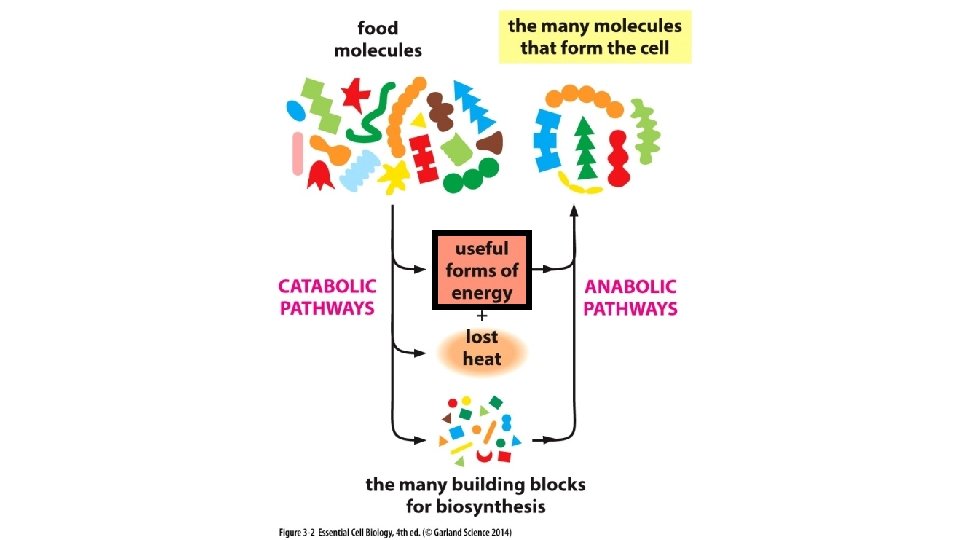
CELLULAR ENERGY CHEMICAL DIRECTIVES Entropy Disorder FoodEnergy Stored



![• Cytoplasmic [NADPH] > > [NADP+] • Cytoplasmic [NADH] < < [NAD+] • Cytoplasmic [NADPH] > > [NADP+] • Cytoplasmic [NADH] < < [NAD+]](https://slidetodoc.com/presentation_image_h2/0282a997fcd3a21ec6f10036ae5f682e/image-18.jpg)


![Potential energy At standard conditions, when [reactant] = [product], DGo = X; X tells Potential energy At standard conditions, when [reactant] = [product], DGo = X; X tells](https://slidetodoc.com/presentation_image_h2/0282a997fcd3a21ec6f10036ae5f682e/image-26.jpg)
![DG < 0 • Without intervention, as Y X reaction continues, [Y] decreases and DG < 0 • Without intervention, as Y X reaction continues, [Y] decreases and](https://slidetodoc.com/presentation_image_h2/0282a997fcd3a21ec6f10036ae5f682e/image-27.jpg)

![[Product] [Reactant] when at equilibrium = the Equilibrium Constant (K) [Product] [Reactant] when at equilibrium = the Equilibrium Constant (K)](https://slidetodoc.com/presentation_image_h2/0282a997fcd3a21ec6f10036ae5f682e/image-31.jpg)




- Slides: 44

CELLULAR ENERGY & CHEMICAL DIRECTIVES
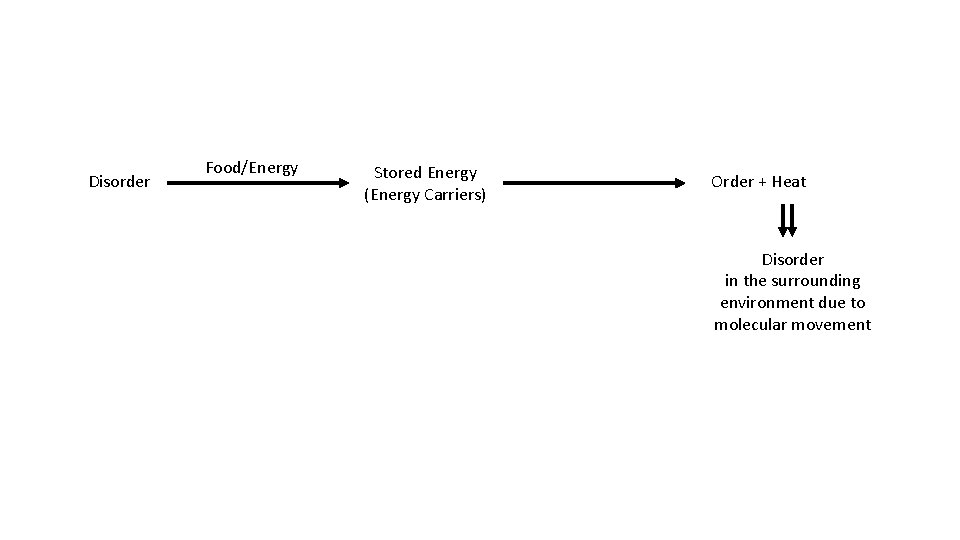
Entropy




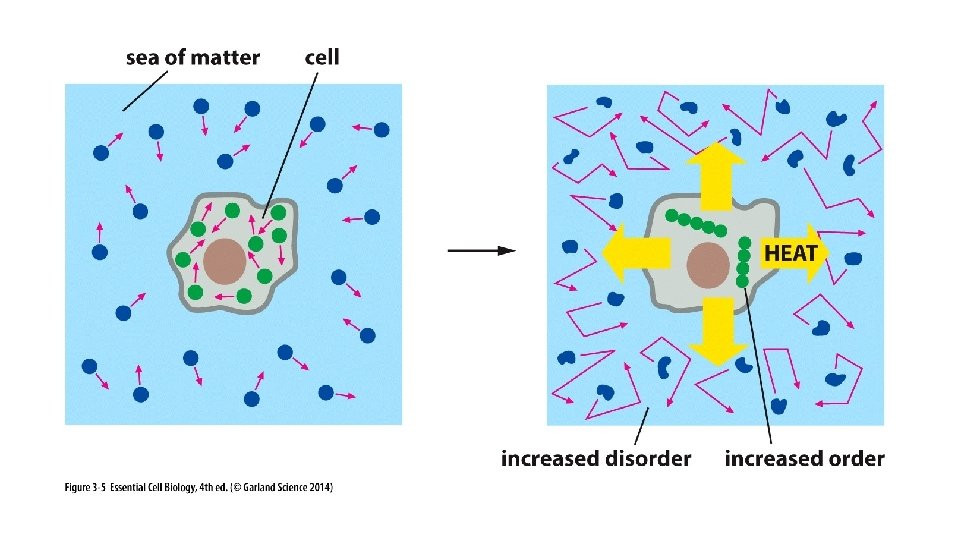
Disorder Food/Energy Stored Energy (Energy Carriers) Order + Heat Disorder in the surrounding environment due to molecular movement

