CELLULAR METABOLISM BIO 137 Anatomy Physiology I Metabolism

CELLULAR METABOLISM BIO 137 Anatomy & Physiology I

Metabolism • The total of all chemical reactions in an organism that are necessary to maintain life • Includes synthesis and breakdown of molecules • These reactions are usually stepwise and are called metabolic pathways • 2 Processes in Metabolism • Catabolism • Anabolism

Metabolic Pathways • Catabolism • Breakdown of larger molecules through Hydrolysis • Exergonic (energy can be used to drive anabolic pathways) • Example: oxidation (breakdown) of glucose in cellular respiration

Metabolic Pathways • Anabolism • Construction of larger molecules (polymers) from monomers through Dehydration • Endergonic – requires energy • Example: building a polypeptide chain and protein from amino acids

Metabolic Pathways • Reactant(s) → Product(s) • Stepwise • Each step in a pathway is catalyzed by a specific enzyme • Enzymes are protein catalysts • A substrate is what an enzyme acts on • Each enzyme is specific for a substrate Enzyme 1 A Enzyme 3 D C B Reaction 1 Starting molecule Enzyme 2 Reaction 3 Product

Activation Energy, EA • In a chemical reaction, bonds are broken in reactants, requiring an initial energy investment • EA – amount of energy needed to break bonds in reactants • EA is usually heat from surroundings

Enzymes • Enzymes are protein catalysts that speed up the rate of a reaction without being consumed • Enzymes are necessary because most reactions proceed very slowly and metabolism would be hindered • A single enzyme can catalyze thousands of reactions a second

Enzyme-Substrate Binding • Enzymes are PROTEINS with specific 3 -dimensional conformations (shape) • Shape of enzyme determines function (what substrate it will bind) • Active site – region of an enzyme that binds a substrate • Substrate ‘fits’ active site, forming enzyme-substrate complex • Lock and key model • In this form, enzyme converts substrate to product**
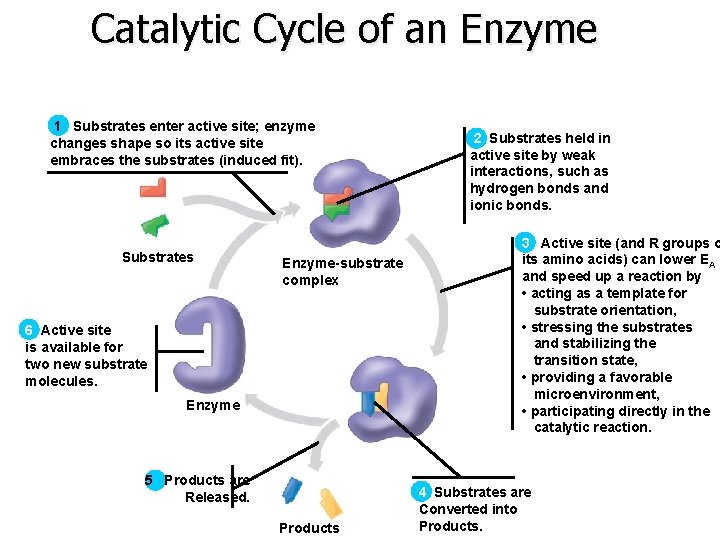
Catalytic Cycle of an Enzyme 1 Substrates enter active site; enzyme changes shape so its active site embraces the substrates (induced fit). Substrates Enzyme-substrate complex 6 Active site is available for two new substrate molecules. Enzyme 5 Products are Released. Products 2 Substrates held in active site by weak interactions, such as hydrogen bonds and ionic bonds. 3 Active site (and R groups o its amino acids) can lower EA and speed up a reaction by • acting as a template for substrate orientation, • stressing the substrates and stabilizing the transition state, • providing a favorable microenvironment, • participating directly in the catalytic reaction. 4 Substrates are Converted into Products.

Enzyme Activity is affected by Environment • Enzymes have optimal conditions under which they work • Optimal conditions favor correct conformation • Any physical or chemical condition that affects an enzyme’s 3 -dimensional shape can affect enzyme activity • Temperature, p. H, chemicals • These conditions can change protein conformation = Denaturation • Makes an enzyme inactive

WHY DO WE CARE SO MUCH ABOUT ENZYMES? Enzyme regulation is vital to the control of metabolism.

Metabolic Regulation • Metabolism is controlled by regulation of enzyme activity 1. Alter gene expression of enzyme 2. Regulate enzymes already present in a cell (Allosteric Regulation)

Energy • Energy is defined as the capacity to do work • Includes kinetic energy and potential energy • Energy can be transformed from one form to another • Energy is used to fuel cellular work

Forms of Energy • Kinetic Energy • Energy of motion • Light (photosynthesis), heat (random movement of atoms and molecules), Pool cue • Potential Energy (PE) • Stored energy due to location or structure • Chemical Energy • PE stored in molecules as a result of the arrangement of atoms in the molecule

Free Energy & Metabolism • Chemical reactions can be classified based on how energy is used • Exergonic • Energy is given off in the reaction • Spontaneous • Endergonic • Energy is required to start the reaction • Not spontaneous

Energy and Metabolism • Nutrients have potential energy (chemical energy) due to the arrangement of atoms • Electrons in the bonds holding atoms together represent energy!! • Energy can be given off when nutrients are broken down • Chemical energy in glucose is converted to ATP energy during Cellular Respiration

