Cellular Respiration A catabolic exergonic oxygen O 2

Cellular Respiration • A catabolic, exergonic, oxygen (O 2) requiring process that uses energy extracted from macromolecules (glucose) to produce energy (ATP) and water (H 2 O). C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy glucose ATP Mag Bio 1

Question: • In what kinds of organisms does cellular respiration take place? • Answer: ALL organisms respire! While some do not respire aerobically (with O 2), all organisms need energy (yes…even prokaryotes!) Mag Bio 2

Eukaryotes: Primary Focus will be on Plants and Animals • Plants - Autotrophs: self-producers. Autotrophs • Animals - Heterotrophs: consumers. Mag Bio 3
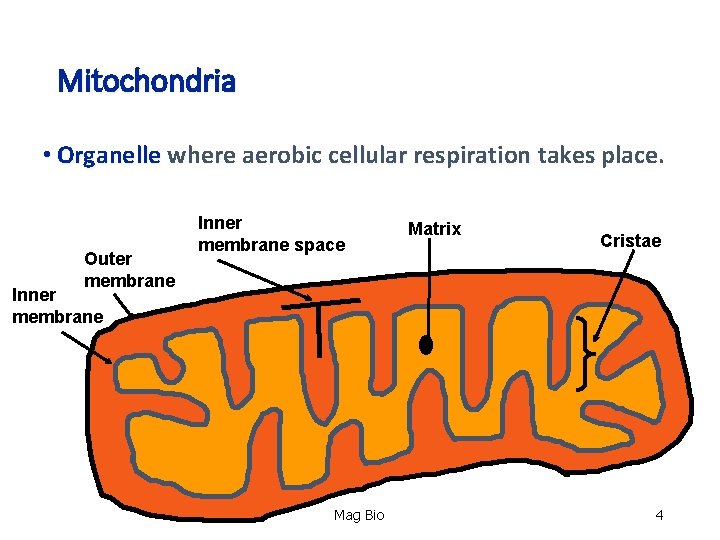
Mitochondria • Organelle where aerobic cellular respiration takes place. Organelle Outer membrane Inner membrane space Matrix Cristae Inner membrane Mag Bio 4

Redox Reaction • Transfer of one or more electrons from one reactant to another. • Two types: 1. Oxidation 2. Reduction Mag Bio 5
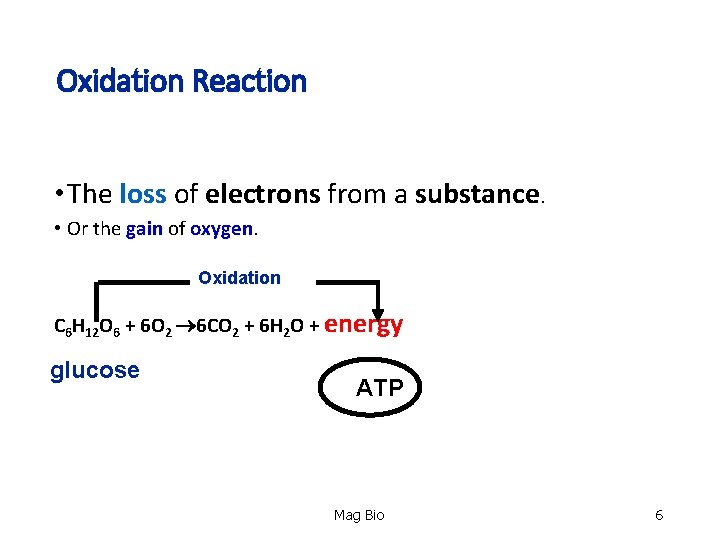
Oxidation Reaction • The loss of electrons from a substance. loss • Or the gain of gain oxygen Oxidation C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy glucose ATP Mag Bio 6
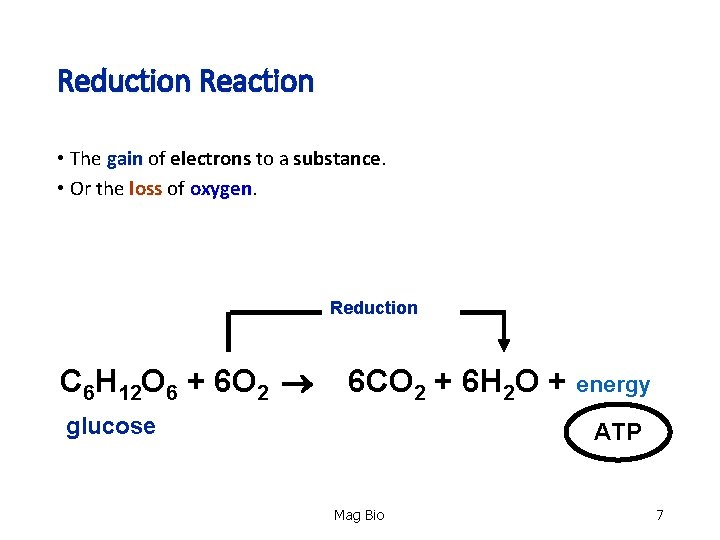
Reduction Reaction • The gain of electrons to a substance. gain • Or the loss of loss oxygen Reduction C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy glucose ATP Mag Bio 7