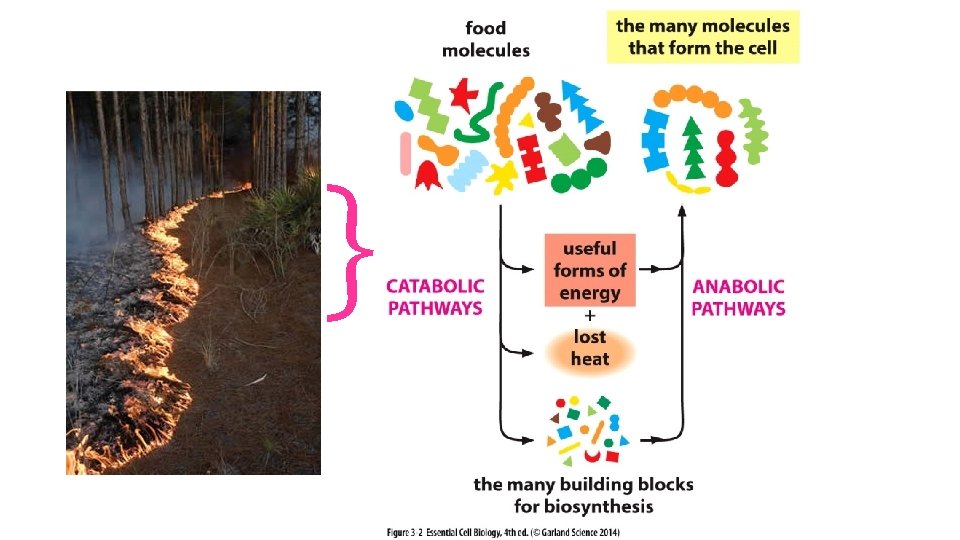
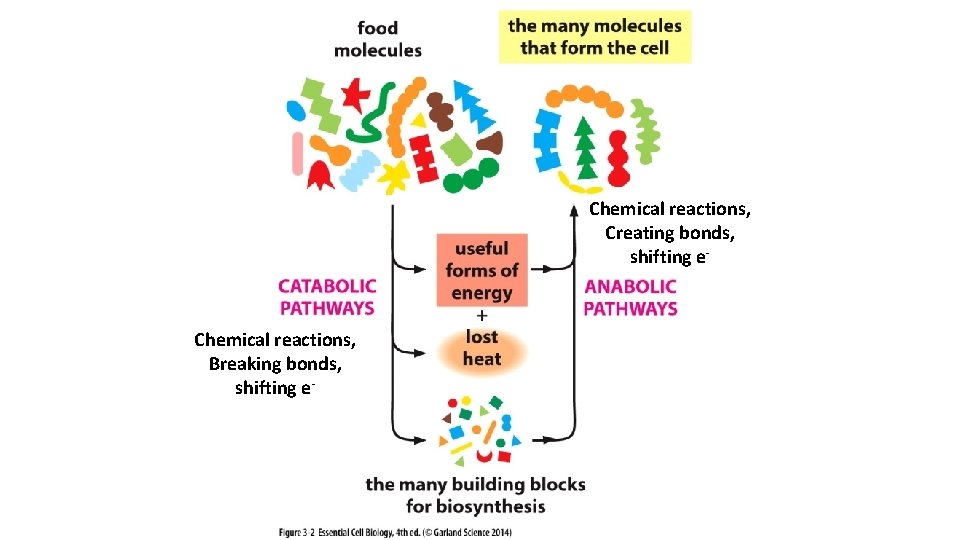
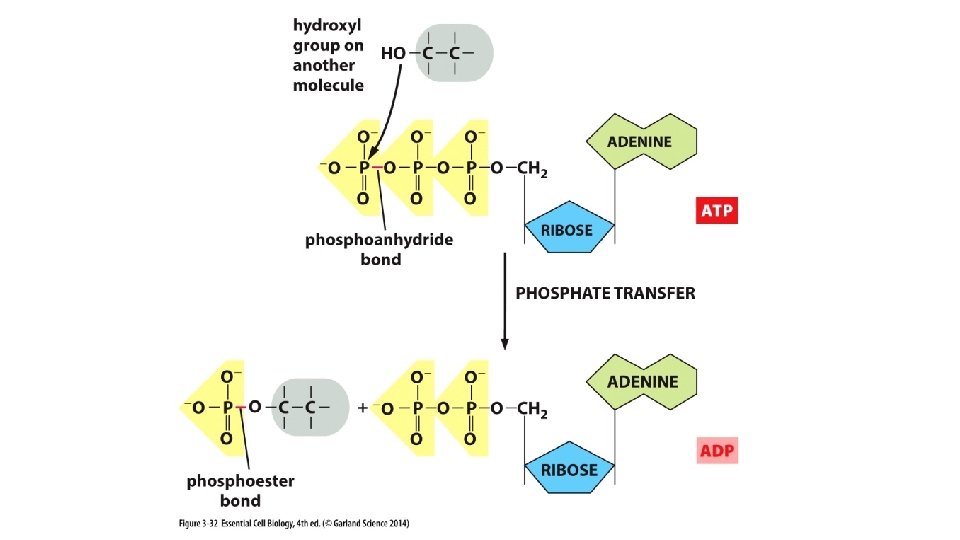
Chemical reactions, Creating bonds, shifting e- Chemical reactions, Breaking bonds, shifting e-

