CELL SIGNALING AND MOTILITY BIOL 3373 Lecture 9

CELL SIGNALING AND MOTILITY (BIOL 3373) Lecture 9 1

CELL MOTILITY 2
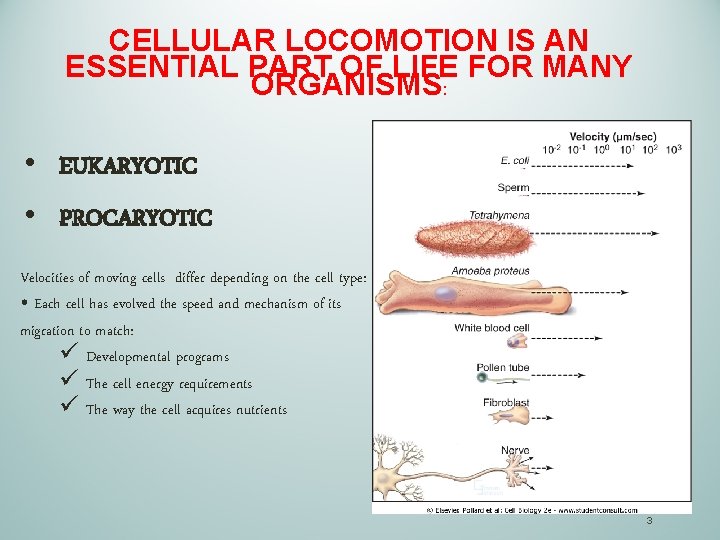
CELLULAR LOCOMOTION IS AN ESSENTIAL PART OF LIFE FOR MANY ORGANISMS: • EUKARYOTIC • PROCARYOTIC Velocities of moving cells differ depending on the cell type: • Each cell has evolved the speed and mechanism of its migration to match: ü Developmental programs ü The cell energy requirements ü The way the cell acquires nutrients 3

Cellular Migration in higher vertebrate are involved in: • • • Tissue and organ development Formation of blood vessels Development of axons Wound healing Protection against infection 4
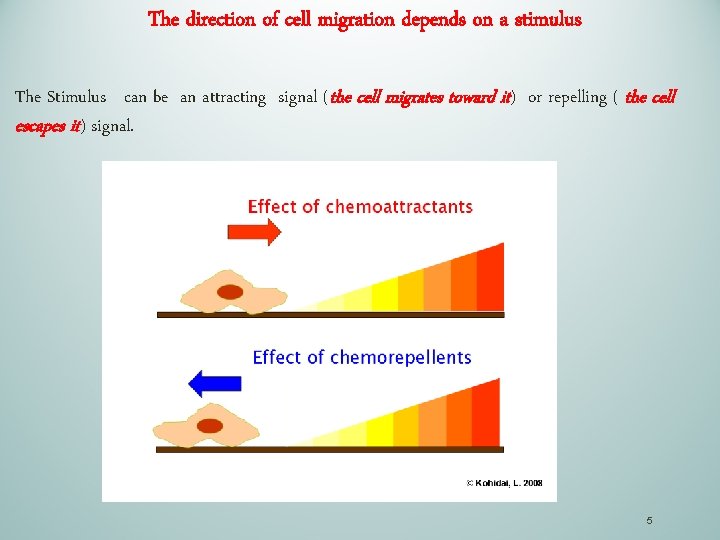
The direction of cell migration depends on a stimulus The Stimulus can be an attracting signal (the cell migrates toward it) or repelling ( the cell escapes it) signal. 5
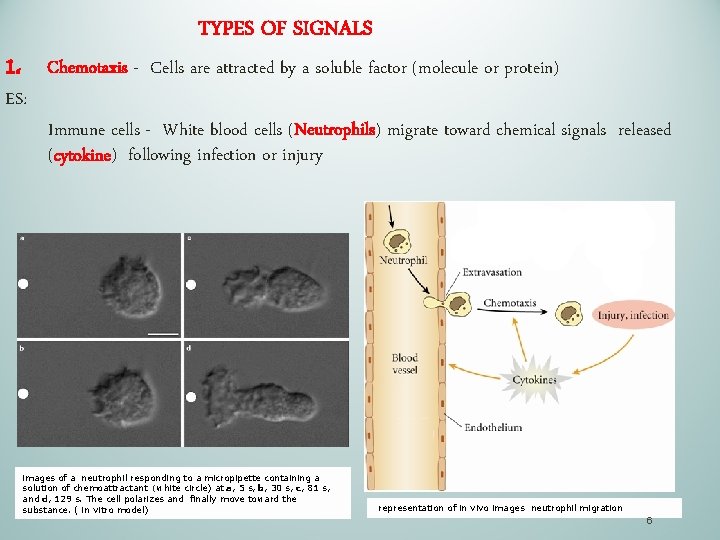
1. TYPES OF SIGNALS ES: Chemotaxis - Cells are attracted by a soluble factor (molecule or protein) Immune cells - White blood cells (Neutrophils) migrate toward chemical signals released (cytokine) following infection or injury images of a neutrophil responding to a micropipette containing a solution of chemoattractant (white circle) at a, 5 s, b, 30 s, c, 81 s, and d, 129 s. The cell polarizes and finally move toward the substance. ( in vitro model) representation of in vivo images neutrophil migration 6
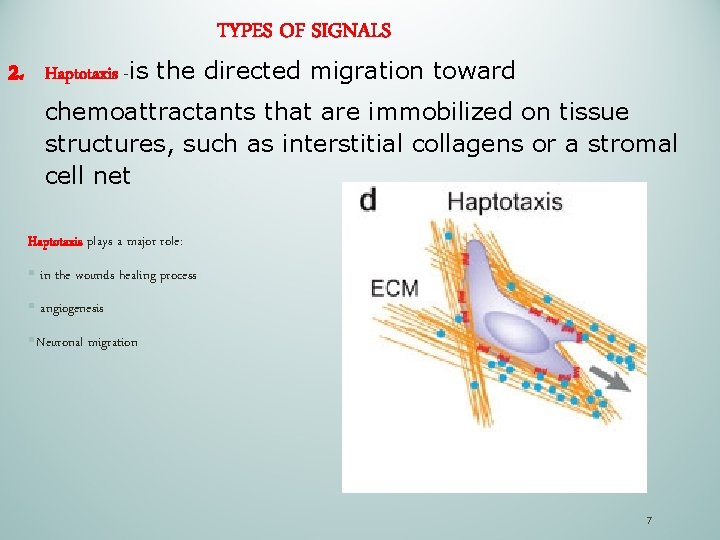
TYPES OF SIGNALS 2. Haptotaxis -is the directed migration toward chemoattractants that are immobilized on tissue structures, such as interstitial collagens or a stromal cell net Haptotaxis plays a major role: § in the wounds healing process § angiogenesis §Neuronal migration 7

TYPES OF SIGNALS 3. Necrotaxis a special type of chemotaxis when the chemoattractant molecules are released from necrotic or apoptotic cells. For Example Neutrophils migrate toward chemical signals from injured, inflamed, and dead tissue. Composition of the substances inducing necrotaxis is rather complex, some of them are still obscure. However, depending on the chemical character of molecules released, necrotaxis can accumulate or repel cells. 8
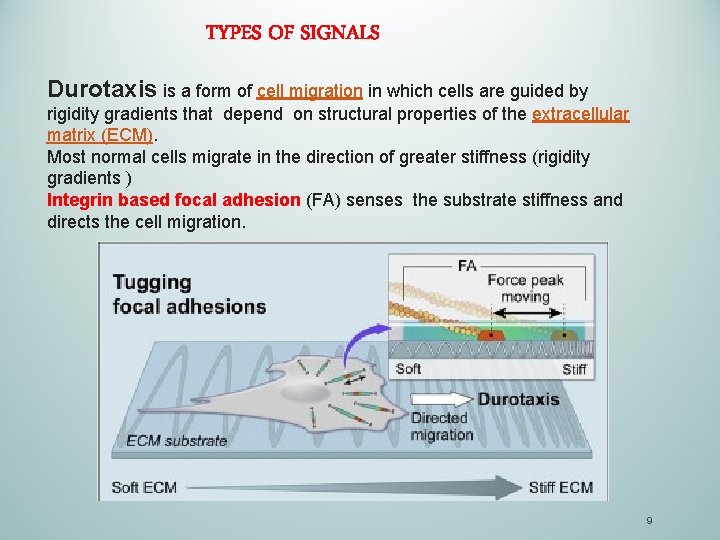
TYPES OF SIGNALS Durotaxis is a form of cell migration in which cells are guided by rigidity gradients that depend on structural properties of the extracellular matrix (ECM). Most normal cells migrate in the direction of greater stiffness (rigidity gradients ) Integrin based focal adhesion (FA) senses the substrate stiffness and directs the cell migration. 9

MIGRATION IS A FORM OF CELL SIGNALING • The Signaling cell is the cell that releases the chemoattractant or chemo repellent. • Target cell is the cell that senses the substance and eventually moves • Cell Migration follows all steps of cell signaling: 1. Reception of signal (target cells sense a chemoattractant or chemo repellent) 2. Transduction of signal (target cells reorganize the cytoskeleton 3. Cellular response Cell motility towards or away from the signal 10

HOW DO CELLS MOVE? 11

HOW DO CELL’S MOVE? The easiest way to study cell migration is to analyze on a single cell that moves over a flat substrate (2 D) Keep in mind that cells in the body do not move over 2 D substrates but over an extracellular matrix, which has a 3 D structure. Also cell usually do not move alone In vivo the migration is a complicated process and it not fully understood yet. 12