Breakdown of Cellular Respiration • Four main parts (reactions). 1. Glycolysis (splitting of sugar) a. cytosol, just outside of mitochondria. 2. ‘Grooming Phase’ (‘Pre-Krebs’) a. migration from cytosol to matrix. Mag Bio 8

Breakdown of Cellular Respiration 3. Krebs Cycle (Citric Acid Cycle) a. mitochondrial matrix 4. Electron Transport Chain (ETC) and Oxidative Phosphorylation a. Also called Chemiosmosis b. inner mitochondrial membrane. Mag Bio 9

Stage 1: Glycolysis • Occurs in the cytosol just outside of mitochondria. • Glycolysis occurs in EVERY organism • Two phases (10 steps): A. Energy investment phase a. Preparatory phase (first 5 steps). B. Energy yielding phase a. Energy payoff phase (second 5 steps). Mag Bio 10
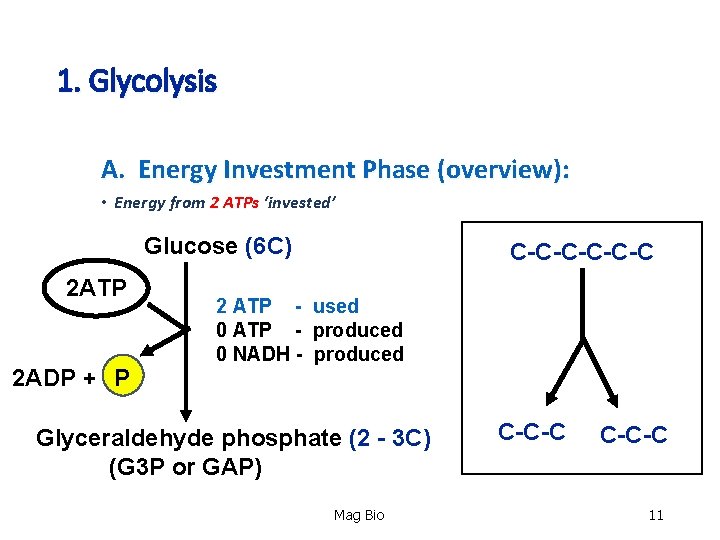
1. Glycolysis A. Energy Investment Phase (overview): • Energy from 2 ATPs ‘invested’ Glucose (6 C) 2 ATP 2 ADP + P C-C-C-C 2 ATP - used 0 ATP - produced 0 NADH - produced Glyceraldehyde phosphate (2 - 3 C) (G 3 P or GAP) Mag Bio C-C-C 11
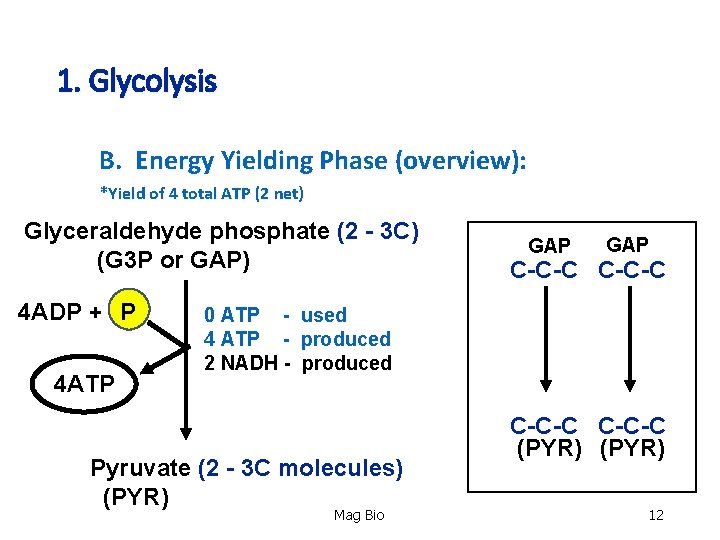
1. Glycolysis B. Energy Yielding Phase (overview): *Yield of 4 total ATP (2 net) Glyceraldehyde phosphate (2 - 3 C) (G 3 P or GAP) 4 ADP + P 4 ATP GAP C-C-C 0 ATP - used 4 ATP - produced 2 NADH - produced Pyruvate (2 - 3 C molecules) (PYR) Mag Bio C-C-C (PYR) 12