Moving electrons

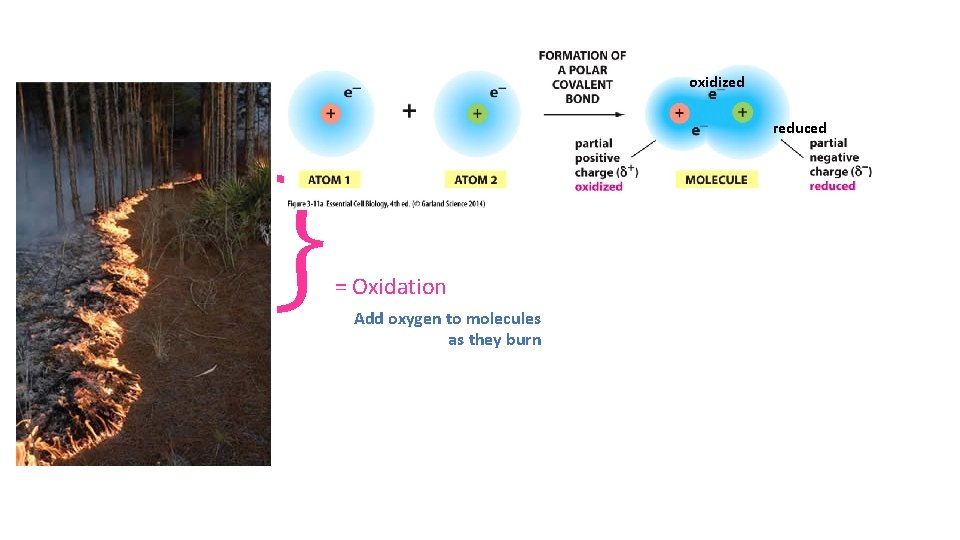
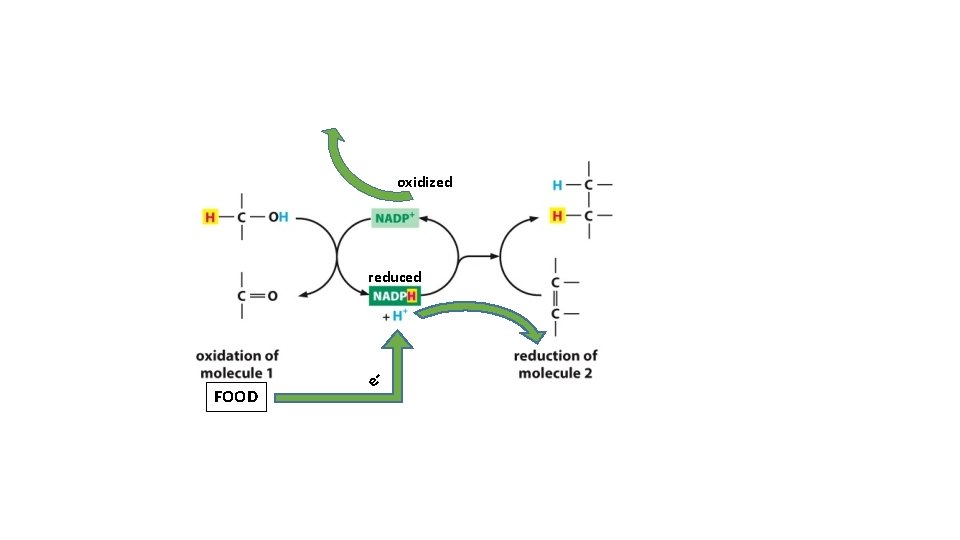
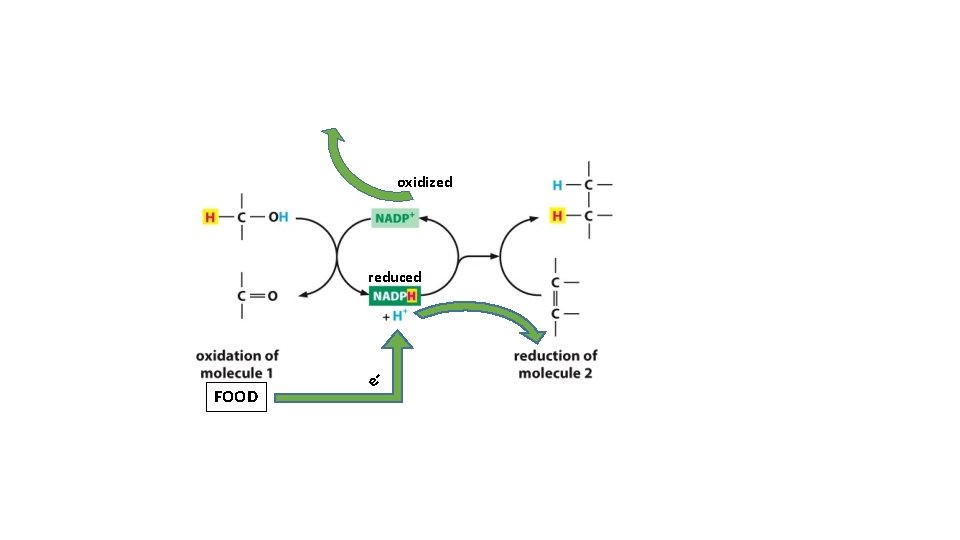
} oxidized reduced = Oxidation Add oxygen to molecules as they burn

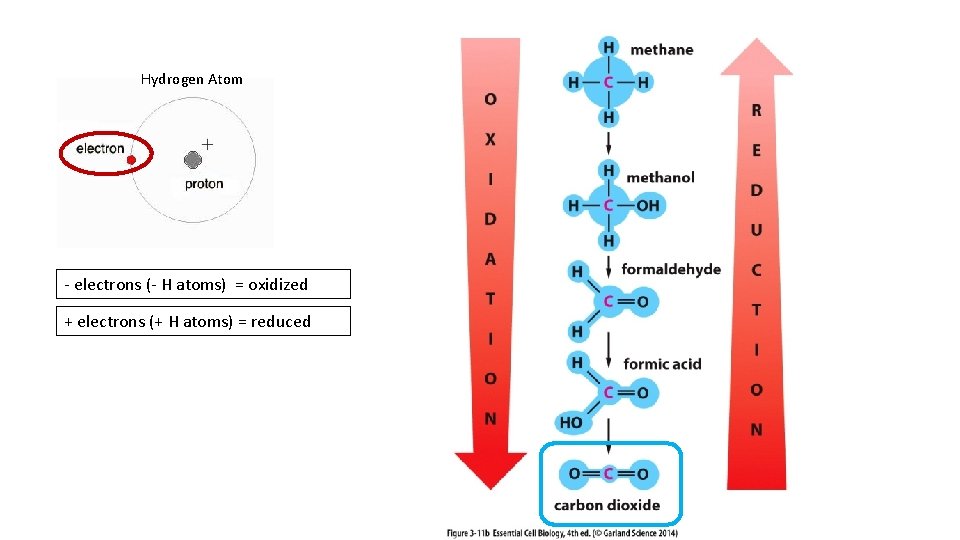
Hydrogen Atom - electrons (- H atoms) = oxidized + electrons (+ H atoms) = reduced