CELLULAR MIGRATION IS A CYCLE OF FOUR STAGES THAT FOLLOW A STIMULATION: 1. Extension- comprises polarization of the cell (defining front vs. back, leading edge vs trailing edge) and formation of protrusions ( lamellipodia and /or filopodia) of the leading edge in the front 2. Formation of adhesive contacts (or focal adhesion) with the surface 3. Translocation 4. De-adhesion and retraction of the trailing edge in the back 13
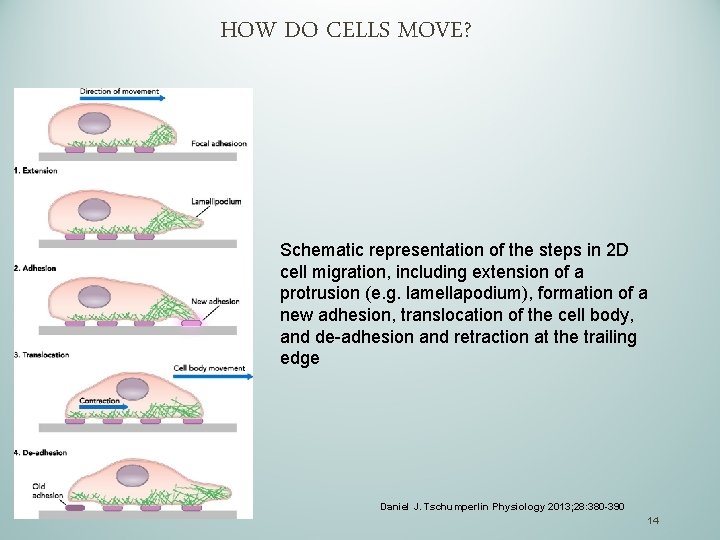
HOW DO CELLS MOVE? Schematic representation of the steps in 2 D cell migration, including extension of a protrusion (e. g. lamellapodium), formation of a new adhesion, translocation of the cell body, and de-adhesion and retraction at the trailing edge Daniel J. Tschumperlin Physiology 2013; 28: 380 -390 14
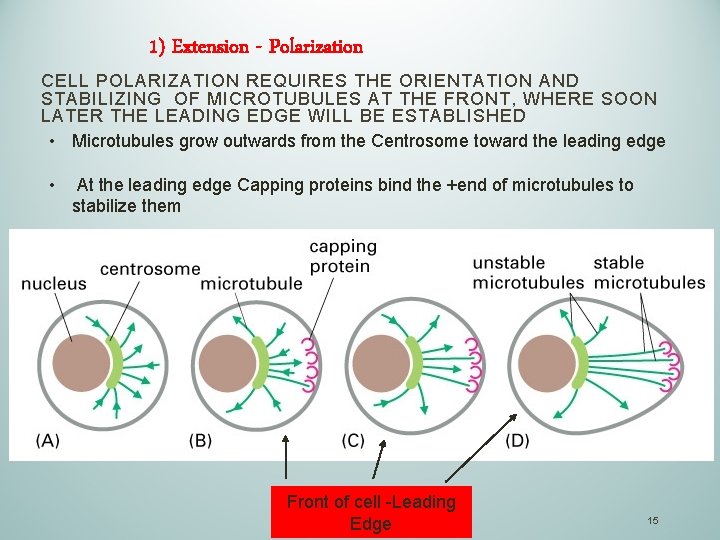
1) Extension - Polarization CELL POLARIZATION REQUIRES THE ORIENTATION AND STABILIZING OF MICROTUBULES AT THE FRONT, WHERE SOON LATER THE LEADING EDGE WILL BE ESTABLISHED • Microtubules grow outwards from the Centrosome toward the leading edge • At the leading edge Capping proteins bind the +end of microtubules to stabilize them Front of cell -Leading Edge 15

1. EXTENSION -PROTRUSION • Protrusion is driven by actin polymerization that produce the leading edge • There are 2 types of protrusive structures in motile cells: lamellipodia (broad flat three-like) and filopodia (finger-like) 16
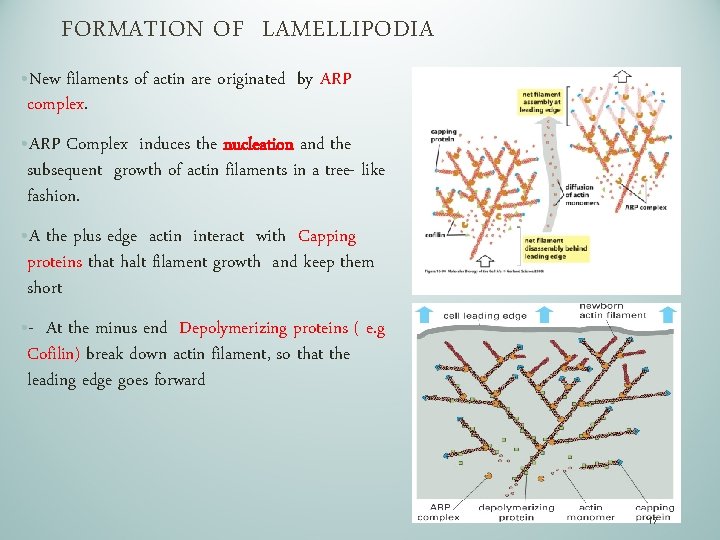
FORMATION OF LAMELLIPODIA • New filaments of actin are originated by ARP complex. • ARP Complex induces the nucleation and the subsequent growth of actin filaments in a tree- like fashion. • A the plus edge actin interact with Capping proteins that halt filament growth and keep them short • - At the minus end Depolymerizing proteins ( e. g Cofilin) break down actin filament, so that the leading edge goes forward 17
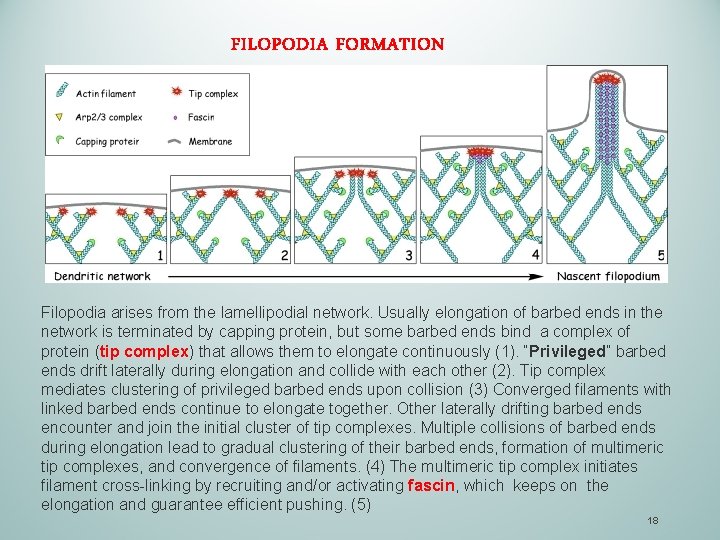
FILOPODIA FORMATION Filopodia arises from the lamellipodial network. Usually elongation of barbed ends in the network is terminated by capping protein, but some barbed ends bind a complex of protein (tip complex) that allows them to elongate continuously (1). “Privileged” barbed ends drift laterally during elongation and collide with each other (2). Tip complex mediates clustering of privileged barbed ends upon collision (3) Converged filaments with linked barbed ends continue to elongate together. Other laterally drifting barbed ends encounter and join the initial cluster of tip complexes. Multiple collisions of barbed ends during elongation lead to gradual clustering of their barbed ends, formation of multimeric tip complexes, and convergence of filaments. (4) The multimeric tip complex initiates filament cross-linking by recruiting and/or activating fascin, which keeps on the elongation and guarantee efficient pushing. (5) 18
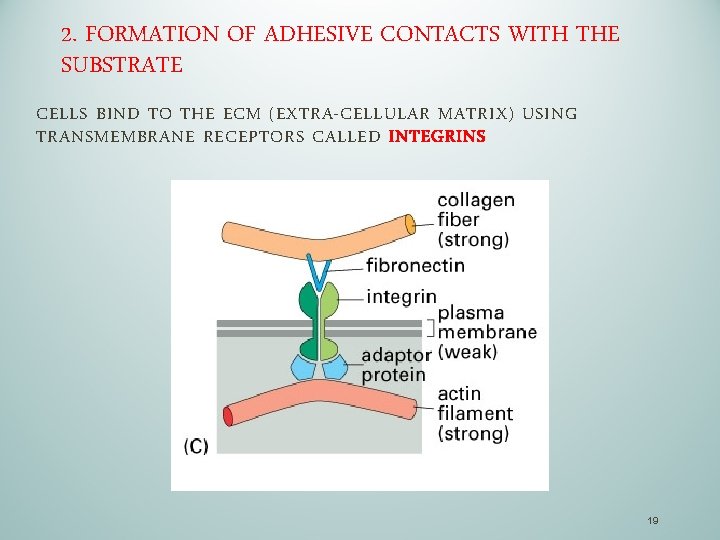
2. FORMATION OF ADHESIVE CONTACTS WITH THE SUBSTRATE CELLS BIND TO THE ECM (EXTRA-CELLULAR MATRIX) USING TRANSMEMBRANE RECEPTORS CALLED INTEGRINS 19
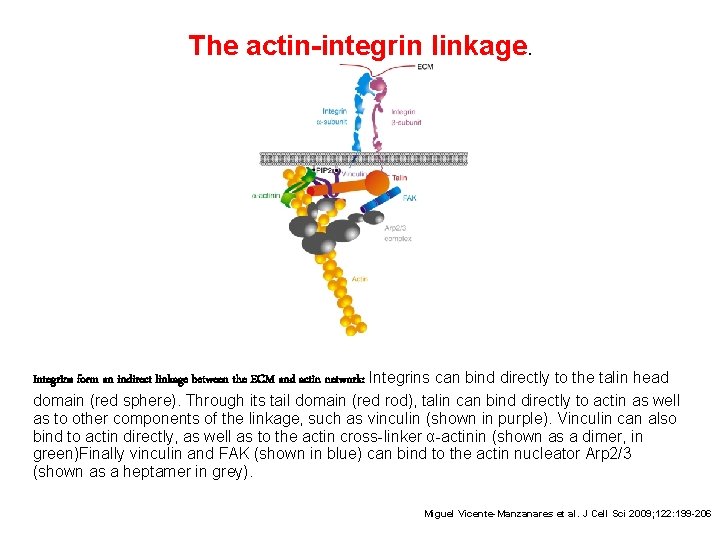
The actin-integrin linkage. Integrins form an indirect linkage between the ECM and actin network: Integrins can bind directly to the talin head domain (red sphere). Through its tail domain (red rod), talin can bind directly to actin as well as to other components of the linkage, such as vinculin (shown in purple). Vinculin can also bind to actin directly, as well as to the actin cross-linker α-actinin (shown as a dimer, in green)Finally vinculin and FAK (shown in blue) can bind to the actin nucleator Arp 2/3 (shown as a heptamer in grey). Miguel Vicente-Manzanares et al. J Cell Sci 2009; 122: 199 -206
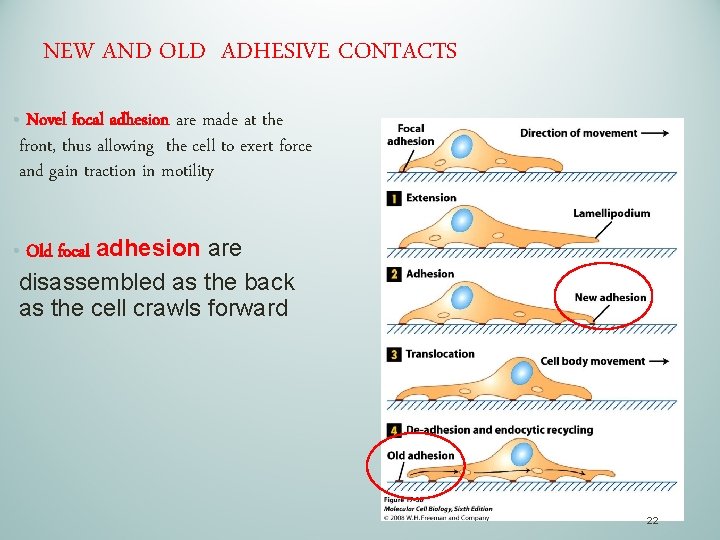
NEW AND OLD ADHESIVE CONTACTS • Novel focal adhesion are made at the front, thus allowing the cell to exert force and gain traction in motility • Old focal adhesion are disassembled as the back as the cell crawls forward 22
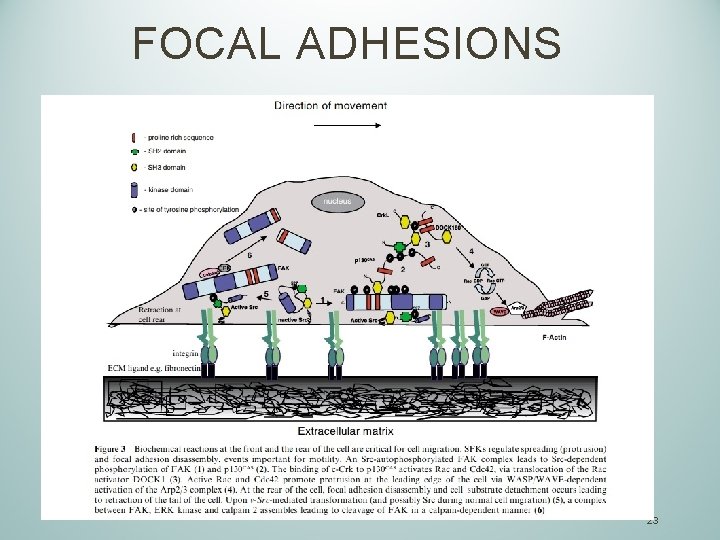
FOCAL ADHESIONS 23