1. Glycolysis • Total Net Yield 2 - 3 C-Pyruvate (PYR) 2 - ATP (by Substrate-level Phosphorylation) 2 - NADH *Keep in mind- the ‘goal’ here is to oxidize glucose and ‘extract’ energy from its bonds. When –OH groups become =O, glucose is being oxidized! So where does the H (from the –OH) and the 2 electrons (between the O and H) go? NAD+ is going to ‘shuttle’ them to the ETC, and gets reduced to NADH. **This is another example of ‘coupling’ Mag Bio 13
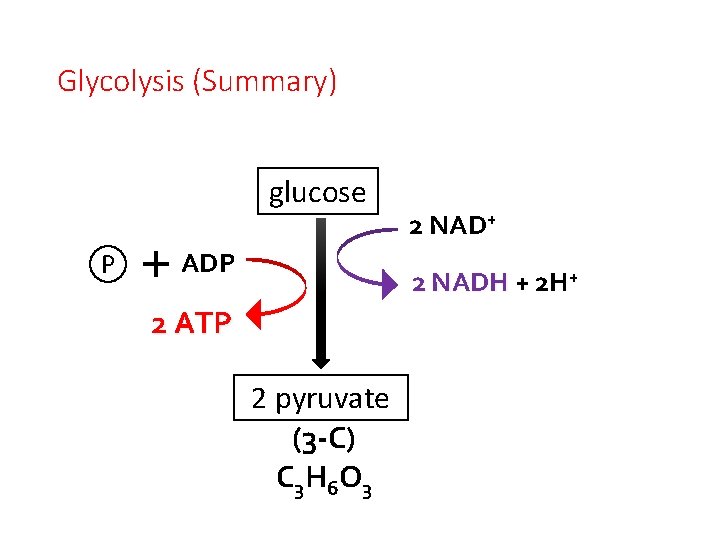
Glycolysis (Summary) glucose P ADP 2 NAD+ 2 NADH + 2 H+ 2 ATP 2 pyruvate (3 -C) C 3 H 6 O 3
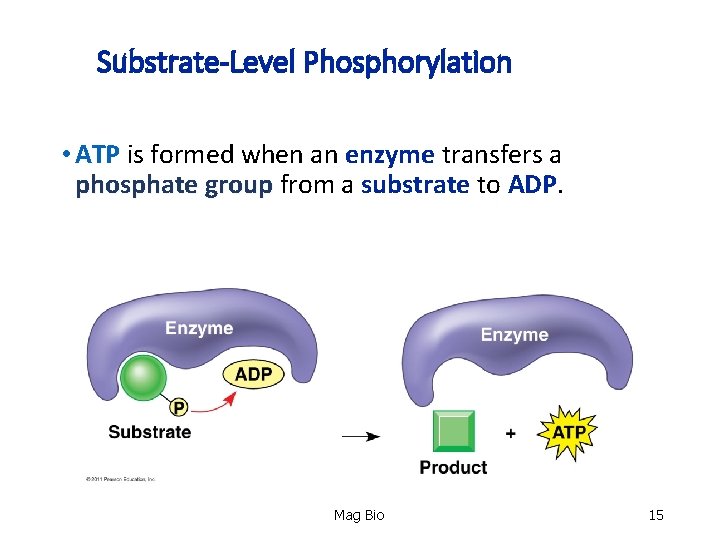
Substrate-Level Phosphorylation • ATP is formed when an enzyme transfers a phosphate group from a substrate to ADP. Mag Bio 15

