Cellular Respiration Harvesting Chemical Energy AP Biology Harvesting

Cellular Respiration Harvesting Chemical Energy AP Biology

Harvesting stored energy § Energy is stored in organic molecules u heterotrophs eat food (organic molecules) § digest organic molecules w serve as raw materials for building & fuels for energy § controlled release of energy w series of step-by-step enzyme-controlled reactions u “burning” fuels § carbohydrates, lipids, proteins, nucleic acids AP Biology 2005 -2006
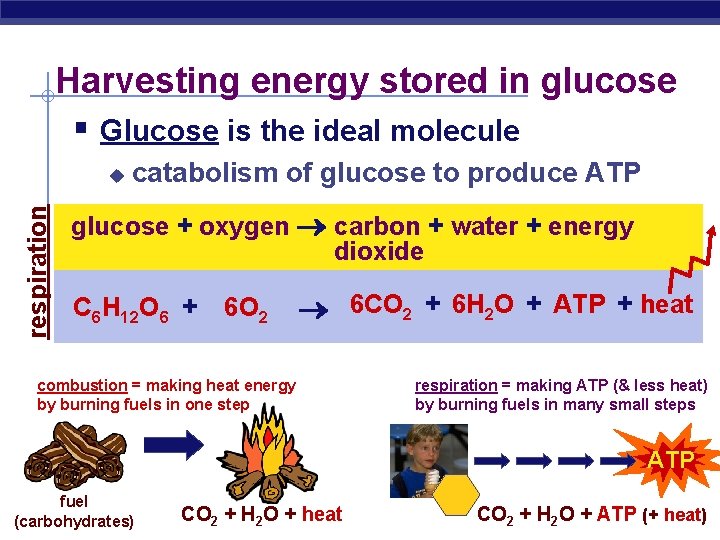
Harvesting energy stored in glucose § Glucose is the ideal molecule respiration u catabolism of glucose to produce ATP glucose + oxygen carbon + water + energy dioxide C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + ATP + heat combustion = making heat energy by burning fuels in one step respiration = making ATP (& less heat) by burning fuels in many small steps ATP fuel AP Biology (carbohydrates) CO 2 + H 2 O + heat CO 2 + H 2 O +2005 -2006 ATP (+ heat)
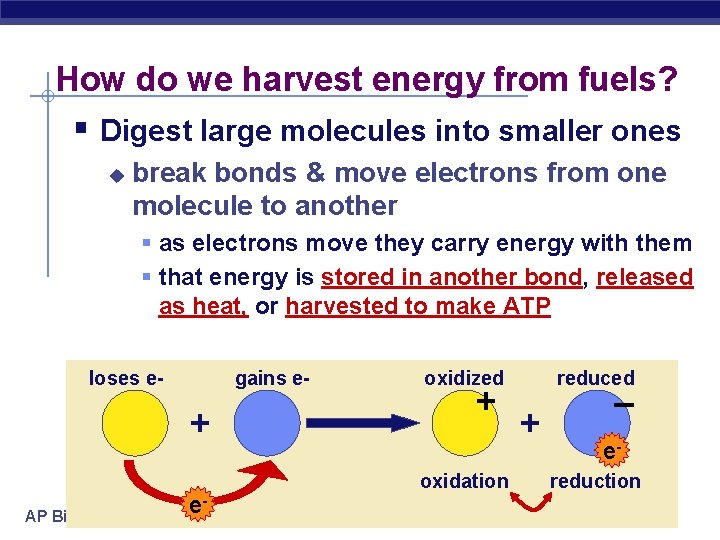
How do we harvest energy from fuels? § Digest large molecules into smaller ones u break bonds & move electrons from one molecule to another § as electrons move they carry energy with them § that energy is stored in another bond, released as heat, or harvested to make ATP loses e- gains e- + AP Biology e- oxidized + oxidation reduced + – ereduction 2005 -2006