3 TRANSLOCATION. Cell body translocation immediately follows protrusion and is independent of actin polymerization. In keratocytes, the cell body 'rolls' behind the front protrusion. This movement is propelled by a coordinated contraction of the actomyosin cytoskeleton, and thus depends on myosin II. 24
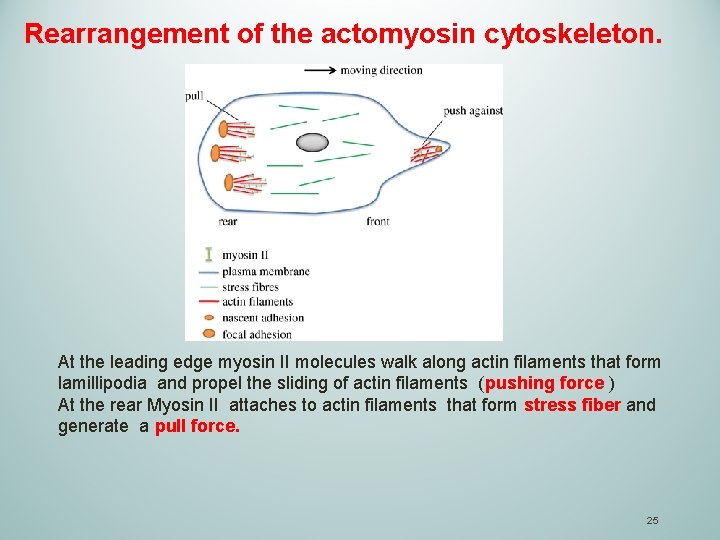
Rearrangement of the actomyosin cytoskeleton. At the leading edge myosin II molecules walk along actin filaments that form lamillipodia and propel the sliding of actin filaments (pushing force ) At the rear Myosin II attaches to actin filaments that form stress fiber and generate a pull force. 25
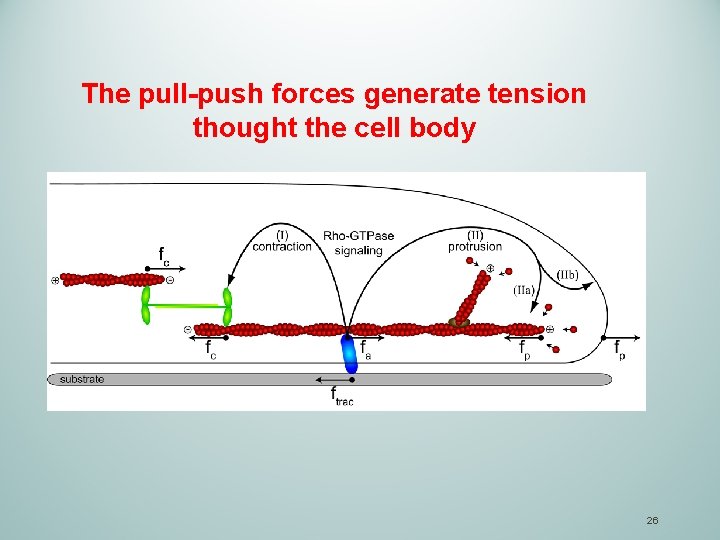
The pull-push forces generate tension thought the cell body 26
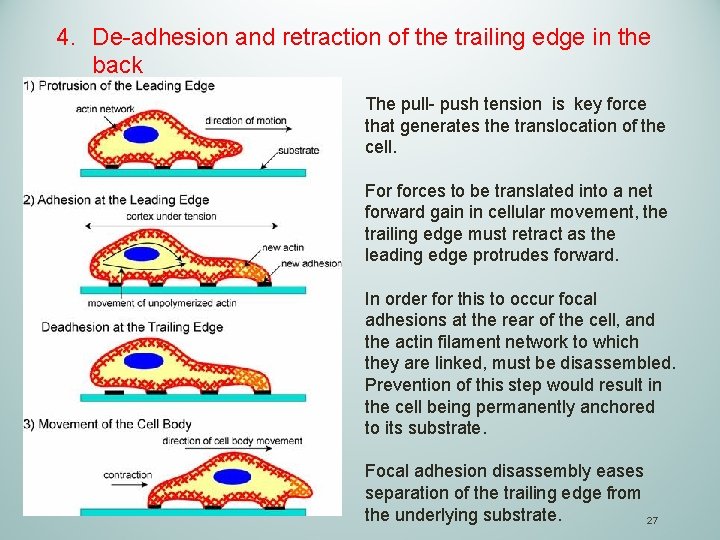
4. De-adhesion and retraction of the trailing edge in the back The pull- push tension is key force that generates the translocation of the cell. For forces to be translated into a net forward gain in cellular movement, the trailing edge must retract as the leading edge protrudes forward. In order for this to occur focal adhesions at the rear of the cell, and the actin filament network to which they are linked, must be disassembled. Prevention of this step would result in the cell being permanently anchored to its substrate. Focal adhesion disassembly eases separation of the trailing edge from the underlying substrate. 27