Electrons and Energy • Loss of electrons in nutrients as they are broken down allows for the production of ATP • Very complicated, do not focus on details • Know that electrons represent stored energy • Electrons are shuttled in a cell by electron carriers and ultimately given to O 2, making H 2 O • CO 2 is lost in several steps along the way • Waste product of metabolism
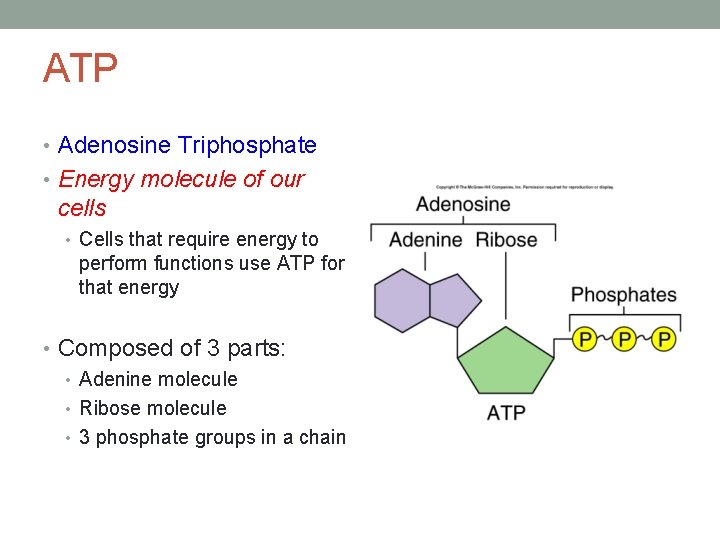
ATP • Adenosine Triphosphate • Energy molecule of our cells • Cells that require energy to perform functions use ATP for that energy • Composed of 3 parts: • Adenine molecule • Ribose molecule • 3 phosphate groups in a chain

Cellular Respiration • Breakdown of nutrients in the presence of oxygen (aerobic) to yield ATP • Involves shuttling of electrons from food to oxygen • C 6 H 12 O 6 + 6 O 2 → 6 CO 2 + 6 H 2 O + (38 ATP) • Breakdown is stepwise

Carbohydrate Metabolism • Glucose is not just an example we happen to choose – it is indeed the body’s preferred source of fuel • During digestion, polysaccharides and disaccharides are hydrolyzed into the monosaccharides glucose (80%), fructose, and galactose • These three monosaccharides are absorbed into the villi of the small intestine and carried to the liver • hepatocytes convert galactose and fructose to glucose

Cellular Respiration • 3 major steps • Glycolysis • Initial breakdown of glucose • Cytosol, anaerobic • Citric Acid Cycle (Krebs) • Matrix of mitochondria, aerobic • Electron Transport Chain (Oxidative Phosphorylation) • Cristae of mitochondria, aerobic • This is where those electrons are used!

Glycolysis • Glucose, C 6 H 12 O 6, is broken down into 2 - pyruvate molecules (3 C) • Stepwise, where electrons are given off to electron carriers • These are used in the Electron Transport Chain • Occurs in the cytosol under Anaerobic conditions • ATP is both consumed and made here
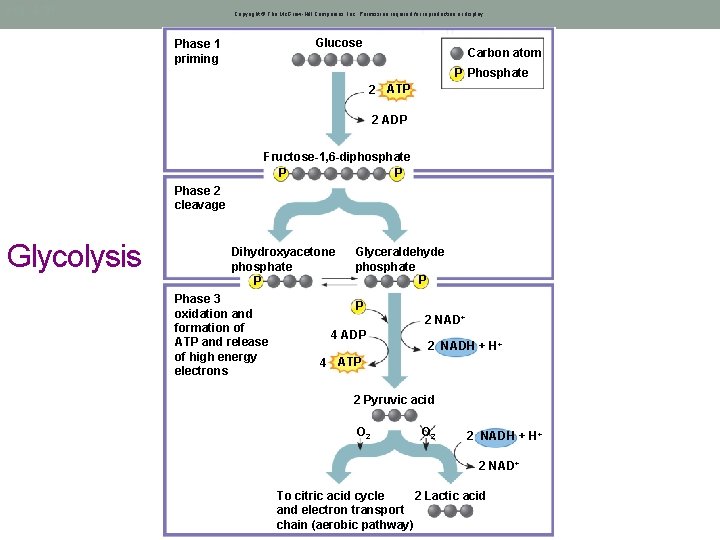
Fig. 4. 10 Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Glucose Phase 1 priming Carbon atom P Phosphate 2 ATP 2 ADP Fructose-1, 6 -diphosphate P P Phase 2 cleavage Glycolysis Dihydroxyacetone phosphate P Phase 3 oxidation and formation of ATP and release of high energy electrons Glyceraldehyde phosphate P P 2 NAD+ 4 ADP 2 NADH + H+ 4 ATP 2 Pyruvic acid Net O 2 2 NADH + H+ 2 NAD+ 2 Lactic acid To citric acid cycle and electron transport chain (aerobic pathway)

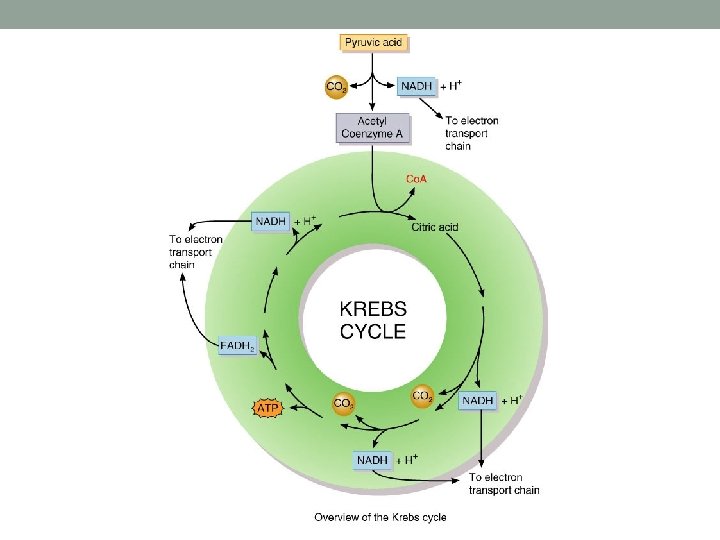
Citric Acid Cycle (Krebs) • Cycle where starting reactants are regenerated • Cycle is completed 2 X per glucose molecule • Stepwise, where electrons are given off to electron carriers • These are used in the Electron Transport Chain • ATP is made • CO 2 is formed as waste

RECALL THAT ELECTRONS REPRESENT STORED ENERGY Now we will use that stored energy to make ATP!