Complex organic molecules Catabolic Pathway Simpler waste products with less E Some E used to do work and dissipated as heat AP Biology
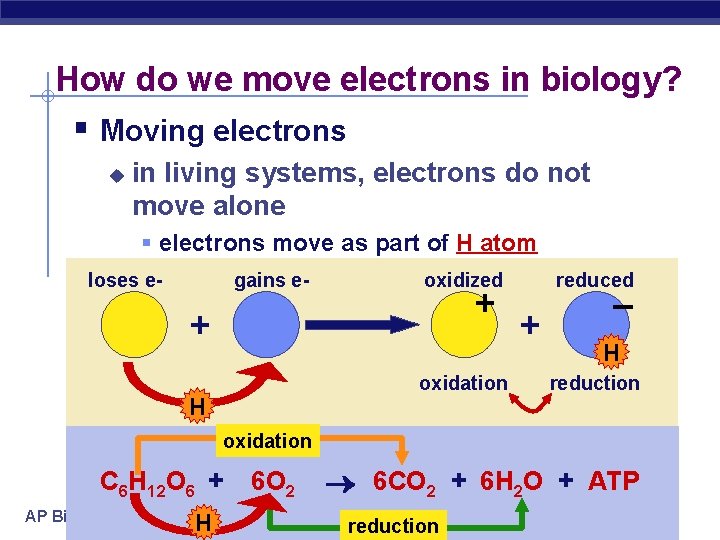
How do we move electrons in biology? § Moving electrons u in living systems, electrons do not move alone § electrons move as part of H atom loses e- gains e- oxidized + + oxidation H reduced + – H reduction oxidation C 6 H 12 O 6 + AP Biology H 6 O 2 6 CO 2 + 6 H 2 O + ATP reduction 2005 -2006
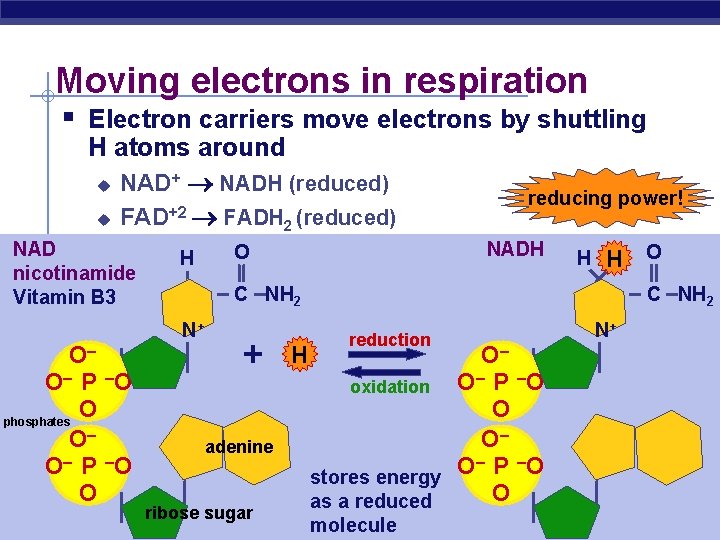
Moving electrons in respiration § Electron carriers move electrons by shuttling H atoms around u NAD+ NADH (reduced) u FAD+2 FADH 2 (reduced) NAD nicotinamide Vitamin B 3 O– O – P –O O phosphates O– O – P –O O AP Biology H reducing power! NADH O H H O C NH 2 N+ + adenine ribose sugar H reduction O– O – P –O oxidation O O– – P –O O stores energy O as a reduced molecule N+ 2005 -2006
Coupling oxidation & reduction § Redox reactions in respiration u release energy as breakdown molecules § break C-C bonds § strip off electrons from C-H bonds by removing H atoms w C 6 H 12 O 6 CO 2 = fuel has been oxidized § electrons attracted to more electronegative atoms w in biology, the most electronegative atom? O 2 w O 2 H 2 O = oxygen has been reduced § release energy to synthesize ATP oxidation C 6 H 12 O 6 + AP Biology 6 O 2 6 CO 2 + 6 H 2 O + ATP reduction 2005 -2006
Oxidation & reduction § Oxidation § Reduction adding O u removing H u loss of electrons u releases energy u exergonic u removing O u adding H u gain of electrons u stores energy u endergonic u oxidation C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + ATP reduction AP Biology

Redox Reactions (oxidation-reduction) oxidation (donor) lose e- Xe- + Y X + Yereduction (acceptor) gain e Oxidation = lose e Reduction = gain e- - Oi. LRi. G or Leo. Ger oxidation C 6 H 12 O 6 + 6 O 2 6 H 2 O + 6 CO 2 + AP Biology reduction ATP
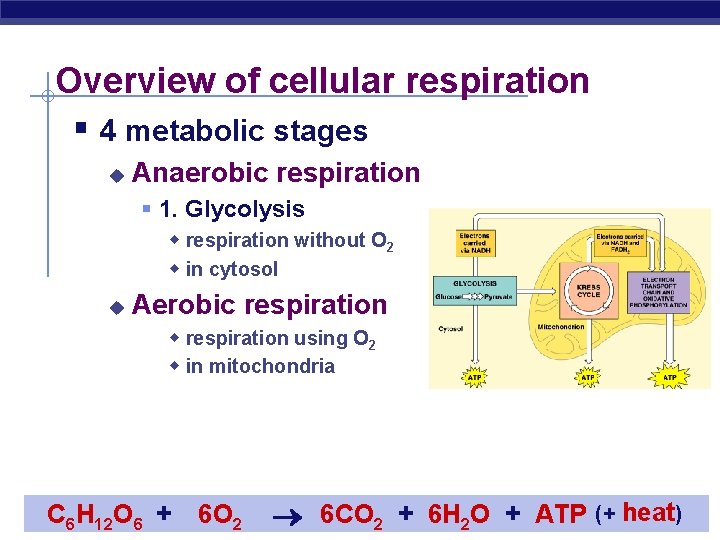
Overview of cellular respiration § 4 metabolic stages u Anaerobic respiration § 1. Glycolysis w respiration without O 2 w in cytosol u Aerobic respiration w respiration using O 2 w in mitochondria C H O 6 + AP Biology 6 12 6 O 2 (+ heat) 6 CO 2 + 6 H 2 O + ATP 2005 -2006
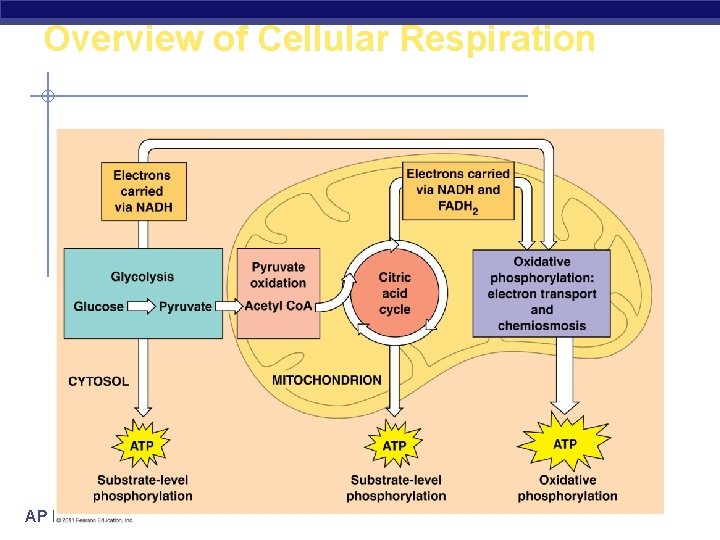
Overview of Cellular Respiration AP Biology