Fermentation • Occurs in cytosol when “NO Oxygen” is present (called anaerobic). • Remember: glycolysis is part of fermentation • Two Types: 1. Alcohol Fermentation 2. Lactic Acid Fermentation Mag Bio 16
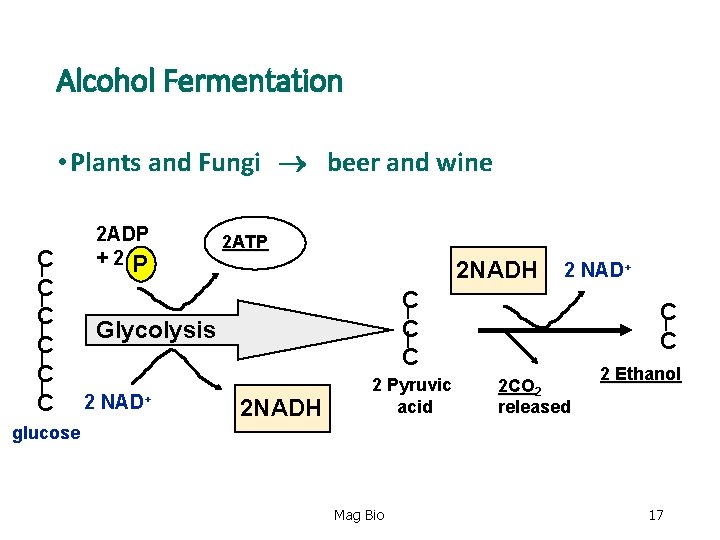
Alcohol Fermentation • Plants and Fungi beer and wine C C C 2 ADP +2 P 2 ATP 2 NADH C C C Glycolysis 2 NAD+ 2 NADH 2 Pyruvic acid C C 2 CO 2 released 2 Ethanol glucose Mag Bio 17

Alcohol Fermentation • End Products: Alcohol fermentation 2 - ATP (substrate-level phosphorylation) 2 - CO 2 2 - Ethanol’s Question- Why does NADH have to put the H and 2 e- back in fermentation? ? Mag Bio 18
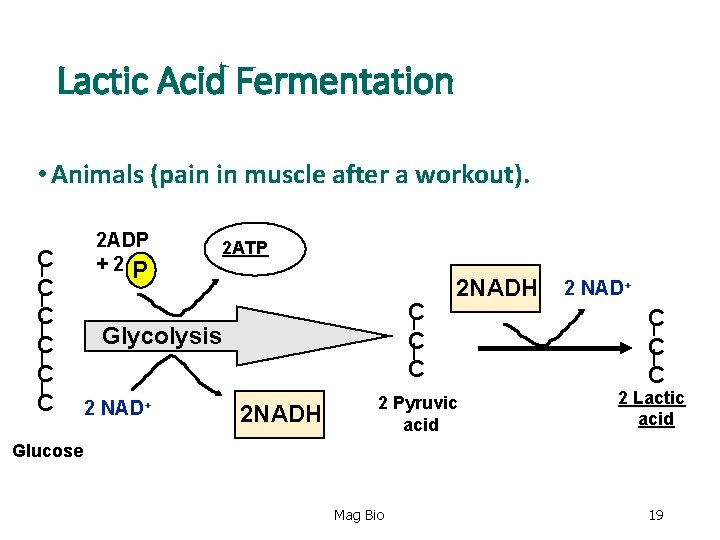
Lactic Acid Fermentation • Animals (pain in muscle after a workout). C C C 2 ADP +2 P 2 ATP C C C Glycolysis 2 NAD+ 2 NADH 2 Pyruvic acid 2 NAD+ C C C 2 Lactic acid Glucose Mag Bio 19

Lactic Acid Fermentation • End Products: Lactic acid fermentation 2 - ATP (substrate-level phosphorylation) 2 - Lactic Acids Mag Bio 20
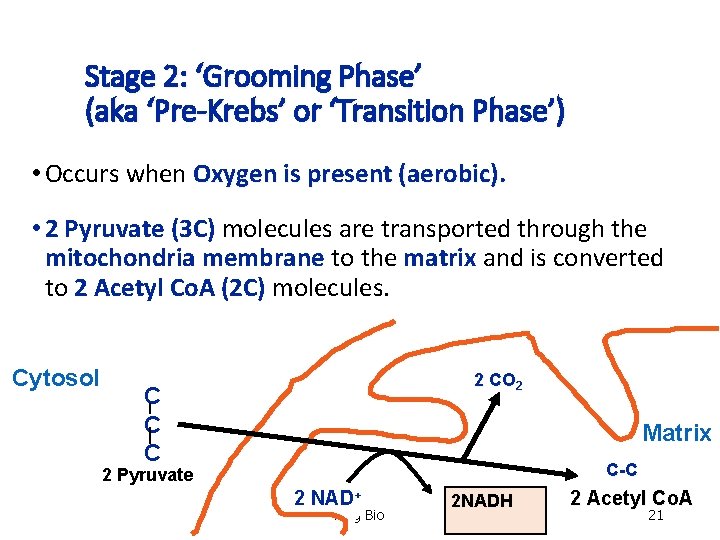
Stage 2: ‘Grooming Phase’ (aka ‘Pre-Krebs’ or ‘Transition Phase’) • Occurs when Oxygen is present (aerobic). • 2 Pyruvate (3 C) molecules are transported through the mitochondria membrane to the matrix and is converted to 2 Acetyl Co. A (2 C) molecules. Cytosol 2 CO 2 C C C Matrix C-C 2 Pyruvate 2 NAD+ Mag Bio 2 NADH 2 Acetyl Co. A 21