Electron Transport Chain • **Energy found in electron carriers is now used to make ATP through oxidative phosphorylation • Occurs on mitochondrial cristae • Electrons are ultimately given to Oxygen and water is formed • Energy given off during this process is used to make ATP!
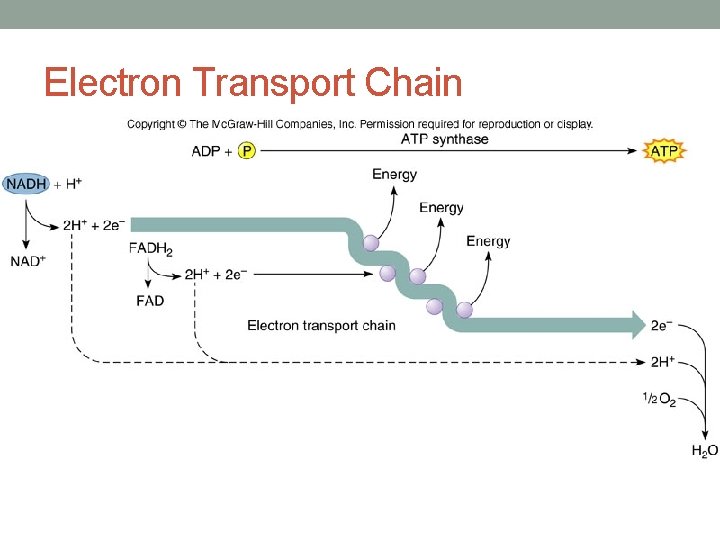
Electron Transport Chain
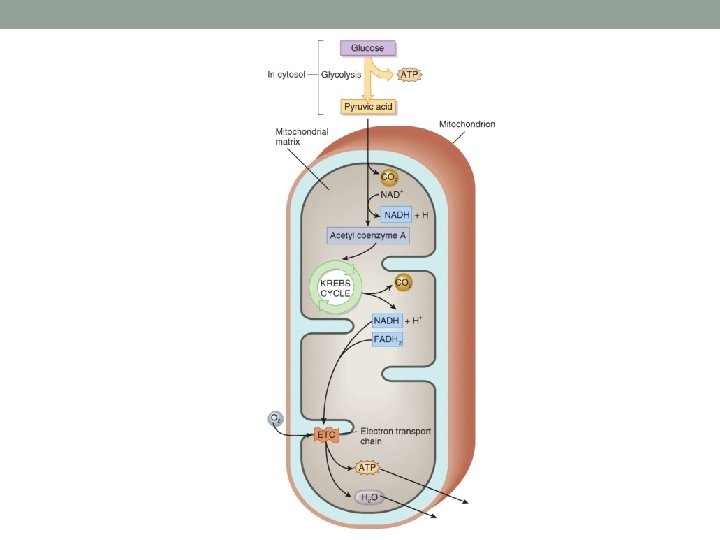
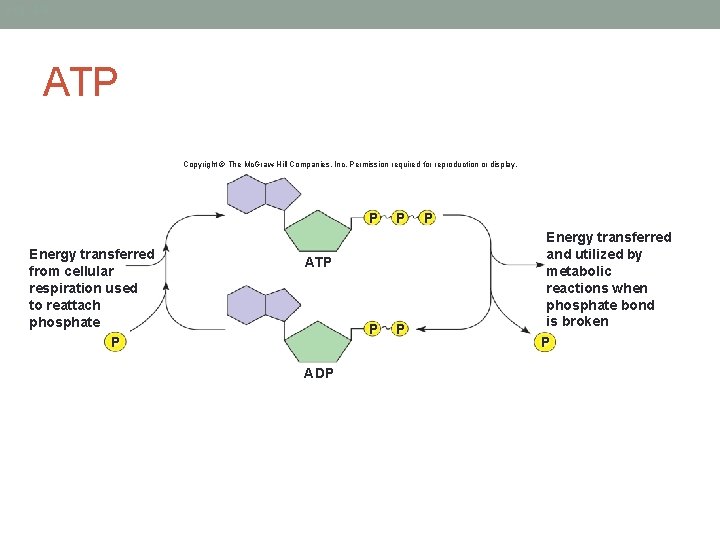
Fig. 4. 8 ATP Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. P Energy transferred from cellular respiration used to reattach phosphate P P ATP P ADP P P Energy transferred and utilized by metabolic reactions when phosphate bond is broken P

