CELL SIGNALING AND MOTILITY BIOL 3373 Lecture 4

CELL SIGNALING AND MOTILITY (BIOL 3373) Lecture 4 1
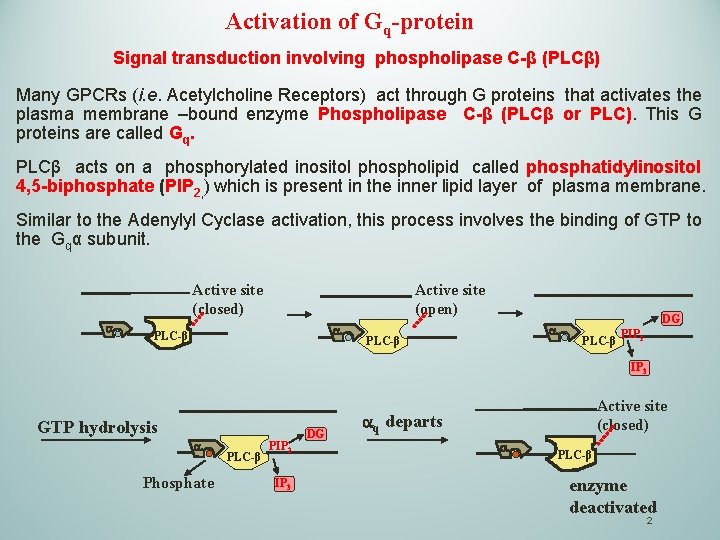
Activation of Gq-protein Signal transduction involving phospholipase C-β (PLCβ) Many GPCRs (i. e. Acetylcholine Receptors) act through G proteins that activates the plasma membrane –bound enzyme Phospholipase C-β (PLCβ or PLC). This G proteins are called Gq. PLCβ acts on a phosphorylated inositol phospholipid called phosphatidylinositol 4, 5 -biphosphate (PIP 2, ) which is present in the inner lipid layer of plasma membrane. Similar to the Adenylyl Cyclase activation, this process involves the binding of GTP to the Gqα subunit. Active site (open) Active site (closed) a a PLC-β DG PLC-β PIP 2 IP 3 GTP hydrolysis a Phosphate PLC-β PIP 2 IP 3 DG Active site (closed) aq departs a PLC-β enzyme deactivated 2

Activation of Gq-protein Signal transduction involving phospholipase C-β (PLCβ) The Phospholipase C-β (PLC) cleaves the phosphatidylinositol 4, 5 biphosphate (PIP 2, ) to generate the inositol 1, 4, 5 -triphospate (IP 3) and Diacylglycerol ( DAG or DG) 3
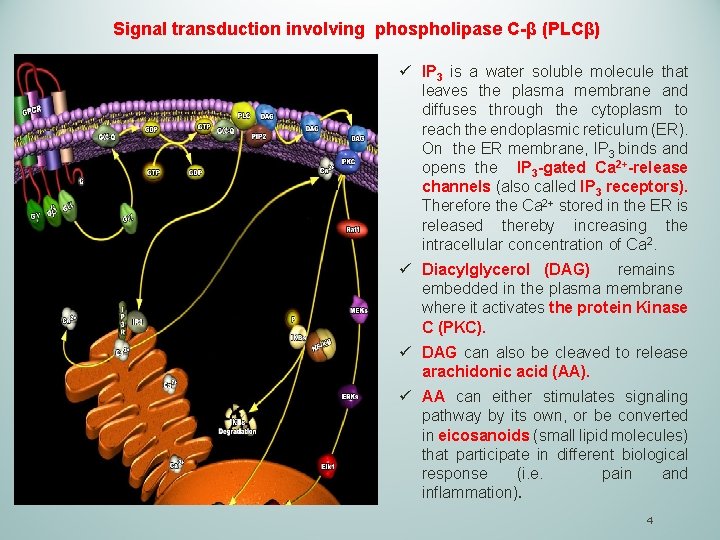
Signal transduction involving phospholipase C-β (PLCβ) ü IP 3 is a water soluble molecule that leaves the plasma membrane and diffuses through the cytoplasm to reach the endoplasmic reticulum (ER). On the ER membrane, IP 3 binds and opens the IP 3 -gated Ca 2+-release channels (also called IP 3 receptors). Therefore the Ca 2+ stored in the ER is released thereby increasing the intracellular concentration of Ca 2. ü Diacylglycerol (DAG) remains embedded in the plasma membrane where it activates the protein Kinase C (PKC). ü DAG can also be cleaved to release arachidonic acid (AA). ü AA can either stimulates signaling pathway by its own, or be converted in eicosanoids (small lipid molecules) that participate in different biological response (i. e. pain and inflammation). 4
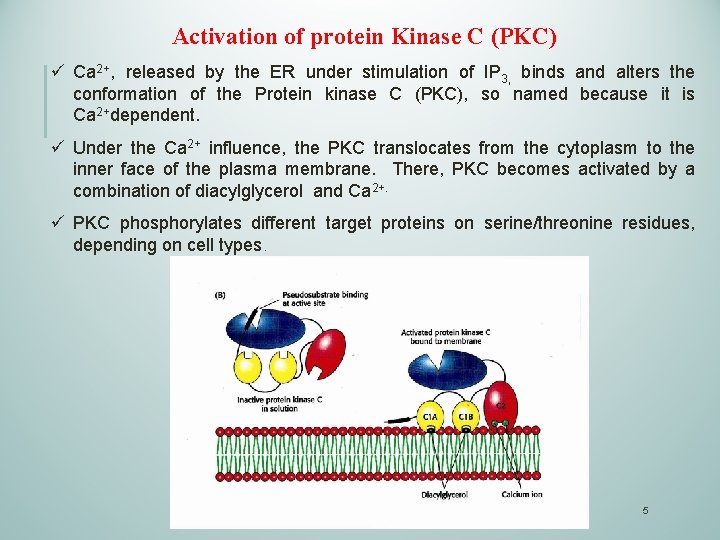