2. Grooming Phase • End Products: grooming phase 2 - NADH 2 - CO 2 2 - Acetyl Co. A (2 C) Mag Bio 22

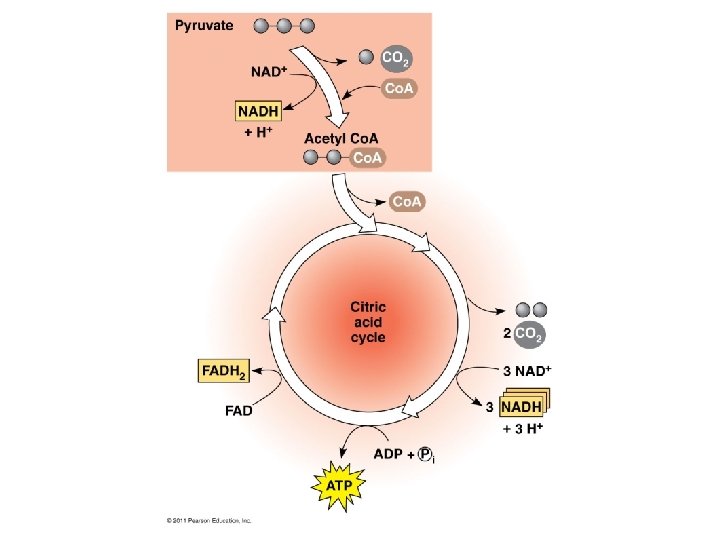
Stage 3: Krebs Cycle (Citric Acid Cycle) • Location: mitochondrial matrix. Location: • Acetyl Co. A (2 C) bonds to Oxalacetic acid (4 C - OAA) to make Citrate (6 C). • It takes 2 turns of the krebs cycle to oxidize 1 glucose molecule. Mitochondrial Matrix Mag Bio 23
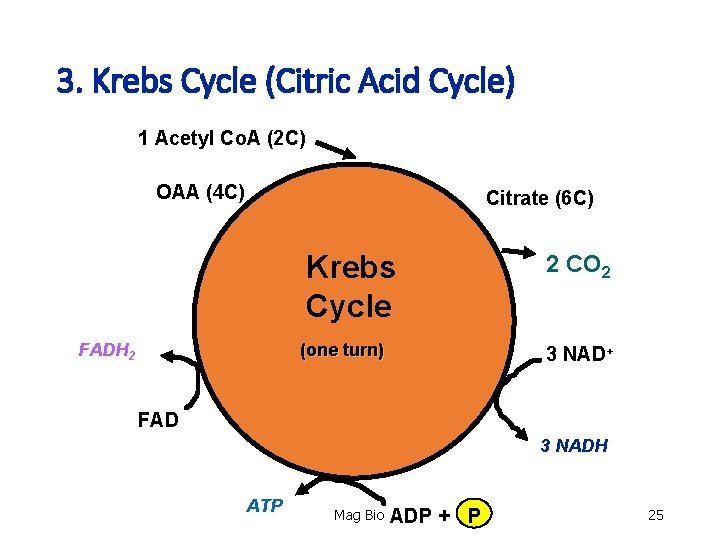
3. Krebs Cycle (Citric Acid Cycle) 1 Acetyl Co. A (2 C) OAA (4 C) Citrate (6 C) Krebs Cycle FADH 2 (one turn) 2 CO 2 3 NAD+ FAD 3 NADH ATP Mag Bio ADP + P 25
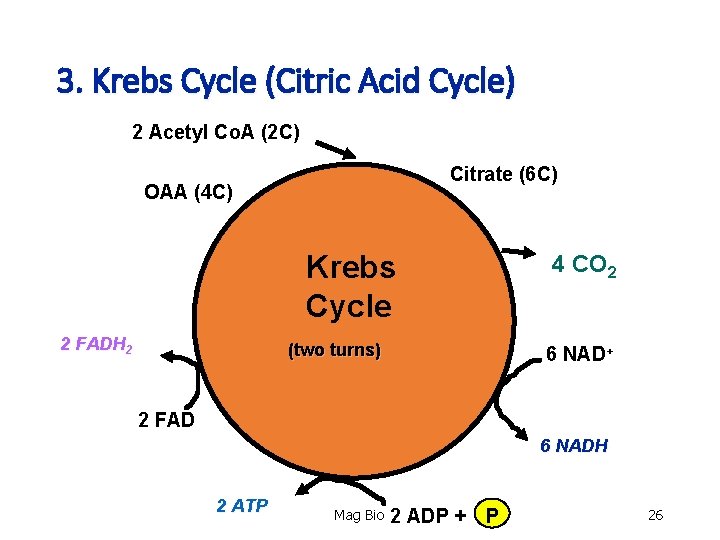
3. Krebs Cycle (Citric Acid Cycle) 2 Acetyl Co. A (2 C) Citrate (6 C) OAA (4 C) Krebs Cycle 2 FADH 2 (two turns) 4 CO 2 6 NAD+ 2 FAD 6 NADH 2 ATP Mag Bio 2 ADP + P 26