Chapter 9. Cellular Respiration STAGE 1: Glycolysis AP Biology

Glycolysis § Breaking down glucose u “glyco – lysis” (splitting sugar) glucose pyruvate 2 x 3 C 6 C u most ancient form of energy capture § starting point for all cellular respiration u inefficient § generate only 2 ATP for every 1 glucose u in cytosol § why does that make evolutionary sense? AP Biology

Glycolysis Stage 1: Energy Investment Stage § Cell uses ATP to phosphorylate compounds of glucose Stage 2: Energy Payoff Stage § Two 3 -C compounds oxidized § For each glucose molecule: 2 Net ATP produced by substrate-level phosphorylation u 2 molecules of NAD+ NADH u AP Biology
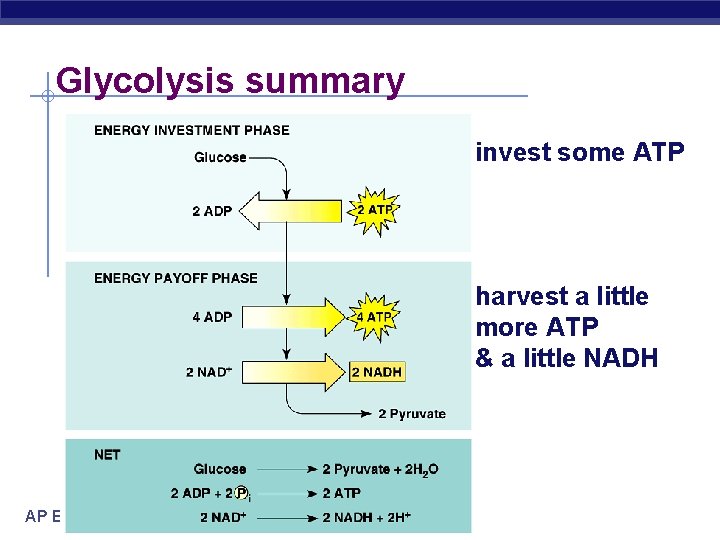
Glycolysis summary invest some ATP harvest a little more ATP & a little NADH AP Biology
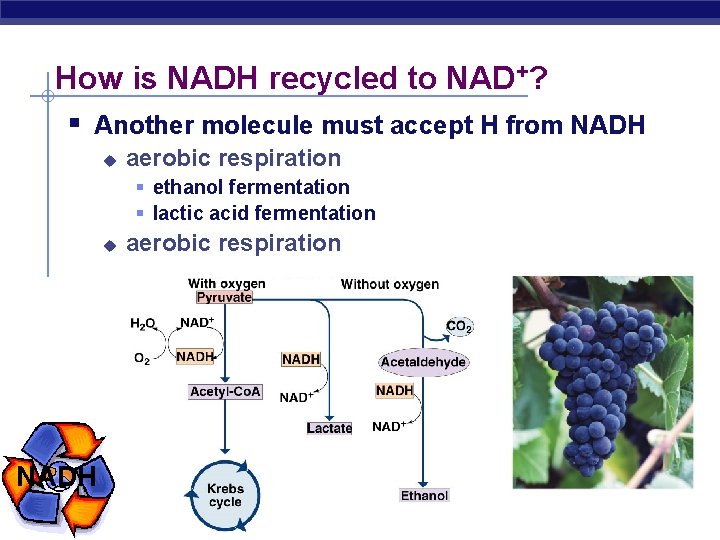
How is NADH recycled to NAD+? § Another molecule must accept H from NADH u aerobic respiration § ethanol fermentation § lactic acid fermentation u NADH AP Biology aerobic respiration
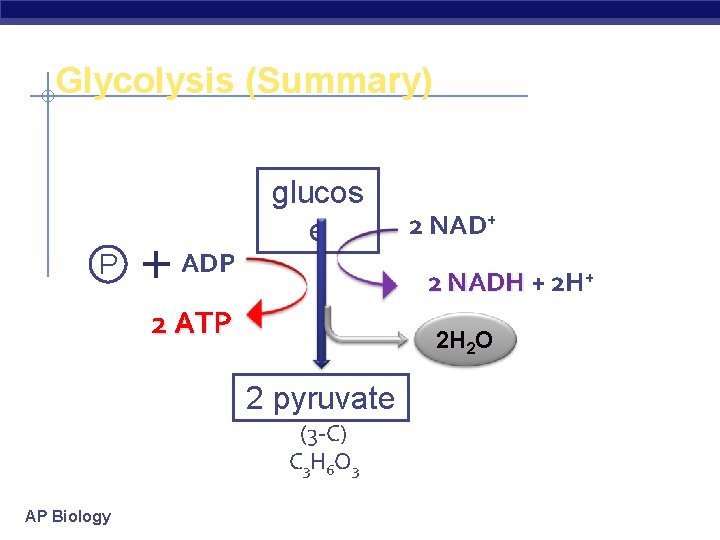
Glycolysis (Summary) P ADP glucos e 2 NADH + 2 H+ 2 ATP 2 H 2 O 2 pyruvate (3 -C) C 3 H 6 O 3 AP Biology 2 NAD+
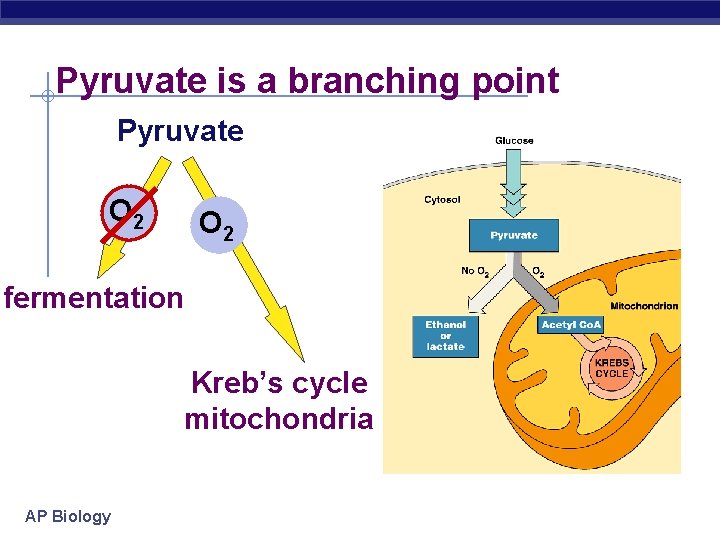
Pyruvate is a branching point Pyruvate O 2 fermentation Kreb’s cycle mitochondria AP Biology