Activation of protein Kinase C (PKC) ü Ca 2+, released by the ER under stimulation of IP 3, binds and alters the conformation of the Protein kinase C (PKC), so named because it is Ca 2+dependent. ü Under the Ca 2+ influence, the PKC translocates from the cytoplasm to the inner face of the plasma membrane. There, PKC becomes activated by a combination of diacylglycerol and Ca 2+. ü PKC phosphorylates different target proteins on serine/threonine residues, depending on cell types. 5
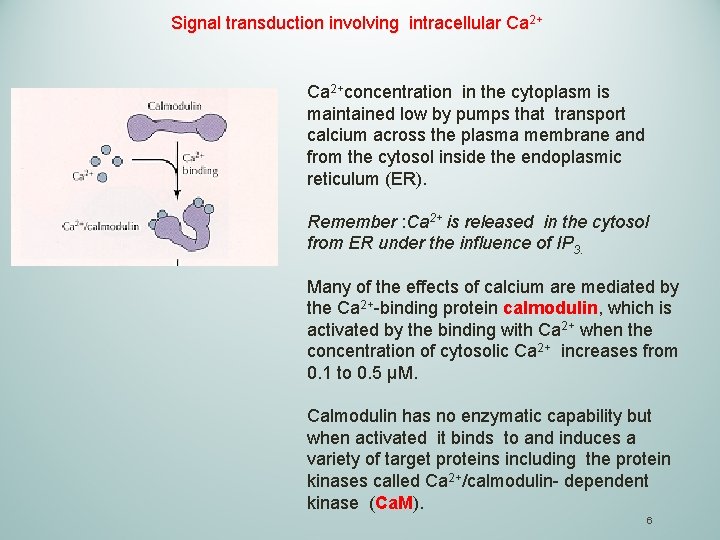
Signal transduction involving intracellular Ca 2+concentration in the cytoplasm is maintained low by pumps that transport calcium across the plasma membrane and from the cytosol inside the endoplasmic reticulum (ER). Remember : Ca 2+ is released in the cytosol from ER under the influence of IP 3. Many of the effects of calcium are mediated by the Ca 2+-binding protein calmodulin, which is activated by the binding with Ca 2+ when the concentration of cytosolic Ca 2+ increases from 0. 1 to 0. 5 μM. Calmodulin has no enzymatic capability but when activated it binds to and induces a variety of target proteins including the protein kinases called Ca 2+/calmodulin- dependent kinase (Ca. M). 6
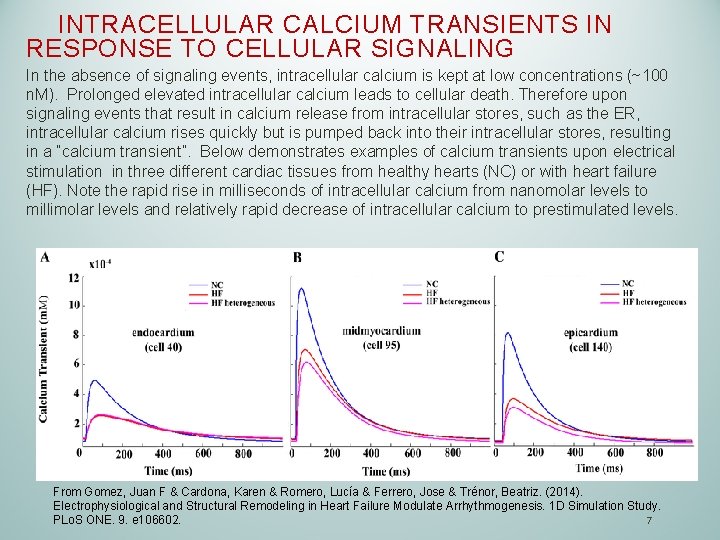
INTRACELLULAR CALCIUM TRANSIENTS IN RESPONSE TO CELLULAR SIGNALING In the absence of signaling events, intracellular calcium is kept at low concentrations (~100 n. M). Prolonged elevated intracellular calcium leads to cellular death. Therefore upon signaling events that result in calcium release from intracellular stores, such as the ER, intracellular calcium rises quickly but is pumped back into their intracellular stores, resulting in a “calcium transient”. Below demonstrates examples of calcium transients upon electrical stimulation in three different cardiac tissues from healthy hearts (NC) or with heart failure (HF). Note the rapid rise in milliseconds of intracellular calcium from nanomolar levels to millimolar levels and relatively rapid decrease of intracellular calcium to prestimulated levels. From Gomez, Juan F & Cardona, Karen & Romero, Lucía & Ferrero, Jose & Trénor, Beatriz. (2014). Electrophysiological and Structural Remodeling in Heart Failure Modulate Arrhythmogenesis. 1 D Simulation Study. 7 PLo. S ONE. 9. e 106602.