3. Krebs Cycle (Citric Acid Cycle) • Total net yield (2 turns of krebs cycle) 1. 2 - ATP (substrate-level phosphorylation) 2. 6 - NADH 3. 2 - FADH 2 4. 4 - CO 2 Mag Bio 27

• reduced forms (NADH and FADH 2) of NAD+ and FAD+, important coenzymes in the cell. Part of this is considered an aerobic pathway (oxygen-requiring) because the NADH and FADH 2 produced must transfer their electrons to the next pathway in the system, which will use oxygen to make WATER! If oxygen is not present, this transfer does not occur. Mag Bio 28

4. Electron Transport Chain (ETC) and Oxidative Phosphorylation (Chemiosmosis) • Location: inner mitochondrial membrane. Location: • Primary ‘goal’- to synthesize a LOT of ATP. So, how? • NADH and FADH 2 deliver the H’s and e- to the ETC. The e - are transported down a series of proteins in the membrane (like a ‘hot potato’). This produces enough energy to pump H+ across the membrane to the intermembrane space. Inner Mitochondrial Membrane Mag Bio 29
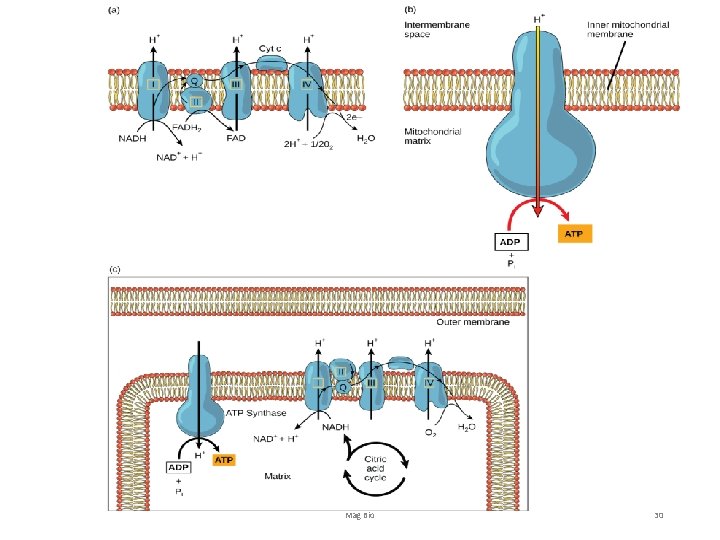
Mag Bio 30

4. Electron Transport Chain (ETC) and Oxidative Phosphorylation (Chemiosmosis) • Location: inner mitochondrial membrane. Location: • Remember: H+ are protons. As they are pumped into the space, they are very energized. Plus they all have + charge, low p. H and are in [high concentration]. This proton motive force causes an enzyme in the membrane, ATP Synthase, to let them diffuse back through (like water over a dam!). This energy transforms many ‘low’ energy ADP + P into ATP. Process is called chemiosmosis. Mag Bio 31

