Cells and Batteries Electricity Circuits Electricity the presence

Cells and Batteries

Electricity & Circuits Electricity: the presence and flow of ____________, usually in the form of ______ through _______ such as copper wires. Electric Circuit: closed path along which ______ that are powered by an energy source can flow. A circuit requires a source of electrical energy which is often a cell or battery.

Battery vs. Cell The word ‘battery’ is used when in actuality, you’re referring to a ‘cell. ’ n. A cell is a device that uses chemical reactions to produce a _____ of electrons. n. A battery is more than one cell connected. n Electric Cells are usually made by separating 2 DIFFERENT metals and putting a liquid that can conduct electricity between them.

Voltaic Cells Voltaic Cell (cell): a source of ____ that generates an electric _____ by chemical reactions involving two different _____ or metal compounds separated by a solution that is a ______. Electric Current: a flow of electric charge. Look familiar? Voltaic Cells are the popular AA and D “batteries”. A battery is made when you place two or more cells in a flashlight.

Parts of a Cell Electrolyte: a ______ or ______ that conducts charge. • In this example, lemons contain acids; acids _______ electricity well. Electrodes: the metal terminals in a cell or battery. n Must be _______ metals with different abilities to ______ electrons. n In this example, the two electrodes are made of aluminum and copper.

First – What is an ion? Ion: an atom in which the total number of electrons is not equal to the total number of protons, giving the atom or molecule a net _______ or _______ electrical charge. Ions can be created, by either chemical or physical means, via ionization. Practice: 1. An oxygen atom, chemical symbol O, gains two electrons. What is its new atomic symbol? 2. A magnesium atom, chemical symbol Mg, loses one electron. What is its new atomic symbol? 3. A nitrogen atom, chemical symbol N, gains three electrons. What is its new atomic symbol? _____
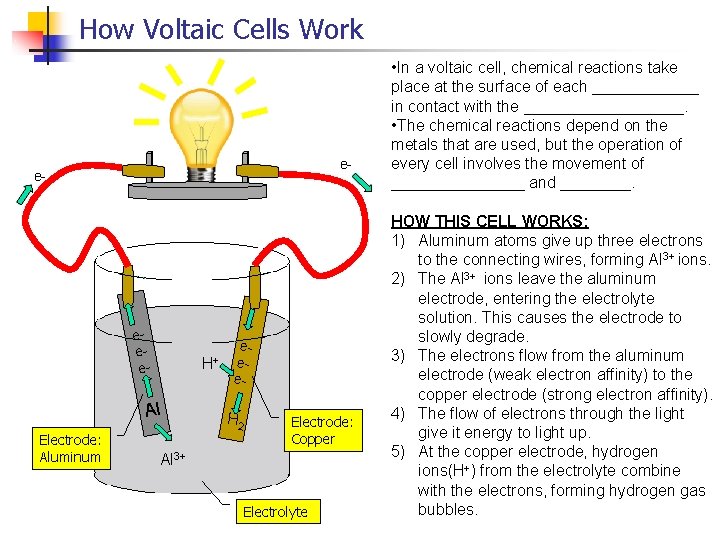
How Voltaic Cells Work e- e- eee- H+ Al Electrode: Aluminum eee- H 2 Electrode: Copper Al 3+ Electrolyte • In a voltaic cell, chemical reactions take place at the surface of each ______ in contact with the _________. • The chemical reactions depend on the metals that are used, but the operation of every cell involves the movement of ________ and ____. HOW THIS CELL WORKS: 1) Aluminum atoms give up three electrons to the connecting wires, forming Al 3+ ions. 2) The Al 3+ ions leave the aluminum electrode, entering the electrolyte solution. This causes the electrode to slowly degrade. 3) The electrons flow from the aluminum electrode (weak electron affinity) to the copper electrode (strong electron affinity). 4) The flow of electrons through the light give it energy to light up. 5) At the copper electrode, hydrogen ions(H+) from the electrolyte combine with the electrons, forming hydrogen gas bubbles.
- Slides: 7