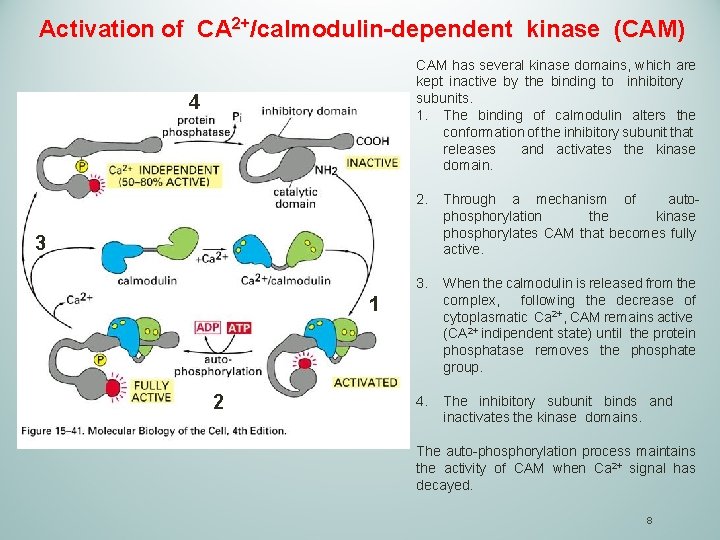
Activation of CA 2+/calmodulin-dependent kinase (CAM) CAM has several kinase domains, which are kept inactive by the binding to inhibitory subunits. 1. The binding of calmodulin alters the conformation of the inhibitory subunit that releases and activates the kinase domain. 4 2. Through a mechanism of autophosphorylation the kinase phosphorylates CAM that becomes fully active. 3. When the calmodulin is released from the complex, following the decrease of cytoplasmatic Ca 2+, CAM remains active (CA 2+ indipendent state) until the protein phosphatase removes the phosphate group. 4. The inhibitory subunit binds and inactivates the kinase domains. 3 1 2 The auto-phosphorylation process maintains the activity of CAM when Ca 2+ signal has decayed. 8

Functions of CA 2+/calmodulin-dependent kinase (CAM) Ca. M is a protein kinase involved in most of the important signaling pathways in the cell. Ca. M targets include: • cyclic nucleotide metabolism (c. AMP) • signal transduction pathways involving phosphorylation and dephosphorylation • calcium transport (carried out by the plasma membrane Ca 2+ pump) • nitric oxide pathway • regulation of cytoskeletal proteins Most of these systems are evolutionarily conserved, with counterparts in vertebrates as well as non-vertebrate species. The conservation of the principle Ca 2+ reactive systems through evolution bespeaks their importance in cell function. 9

SIGNAL TERMINATION • Signal Generation/Reception • Signal Transduction • Signal Termination Proper regulated cell signaling also requires that the original signal be terminated. Inability or improper signal termination results in over-stimulation of a receptorial system and can be seen pathologically as effects like tremor (neurotransmission as in tremor), unregulated proliferation (cancer), decreased drug effect (tolerance), diarrhea (cholera and pertussis toxin) 10

1. 1. 2. 3. MECHANISMS OF SIGNAL TERMINATION Removal of Signal/Stimulus Enzymatic degradation of neurotransmitter in synapatic cleft Reuptake of neurotransmitter in synaptic cleft Physical removal of signal: e. g. T cell, cell communication 2. Uncoupling/Degradation of Transducer 1. 2. G-protein receptor kinase/arrestins Ubiquitination/ Proteosomal degradation 3. Removal/Degradation of second messenger 1. 2. Phosphodiesterases: decrease c. AMP, c. GMP Sequestration of calcium 4. Inactivation of Receptorial System 1. 2. 3. 4. 5. Phosphatases: PTEN Protein phosphatases: Ubiquitination Transcription of repressors Removal of Receptor 1. Receptor endocytosis 2. Decreased transcription of receptor 11
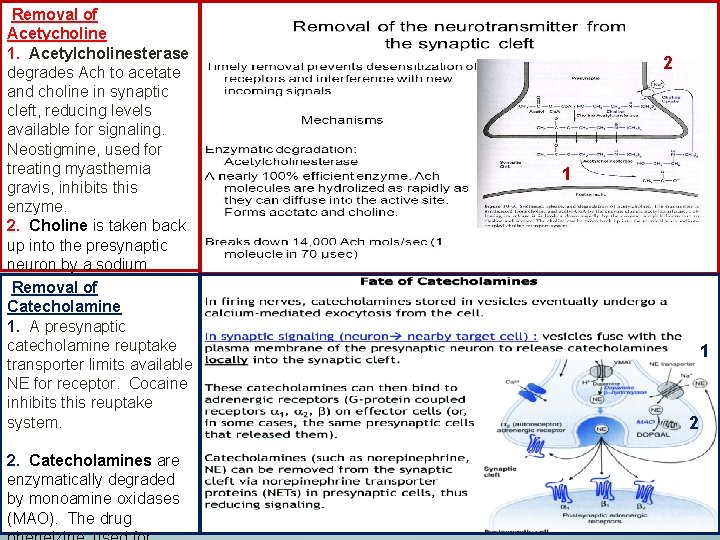
Removal of Acetycholine 1. Acetylcholinesterase degrades Ach to acetate and choline in synaptic cleft, reducing levels available for signaling. Neostigmine, used for treating myasthemia gravis, inhibits this enzyme. 2. Choline is taken back up into the presynaptic neuron by a sodium dependent transporter. Removal of Tetanus toxin can inhibit Catecholamine this transporter. 1. A presynaptic catecholamine reuptake transporter limits available NE for receptor. Cocaine inhibits this reuptake system. 2. Catecholamines are enzymatically degraded by monoamine oxidases (MAO). The drug 2 1 1 2 12
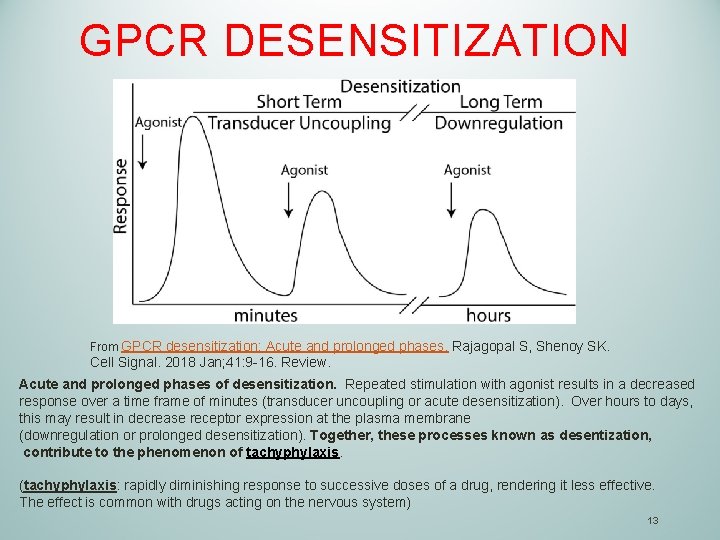
