Cellular Respiration Breakdown of glucose to carbon dioxide

Cellular Respiration Breakdown of glucose to carbon dioxide and water

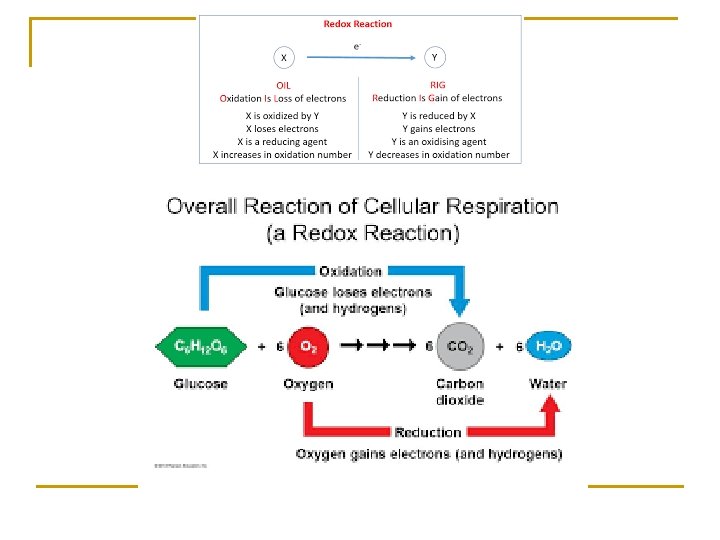
Redox reaction n n Hydrogen atoms consist of a hydrogen ion and an electron (H+ and e-) Glucose is oxidized when the hydrogen is removed Oxygen is reduced when it gains hydrogen and becomes water Exergonic reaction – glucose is a high energy molecule water and carbon dioxide are low energy molecules

ATP n n Build up of ATP = endergonic Glucose is broken down slowly, ATP produced gradually Maximum production of ATP = 36 – 38 39% efficiency from glucose to ATP

Coenzymes n NAD+ (nicotinamide adenine dinucleotide) q n Accepts 2 electrons and 1 hydrogen = NADH FAD (flavin adenine dinucleotide) q Accepts 2 electrons and 2 hydrogens = FADH 2

Making of ATP n Oxidative phosphorylation – ATP made by the redox reactions of the Electron Transport Chain (ETC and Chemiosmosis) q n Accounts for 90% of the ATP generated in respiration Substrate Level Phosphorylation – enzyme transfer a P group from a substrate molecule to ATP

Phases of cellular respiration n n Glycolysis – outside mitochondria, anaerobic, splitting of glucose – 2 pyruvate, yield 2 ATP Preparatory reaction – in mitochondria, pyruvate oxidized to 2 – C acetyl group, preps for citric acid cycle Citric acid cycle – (Krebs) in matrix of mitochondria, yield 2 ATP Electron transport chain – cristae, oxygen is final electron acceptor and forms water, result in 32 – 34 ATP

Glycolysis –see handout/text n n n Outside of mitochondria in cytoplasm Glucose 2 pyruvate Does not require oxygen, anaerobic
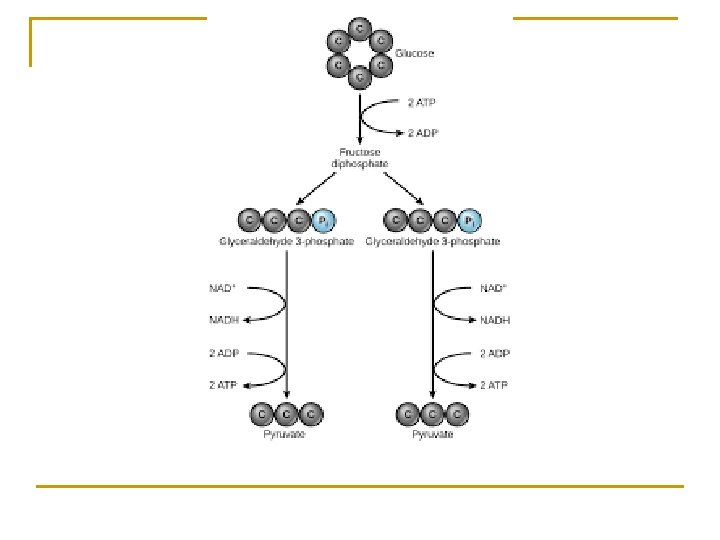

Energy investment n 2 ATP are used to activate glucose and break down into 2 C 3 molecules of G 3 P.

Energy harvesting n n Electrons removed (oxidation) with hydrogen ions. NAD picks up Hydrogen and electrons = NADH Substrate level phosphorylation – production of ATP via an enzyme passing a high energy phosphate to ADP = 4 ATP Investment of 2 ATP minus 4 ATP = 2 net ATP