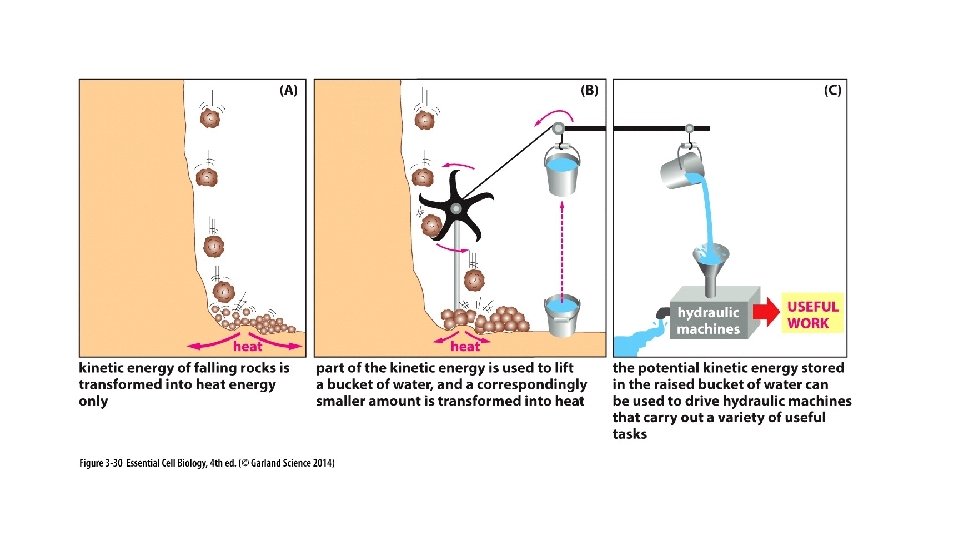
Usable (stored) Energy



oxidized reduced FOOD e-

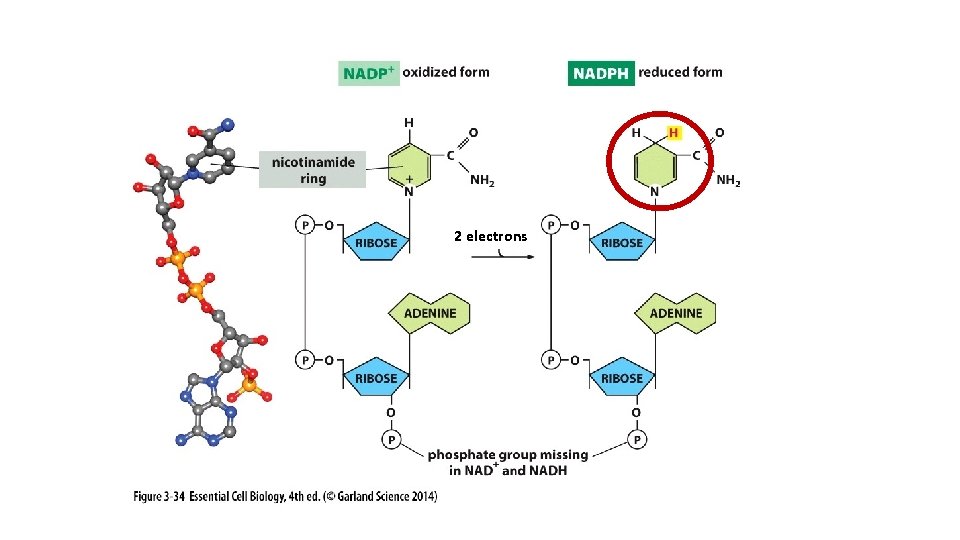
2 electrons

oxidized reduced FOOD e-

![Cytoplasmic NADPH NADP Cytoplasmic NADH NAD • Cytoplasmic [NADPH] > > [NADP+] • Cytoplasmic [NADH] < < [NAD+]](https://slidetodoc.com/presentation_image_h2/0282a997fcd3a21ec6f10036ae5f682e/image-18.jpg)
• Cytoplasmic [NADPH] > > [NADP+] • Cytoplasmic [NADH] < < [NAD+]