CELL MOTILITY https: //www. youtube. com/watch? v=t 3 u 2_p. AEB 94 28

Composite describing how tumorigenesis and mesenchymal stroma progression are two processes which affect and incite each other. The photographs represent confocal 3 D reconstructions of fibroblast-derived ECMs (in brown) and cell nuclei (in green) where the disorganized in vitro stroma is shown in the left panel while the parallel patterned architecture of tumor-associated stromal matrix is evident in the right panel. The gradient progression bar in brown, at the bottom of the composite, represents the increased stiffness and architectural patterning of the stroma during these joint progression processes. The two confocal images are a variation from images published in Amatangelo, Bassi et al. 2005 [20]. From: Cukierman E, Bassi DE. Physico-mechanical aspects of extracellular matrix influences on
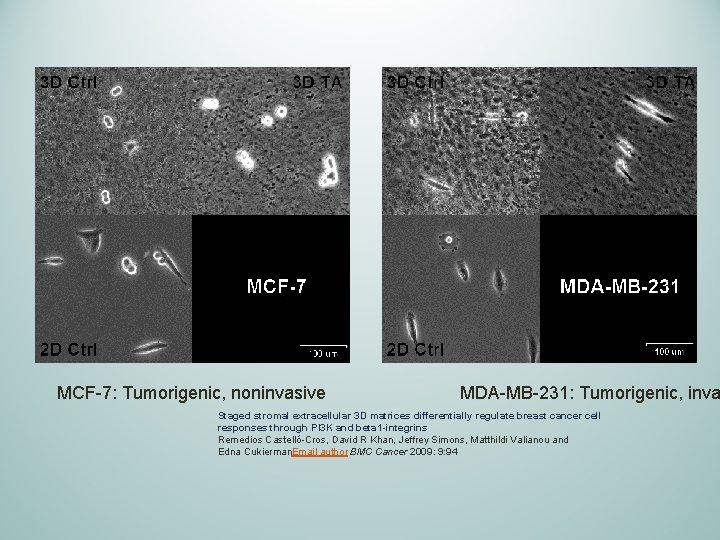
MCF-7: Tumorigenic, noninvasive MDA-MB-231: Tumorigenic, inva Staged stromal extracellular 3 D matrices differentially regulate breast cancer cell responses through PI 3 K and beta 1 -integrins Remedios Castelló-Cros, David R Khan, Jeffrey Simons, Matthildi Valianou and Edna Cukierman. Email author BMC Cancer 2009: 9: 94
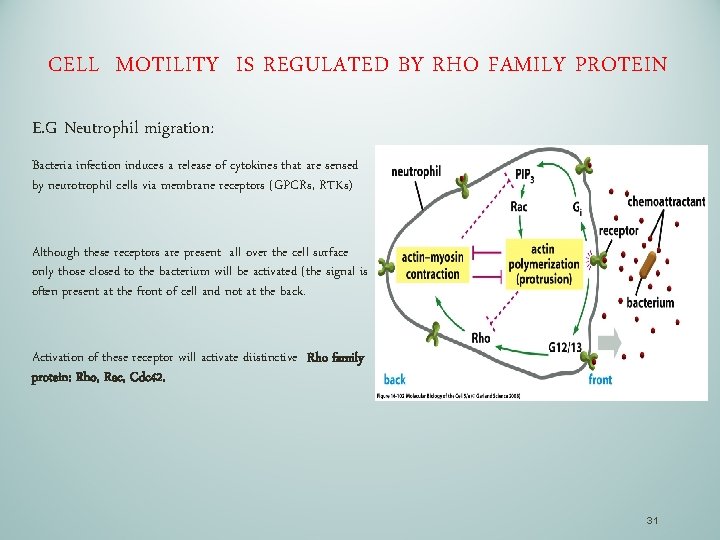
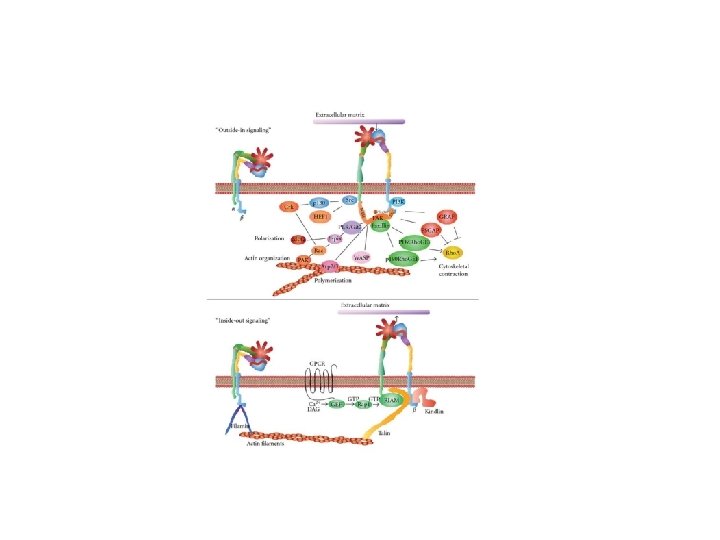
CELL MOTILITY IS REGULATED BY RHO FAMILY PROTEIN E. G Neutrophil migration: Bacteria infection induces a release of cytokines that are sensed by neurotrophil cells via membrane receptors (GPCRs, RTKs) Although these receptors are present all over the cell surface only those closed to the bacterium will be activated (the signal is often present at the front of cell and not at the back. Activation of these receptor will activate diistinctive Rho family protein: Rho, Rac, Cdc 42. 31
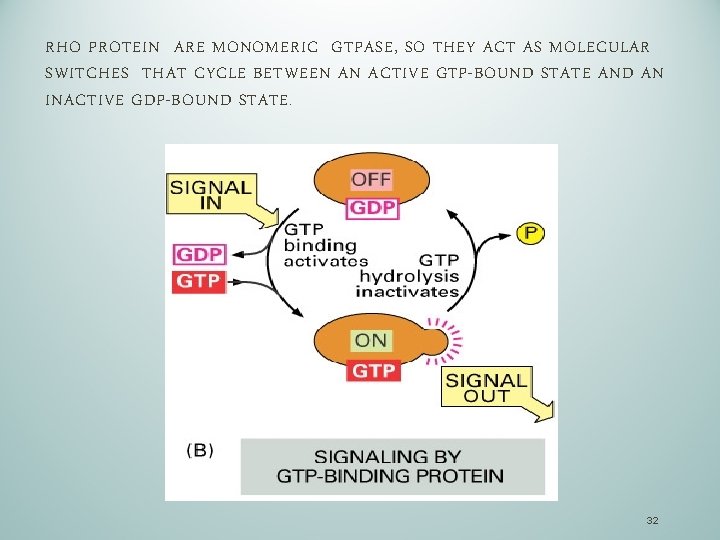
RHO PROTEIN ARE MONOMERIC GTPASE, SO THEY ACT AS MOLECULAR SWITCHES THAT CYCLE BETWEEN AN ACTIVE GTP-BOUND STATE AND AN INACTIVE GDP-BOUND STATE. 32
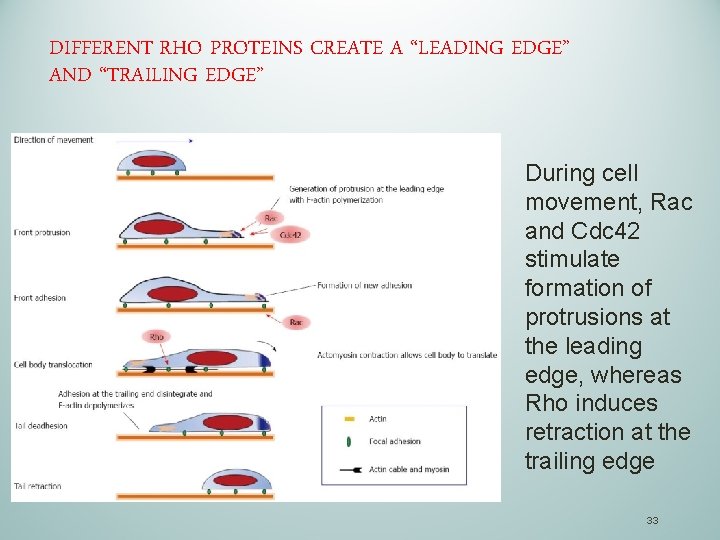
DIFFERENT RHO PROTEINS CREATE A “LEADING EDGE” AND “TRAILING EDGE” During cell movement, Rac and Cdc 42 stimulate formation of protrusions at the leading edge, whereas Rho induces retraction at the trailing edge 33
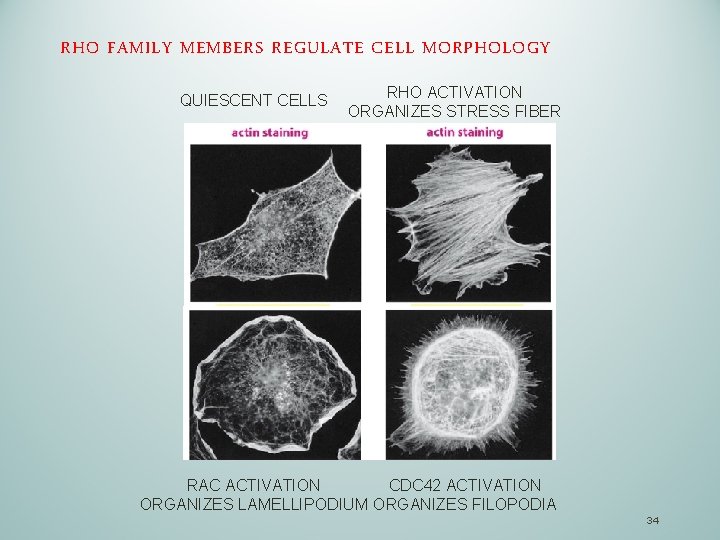
RHO FAMILY MEMBERS REGULATE CELL MORPHOLOGY QUIESCENT CELLS RHO ACTIVATION ORGANIZES STRESS FIBER RAC ACTIVATION CDC 42 ACTIVATION ORGANIZES LAMELLIPODIUM ORGANIZES FILOPODIA 34

EXAMPLES OF PROCESSES INVOLVING CELL MOTILITY 35

Embryogenesis • Embryogenesis is the process that leads the creation of the embryo, and culminate with its morphogenesis ( formation of organ and tissue) • Embryogenesis differs among organism classes ( fish, amphibian, mammalian) 36
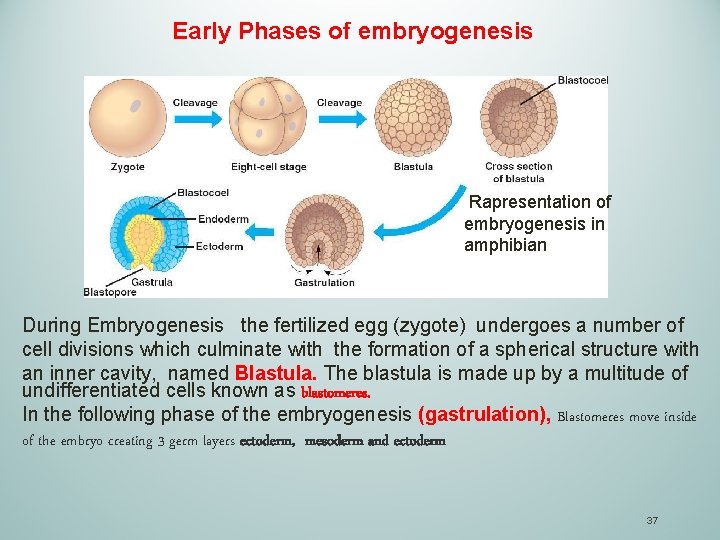
Early Phases of embryogenesis Rapresentation of embryogenesis in amphibian During Embryogenesis the fertilized egg (zygote) undergoes a number of cell divisions which culminate with the formation of a spherical structure with an inner cavity, named Blastula. The blastula is made up by a multitude of undifferentiated cells known as blastomeres. In the following phase of the embryogenesis (gastrulation), Blastomeres move inside of the embryo creating 3 germ layers ectoderm, mesoderm and ectoderm 37

During Gastrulation cells move as a group or as single entities One important aspect of the gastrulation is the migration of cells inside the blastula to create the 3 germ layer The gastrulation culminates with the formation of 3 germ layers: Ø The inner layers is called the endoderm. The endoderm will give rise to the lining of the digestive tract, respiratory tract, and circulatory system and make up other internal tissues such as the pancreas and liver. Ø The cells remaining on the outside of the embryo become the outer germ layer, the ectoderm. The ectoderm will give rise to the nervous system, the skin, hair, and nails, sweat glands, oil glands, and milk secretory ducts. Ø Other cells migrate between the endoderm and the ectoderm to become the middle germ layer, or mesoderm. The mesoderm will contribute tissues to many organs, including blood vessels, muscle, bones, liver, and heart. 38

Frog Gastrulation Development Biology - Amphibian Development Biology https: //youtu. be/OZm. Ty 4 yxy. Vk 39

MISREGULATION OF CELL MIGRATION CONTRIBUTES TO DISEASE • Congenital birth defects • Chronic inflammatory diseases (asthma & arthritis) • Cancer (metastasis) • Atherosclerosis & heart disease 40
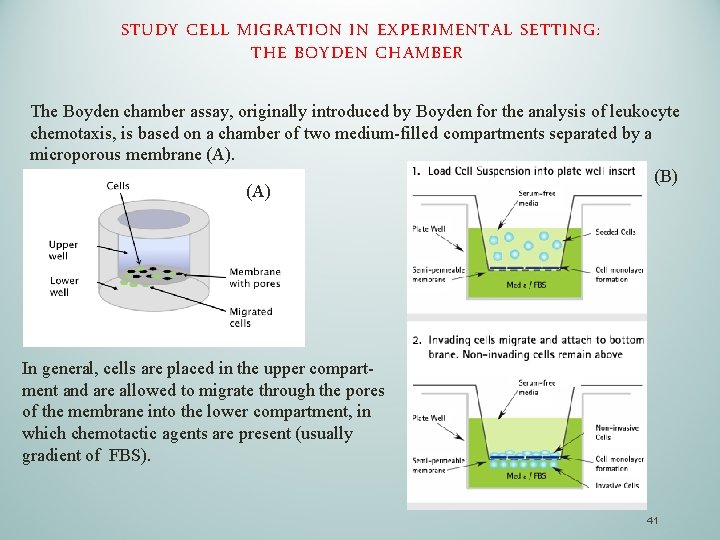
STUDY CELL MIGRATION IN EXPERIMENTAL SETTING: THE BOYDEN CHAMBER The Boyden chamber assay, originally introduced by Boyden for the analysis of leukocyte chemotaxis, is based on a chamber of two medium-filled compartments separated by a microporous membrane (A). (B) (A) In general, cells are placed in the upper compart- ment and are allowed to migrate through the pores of the membrane into the lower compartment, in which chemotactic agents are present (usually gradient of FBS). 41

