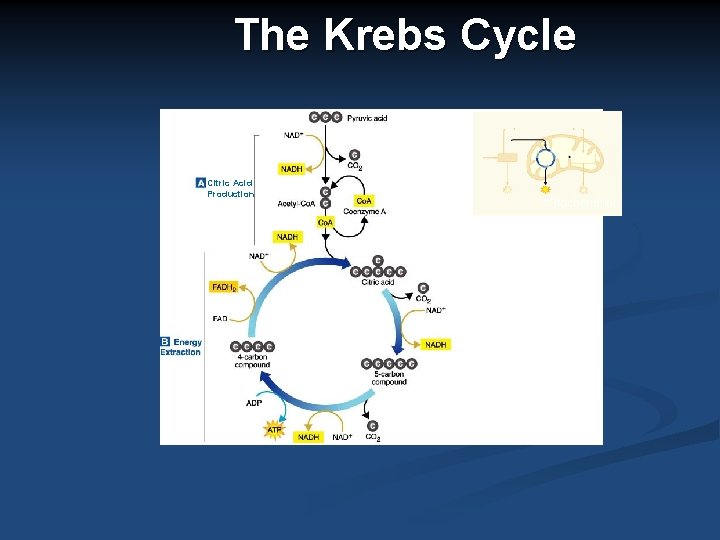
The Krebs Cycle Citric Acid Production Mitochondrion
Aerobic Respiration Glycolysis Aerobic ATP Krebs ATP Electron Transport Chain ATP

n In the inner membranes of the mitochondria in your cells, hundreds of little cellular machines are busy working to transfer energy. n The machines are called electron transfer chains. They are made of a team of proteins that sit in the membranes, transferring energy and electrons.

The coenzymes NADH and FADH 2 carry energy and electrons from glycolysis and the Kreb’s cycle to the electron transport chain. n The coenzymes transfer the electrons to the proteins of the electron transport chain, which pass the electrons down the chain. n

n Oxygen collects the electrons at the end of the chain. While oxygen accepts the electron, it also picks up protons (H+) and forms H 2 O. While electrons are transferred along the electron transport chain, the proteins move protons (H+) across the membranes of the mitochondria. n These protons flow back across the membrane through a protein called ATP synthase. n

It adds phosphate molecules to ADP forming ATP. n Process is also called oxidative phosphorylation. n At the end of this entire process, the energy transferred from glucose is 36 to 38 molecules of ATP. n

Electron Transport Chain Where? Inner membrane of mitochondria n Oxygen required? Yes n Energy Yield Total of 32 ATP n O 2 combines with TWO H+ to form H 2 O n Exhale - CO 2, H 2 O comes from cellular respiration n In n Out 32 ATP NADH electron carriers FADH 2 n
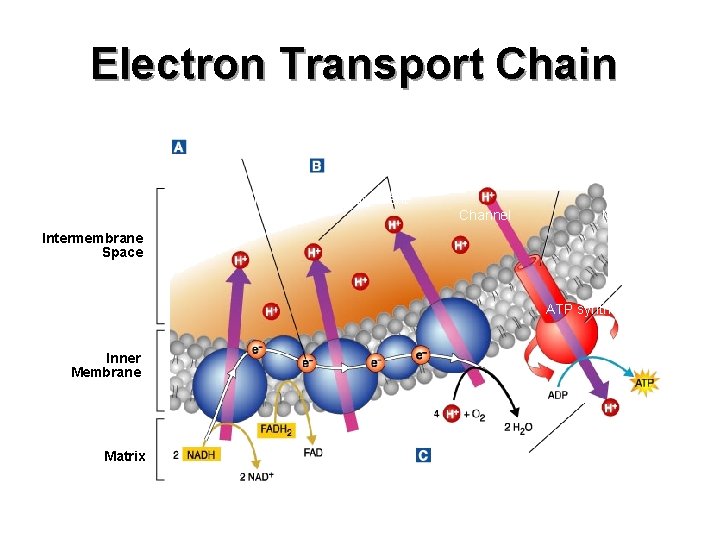
Electron Transport Chain Electron Transport Hydrogen Ion Movement Channel Mitochondrion Intermembrane Space ATP synthase Inner Membrane Matrix ATP Production

Summary of Aerobic Respiration n ATP is then used to move muscles and complete other cellular activities. n Glycolysis, Krebs and Electron transport chain together make up aerobic respiration, starting with glucose and ending with ATP. Up to 36 ATP

Aerobic Respiration Glycolysis 2 ATP Aerobic Krebs Total of up to 36 ATP 2 ATP Electron Transport Chain Up to 32 ATP

The first two processes Glycolysis and the Krebs cycle break down food molecules. n The third pathway oxidative phosphorylation transfers energy from the food molecules to ATP. n
Anaerobic Respiration Cellular Respiration Glycolysis ATP Anaerobic Alcohol Fermentation ATP Lactic Acid Fermentation ATP

Cellular Respiration Fermentation in the Absence of Oxygen • Fermentation When oxygen is not present, fermentation follows. • Lactic Acid Fermentation In lactic acid fermentation, pyruvate is converted to lactate.