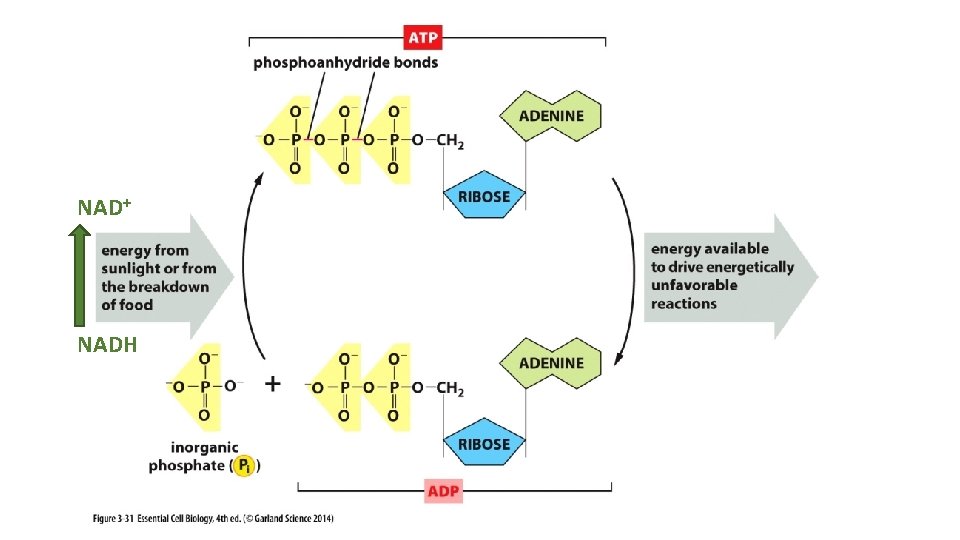
NAD+ NADH


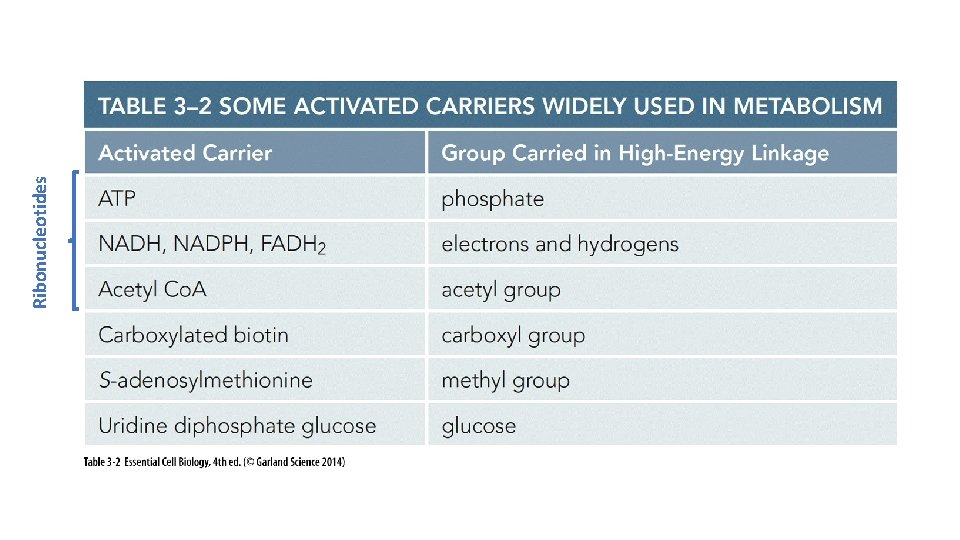
Ribonucleotides

When cells can use energy

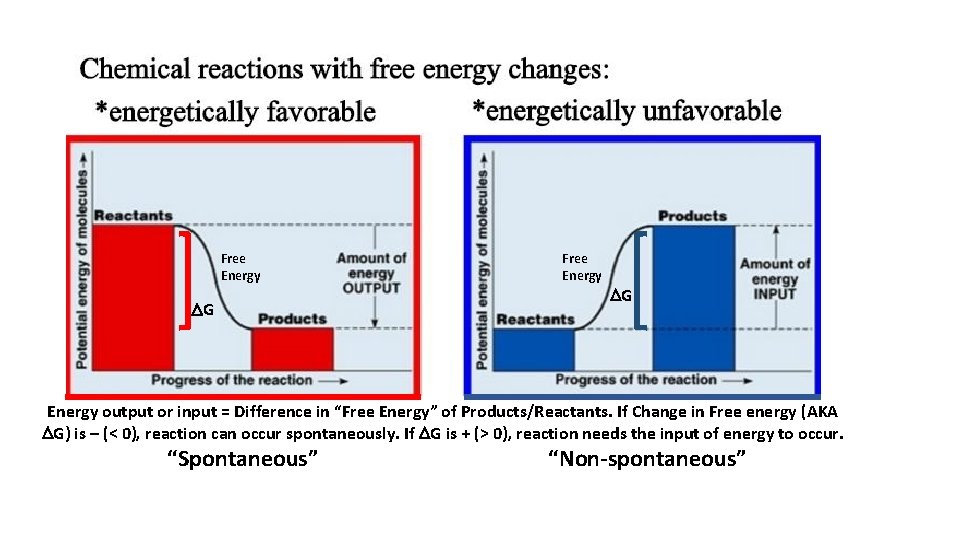
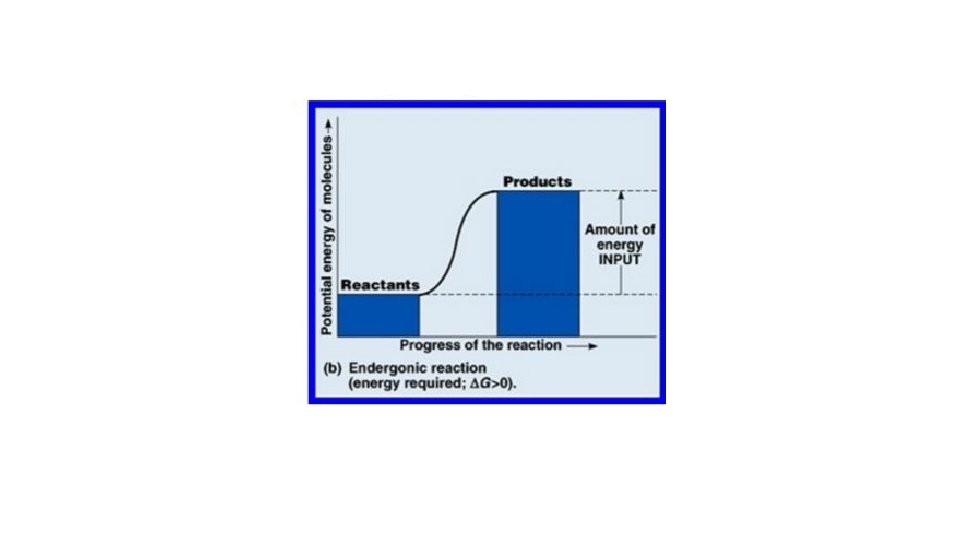
Free Energy DG Energy output or input = Difference in “Free Energy” of Products/Reactants. If Change in Free energy (AKA DG) is – (< 0), reaction can occur spontaneously. If DG is + (> 0), reaction needs the input of energy to occur. “Spontaneous” “Non-spontaneous”

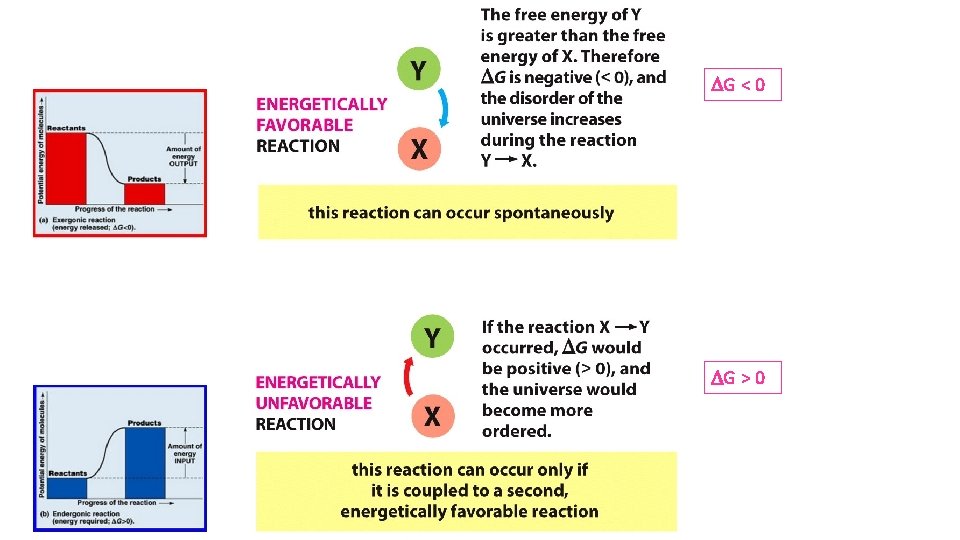
DG < 0 DG > 0

• DG helps us predict whether a reaction will occur spontaneously. • DG also tells us how much energy will be produced in a spontaneous reaction (or will be needed to drive a nonspontaneous reaction).
![Potential energy At standard conditions when reactant product DGo X X tells Potential energy At standard conditions, when [reactant] = [product], DGo = X; X tells](https://slidetodoc.com/presentation_image_h2/0282a997fcd3a21ec6f10036ae5f682e/image-26.jpg)
Potential energy At standard conditions, when [reactant] = [product], DGo = X; X tells us how favorable (how much energy will be produced) a reaction is. Reaction progress
![DG 0 Without intervention as Y X reaction continues Y decreases and DG < 0 • Without intervention, as Y X reaction continues, [Y] decreases and](https://slidetodoc.com/presentation_image_h2/0282a997fcd3a21ec6f10036ae5f682e/image-27.jpg)
DG < 0 • Without intervention, as Y X reaction continues, [Y] decreases and [X] increases. • Therefore, DG changes (becomes more +) as the reaction proceeds. • Thus, reactions become less favorable as they proceed. • When a reaction reaches equilibrium, DG = 0. DG > 0