Anaerobic ethanol fermentation § Bacteria, yeast pyruvate ethanol + CO 2 3 C NADH 2 C 1 C NAD+ § beer, wine, bread § at ~12% ethanol, kills yeast § Animals, some fungi pyruvate lactic acid 3 C NADH 3 C NAD+ § cheese, yogurt, anaerobic exercise (no O 22005 -2006 ) AP Biology

Types of Fermentation Alcohol fermentation § Pyruvate Ethanol + § § CO 2 Ex. bacteria, yeast Used in brewing, winemaking, baking Lactic acid fermentation § Pyruvate Lactate § Ex. fungi, bacteria, human § § AP Biology muscle cells Used to make cheese, yogurt, acetone, methanol Note: Lactate build-up does NOT causes muscle fatigue and pain (old idea)
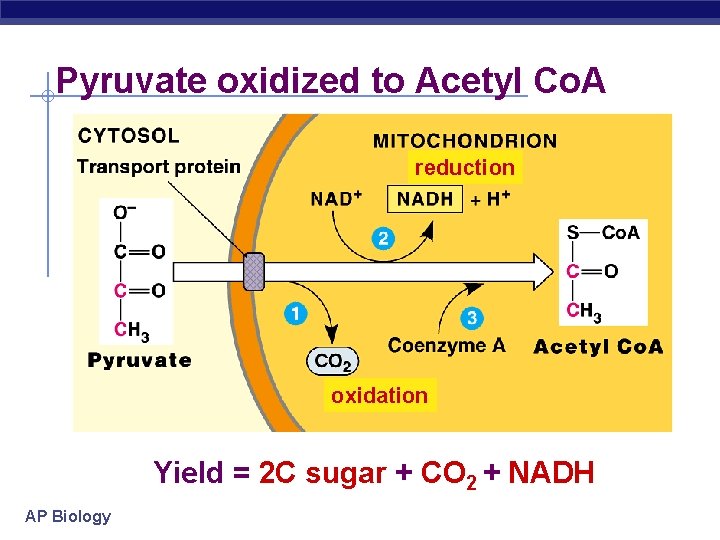
Pyruvate oxidized to Acetyl Co. A reduction oxidation Yield = 2 C sugar + CO 2 + NADH AP Biology

