Cell suspension culture Cell suspension culture When callus











































- Slides: 51

Cell suspension culture

Cell suspension culture • When callus pieces are agitated in a liquid medium, they tend to break up. • Suspensions are much easier to bulk up than callus since there is no manual transfer or solid support. • Large scale (50, 000 l) commercial fermentations for Shikonin and Berberine.



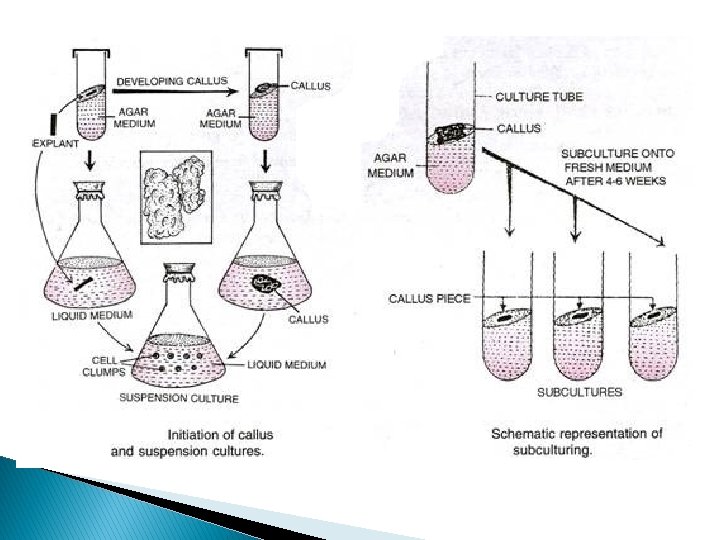
� Aggregates of cells suspended in a liquid medium. � Transfer of callus to liquid media. � Then they are agitated.


FRIABILITY � Separation of cells following cell division. � Good suspension : � Culture consisting of a high percentage of single cells and small clusters of cells. � Different requirements for different cases. � The choice of Suitable conditions is largely determined by trial and error.

Characteristics of plant cells • Large (10 -100 M long) • Tend to occur in aggregates • Shear-sensitive • Slow growing • Easily contaminated • Low oxygen demand (kla of 5 -20) • Will not tolerate anaerobic conditions • Can grow to high cell densities (>300 g/l fresh weight). • Can form very viscous solutions

� Large amount of callus is required. � 2 -3 gm for 100 cm 3 � Three phases will be observed � Lag phase � Cell division � Stationary phase � The cells should be subcultured early during the stationary phase.

� Different time periods for subculturing � Some plants max. cell density is reached within about 18 -25 days. � In some plants as short as 6 -9 days � Nylon net or stainless steel filter is used to remove larger cell aggregates.

� Small portion is withdrawn and cell density will be checked � 9 -15× 103 cells / cm 3 for sycamore. � The best speed for 100 -120 rpm � Liquid should be filled 20% of the size of the flask for adequate aeration.

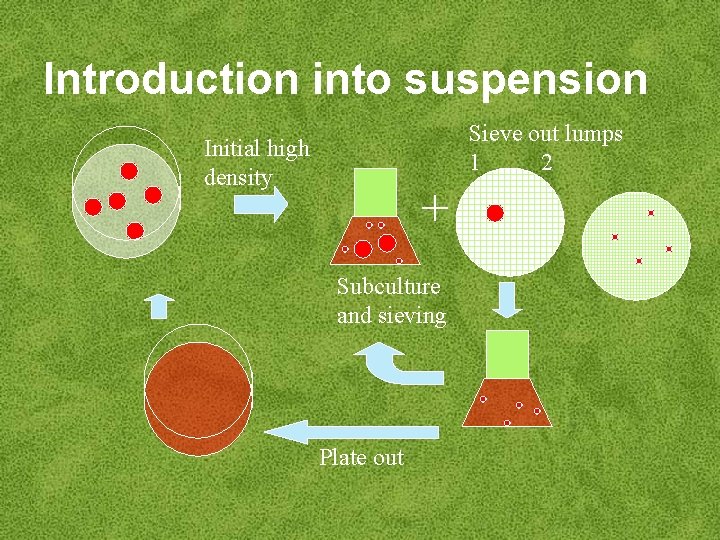
Introduction of callus into suspension • ‘Friable’ callus goes easily into suspension – – 2, 4 -D low cytokinin semi-solid medium enzymic digestion with pectinase – blending • Removal of large cell aggregates by sieving • Plating of single cells and small cell aggregates - only viable cells will grow and can be reintroduced into suspension