STUDY CELL MIGRATION IN EXPERIMENTAL SETTING: THE BOYDEN CHAMBER In a standard Boyden assay, the pore diameter of the membrane is typically 3 to 12 μm, ( most used is 8 μm) and is selected to suit the size of the cells analyzed. Smaller pore size results in a greater challenge for the migrating cell. Most cells range in size from 3050 μm and can migrate efficiently through 8 μm pores, whereas, lymphocytes (10 μm) can migrate through pores as small as 0. 3 μm. For modelling in vivo conditions, several protocols prefer coverage of filter with molecules of extracellular matrix (collagen, elastin etc. ) Cancer Cell are invasive: with Boyden Chamber we can study invasive behavior of the cells. In this case the membrane is covered by a surrogate of ECM called matrigel. Matrigel clogs up membrane pores so normal cells can not migrate unless they are able to digest the matrigel. Cancer cells release matrix metalloproteinase (MMP) able to destroy ECM in vivo or matrigel in vitro 42
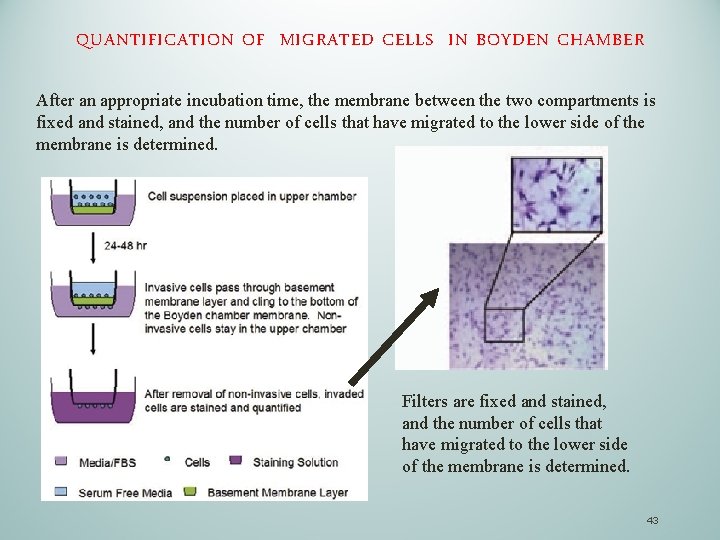
QUANTIFICATION OF MIGRATED CELLS IN BOYDEN CHAMBER After an appropriate incubation time, the membrane between the two compartments is fixed and stained, and the number of cells that have migrated to the lower side of the membrane is determined. Filters are fixed and stained, and the number of cells that have migrated to the lower side of the membrane is determined. 43

WNT SİGNALİNG PATHWAYS Lecture 10 44

WNT SIGNALING BIOLOGY 1 • Wnt ligands are a large family (19 in human) of secreted glycoproteins that are cysteine-rich and highly hydrophobic. Wnts are produced as precursor proteins that contain a short N -terminal signal sequence and a mature segment that varies in length from approximately 320 to 400 amino acids. • Wnt signaling pathways use either nearby cell-cell communication (paracrine) or same-cell communication (autocrine). They are highly evolutionarily conserved in animals. Wnt signaling was first identified for its role in carcinogenesis, then for its function in embryonic development. The embryonic processes it controls include body axis patterning, cell fate specification, cell proliferation and cell migration. These processes are necessary for proper formation of important tissues including bone, heart and muscle. Its role in embryonic development was discovered when genetic mutations in Wnt pathway proteins produced abnormal fruit fly embryos. • Wnt signaling also controls tissue regeneration in adult bone marrow, skin and intestine (stem cells) Later research found that the genes responsible for these abnormalities also influenced breast cancer development in mice. 45
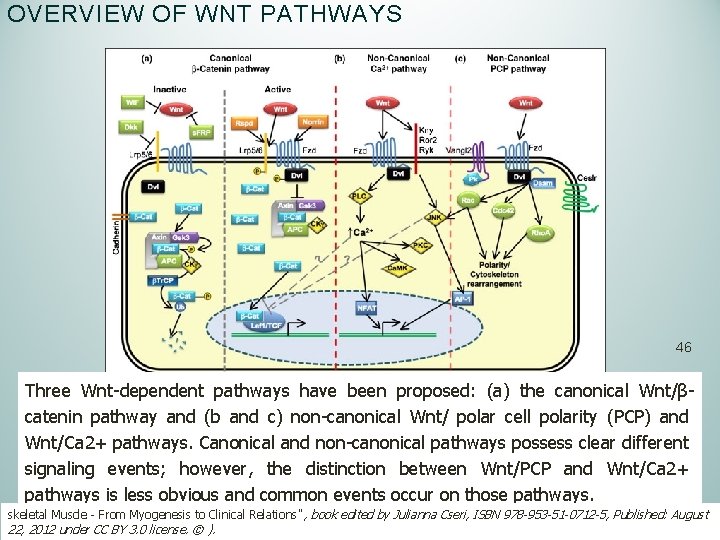
OVERVIEW OF WNT PATHWAYS 46 Three Wnt-dependent pathways have been proposed: (a) the canonical Wnt/βcatenin pathway and (b and c) non-canonical Wnt/ polar cell polarity (PCP) and Wnt/Ca 2+ pathways. Canonical and non-canonical pathways possess clear different signaling events; however, the distinction between Wnt/PCP and Wnt/Ca 2+ pathways is less obvious and common events occur on those pathways. skeletal Muscle - From Myogenesis to Clinical Relations" , book edited by Julianna Cseri, ISBN 978 -953 -51 -0712 -5, Published: August 22, 2012 under CC BY 3. 0 license. © ).
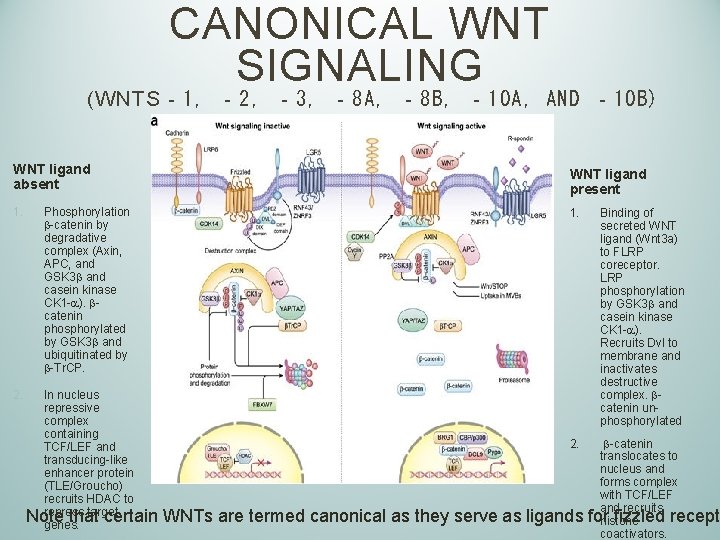
CANONICAL WNT SIGNALING (WNTS‐ 1, ‐ 2, ‐ 3, ‐ 8 A, ‐ 8 B, ‐ 10 A, AND ‐ 10 B) WNT ligand absent WNT ligand present 1. 2. Phosphorylation -catenin by degradative complex (Axin, APC, and GSK 3 and casein kinase CK 1 - ). catenin phosphorylated by GSK 3 and ubiquitinated by -Tr. CP. Binding of secreted WNT ligand (Wnt 3 a) to FLRP coreceptor. LRP phosphorylation by GSK 3 and casein kinase CK 1 - ). Recruits Dvl to membrane and inactivates destructive complex. catenin unphosphorylated In nucleus repressive complex containing 2. -catenin TCF/LEF and translocates to transducing-like nucleus and enhancer protein forms complex (TLE/Groucho) with TCF/LEF recruits HDAC to and recruits repress target Note that certain WNTs are termed canonical as they serve as ligands for fizzled recept histone genes. coactivators.

WNT PROTEINS Large family of secreted molecules 350 to 400 amino acids Highly conserved in evolution (conserved cysteine residues) 48

WNT PROTEINS FUNCTION Involved in intercellular signaling during development: n Early mesodermal formation of embryo n Morphogenesis of brain and kidneys 49

WNT PROTEINS DISCOVERY Wnt name derived from first 2 members discovered: n. Drosophila n. Mouse cancer) Wingless (morphogenesis of wing) int-1 (it induces the development of breast 50