GPCR DESENSITIZATION From GPCR desensitization: Acute and prolonged phases. Rajagopal S, Shenoy SK. Cell Signal. 2018 Jan; 41: 9 -16. Review. Acute and prolonged phases of desensitization. Repeated stimulation with agonist results in a decreased response over a time frame of minutes (transducer uncoupling or acute desensitization). Over hours to days, this may result in decrease receptor expression at the plasma membrane (downregulation or prolonged desensitization). Together, these processes known as desentization, contribute to the phenomenon of tachyphylaxis. (tachyphylaxis: rapidly diminishing response to successive doses of a drug, rendering it less effective. The effect is common with drugs acting on the nervous system) 13
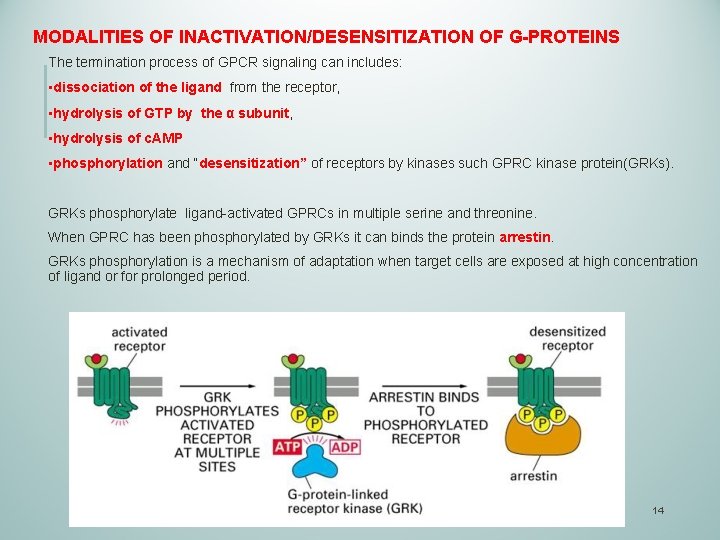
MODALITIES OF INACTIVATION/DESENSITIZATION OF G-PROTEINS The termination process of GPCR signaling can includes: • dissociation of the ligand from the receptor, • hydrolysis of GTP by the α subunit, • hydrolysis of c. AMP • phosphorylation and “desensitization” of receptors by kinases such GPRC kinase protein(GRKs). GRKs phosphorylate ligand-activated GPRCs in multiple serine and threonine. When GPRC has been phosphorylated by GRKs it can binds the protein arrestin. GRKs phosphorylation is a mechanism of adaptation when target cells are exposed at high concentration of ligand or for prolonged period. 14
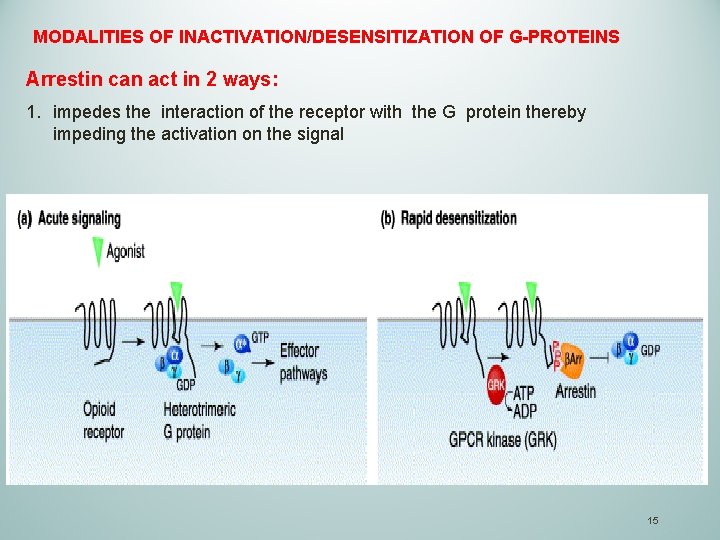
MODALITIES OF INACTIVATION/DESENSITIZATION OF G-PROTEINS Arrestin can act in 2 ways: 1. impedes the interaction of the receptor with the G protein thereby impeding the activation on the signal 15
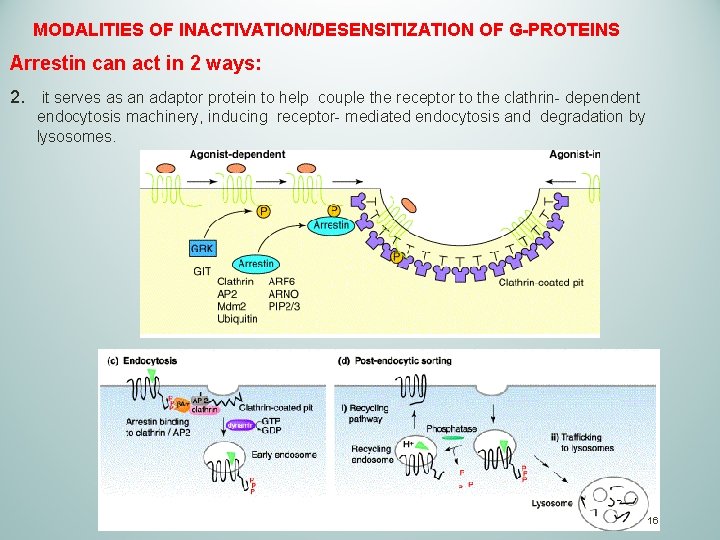
MODALITIES OF INACTIVATION/DESENSITIZATION OF G-PROTEINS Arrestin can act in 2 ways: 2. it serves as an adaptor protein to help couple the receptor to the clathrin- dependent endocytosis machinery, inducing receptor- mediated endocytosis and degradation by lysosomes. 16

MODALITIES OF INACTIVATION/DESENSITIZATION OF G-PROTEINS Robert Joseph Lefkowitz, M. D. • Won Nobel Prize in Chemistry for discovery, cloning and determination of structure/function of GPCRs ( AR), GPCR kinases ( ARK: GRK 2), -arrestin • Characterized receptor desensitization Please Double Click Below for Video https: //www. youtube. com/watch? v=OPYqp. YZ 7 x. L 4 17
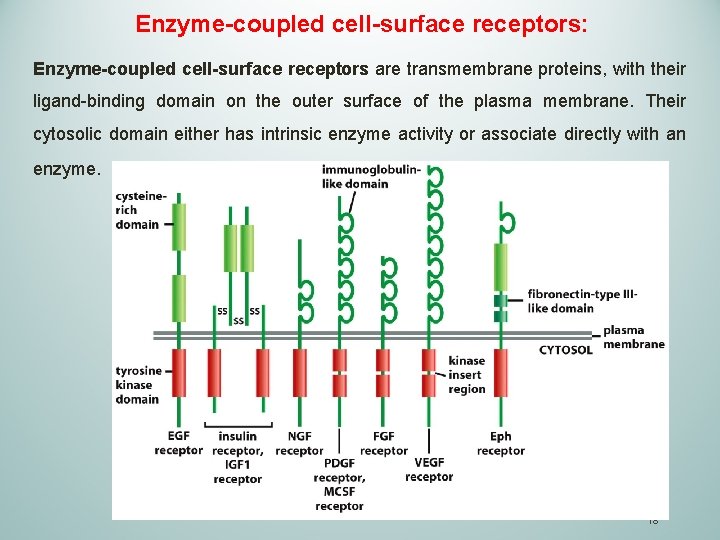
Enzyme-coupled cell-surface receptors: Enzyme-coupled cell-surface receptors are transmembrane proteins, with their ligand-binding domain on the outer surface of the plasma membrane. Their cytosolic domain either has intrinsic enzyme activity or associate directly with an enzyme. 18

Enzyme-coupled cell-surface receptors 1. Receptor tyrosine kinases – phosphorylate specific tyrosines on specific intracellular signaling proteins. (EGFR) 1. Tyrosine-kinase-associated receptors – these enzymes associate with intracellular proteins that have tyrosine kinase activity. (Cytokine/Jak. Stat) 1. Receptor-like tyrosine phosphatases – remove phosphate groups from tyrosines of specific intracellular proteins. 2. Receptor serine/threonine kinases – phosphorylate specific serines or threonines on associated gene regulatory proteins. (TGF-b/Smad) 3. Receptor guanylyl cyclases – directly catalyze the production of cyclic GMP in the cytosol. (Natriuretic peptides receptor) 4. Histidine-kinase-associated receptors- activate a 2 -component signaling pathway whereby the kinase phosphorylates itself on histidine (autophosphorylation) and then immediately transfers the phosphate to a second intracellular protein. Only occurs in yeasts and 19 plants involved in chemotaxis.