Introduction into suspension Sieve out lumps 1 2 Initial high density + Subculture and sieving Plate out

Growth kinetics 1. Initial lag dependent on dilution 2. Exponential phase (dt 1 -30 d) 3. Linear/deceleration phase (declining nutrients) 4. Stationary (nutrients exhausted) 3 1 2 4

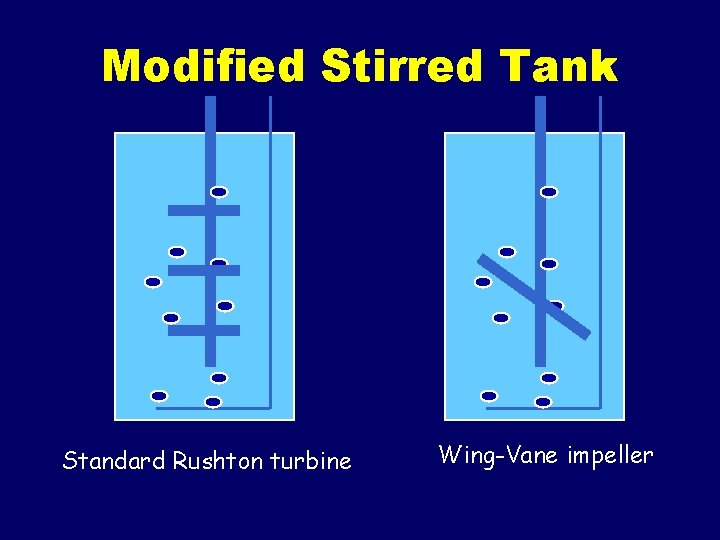
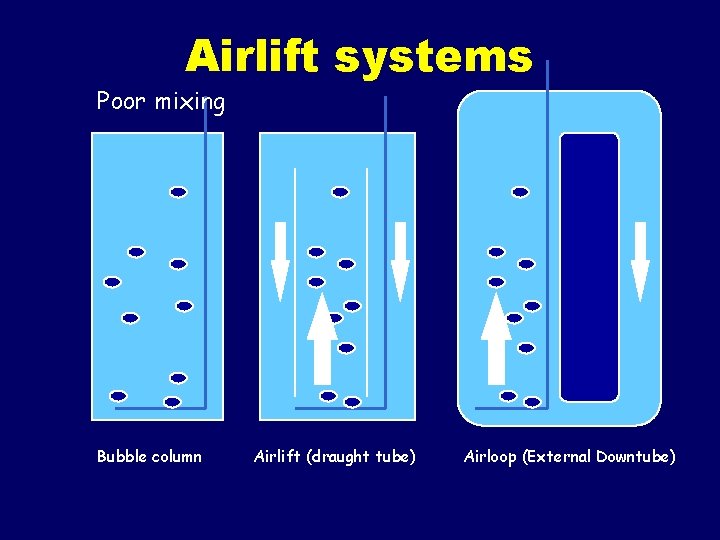
Reactors for plant suspension cultures • • • Modified stirred tank Air-lift Air loop Bubble column Rotating drum reactor

Modified Stirred Tank Standard Rushton turbine Wing-Vane impeller

Airlift systems Poor mixing Bubble column Airlift (draught tube) Airloop (External Downtube)

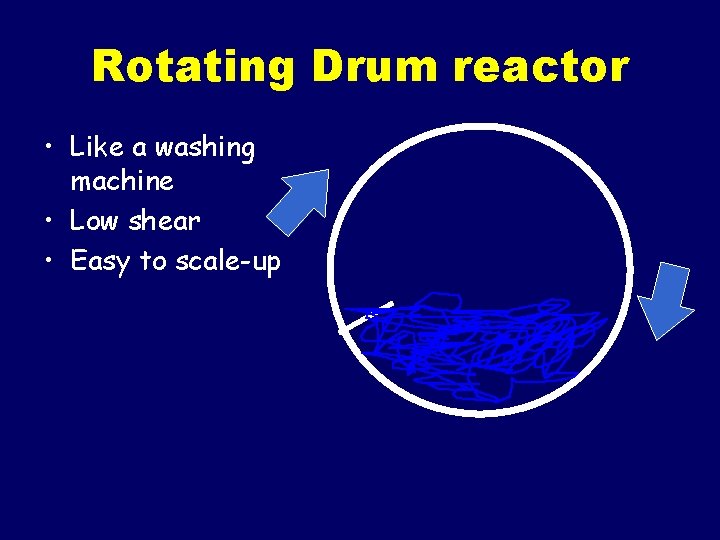
Rotating Drum reactor • Like a washing machine • Low shear • Easy to scale-up