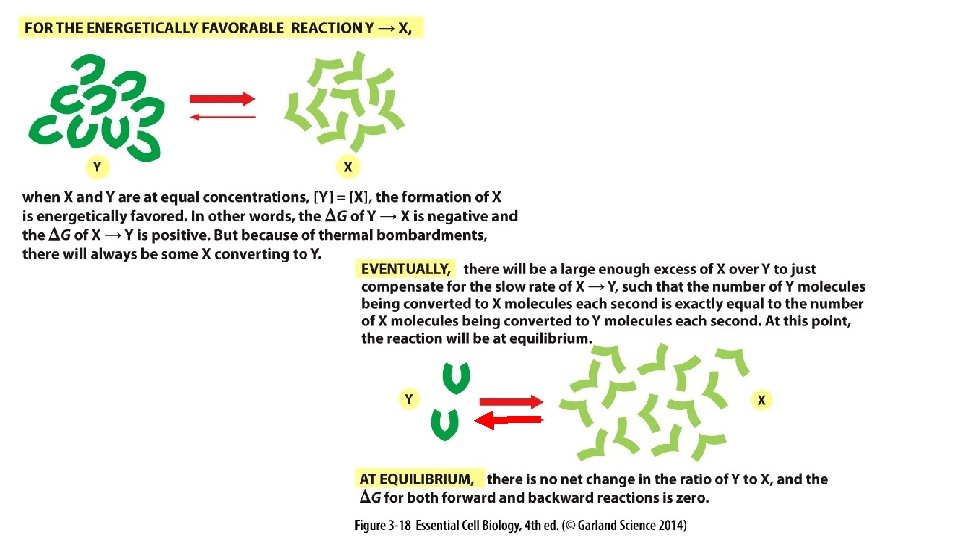
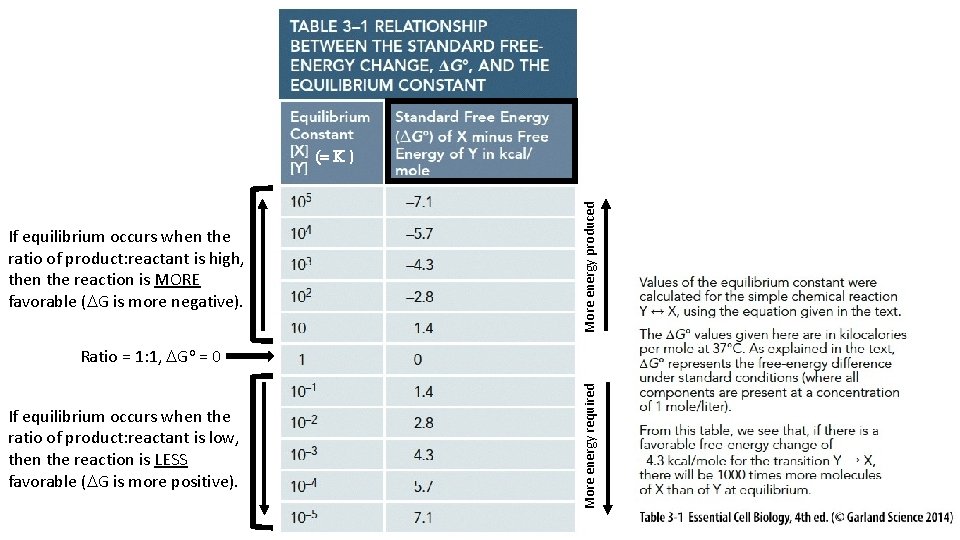
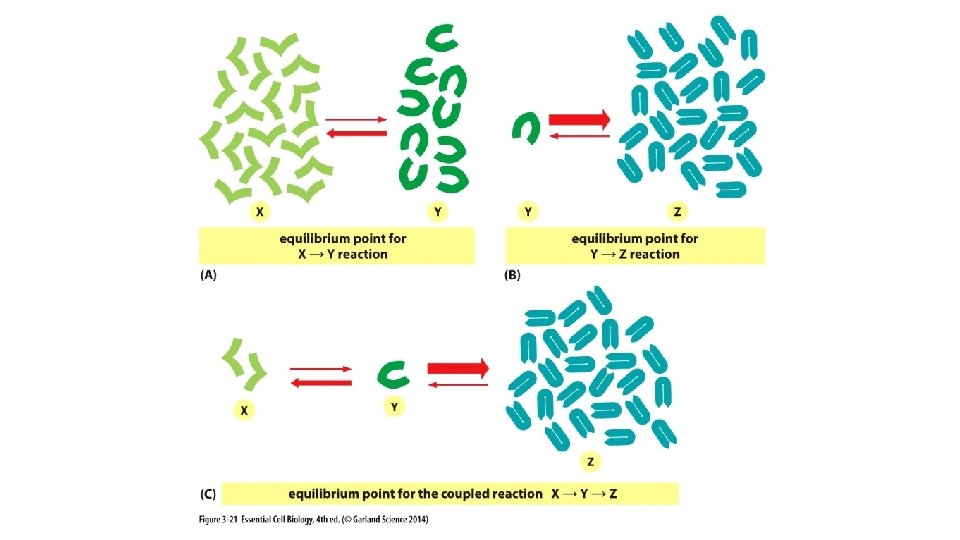
If equilibrium occurs when the ratio of product: reactant is high, then the reaction is MORE favorable (DG is more negative). More energy produced (= K ) If equilibrium occurs when the ratio of product: reactant is low, then the reaction is LESS favorable (DG is more positive). More energy required Ratio = 1: 1, DGo = 0
![Product Reactant when at equilibrium the Equilibrium Constant K [Product] [Reactant] when at equilibrium = the Equilibrium Constant (K)](https://slidetodoc.com/presentation_image_h2/0282a997fcd3a21ec6f10036ae5f682e/image-31.jpg)
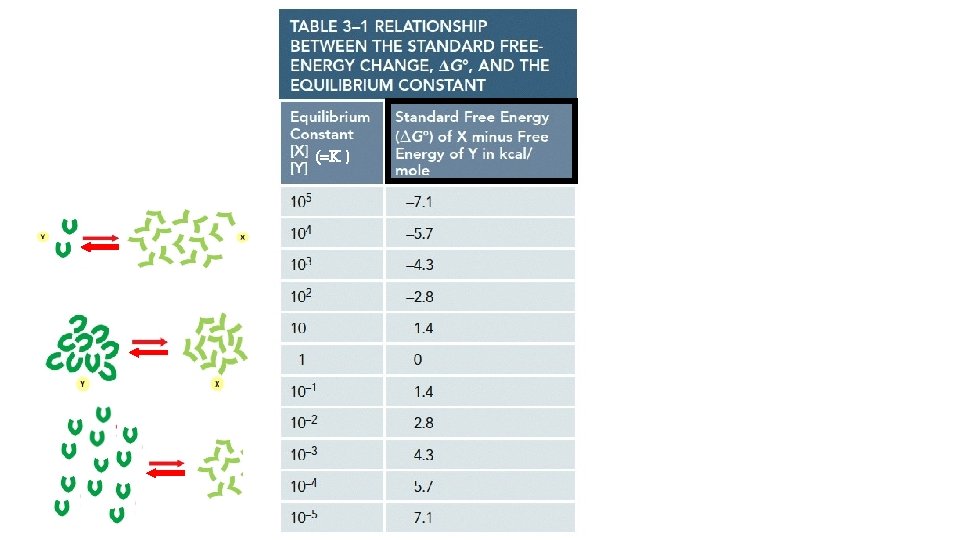
[Product] [Reactant] when at equilibrium = the Equilibrium Constant (K)