Receptor tyrosine kinases (RTKs) are transmembrane proteins 3 major features: § extracellular domain (ectodomain) § transmembrane region § intracellular domain with tyrosine kinase activity 20

Receptor tyrosine kinases (RTKs) 21
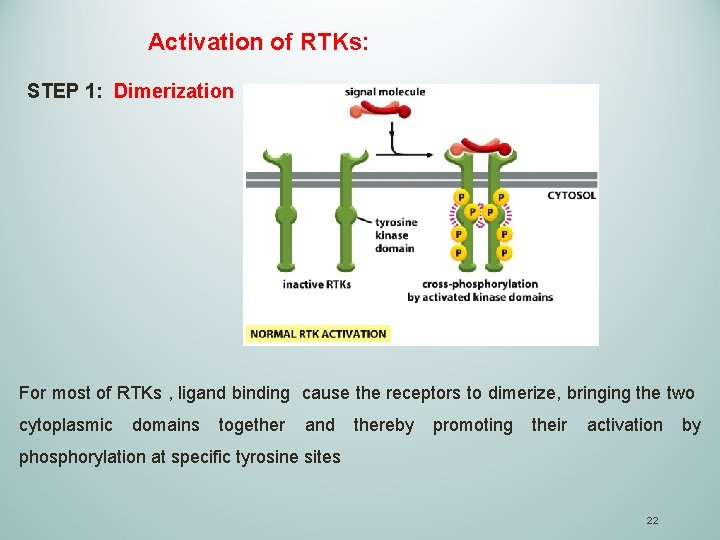
Activation of RTKs: STEP 1: Dimerization For most of RTKs , ligand binding cause the receptors to dimerize, bringing the two cytoplasmic domains together and thereby promoting their activation by phosphorylation at specific tyrosine sites 22
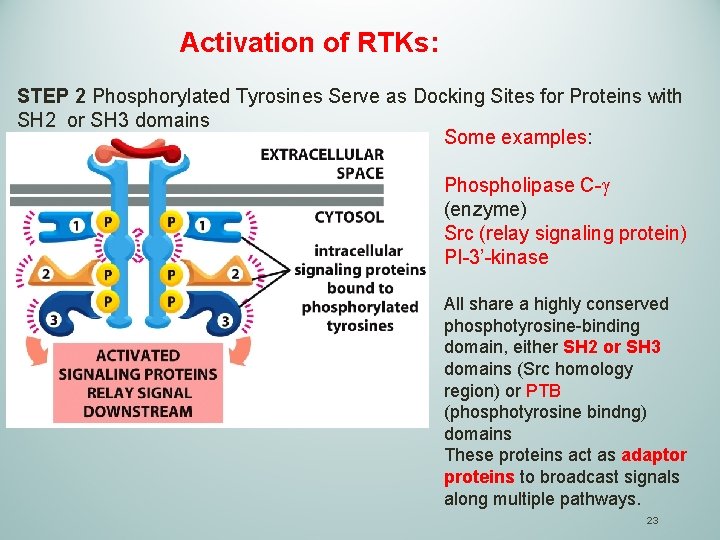
Activation of RTKs: STEP 2 Phosphorylated Tyrosines Serve as Docking Sites for Proteins with SH 2 or SH 3 domains Some examples: Phospholipase C-g (enzyme) Src (relay signaling protein) PI-3’-kinase All share a highly conserved phosphotyrosine-binding domain, either SH 2 or SH 3 domains (Src homology region) or PTB (phosphotyrosine bindng) domains These proteins act as adaptor proteins to broadcast signals along multiple pathways. 23

Adaptor Proteins : How They Function? 24
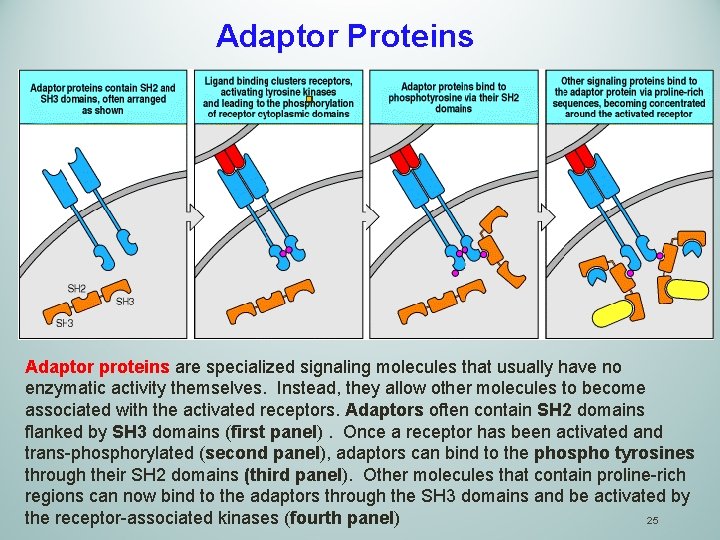
Adaptor Proteins Adaptor proteins are specialized signaling molecules that usually have no enzymatic activity themselves. Instead, they allow other molecules to become associated with the activated receptors. Adaptors often contain SH 2 domains flanked by SH 3 domains (first panel). Once a receptor has been activated and trans-phosphorylated (second panel), adaptors can bind to the phospho tyrosines through their SH 2 domains (third panel). Other molecules that contain proline-rich regions can now bind to the adaptors through the SH 3 domains and be activated by the receptor-associated kinases (fourth panel) 25
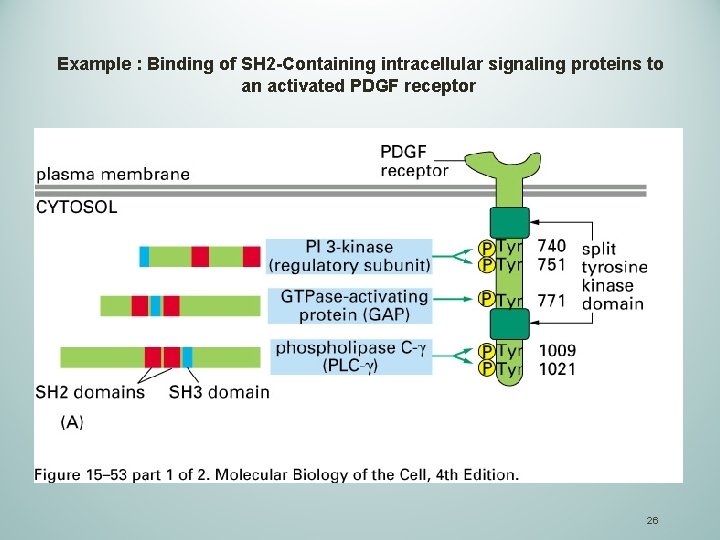
Example : Binding of SH 2 -Containing intracellular signaling proteins to an activated PDGF receptor 26