4. Electron Transport Chain (ETC) and Oxidative Phosphorylation (Chemiosmosis) -So what happened to all the e- being passed down the chain? Plus where are the H’s that just passed thru ATP Synthase? -As the e- are passed from protein to protein (think of each successive protein as being more electronegative than the other) there’s something waiting on them that’s more electronegative than all of them…Oxygen (O 2). So now we have oxygen Electrons and H+. What can we make out of that? Answer: WATER! Oxygen is the FINAL receptor of electrons!! Mag Bio 32

4. Summary of ETC & the Process of Chemiosmosis: • The high concentration of H+ in intermembrane space used Proton Motive Force through ATP Synthase to make ATP. • All NADH and FADH 2 delivers H’s and e- to ETC. As a result, they become oxidized (“OXIDATIVE) to NAD+ and FAD. The proteins in the chain became reduced as the e- were shuttled down the chain. • How much ATP gets made? (“PHOSPHORILATION”) -Each NADH converts to ~ 3 ATP. -Each FADH 2 converts to ~2 ATP (enters the ETC ‘downstream’ from NADH). 33

Summary • Finally, water is made from H’s, e- and oxygen as a result of the ETC and chemiosmosis • Let’s look at the process using some diagrams (then we’ll watch some good animations) Mag Bio 34
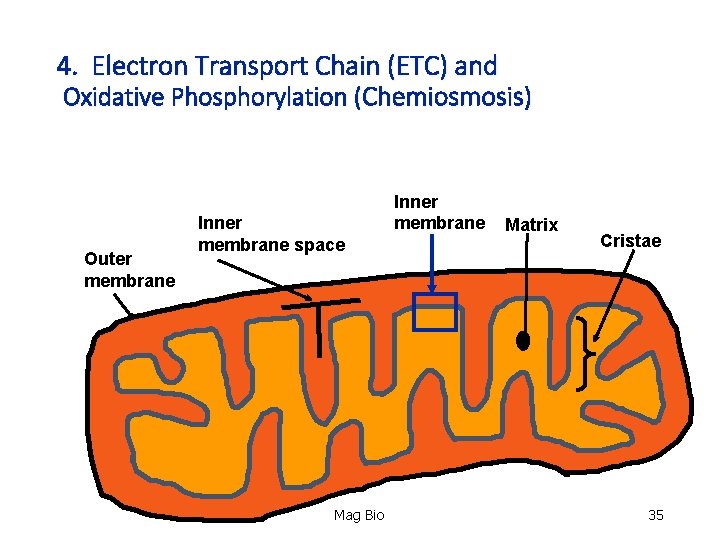
4. Electron Transport Chain (ETC) and Oxidative Phosphorylation (Chemiosmosis) Outer membrane Inner membrane space Mag Bio Inner membrane Matrix Cristae 35
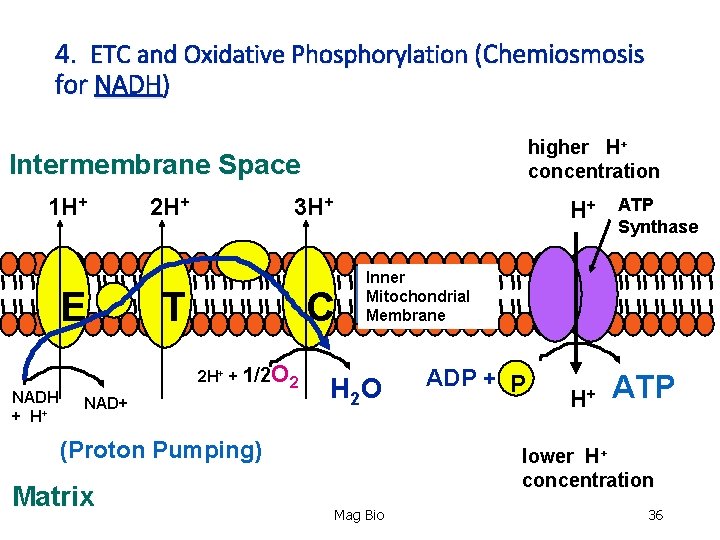
4. ETC and Oxidative Phosphorylation (Chemiosmosis for NADH) higher H+ concentration Intermembrane Space 1 H+ E NADH + H+ 2 H+ 3 H+ T C 2 H+ + 1/2 O 2 NAD+ H+ Inner Mitochondrial Membrane H 2 O (Proton Pumping) Matrix ATP Synthase ADP + P H+ ATP lower H+ concentration Mag Bio 36
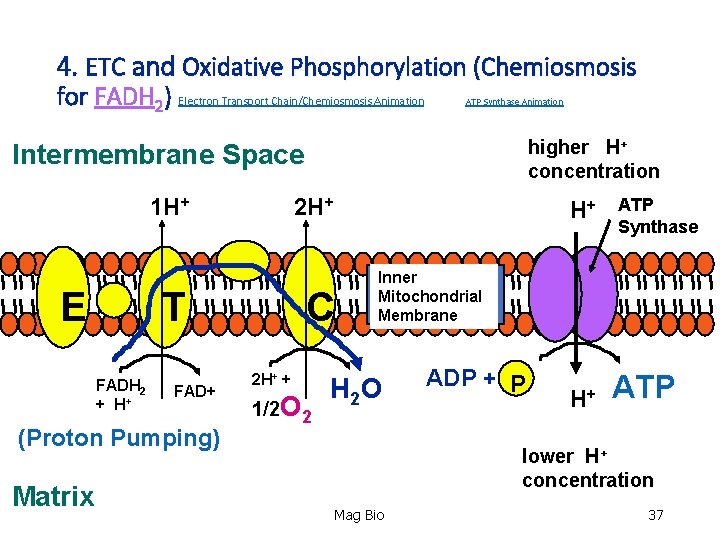
4. ETC and Oxidative Phosphorylation (Chemiosmosis for FADH 2) Electron Transport Chain/Chemiosmosis Animation ATP Synthase Animation higher H+ concentration Intermembrane Space 1 H+ E T FADH 2 + H+ FAD+ (Proton Pumping) Matrix 2 H+ C 2 H+ + 1/2 O 2 H+ ATP Synthase Inner Mitochondrial Membrane H 2 O ADP + P H+ ATP lower H+ concentration Mag Bio 37