Fermentation • If oxygen is not present after glycolysis, pyruvate is fermented • Alcohol Fermentation • Lactic Acid Fermentation • Yeasts and some bacteria • In animals • Pyruvate is converted to ethanol lactic acid • Accumulates and causes muscle fatigue and soreness
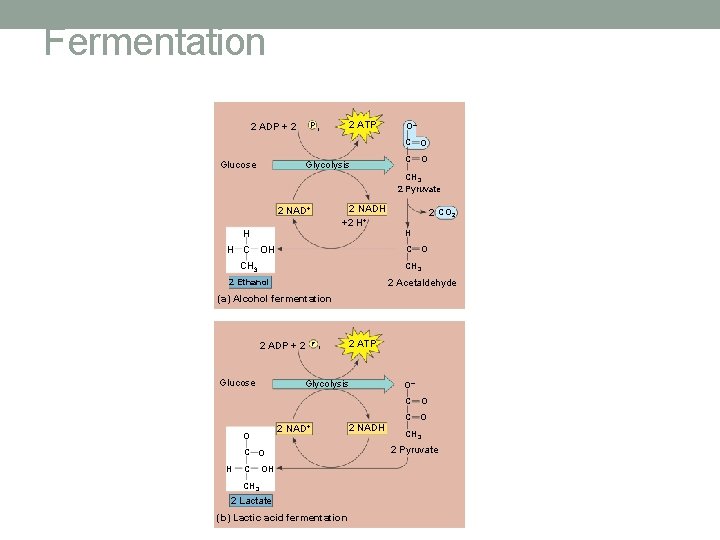
Fermentation 2 ADP + 2 Glucose 2 ATP Pi Glycolysis O– C O CH 3 2 Pyruvate 2 NADH +2 H+ 2 NAD+ H 2 CO 2 H H C OH C CH 3 O CH 3 2 Acetaldehyde 2 Ethanol (a) Alcohol fermentation 2 ADP + 2 Glucose P i Glycolysis 2 NAD+ O C O H C 2 ATP OH CH 3 2 Lactate (b) Lactic acid fermentation 2 NADH O– C O CH 3 2 Pyruvate
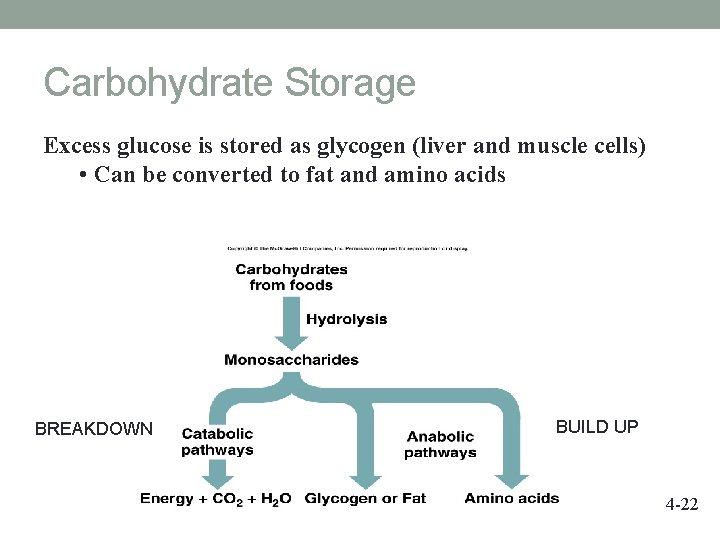
Carbohydrate Storage Excess glucose is stored as glycogen (liver and muscle cells) • Can be converted to fat and amino acids BREAKDOWN BUILD UP 4 -22
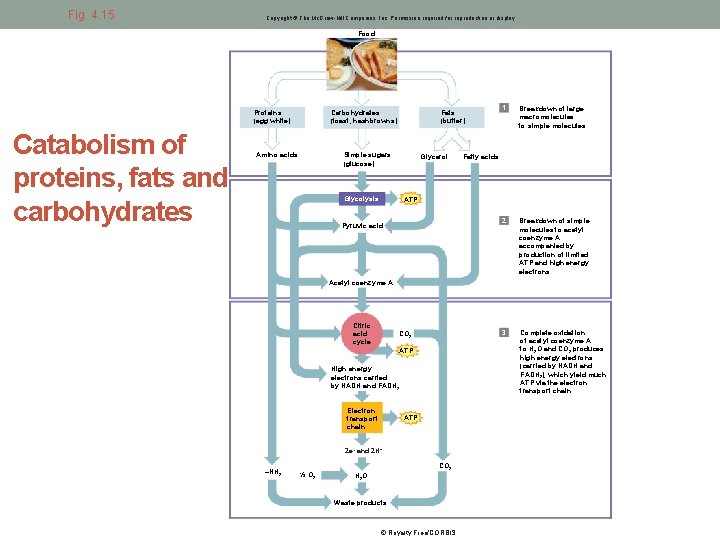
Fig. 4. 15 Fig. Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Food Proteins (egg white) Catabolism of proteins, fats and carbohydrates Carbohydrates (toast, hashbrowns) Amino acids Fats (butter) Simple sugars (glucose) Glycerol Glycolysis 1 Breakdown of large macromolecules to simple molecules 2 Breakdown of simple molecules to acetyl coenzyme A accompanied by production of limited ATP and high energy electrons 3 Complete oxidation of acetyl coenzyme A to H 2 O and CO 2 produces high energy electrons (carried by NADH and FADH 2), which yield much ATP via the electron transport chain Fatty acids ATP Pyruvic acid Acetyl coenzyme A Citric acid cycle CO 2 ATP High energy electrons carried by NADH and FADH 2 Electron transport chain ATP 2 e– and 2 H+ –NH 2 ½ O 2 CO 2 H 2 O Waste products © Royalty Free/CORBIS.
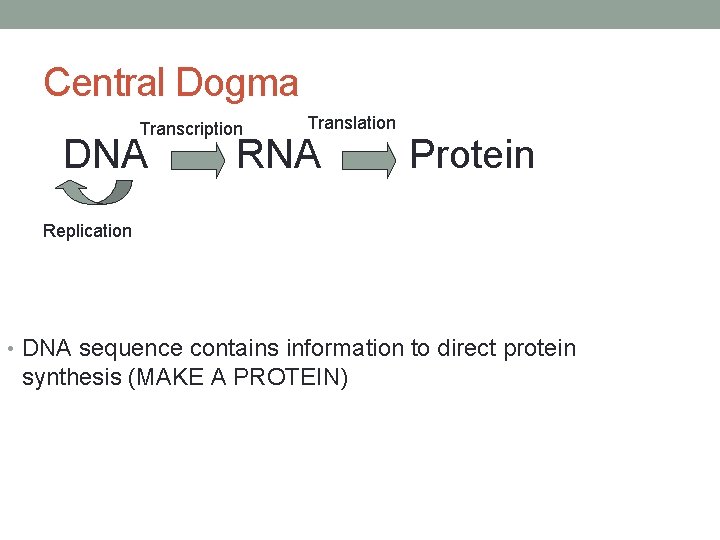
Central Dogma Transcription DNA Translation RNA Protein Replication • DNA sequence contains information to direct protein synthesis (MAKE A PROTEIN)