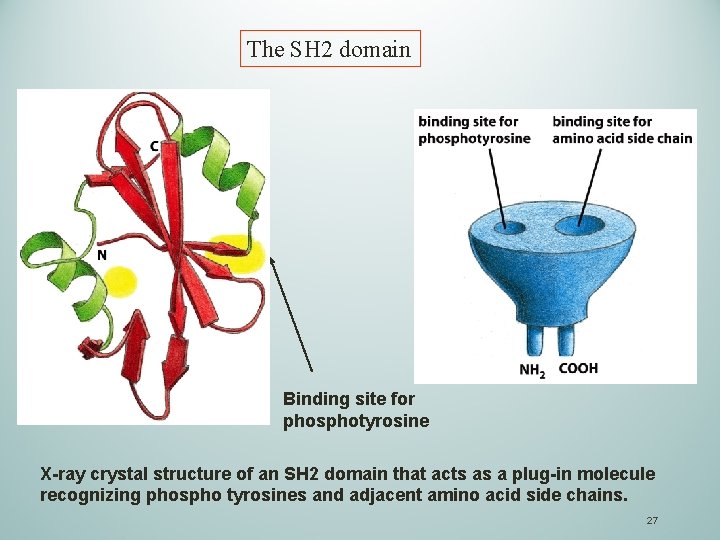
The SH 2 domain Binding site for phosphotyrosine X-ray crystal structure of an SH 2 domain that acts as a plug-in molecule recognizing phospho tyrosines and adjacent amino acid side chains. 27
Please Double Click Below for Video https: //www. youtube. com/watch? v=Ia 6 Ojv. Baz. GE 28
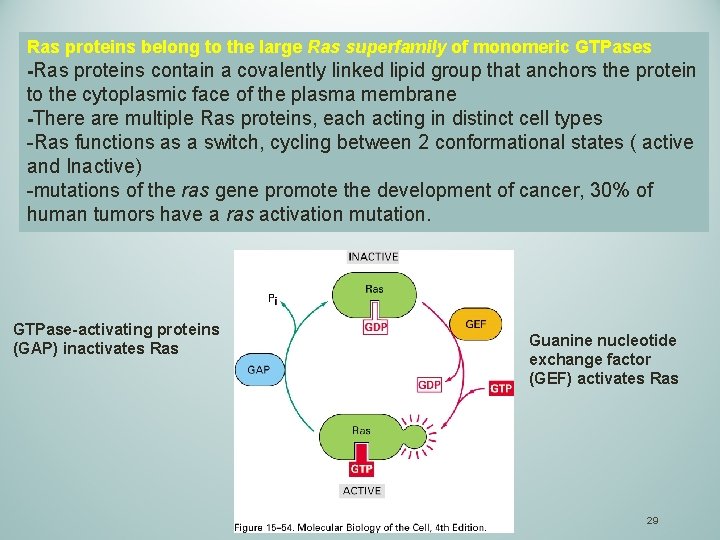
Ras proteins belong to the large Ras superfamily of monomeric GTPases -Ras proteins contain a covalently linked lipid group that anchors the protein to the cytoplasmic face of the plasma membrane -There are multiple Ras proteins, each acting in distinct cell types -Ras functions as a switch, cycling between 2 conformational states ( active and Inactive) -mutations of the ras gene promote the development of cancer, 30% of human tumors have a ras activation mutation. GTPase-activating proteins (GAP) inactivates Ras Guanine nucleotide exchange factor (GEF) activates Ras 29
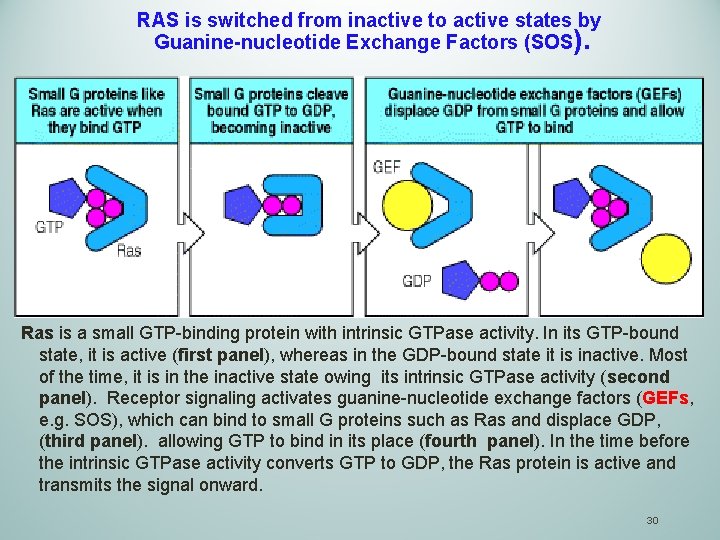
RAS is switched from inactive to active states by Guanine-nucleotide Exchange Factors (SOS). Ras is a small GTP-binding protein with intrinsic GTPase activity. In its GTP-bound state, it is active (first panel), whereas in the GDP-bound state it is inactive. Most of the time, it is in the inactive state owing its intrinsic GTPase activity (second panel). Receptor signaling activates guanine-nucleotide exchange factors (GEFs, e. g. SOS), which can bind to small G proteins such as Ras and displace GDP, (third panel). allowing GTP to bind in its place (fourth panel). In the time before the intrinsic GTPase activity converts GTP to GDP, the Ras protein is active and transmits the signal onward. 30
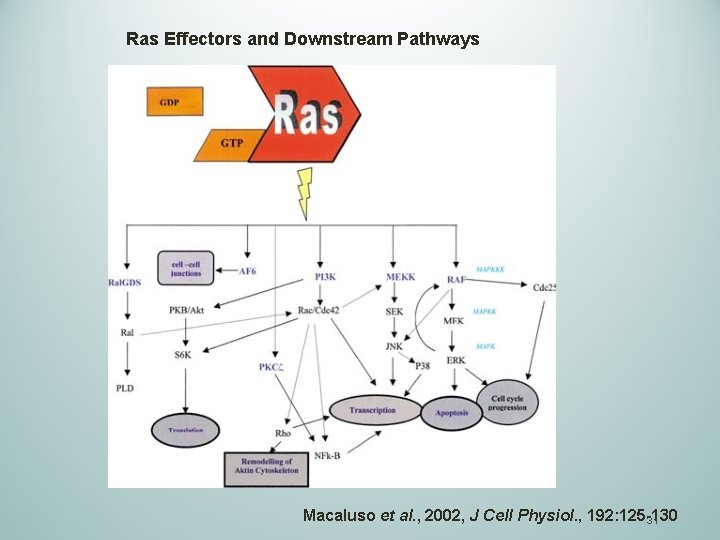
Ras Effectors and Downstream Pathways Macaluso et al. , 2002, J Cell Physiol. , 192: 125 -130 31