Anaerobic Respiration 23

25 Anaerobic respiration can be represented by the equation C 6 H 12 O 6 glucose energy 2 C 2 H 5 OH alcohol + 2 CO 2 The energy released by anaerobic respiration is considerably less than the energy from aerobic respiration. Anaerobic respiration takes place at some stage in the cells of most living organisms. For example, our own muscles resort to anaerobic respiration when oxygen is not delivered to them fast enough.

Anaerobic Respiration (low oxygen or no oxygen) 3 types of anaerobic respiration: 1. Lactic Acid Fermentation – occurs when you run without proper warm-up. Carbon dioxide and lactic acid are produced from pyruvic acid. Results in muscle soreness.

n 2. Alcohol Fermentation – pyruvic acid is broken down into ethyl alcohol (ethanol) and carbon dioxide. Results in food/beverages that are high in calories. n 3. Acetic Acid Fermentation – Pyruvic acid is broken down into acetic acid (vinegar) and carbon dioxide. Cabbage becomes sauerkraut.

Alcohol fermentation Used by bacteria and yeast (a type of fungus). Alcohol and CO 2 are products of this reaction. n For thousands of years, humans have used yeast in brewing, winemaking and baking. The bubbles (CO 2) generated by baker’s yeast allow n

n Lactic Acid Fermentation Lactic acid fermentation is used by certain fungi and bacteria in the dairy industry to make cheese and yogurt. Lactate is the product of this reaction.

Lactic acid fermentation is also used by human muscles. n Muscles make ATP by lactic acid fermentation when oxygen is scarce. n (For instance, opening and closing your hand many times over short periods of time, or lifting a weight for many repetitions. ) n
n As your muscles run out of oxygen, they need a quick way to make more ATP. Lactic acid fermentation provides the ATP but produces lactate which accumulates causing muscle soreness. Eventually, the lactate is carried away by blood to the liver where it is converted back to
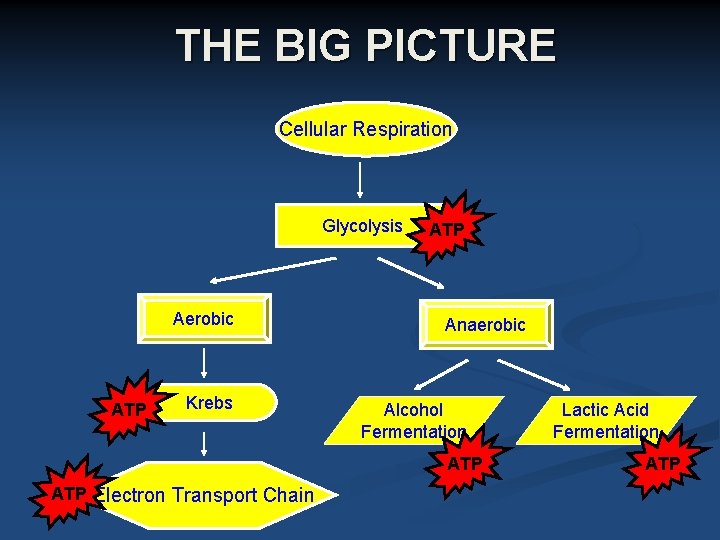
THE BIG PICTURE Cellular Respiration Glycolysis Aerobic ATP Krebs ATP Anaerobic Alcohol Fermentation ATP Electron Transport Chain Lactic Acid Fermentation ATP

Aerobic vs. Anaerobic n Anaerobic DOES NOT require oxygen Simple n fast n produces smaller amounts of energy (ATP) n n Aerobic requires oxygen Yields large amounts of energy n What is this energy molecule? n ATP, ATP n

Lactic Acid Fermentation n bacteria, plants and most animals n After glycolysis n n 2 pyruvic acid changed to lactic acid Sometimes happens in your muscles, cramps-----Exercise

Alcoholic Fermentation n Bacteria and fungi (yeast) n Ethyl alcohol and carbon dioxide are the end products Process used to form beer, wine, and other alcoholic beverages n Also used to raise dough, bread n
- Slides: 68