Genetics • Genetic information inherited from our parents is found in our DNA • Gene • Sequence of DNA nucleotides that codes for a protein • DNA sequence contains information to direct protein synthesis • Gene product = A protein

Genetics • A Protein performs the function of the gene • All of the DNA in a cell constitutes its genome
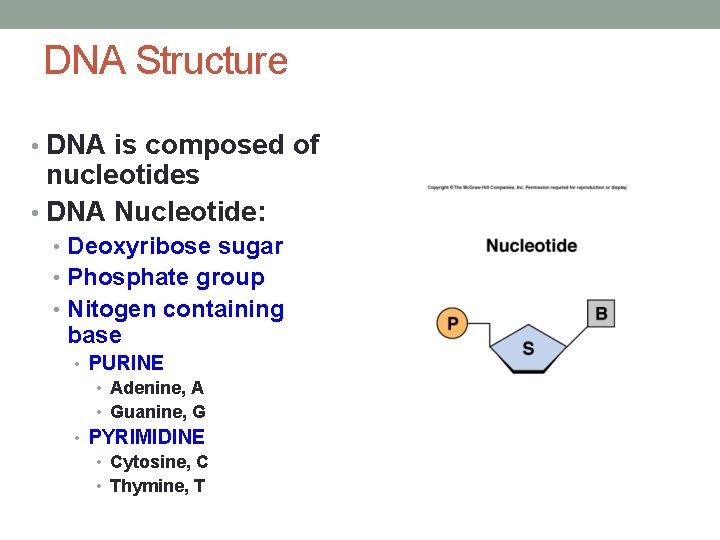
DNA Structure • DNA is composed of nucleotides • DNA Nucleotide: • Deoxyribose sugar • Phosphate group • Nitogen containing base • PURINE • Adenine, A • Guanine, G • PYRIMIDINE • Cytosine, C • Thymine, T
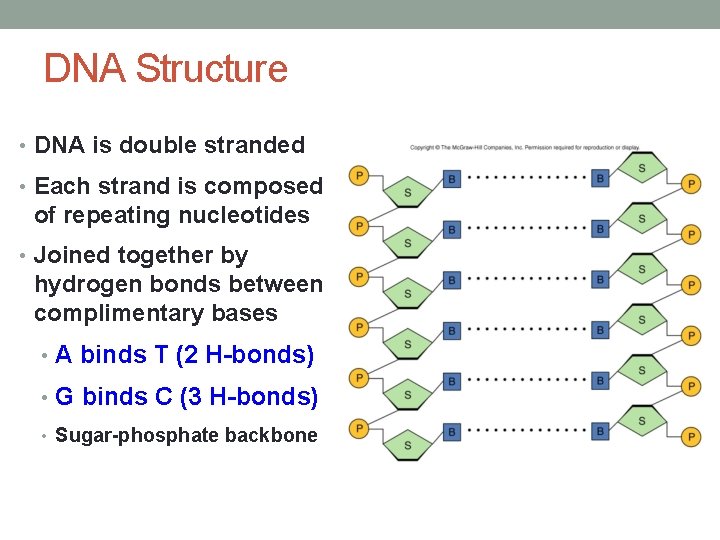
DNA Structure • DNA is double stranded • Each strand is composed of repeating nucleotides • Joined together by hydrogen bonds between complimentary bases • A binds T (2 H-bonds) • G binds C (3 H-bonds) • Sugar-phosphate backbone
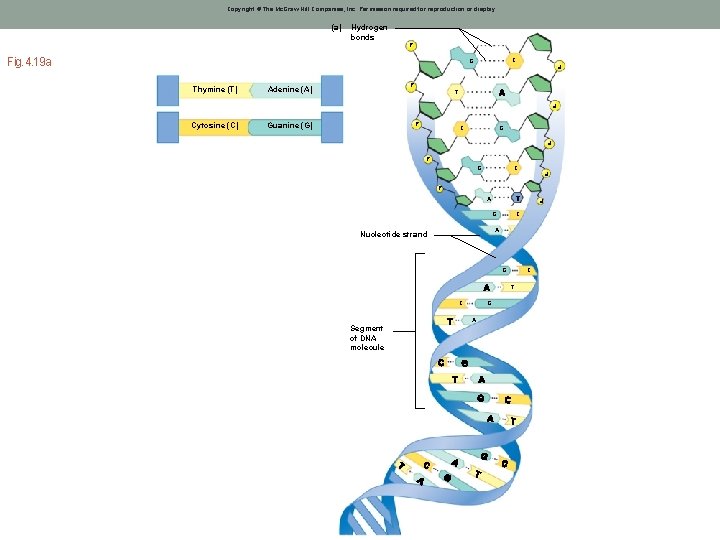
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Hydrogen bonds P P Fig. 4. 19 a CC G Thymine (T) Adenine (A) Cytosine (C) Guanine (G) P P (a) TT P P P CC GG P P P C P G P A P G C A Nucleotide strand G C T C G A Segment of DNA molecule
DNA Replication • Occurs in the nucleus • DNA unwinds and is replicated before a cell divides • Makes an identical copy of DNA using parental DNA as a template