Activation of RTKs: STEP 3 ACTIVATION of RAS Grb-2 is an adaptor protein linking The receptor tyrosine kinase to Ras via the GEF called Sos. This is only one way of activating Ras. Adaptor protein (GRB 2) RAS-GEF (SOS) GRB 2 recognizes phosphorylated tyrosine on the activated receptor and recruit SOS stimulates the inactive RAS to replace its bound GDP by GTP, which activates RAS to relay the signal downstream. Activation of ras is short-lived because it is inactivated by phosphatases and GAPS. Once activated, Ras activates other signaling proteins to relay the signal downstream along several pathways. 32
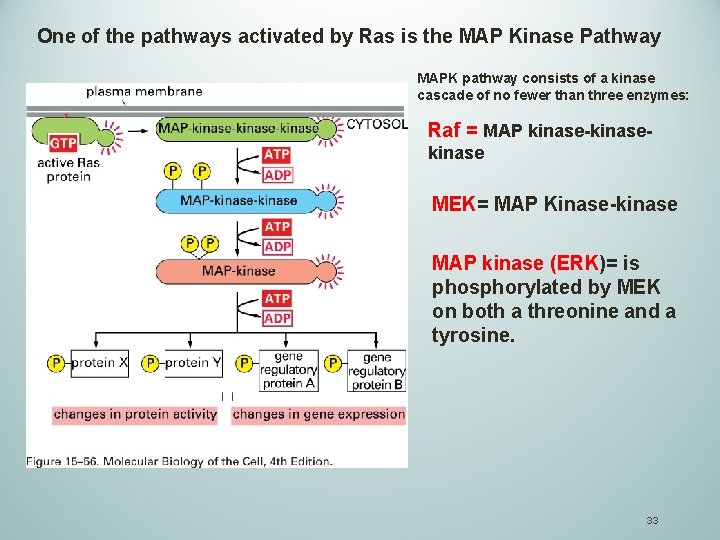
One of the pathways activated by Ras is the MAP Kinase Pathway MAPK pathway consists of a kinase cascade of no fewer than three enzymes: Raf = MAP kinase-kinase MEK= MAP Kinase-kinase MAP kinase (ERK)= is phosphorylated by MEK on both a threonine and a tyrosine. 33
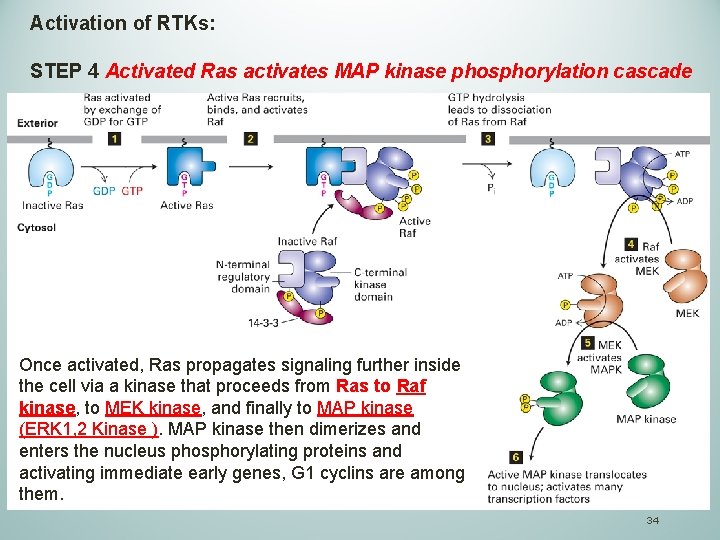
Activation of RTKs: STEP 4 Activated Ras activates MAP kinase phosphorylation cascade Once activated, Ras propagates signaling further inside the cell via a kinase that proceeds from Ras to Raf kinase, to MEK kinase, and finally to MAP kinase (ERK 1, 2 Kinase ). MAP kinase then dimerizes and enters the nucleus phosphorylating proteins and activating immediate early genes, G 1 cyclins are among them. 34
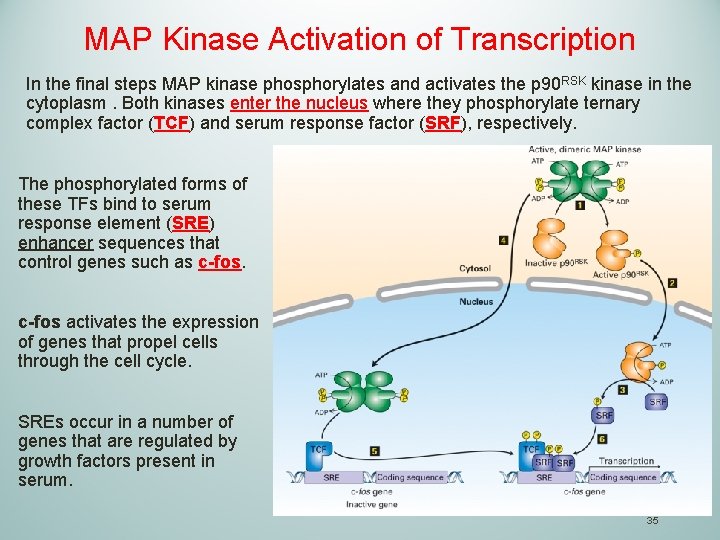
MAP Kinase Activation of Transcription In the final steps MAP kinase phosphorylates and activates the p 90 RSK kinase in the cytoplasm. Both kinases enter the nucleus where they phosphorylate ternary complex factor (TCF) and serum response factor (SRF), respectively. The phosphorylated forms of these TFs bind to serum response element (SRE) enhancer sequences that control genes such as c-fos activates the expression of genes that propel cells through the cell cycle. SREs occur in a number of genes that are regulated by growth factors present in serum. 35