WNT PROTEINS IS PRODUCED BY SIGNALING CELLS WNT is secreted as insoluble (hydrophobic) molecule: Wnt is Palmitoylated (addition of Palmitic acid on the cysteine residue). Palmitate is saturated 16 carbon fatty acid – first product of lipogenesis) Enzyme responsible for the palmitoylation of WNT: Porcupine acyl-transferase (por). 51
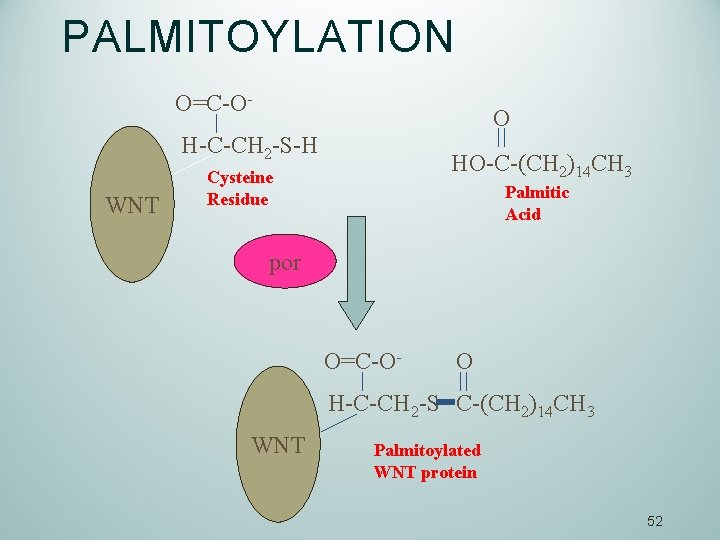
PALMITOYLATION O=C-O- O H-C-CH 2 -S-H WNT HO-C-(CH 2)14 CH 3 Cysteine Residue Palmitic Acid por O=C-O- O H-C-CH 2 -S C-(CH 2)14 CH 3 WNT Palmitoylated WNT protein 52
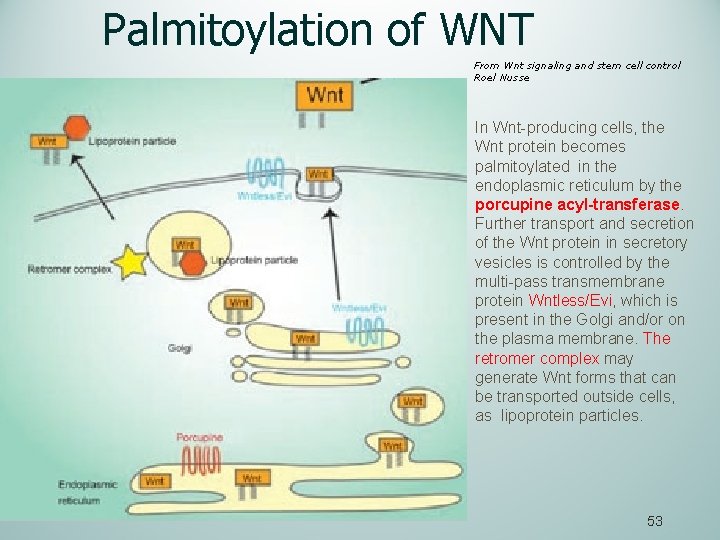
Palmitoylation of WNT From Wnt signaling and stem cell control Roel Nusse In Wnt-producing cells, the Wnt protein becomes palmitoylated in the endoplasmic reticulum by the porcupine acyl-transferase. Further transport and secretion of the Wnt protein in secretory vesicles is controlled by the multi-pass transmembrane protein Wntless/Evi, which is present in the Golgi and/or on the plasma membrane. The retromer complex may generate Wnt forms that can be transported outside cells, as lipoprotein particles. 53

Palmitoylation of WNT • Essential for function and signaling of WNT Mutation of cysteine or Removal of palmitate Inactive WNT Protein • Palmitoylation is necessary for membrane targeting 54
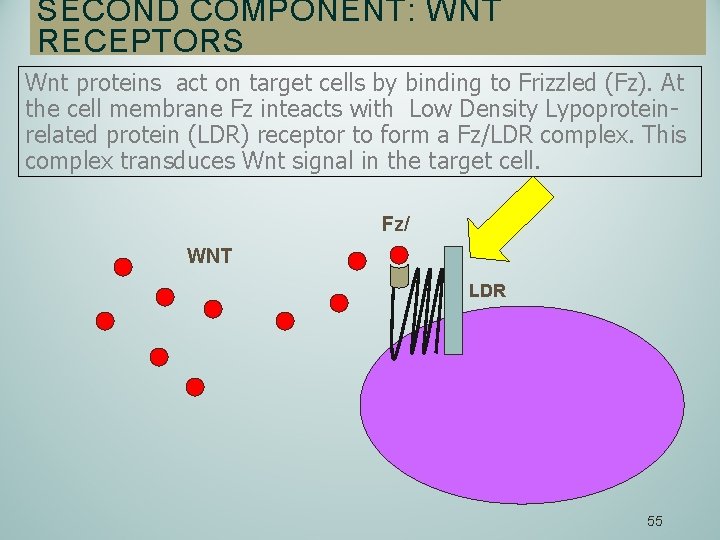
SECOND COMPONENT: WNT RECEPTORS Wnt proteins act on target cells by binding to Frizzled (Fz). At the cell membrane Fz inteacts with Low Density Lypoproteinrelated protein (LDR) receptor to form a Fz/LDR complex. This complex transduces Wnt signal in the target cell. Fz/ WNT LDR 55
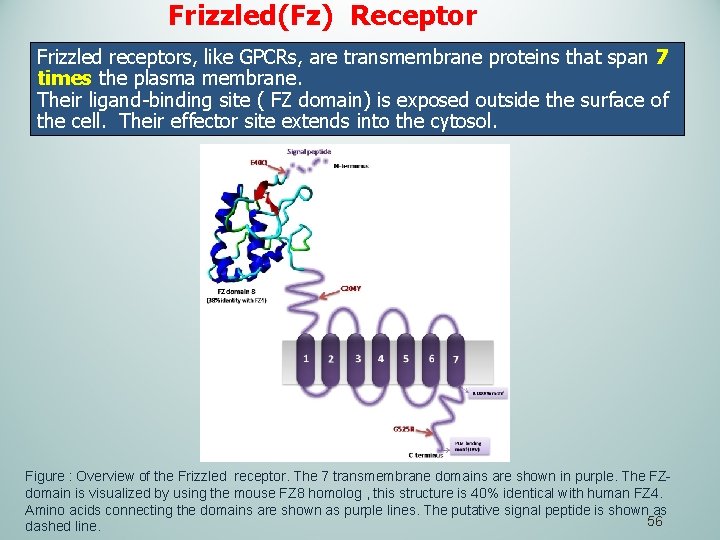
Frizzled(Fz) Receptor Frizzled receptors, like GPCRs, are transmembrane proteins that span 7 times the plasma membrane. Their ligand-binding site ( FZ domain) is exposed outside the surface of the cell. Their effector site extends into the cytosol. Figure : Overview of the Frizzled receptor. The 7 transmembrane domains are shown in purple. The FZdomain is visualized by using the mouse FZ 8 homolog , this structure is 40% identical with human FZ 4. Amino acids connecting the domains are shown as purple lines. The putative signal peptide is shown as 56 dashed line.

EXTRACELLULAR BINDING OF WNT Extracellular inhibitors: SFRP – Secreted Frizzled-related protein Resembles ligand-binding domain of Frizzled WIF – Wnt inhibitory factor Secreted molecules resembling extracellular portion of the receptor 57
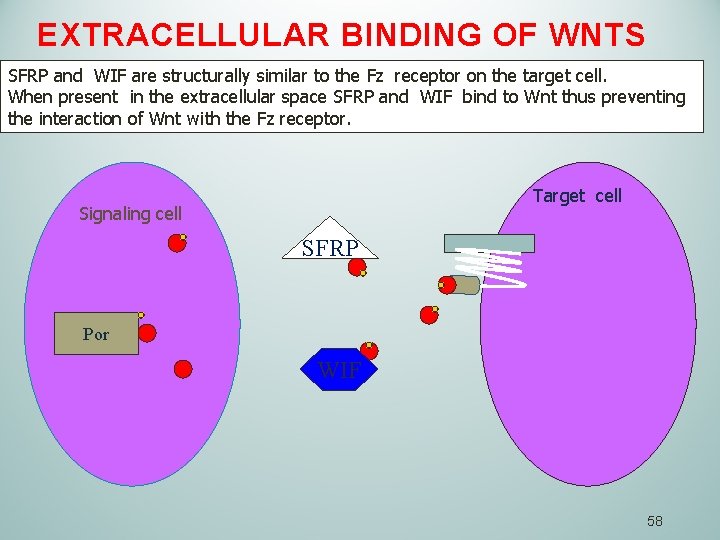
EXTRACELLULAR BINDING OF WNTS SFRP and WIF are structurally similar to the Fz receptor on the target cell. When present in the extracellular space SFRP and WIF bind to Wnt thus preventing the interaction of Wnt with the Fz receptor. Target cell Signaling cell SFRP Por WIF 58

Why WNTs form complexes with SFRP/ WIF? n n SFRP/WIF regulates WNT signaling Wnt signal induces the production of SFRP/WIF, that can activate feedback loop, thus turning the response off. 59

WNT SIGNAL TRANSDUCTION: 1. Canonical Signaling 2. Non-canonical WNT signaling 60
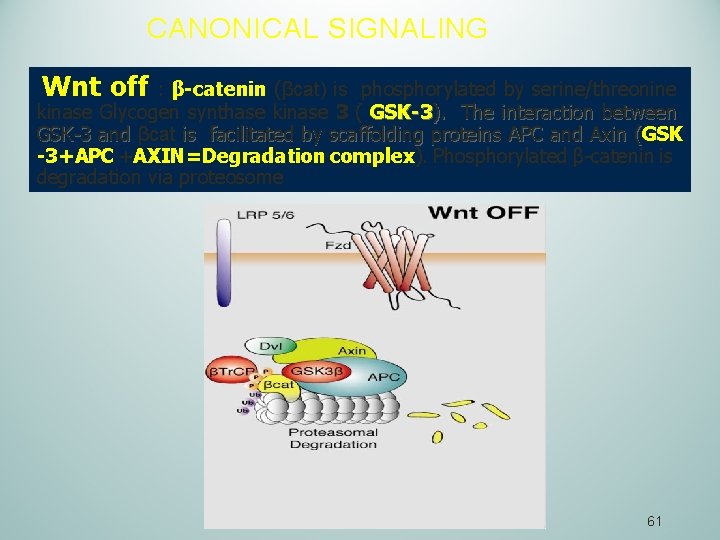
CANONICAL SIGNALING Wnt off : β-catenin (βcat) is phosphorylated by serine/threonine kinase Glycogen synthase kinase 3 ( GSK-3). The interaction between GSK-3 and βcat is facilitated by scaffolding proteins APC and Axin (GSK -3+APC +AXIN=Degradation complex). Phosphorylated β-catenin is degradation via proteosome 61
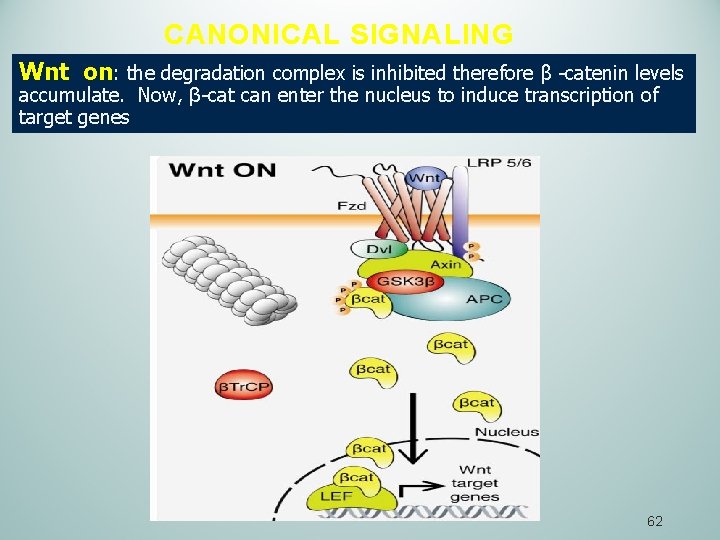
CANONICAL SIGNALING Wnt on: the degradation complex is inhibited therefore β -catenin levels accumulate. Now, β-cat can enter the nucleus to induce transcription of target genes 62