DNA Replication • DNA Replication is semi-conservative • Resulting DNA is half-old, half-new • Parental DNA (template) and newly synthesized DNA • DNA Polymerase enzyme responsible for addition of nucleotides • A binds T (2 H-bonds) • G binds C (3 H-bonds)

DNA Replication

Replication Example • TACAGTCCATTCACCTAGGATATT
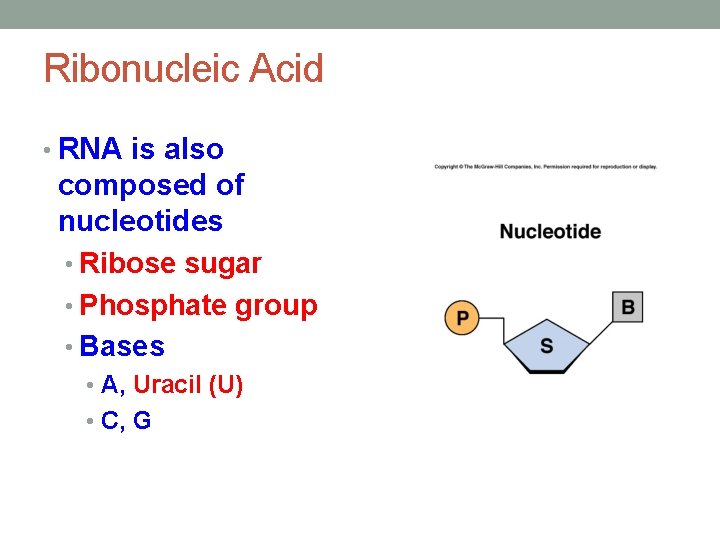
Ribonucleic Acid • RNA is also composed of nucleotides • Ribose sugar • Phosphate group • Bases • A, Uracil (U) • C, G
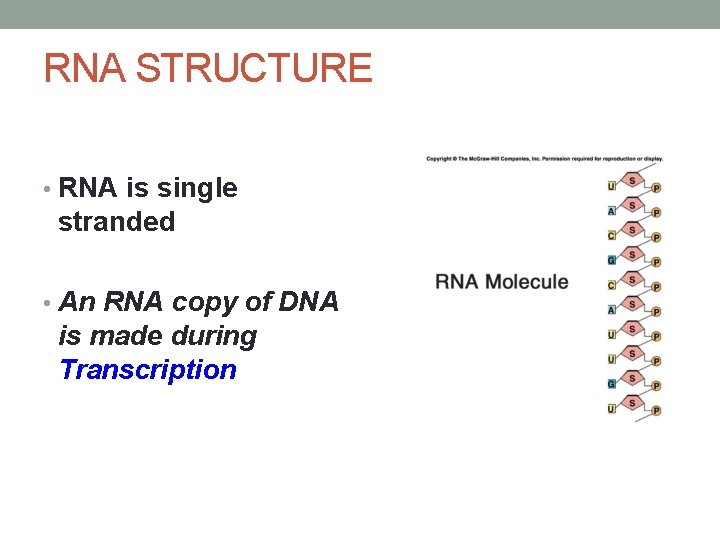
RNA STRUCTURE • RNA is single stranded • An RNA copy of DNA is made during Transcription
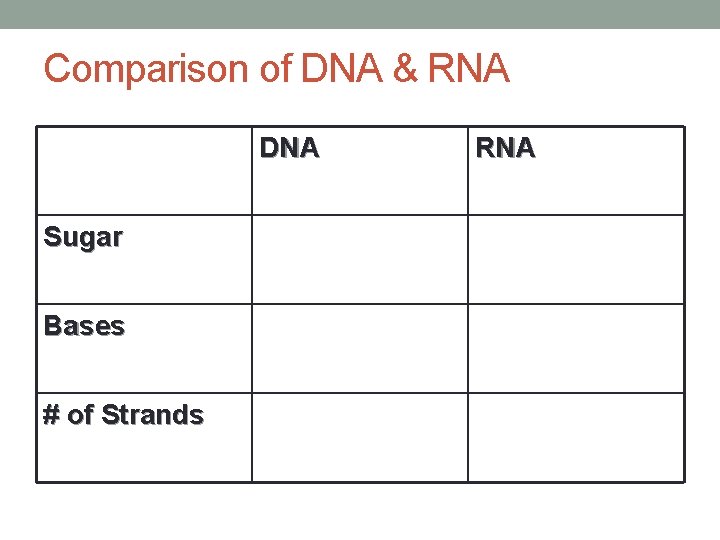
Comparison of DNA & RNA DNA Sugar Bases # of Strands RNA

Types of RNA Messenger RNA, m. RNA Ø Carries code (message) for protein to be synthesized • Transfer RNA, t. RNA Ø Carries appropriate amino acid to ribosome to be incorporated into protein Ribosomal RNA, r. RNA Ø The RNA component of the ribosome (recall that a ribosome is composed of RNA plus protein)

Transcription • Occurs in the nucleus • Make a messenger RNA copy of the DNA (gene) • RNA Polymerase enzyme copies the DNA • Base Pairing • DNA • T • C • G RNA U A G C
Transcription • **Only transcribe a gene when it is needed • All cells have the same genes but have differential expression of those genes

Transcription • Transcribe the following DNA sequence: • TACAGTCCATTCACCTAGGATATT

Following transcription, the m. RNA leaves the nucleus and enters the cytosol where it is threaded through a ribosome to undergo translation.