Citric Acid Cycle (Krebs) § Occurs in mitochondrial matrix § Acetyl Co. A Citrate released CO § Net gain: 2 ATP, 6 NADH, 22 FADH 2 (electron § carrier) ATP produced by substrate-level phosphorylation AP Biology
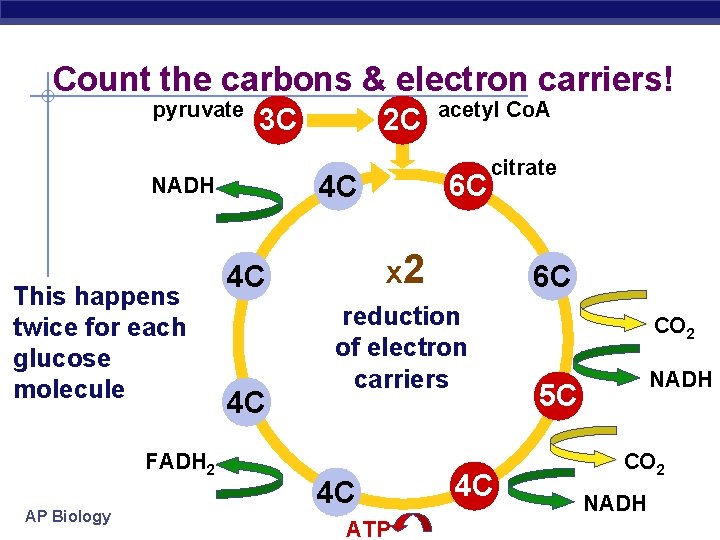
Count the carbons & electron carriers! pyruvate 3 C FADH 2 AP Biology citrate x 2 4 C 4 C acetyl Co. A 6 C 4 C NADH This happens twice for each glucose molecule 2 C 6 C reduction of electron carriers 4 C ATP 4 C CO 2 NADH 5 C CO 2 NADH
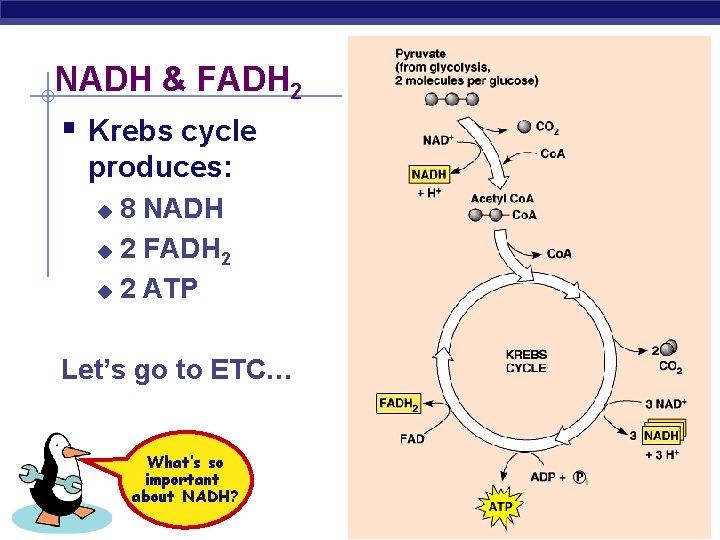
NADH & FADH 2 § Krebs cycle produces: 8 NADH u 2 FADH 2 u 2 ATP u Let’s go to ETC… What’s so important about NADH? AP Biology 2005 -2006
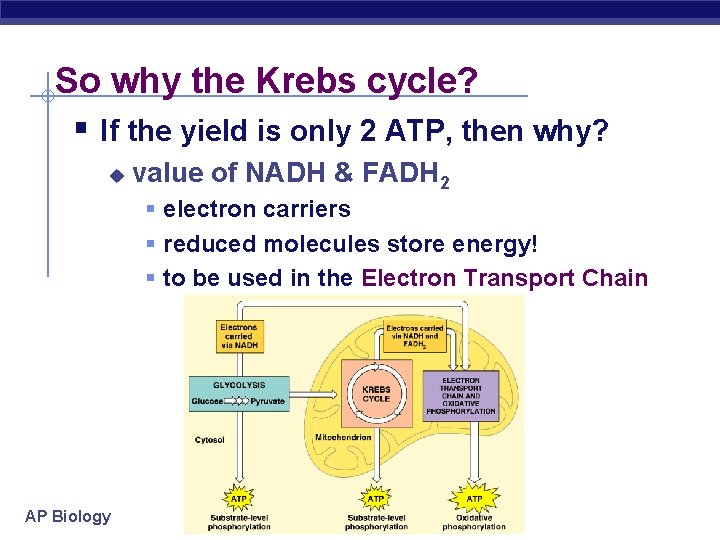
So why the Krebs cycle? § If the yield is only 2 ATP, then why? u value of NADH & FADH 2 § electron carriers § reduced molecules store energy! § to be used in the Electron Transport Chain AP Biology

ATP accounting so far… § Glycolysis 2 ATP § Kreb’s cycle 2 ATP § Life takes a lot of energy to run, need to extract more energy than 4 ATP! Why stop here… AP Biology There’s got to be more to life than this.

Last stop and most important! § Electron Transport Chain u series of molecules built into inner mitochondrial membrane § mostly transport (integral) proteins transport of electrons down ETC linked to ATP synthesis u yields ~34 ATP from 1 glucose! u only in presence of O 2 (aerobic) u AP Biology That sounds more like it!
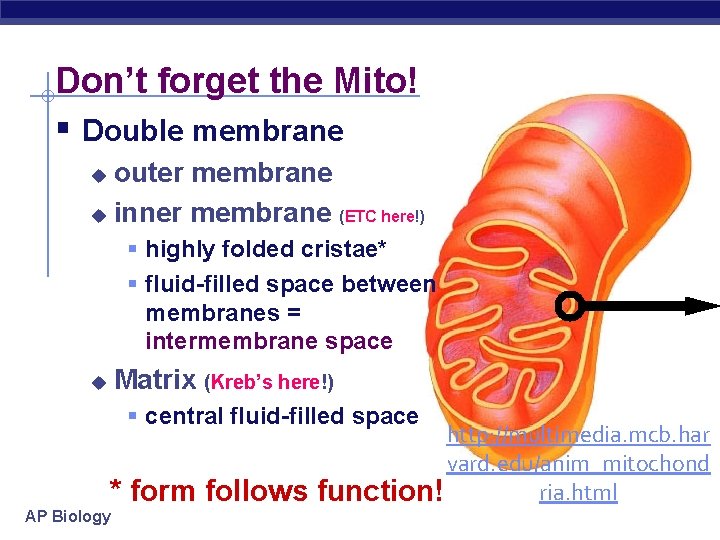
Don’t forget the Mito! § Double membrane outer membrane u inner membrane (ETC here!) u § highly folded cristae* § fluid-filled space between membranes = intermembrane space Matrix (Kreb’s here!) u § central fluid-filled space * form follows AP Biology http: //multimedia. mcb. har vard. edu/anim_mitochond ria. html function!