WHY Β-CATENIN ACCUMULATE IN PRESENCE OF WNT? Wnt signal enables β-catenin accumulation by three mechanisms: 63
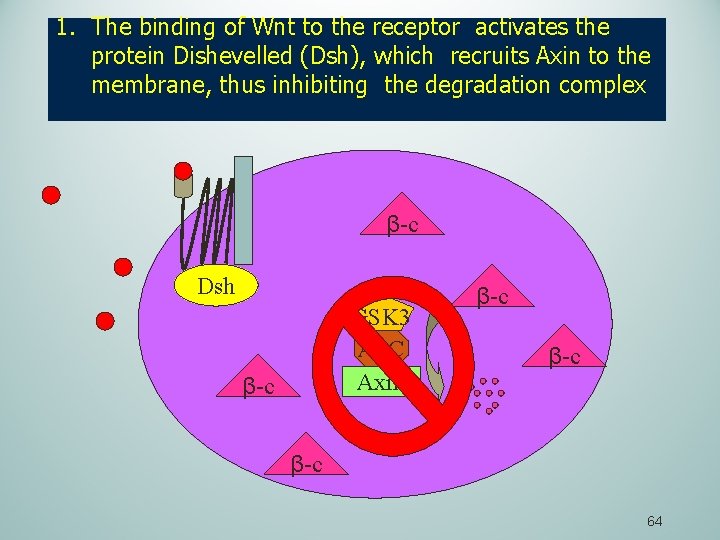
1. The binding of Wnt to the receptor activates the protein Dishevelled (Dsh), which recruits Axin to the membrane, thus inhibiting the degradation complex β-c Dsh GSK 3 APC Axin β-c β-c 64
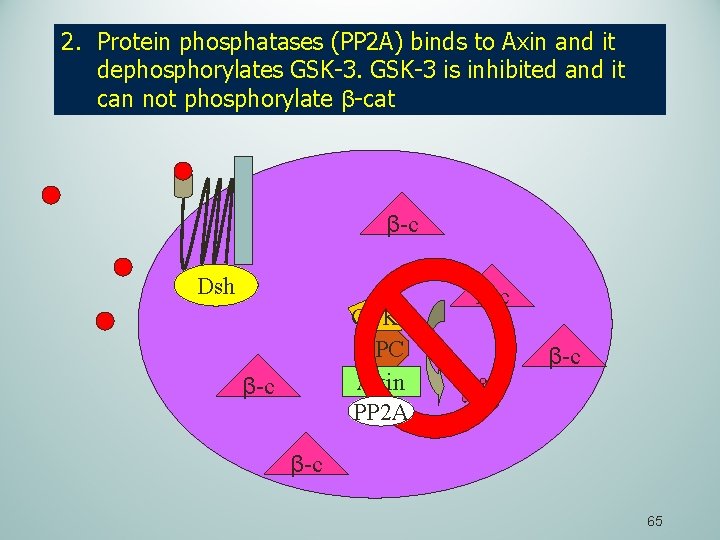
2. Protein phosphatases (PP 2 A) binds to Axin and it dephosphorylates GSK-3 is inhibited and it can not phosphorylate β-cat β-c Dsh GSK 3 APC Axin PP 2 A β-c Β-c β-c 65
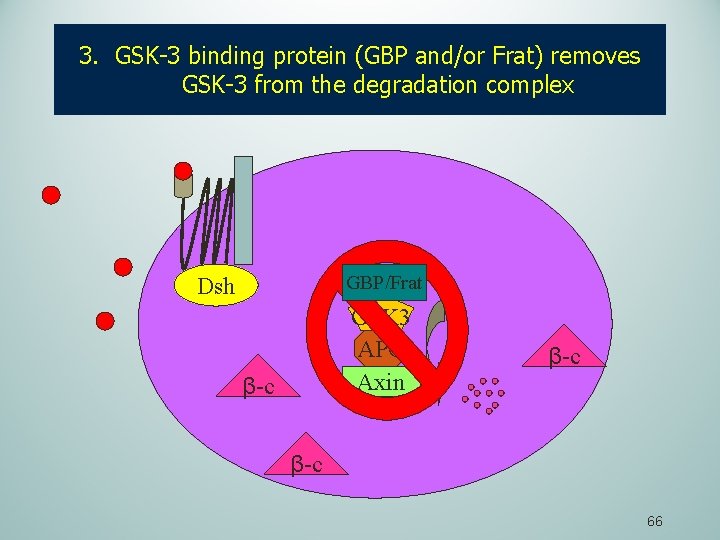
3. GSK-3 binding protein (GBP and/or Frat) removes GSK-3 from the degradation complex GBP/Frat Dsh GSK 3 APC Axin β-c β-c 66
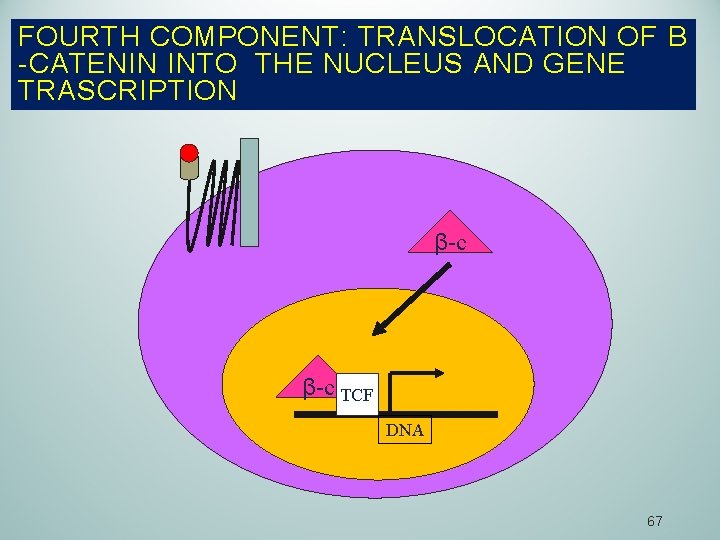
FOURTH COMPONENT: TRANSLOCATION OF Β -CATENIN INTO THE NUCLEUS AND GENE TRASCRIPTION β-c TCF DNA 67
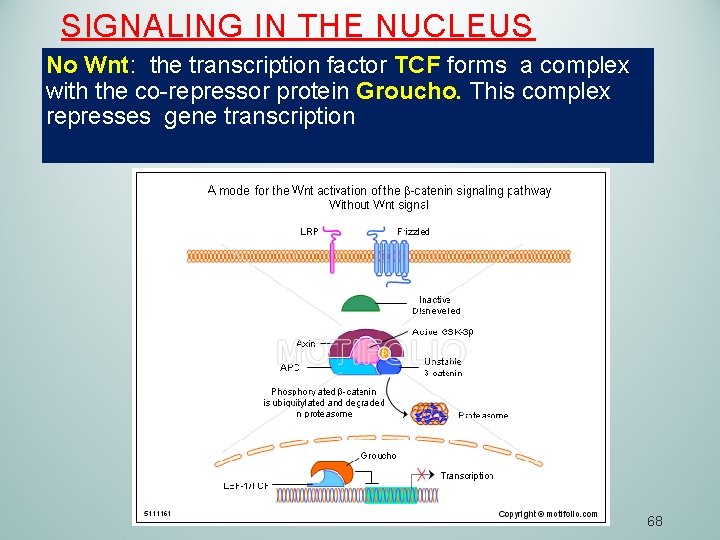
SIGNALING IN THE NUCLEUS No Wnt: the transcription factor TCF forms a complex with the co-repressor protein Groucho. This complex represses gene transcription 68
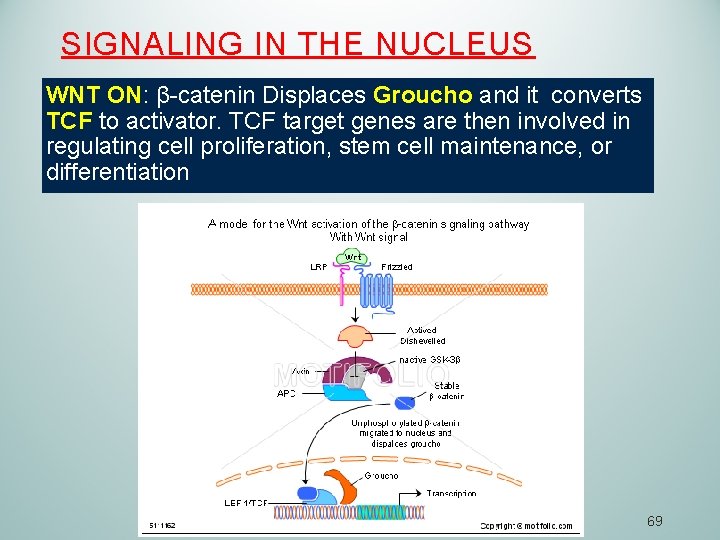
SIGNALING IN THE NUCLEUS WNT ON: β-catenin Displaces Groucho and it converts TCF to activator. TCF target genes are then involved in regulating cell proliferation, stem cell maintenance, or differentiation 69
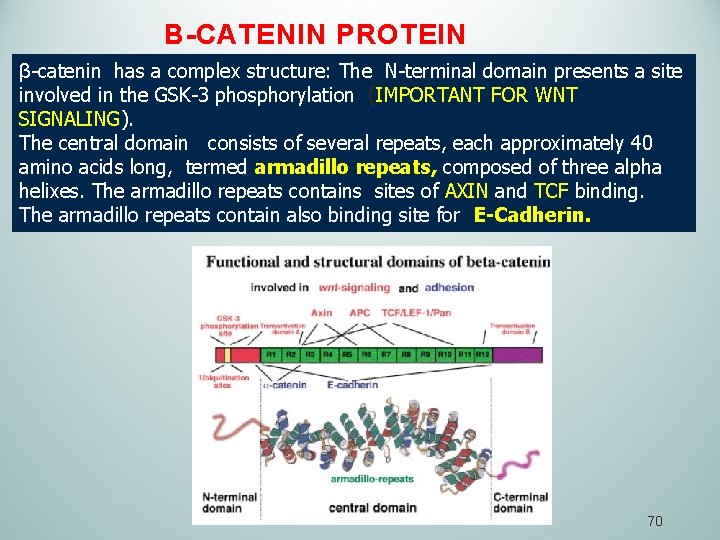
B-CATENIN PROTEIN β-catenin has a complex structure: The N-terminal domain presents a site involved in the GSK-3 phosphorylation (IMPORTANT FOR WNT SIGNALING). The central domain consists of several repeats, each approximately 40 amino acids long, termed armadillo repeats, composed of three alpha helixes. The armadillo repeats contains sites of AXIN and TCF binding. The armadillo repeats contain also binding site for E-Cadherin. 70
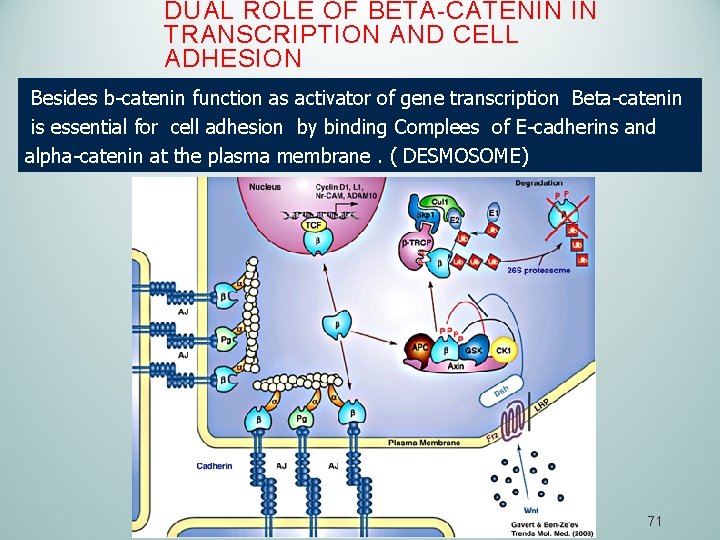
DUAL ROLE OF BETA-CATENIN IN TRANSCRIPTION AND CELL ADHESION Besides b-catenin function as activator of gene transcription Beta-catenin is essential for cell adhesion by binding Complees of E-cadherins and alpha-catenin at the plasma membrane. ( DESMOSOME) 71