Translation • m. RNA is translated into protein • Occurs on the ribosome • m. RNA is read 3 bases at a time • These are called codons • Each codon corresponds to an amino acid

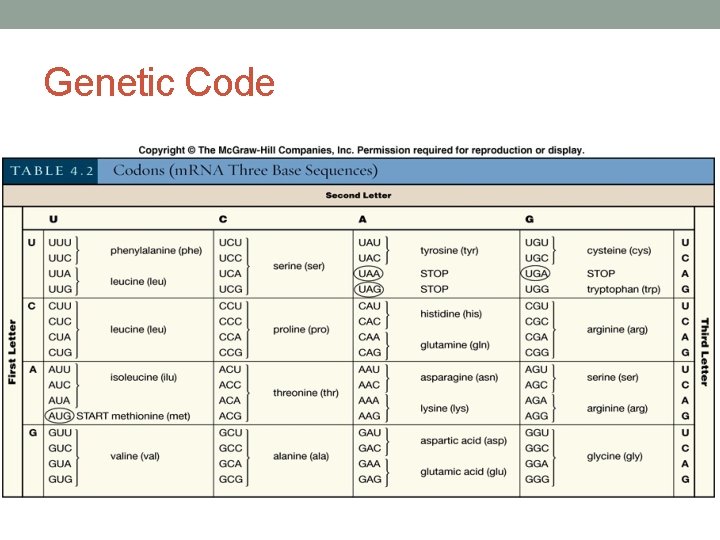
The Genetic Code • There are 64 codons that make up the genetic code • Each codon corresponds to an amino acid • 20 amino acids in nature • Code is redundant • 1 START codon: AUG • 3 STOP codons: UAA, UAG, UGA • Amino acids are attached to a specific t. RNA • t. RNA carries the amino acid to the growing polypeptide chain
Genetic Code

Translation • 1 st codon of every gene is always AUG • START codon • Translation begins at AUG • Translation ends when a STOP codon is reached • UAA, UAG, UGA • Remember, Amino acids are attached to a specific t. RNA • Has anticodon sequence • If m. RNA is UAA, t. RNA anticodon is AUU

Translate the m. RNA sequence from before

Translation • Each time a codon is read, a new amino acid is added to a growing chain • Peptide bonds form between each amino acid • When a STOP codon is reached, the protein is released
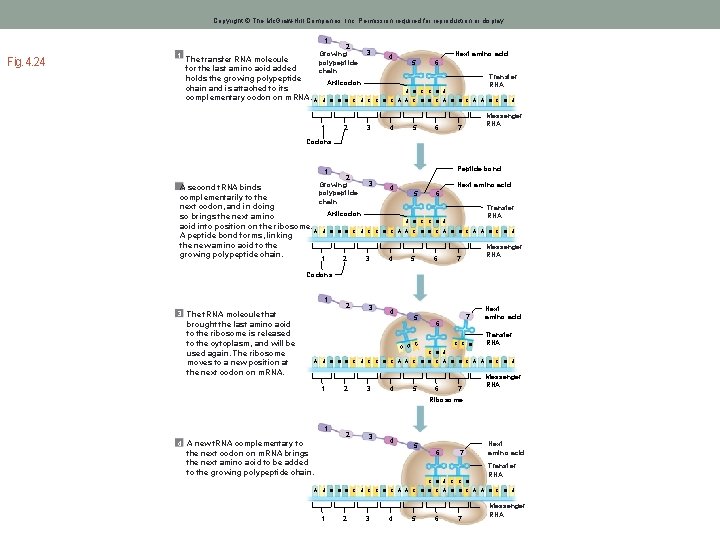
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. 1 1 Fig. 4. 24 2 Growing The transfer RNA molecule polypeptide for the last amino acid added chain holds the growing polypeptide Anticodon chain and is attached to its complementary codon on m. RNA. A U G G G C U 1 2 3 4 Next amino acid 5 6 Transfer RNA U G C C G U C C G C A A C G G C A A G C G U 3 4 5 6 Messenger RNA 7 Codons Peptide bond 1 2 2 Growing polypeptide chain 3 A second t. RNA binds complementarily to the next codon, and in doing Anticodon so brings the next amino acid into position on the ribosome. A U G G G C U C A peptide bond forms, linking the new amino acid to the growing polypeptide chain. 1 2 3 4 Next amino acid 5 6 Transfer RNA U G C C G U C G C A A C G G C A A G C G U 4 5 6 Messenger RNA 7 Codons 1 2 3 The t. RNA molecule that brought the last amino acid to the ribosome is released to the cytoplasm, and will be used again. The ribosome moves to a new position at the next codon on m. RNA. 3 4 5 C U G 7 6 C C G Next amino acid Transfer RNA C G U A U G G G C U C C G C A A C G G C A A G C G U 1 2 3 4 5 6 7 Messenger RNA Ribosome 1 2 4 A new t. RNA complementary to 3 4 the next codon on m. RNA brings the next amino acid to be added to the growing polypeptide chain. 5 6 7 Next amino acid Transfer RNA C G U C C G A U G G G C U C C G C A A C G G C A A G C G U 1 2 3 4 5 6 7 Messenger RNA
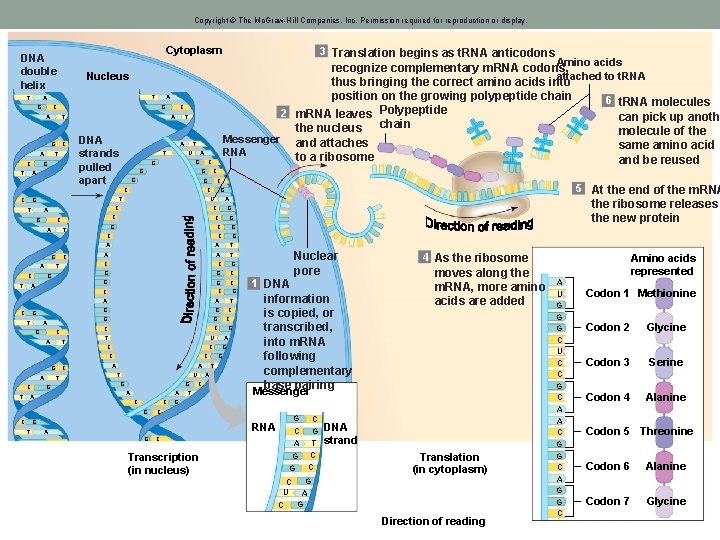
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Cytoplasm DNA double helix T Nucleus T A G T C T G A C A T A C A G C DNA strands pulled apart T A T T U A G G G C C C T G C A T A C G A C T G C G T A G C A T G C T G A T C C G A T G C G G C C C A A T U G G A A C C A T A C T G 1 DNA information is copied, or transcribed, into m. RNA following complementary base pairing moves along the m. RNA, more amino acids are added Messenger C RNA G 4 As the ribosome G G T G A G Nuclear pore G C U T G C G G A C A G C G the ribosome releases the new protein G C C A G 5 At the end of the m. RNA G U C C A Amino acids recognize complementary m. RNA codons, attached to t. RNA thus bringing the correct amino acids into position on the growing polypeptide chain 6 t. RNA molecules Polypeptide 2 m. RNA leaves can pick up anoth the nucleus chain molecule of the Messenger and attaches same amino acid RNA to a ribosome and be reused C G C 3 Translation begins as t. RNA anticodons C Transcription (in nucleus) G C G T C A G G C C DNA strand Translation (in cytoplasm) G C U A G C Direction of reading Amino acids represented A U G G G C U C C G C A A C G G C A G G C Codon 1 Methionine Codon 2 Glycine Codon 3 Serine Codon 4 Alanine Codon 5 Threonine Codon 6 Alanine Codon 7 Glycine