Electron Transport Chain (ETC) § Collection of molecules § § embedded in inner membrane of mitochondria Tightly bound protein + nonprotein components Alternate between reduced/oxidized states as accept/donate e. Does not make ATP directly Ease fall of e- from food to O 2 2 H+ + ½ O 2 H 2 O § § §AP Biology
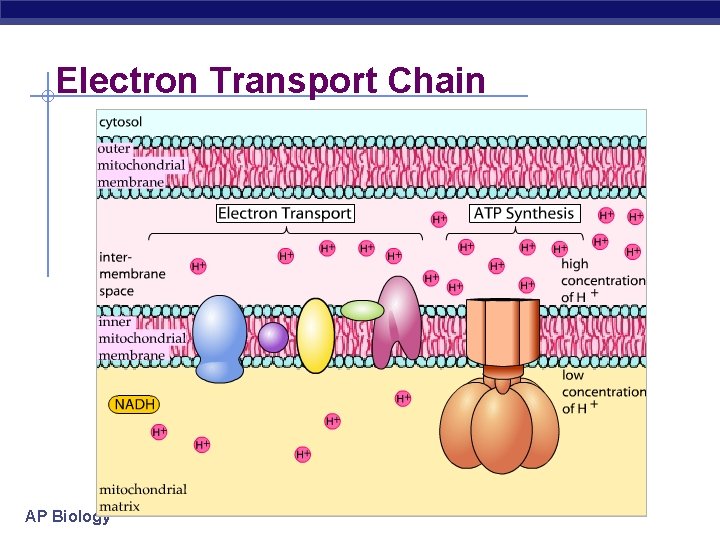
Electron Transport Chain AP Biology
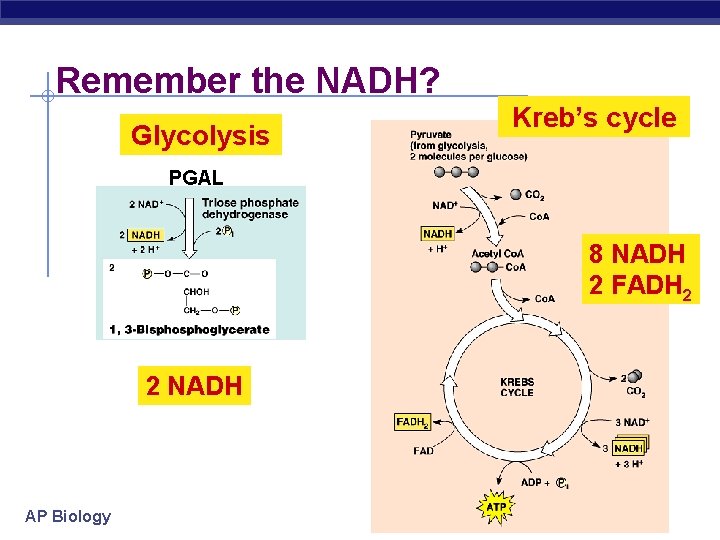
Remember the NADH? Glycolysis Kreb’s cycle PGAL 8 NADH 2 FADH 2 2 NADH AP Biology 2005 -2006
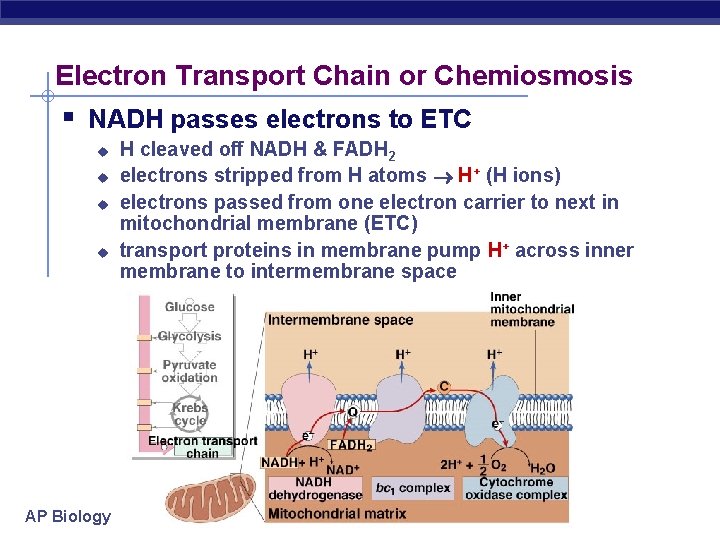
Electron Transport Chain or Chemiosmosis § NADH passes electrons to ETC u u AP Biology H cleaved off NADH & FADH 2 electrons stripped from H atoms H+ (H ions) electrons passed from one electron carrier to next in mitochondrial membrane (ETC) transport proteins in membrane pump H+ across inner membrane to intermembrane space
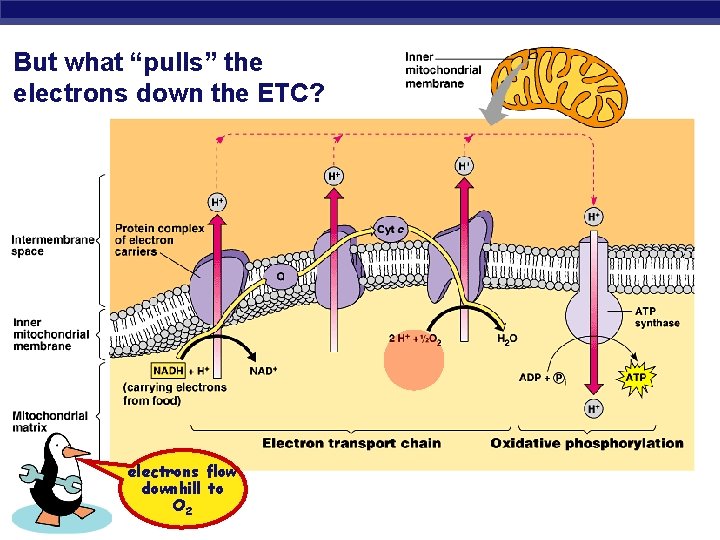
But what “pulls” the electrons down the ETC? AP Biology electrons flow downhill to O 2
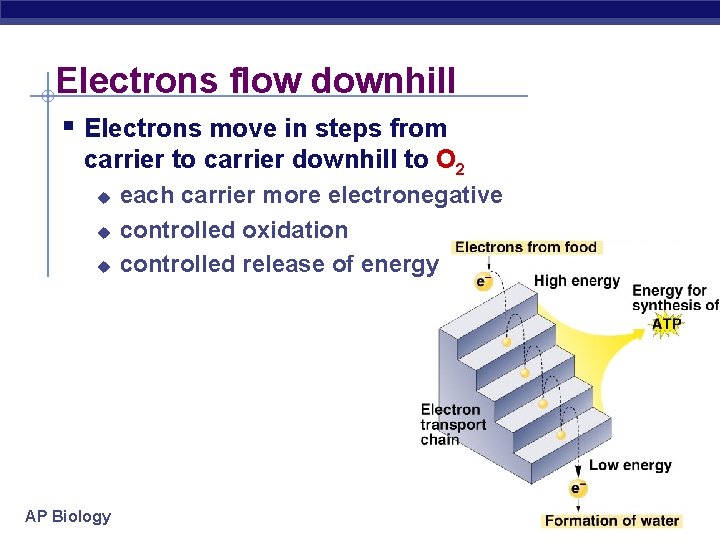
Electrons flow downhill § Electrons move in steps from carrier to carrier downhill to O 2 u u u AP Biology each carrier more electronegative controlled oxidation controlled release of energy 2005 -2006