Ways to increase product formation • Select • Start off with a producing part • Modify media for growth and product formation. • Feed precursors or feed intermediates (bioconversion) • Produce ‘plant-like’ conditions (immobilisation)

Synchronization • Cold treatment: 4 o. C • Starvation: deprivation of an essential growth compound, e. g. N →accumulation in G 1 • Use of DNA synthesis inhibitors: thymidine, 5 fluorodeoxyuridine, hydroxyurea • Colchicine method: arresting the cells in metaphase stage, measured in terms of mitotic index (% cells in the mitotic bphase)

Selection • Select at the level of the intact plant • Select in culture – single cell is selection unit – possible to plate up to 1, 000 cells on a Petri-dish. – Progressive selection over a number of phases

Selection Strategies • • Positive Negative Visual Analytical Screening

Positive selection • Add into medium a toxic compound e. g. hydroxy proline, kanamycin • Only those cells able to grow in the presence of the selective agent give colonies • Plate out and pick off growing colonies. • Possible to select one colony from millions of plated cells in a days work. • Need a strong selection pressure - get escapes

Negative selection • Add in an agent that kills dividing cells e. g. chlorate / BUd. R. • Plate out leave for a suitable time, wash out agent then put on growth medium. • All cells growing on selective agent will die leaving only non-growing cells to now grow. • Useful for selecting auxotrophs.

Visual selection • Only useful for colored or fluorescent compounds e. g. shikonin, berberine, some alkaloids • Plate out at about 50, 000 cells per plate • Pick off colored / fluorescent-expressing compounds (cell compounds? ) • Possible to screen about 1, 000 cells in a days work.

Analytical Screening • Cut each piece of callus in half • One half subcultured • Other half extracted and amount of compound determined analytically (HPLC/ GCMS/ ELISA)

Embryo Culture

Embryo Culture Uses • Rescue F 1 hybrids from wide crosses • Overcome seed dormancy, usually with addition of hormone (GA) to medium • To overcome immaturity in seeds – To speed generations in a breeding program – To rescue a cross or self (valuable genotype)

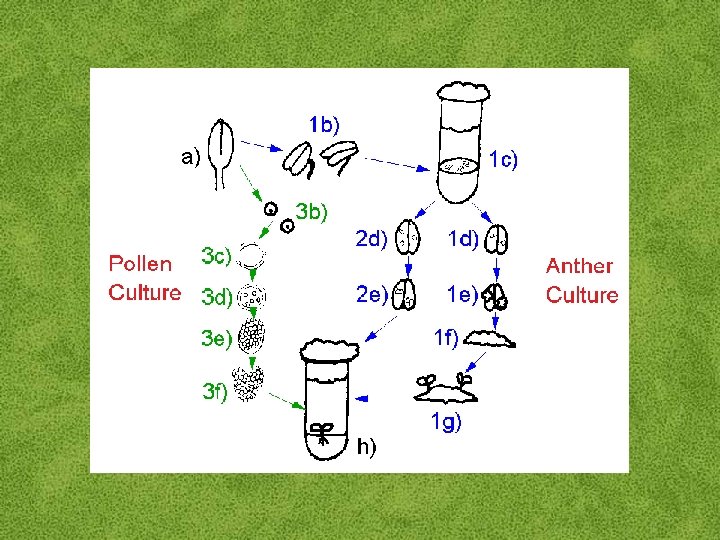
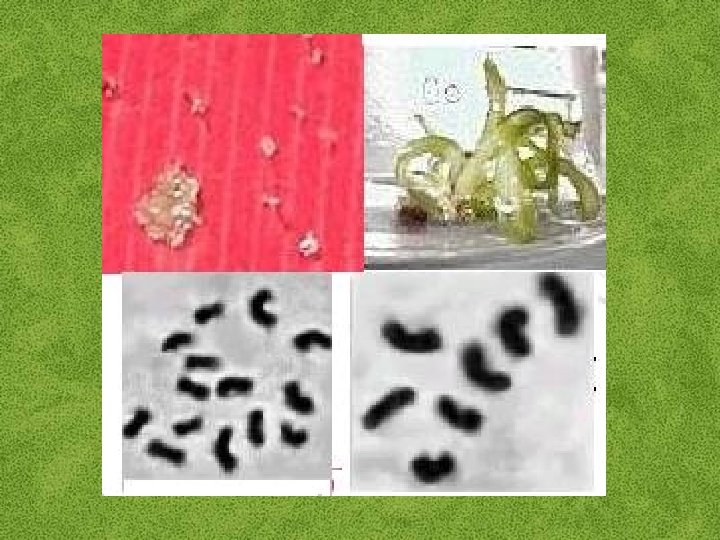
Haploid Plant Production • Embryo rescue of interspecific crosses – Bulbosum method • Anther culture/Microspore culture – Culturing of anthers or pollen grains (microspores) – Derive a mature plant from a single microspore • Ovule culture – Culturing of unfertilized ovules (macrospores)