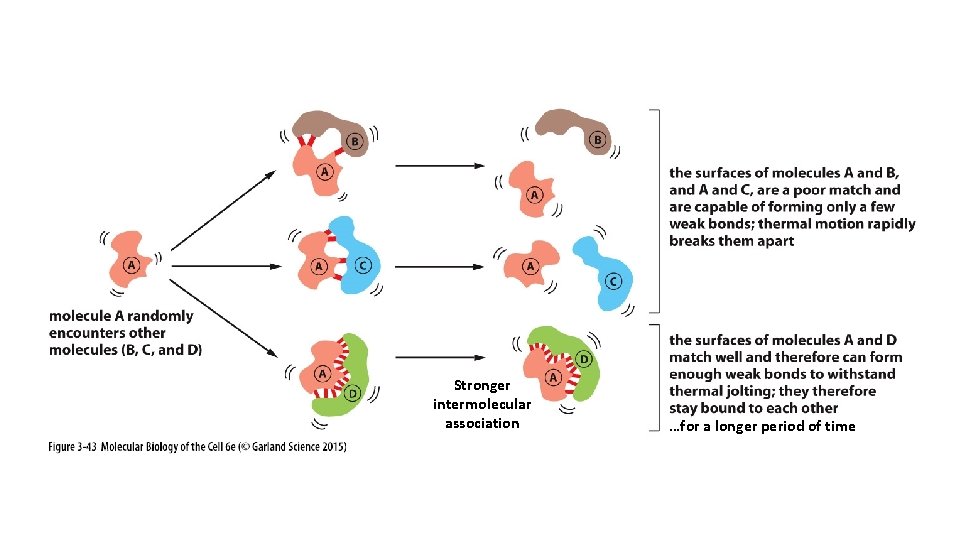
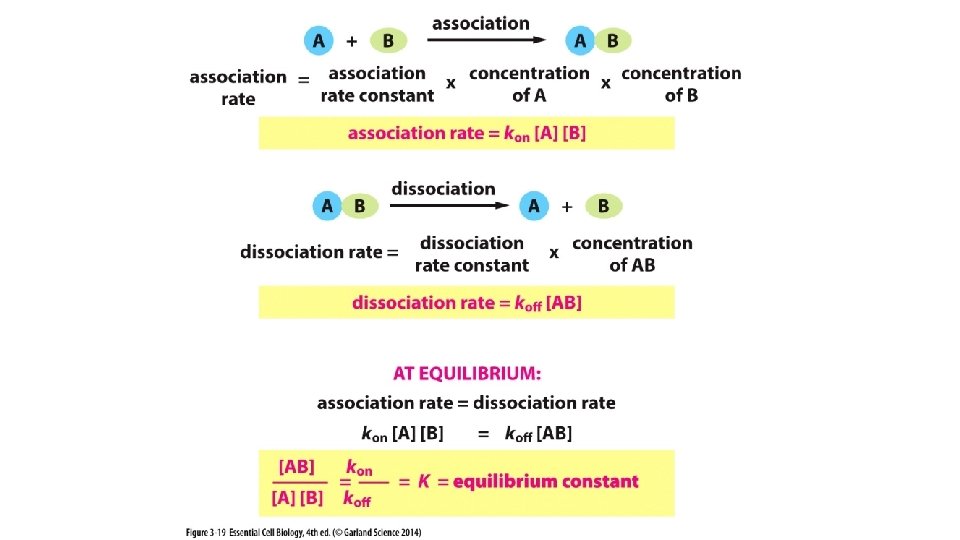
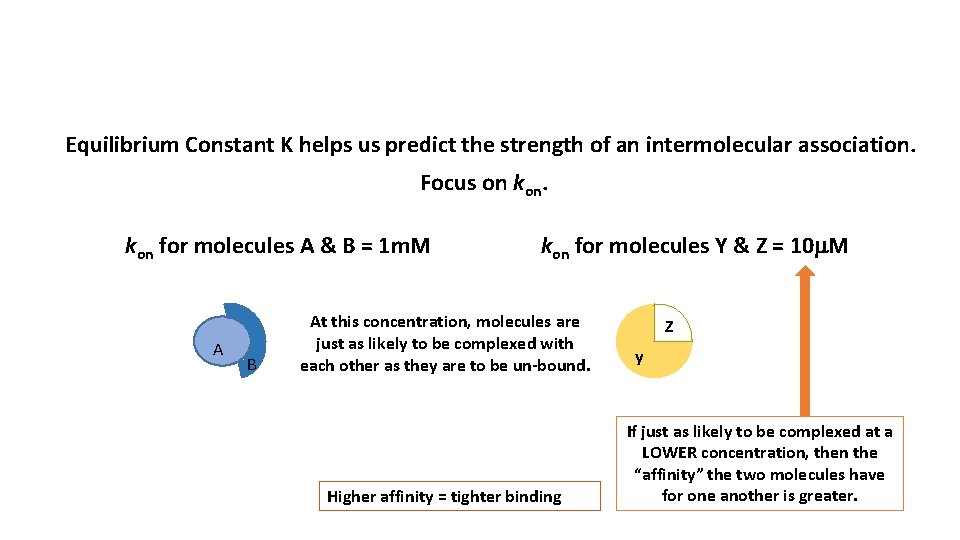
Equilibrium Constant K helps us predict the strength of an intermolecular association.