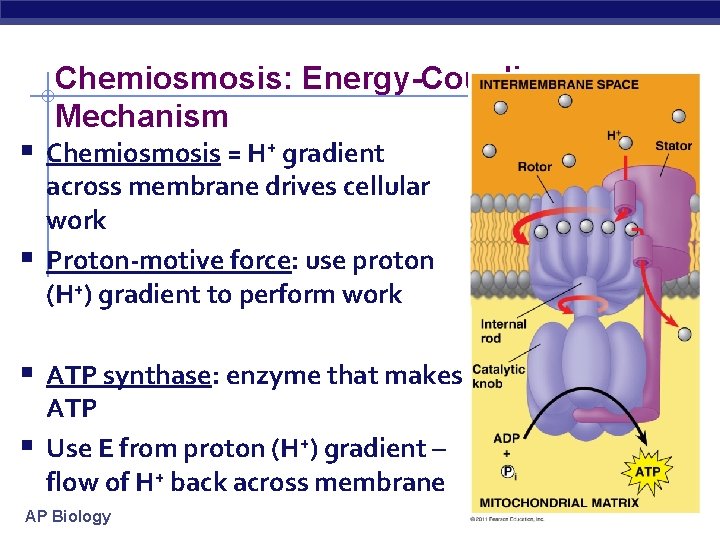
Chemiosmosis: Energy-Coupling Mechanism § Chemiosmosis = H+ gradient § across membrane drives cellular work Proton-motive force: use proton (H+) gradient to perform work § ATP synthase: enzyme that makes § ATP Use E from proton (H+) gradient – flow of H+ back across membrane AP Biology
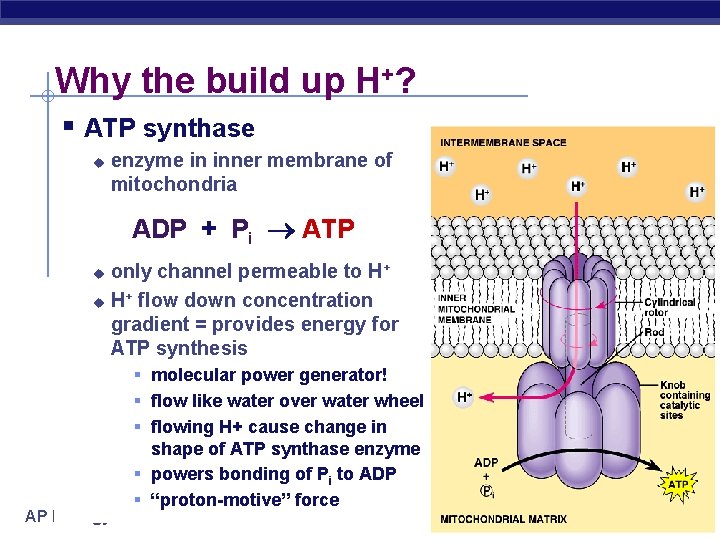
Why the build up H+? § ATP synthase u enzyme in inner membrane of mitochondria ADP + Pi ATP only channel permeable to H+ u H+ flow down concentration gradient = provides energy for ATP synthesis u AP Biology § molecular power generator! § flow like water over water wheel § flowing H+ cause change in shape of ATP synthase enzyme § powers bonding of Pi to ADP § “proton-motive” force 2005 -2006
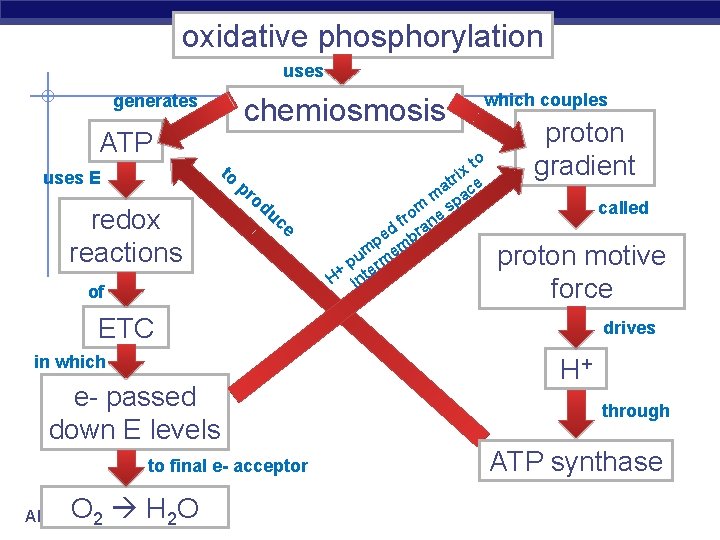
oxidative phosphorylation uses generates chemiosmosis ATP to uses E redox reactions pr od uc e of which couples to x i r at ce m pa s m fro ane d r pe mb m e pu erm H+ int proton gradient called proton motive force ETC in which e- passed down E levels to final e- acceptor O H 2 O AP Biology 2 drives H+ through ATP synthase
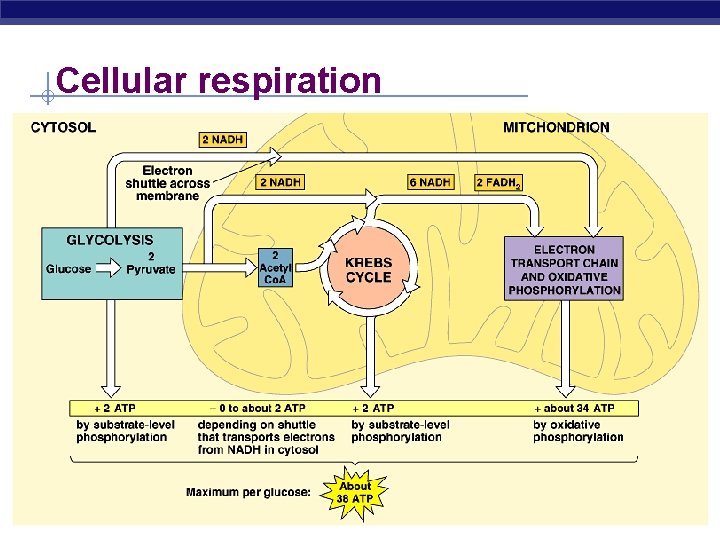
Cellular respiration AP Biology 2005 -2006