Anther/Microspore Culture Factors • Genotype • Optimum growth of mother plant • Correct stage of pollen development – Need to be able to switch pollen development from gametogenesis to embryogenesis • Pretreatment of anthers – Cold and heat have been effective • Culture medium – Additives – Agar vs. ‘Floating’

Ovule Culture for Haploid Production • Essentially the same as embryo culture – difference is an unfertilized ovule instead of a fertilized embryo • Effective for crops that do not yet have an efficient microspore culture system – e. g. : melon, onion

Haploids • Weak, sterile plant • Usually want to double the chromosomes, creating a dihaploidbplant with normal growth & fertility – Chromosomes can be doubled by – Colchicine treatment – Spontaneous doubling

Germplasm Preservation Extension of micropropagation techniques: Two methods: • 1. Slow growth techniques – ↓Temp. , ↓Light, media supplements (osmotic inhibitors, growth retardants), tissue dehydration, etc – Medium-term storage (1 to 4 years) • 2. Cryopreservation – Ultra low temperatures. Stops cell division & metabolic processes – Very long-term (indefinite? )

Most economical germplasm storage – Why not seeds? • Some crops do not produce viable seeds • Some seeds remain viable for a limited duration only and are recalcitrant to storage • Seeds of certain species deteriorate rapidly due to seed borne pathogen • Some seeds are very heterozygous not suitable for maintaining true to type genotypes • Effective approach to circumvent the above problems may be application of cryopreservation technology

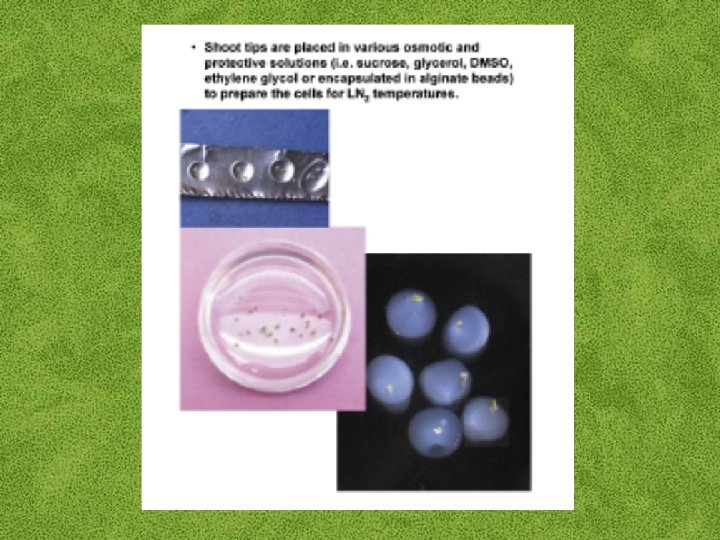
Cryogenic explants: • • Undifferentiated plant cells Embryonic suspension Callus Pollen Seeds Somatic embryos Shoot apices

Preparation • • Pretreatment Cryopreservation method Thawing method Recovery method is critical







Cryobiology • Is the study of the effects of extremely low temperatures on biological systems, such as cells or organisms. • Cryopreservation – an applied aspect of cryobiology – has resulted in methods that permit low temperature maintenance of a diversity of cells

Cryopreservation Requirements: • Preculturing–Usually a rapid growth rate to create cells with small vacuoles and low water content • Cryoprotection–Glycerol, DMSO, PEG, etc…, to protect against ice damage and alter the form of ice crystals • Freezing–The most critical phase; one of two methods: • Slow freezing allows for cytoplasmic dehydration • Quick freezing results in fast intercellular freezing with little dehydration

Cryopreservation Requirements • Storage–Usually in liquid nitrogen (-96 C) to avoid changes in ice crystals that occur above 100 C • Thawing–Usually rapid thawing to avoid damage from ice crystal growth • Recovery – – Thawed cells must be washed of cryoprotectants and nursed back to normal growth– – Callus production avoided to maintain genetic stability