TOTAL ATP YIELD 1. 04 ATP - substrate-level phosphorylation 2. ~32 -34 ATP - ETC & oxidative phosphorylation ~36 -38 ATP - TOTAL YIELD ATP Mag Bio 38

Let’s add up that energy for eukaryotes (you fill in the values) • Total ATP Yield ATP - glycolysis (substrate-level phosphorylation) ATP - converted from 2 NADH - glycolysis ATP - converted from 2 NADH - grooming phase ATP - Krebs cycle (substrate-level phosphorylation) ATP - converted from 6 NADH - Krebs cycle ATP - converted from 2 FADH 2 - Krebs cycle ATP - TOTAL Mag Bio 39
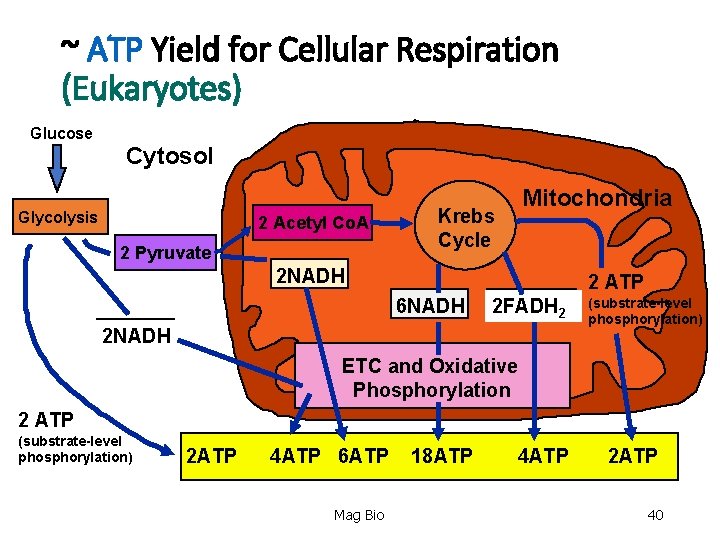
~ ATP Yield for Cellular Respiration (Eukaryotes) Glucose Cytosol Glycolysis 2 Acetyl Co. A 2 Pyruvate Mitochondria Krebs Cycle 2 NADH 2 ATP 6 NADH 2 FADH 2 2 NADH (substrate-level phosphorylation) ETC and Oxidative Phosphorylation 2 ATP (substrate-level phosphorylation) 2 ATP 4 ATP 6 ATP Mag Bio 18 ATP 4 ATP 2 ATP 40

Question: • In addition to glucose, what other various food molecules are use in Cellular Respiration? Mag Bio 41

Catabolism of Various Food Molecules • Other organic molecules used for fuel. 1. Carbohydrates: polysaccharides 2. Fats: glycerol’s and fatty acids 3. Proteins: amino acids Mag Bio 42
- Slides: 42