Bio. Flix: Cellular Respiration AP Biology
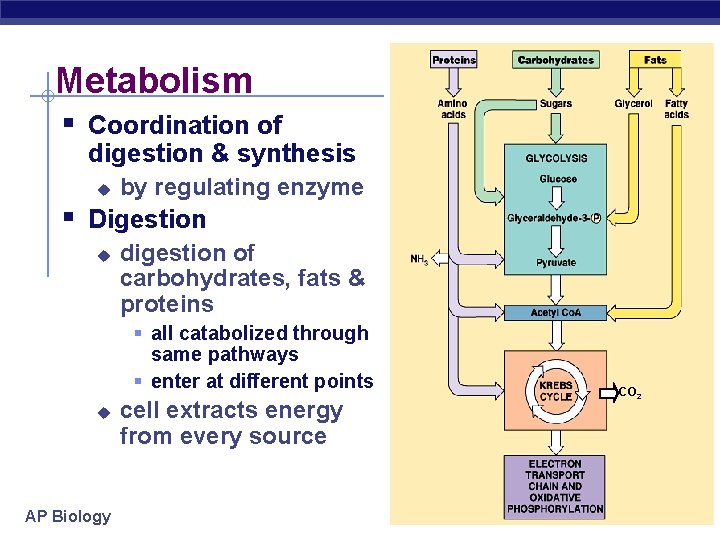
Metabolism § Coordination of digestion & synthesis u by regulating enzyme § Digestion u digestion of carbohydrates, fats & proteins § all catabolized through same pathways § enter at different points u AP Biology cell extracts energy from every source CO 2 2005 -2006
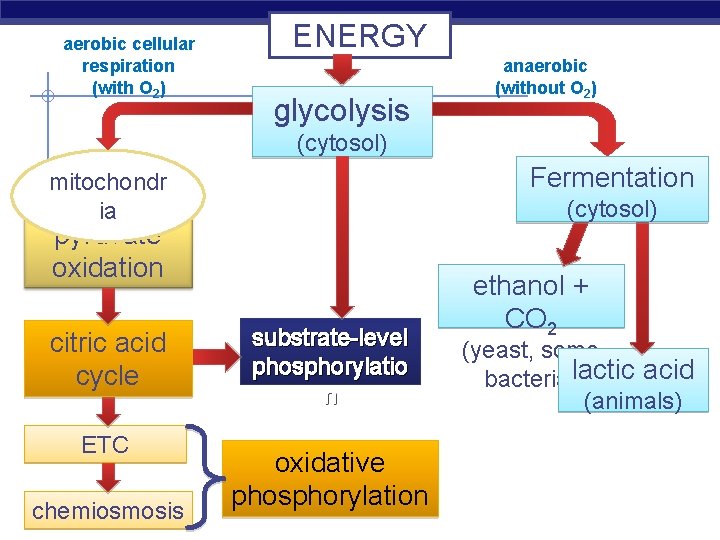
aerobic cellular respiration (with O 2) ENERGY glycolysis anaerobic (without O 2) (cytosol) Fermentation mitochondr ia (cytosol) pyruvate oxidation citric acid cycle ETC chemiosmosis AP Biology substrate-level phosphorylatio n oxidative phosphorylation ethanol + CO 2 (yeast, some bacteria)lactic acid (animals)

Summary of cellular respiration C 6 H 12 O 6 + 6 O 2 § § § § 6 CO 2 + 6 H 2 O + ~36 ATP Where did the glucose come from? Where did the O 2 come from? Where did the CO 2 come from? Where did the H 2 O come from? Where did the ATP come from? What else is produced that is not listed in this equation? Why do we breathe? AP Biology

Taking it beyond… § What is the final electron acceptor in electron transport chain? O 2 § So what happens if O 2 unavailable? § ETC backs up § ATP production ceases § cells run out of energy § and you die! AP Biology
- Slides: 44