Mutations • Result from an error in DNA sequence • Caused by many things: • Chemicals, error in replication, sunlight, X-rays • Mutations affect the protein product of a gene • Not made • Made, but wrong conformation • Non-functional • Protein Made in excess

Mutations Affect Protein Product • Sickle-cell anemia • Results from a single amino acid change in the gene that codes for hemoglobin • This defect causes RBCs to become sickle-shaped in low oxygen situations
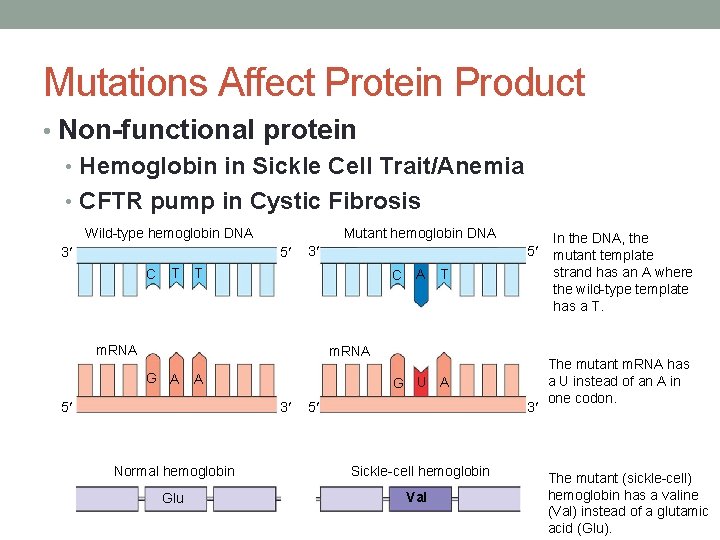
Mutations Affect Protein Product • Non-functional protein • Hemoglobin in Sickle Cell Trait/Anemia • CFTR pump in Cystic Fibrosis Wild-type hemoglobin DNA 3 Mutant hemoglobin DNA 5 C T T In the DNA, the mutant template strand has an A where the wild-type template has a T. G U A The mutant m. RNA has a U instead of an A in one codon. 3 5 T C A m. RNA G A A 5 3 Normal hemoglobin Sickle-cell hemoglobin Glu Val The mutant (sickle-cell) hemoglobin has a valine (Val) instead of a glutamic acid (Glu).
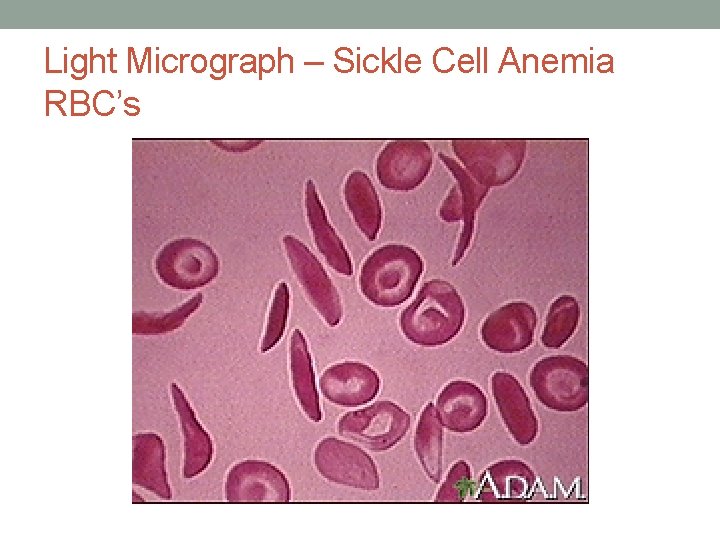
Light Micrograph – Sickle Cell Anemia RBC’s
- Slides: 64