Stronger intermolecular association …for a longer period of time


Equilibrium Constant K helps us predict the strength of an intermolecular association. Focus on kon for molecules A & B = 1 m. M A B kon for molecules Y & Z = 10 m. M At this concentration, molecules are just as likely to be complexed with each other as they are to be un-bound. Higher affinity = tighter binding Z y If just as likely to be complexed at a LOWER concentration, then the “affinity” the two molecules have for one another is greater.

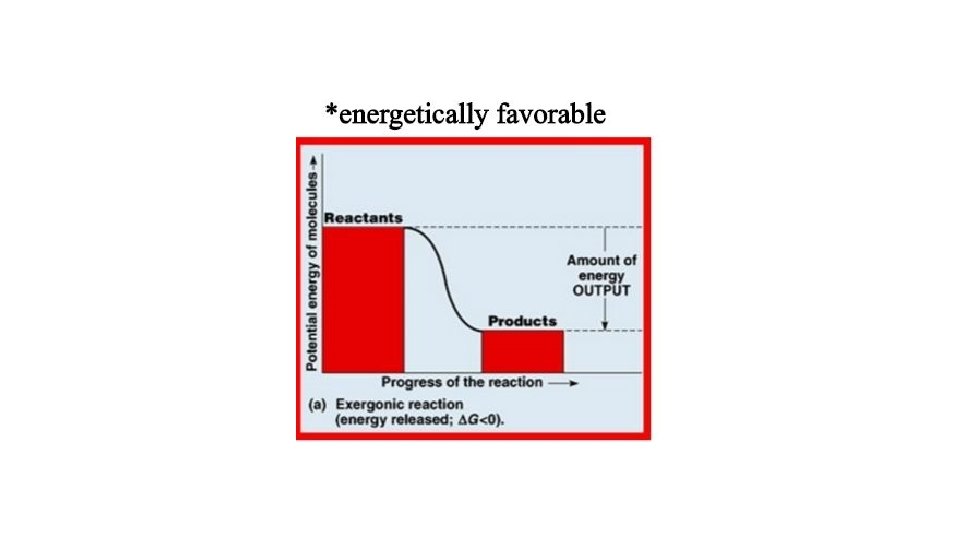
Finally, not all energetically favorable, “spontaneous” reactions are actually spontaneous.






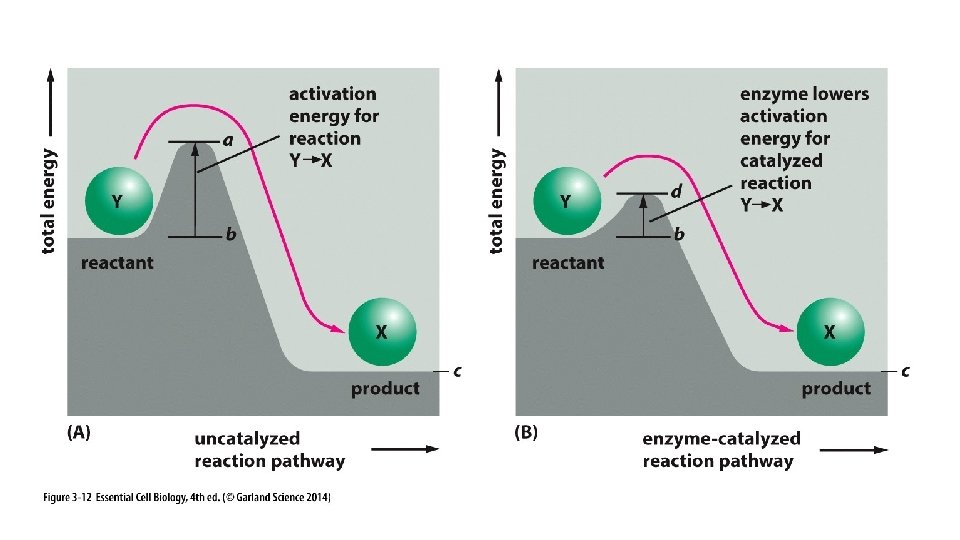
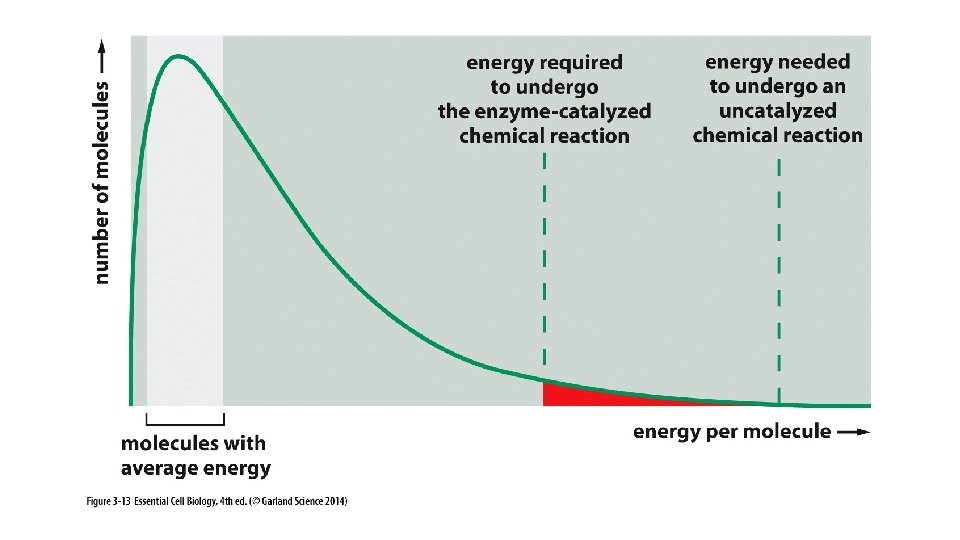
“Take Home” messages: • DG (free energy) is used to predict whether a reaction will occur spontaneously. • DG tells us how much energy will be produced if a reaction is spontaneous and if not, how much energy will be required to drive it. • DG is related to K, the equilibrium constant, which helps us predict the affinity between reactants. • Spontaneous reactions often must overcome an “activation energy”; this is facilitated by enzymes.