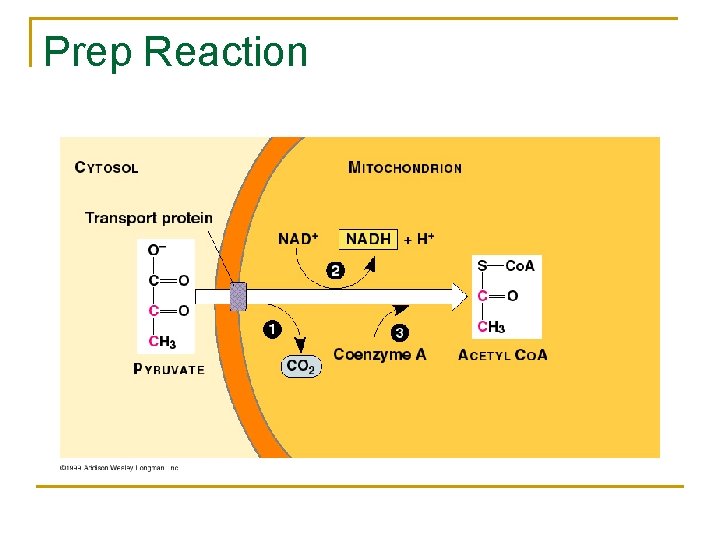
Prep Reaction n n Pyruvate (2 C 3 molecules) is converted to an acetyl group (C 2) that is attached to coenzyme A (Co. A) CO 2 is a product Oxidation occurs, electrons from pyruvate are removed, NADH Reaction occurs twice since there are 2 pyruvate per glucose molecule
Prep Reaction

Prep reaction Inputs: 2 pyruvate, 2 NAD+, 2 Co. A n Outputs: 2 CO 2 (Product), 2 NADH, 2 acetyl Co. A n

Citric Acid Cycle n n Matrix of mitochondria C 2 (acetyl Co. A) joins with C 4 molecule = C 6 citrate molecule. Each acetyl group is oxidized to 2 CO 2 molecules = 4 CO 2 total released Cycle goes around 2 times

Citric Acid Cycle n n n Substrate Level phosphorylation - An enzyme passes a high energy phosphate to ADP = ATP Inputs: 2 acetyl Co. A, 6 NAD, 2 ADP/P, 2 FAD Outputs: 4 CO 2 (product), 6 NADH, 2 FADH 2, 2 ATP
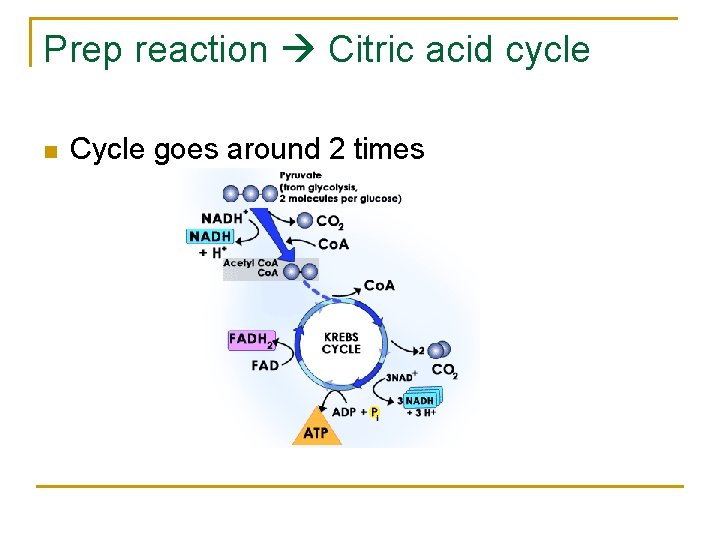
Prep reaction Citric acid cycle n Cycle goes around 2 times

Electron Transport Chain n Electrons are carried by NADH and FADH 2 Oxidation-reduction reaction starts the ETC. High energy electrons enter the chain, low energy electrons leave. There is a series of carriers that transport the electrons, first reduced when it accepts the electrons, then oxidized when it releases them.

Carriers n n n 3 protein complexes (NADH-Q reductase, cytochrome reductase and cytochrome oxidase) 2 carriers that transport electrons on ETC– coenzyme Q and cytochrome c Cytochrome – protein that has a tightly bound heme group with a central atom of iron.
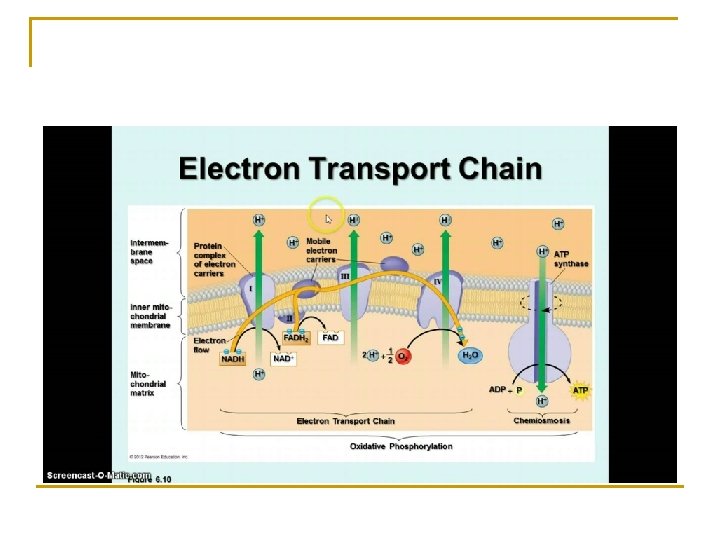

Oxidative phosphorylation n Hydrogen ions are pumped out of the matrix into the intermembrane space, H+ flow back through membrane due to H+ gradient = chemiosmosis n Production of ATP as a result of energy released by the ETC = 32 – 34 ATP n Production of ATP via substrate phosphorylation is 4 ATP Total ATP = 36 – 38 ATP 39% efficiency of cellular resp. n

ETC n n For each NADH that is oxidized, 3 ATP molecules are produced For each FADH 2 that is oxidized, 2 ATP are produced, (due to the fact that they follow NADH and electrons are at a lower energy level).

Electron transport chain n n Oxygen is the final electron acceptor in the ETC Receives last electrons and combines with the hydrogen ions to form water (product)
- Slides: 23