WNT SIGNALING IN CANCER AND HUMAN DISEASE In adults, mis-regulation of the Wnt pathway leads to a variety of abnormalities and degenerative diseases 72

73
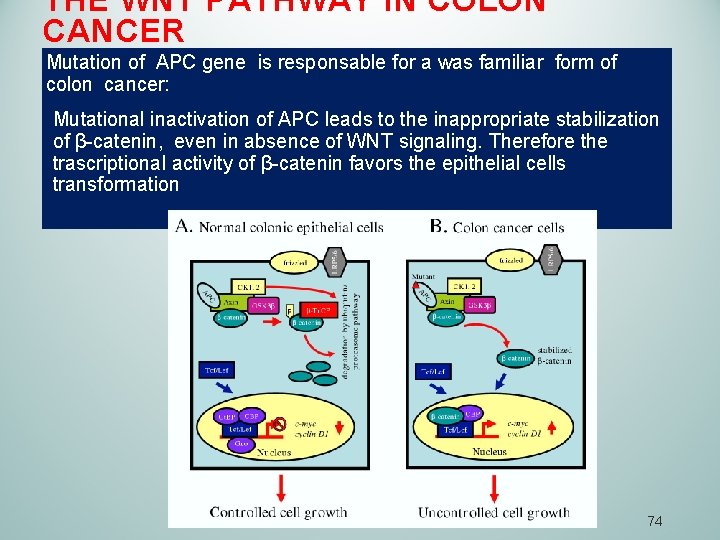
THE WNT PATHWAY İN COLON CANCER Mutation of APC gene is responsable for a was familiar form of colon cancer: Mutational inactivation of APC leads to the inappropriate stabilization of β-catenin, even in absence of WNT signaling. Therefore the trascriptional activity of β-catenin favors the epithelial cells transformation 74

WNT SIGNALING IN NORMAL AND CANCER CELL https: //www. youtube. com/watch? v=Yu. Vk. RTOYOl. Q 75
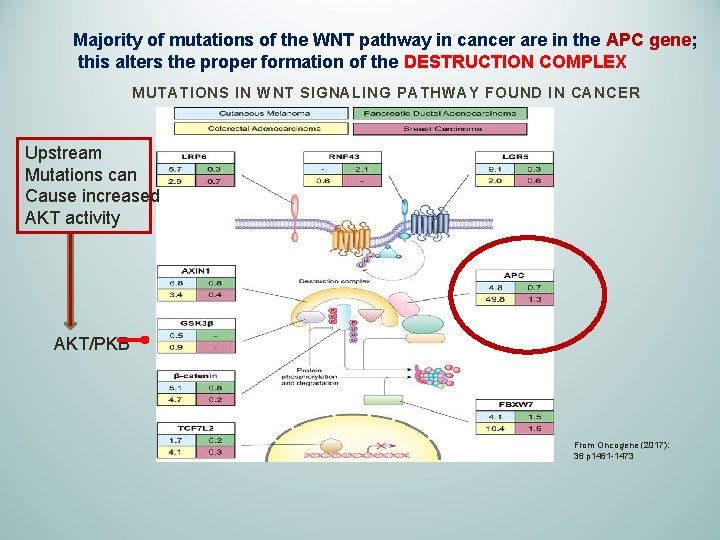
Majority of mutations of the WNT pathway in cancer are in the APC gene; this alters the proper formation of the DESTRUCTION COMPLEX MUTATIONS IN WNT SIGNALING PATHWAY FOUND IN CANCER Upstream Mutations can Cause increased AKT activity AKT/PKB From Oncogene (2017): 36 p 1461 -1473

“NON-CANONICAL” WNT SIGNALING 77
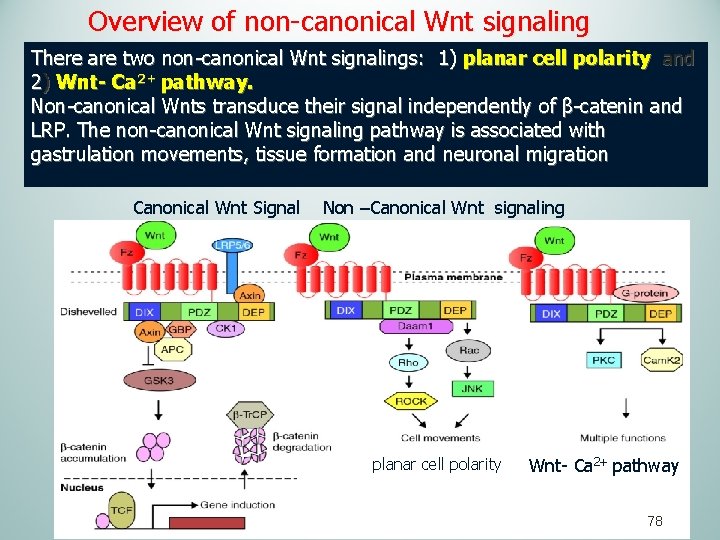
Overview of non-canonical Wnt signaling There are two non-canonical Wnt signalings: 1) planar cell polarity and 2) Wnt- Ca 2+ pathway. Non-canonical Wnts transduce their signal independently of β-catenin and LRP. The non-canonical Wnt signaling pathway is associated with gastrulation movements, tissue formation and neuronal migration Canonical Wnt Signal Non –Canonical Wnt signaling planar cell polarity Wnt- Ca 2+ pathway 78
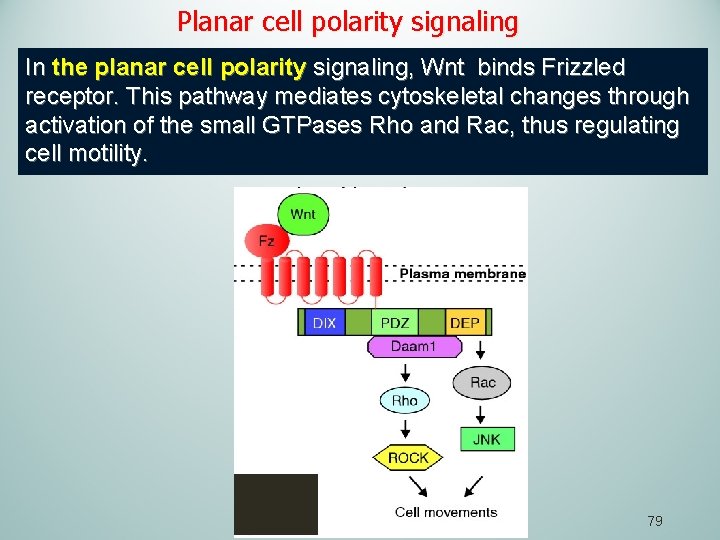
Planar cell polarity signaling In the planar cell polarity signaling, Wnt binds Frizzled receptor. This pathway mediates cytoskeletal changes through activation of the small GTPases Rho and Rac, thus regulating cell motility. 79
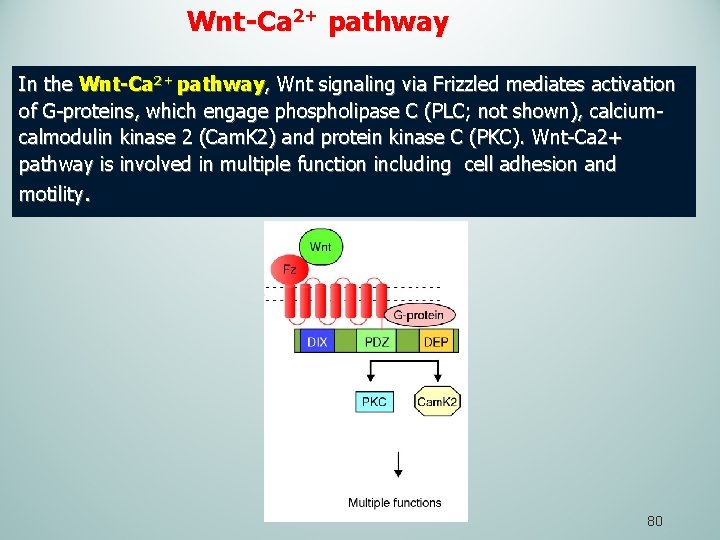
Wnt-Ca 2+ pathway In the Wnt-Ca 2+ pathway, Wnt signaling via Frizzled mediates activation of G-proteins, which engage phospholipase C (PLC; not shown), calciumcalmodulin kinase 2 (Cam. K 2) and protein kinase C (PKC). Wnt-Ca 2+ pathway is involved in multiple function including cell adhesion and motility. 80
- Slides: 80