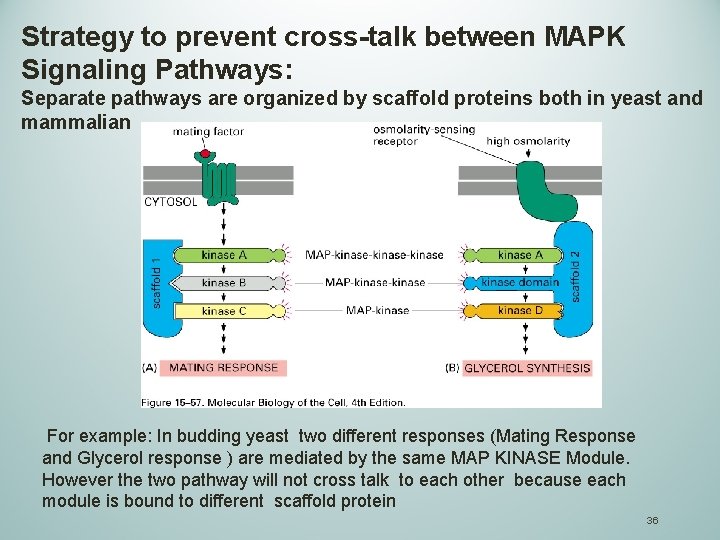
Strategy to prevent cross-talk between MAPK Signaling Pathways: Separate pathways are organized by scaffold proteins both in yeast and mammalian For example: In budding yeast two different responses (Mating Response and Glycerol response ) are mediated by the same MAP KINASE Module. However the two pathway will not cross talk to each other because each module is bound to different scaffold protein 36

RAS MAPK Signaling Pathway Please Double Click Below for Video https: //www. youtube. com/watch? v=r 7 Go. Z 9 v. FCY 8 37

Phosphatidylinostitol 3 -kinase (PI 3 K) Several growth factors and signaling compounds, including fibroblast growth factor (FGF), vascular endothelial cell growth factor (VEGF), human growth factor (HGF), angiopoietin I (Ang 1), and insulin activates a RTK, that recruits and activate the Phosphatidylinostitol 3 -kinase (PI 3 K), instead of SOS-RAS signaling PI 3 Ks are heterodimers composed of a regulatory and a catalytic subunit: • The regulatory subunit contains SH 2 and SH 3 domains and is referred to as p 85. • Catalytic subunits are referred to as p 110. Activation of PI 3 K places it close to its substrates at the inner leaflet of the plasma membrane. Several growth factors can initiate the process of activating PI 3 K. All of these molecules activate receptor tyrosine kinases (RTK), leading to autophosphorylation. The phosphorylated residue on the receptor becomes a docking site for the p 85 subunit of the heterodimeric PI 3 K. In some cases, however, receptor phosphorylation mediates the recruitment of an adaptor protein. 38
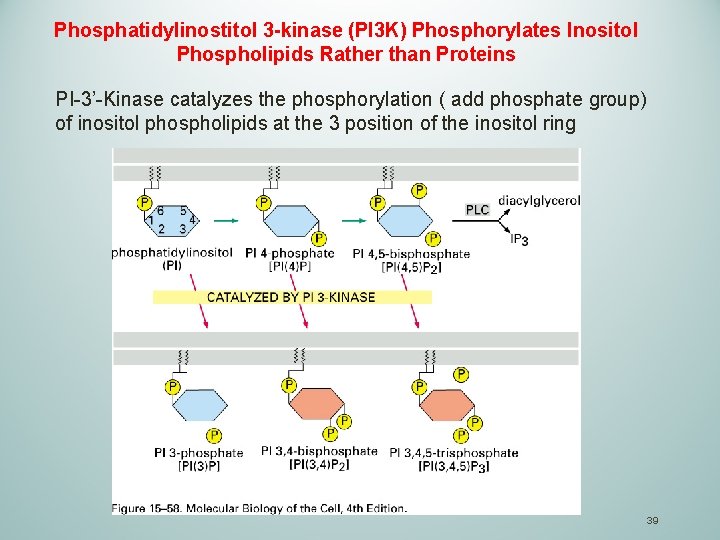
Phosphatidylinostitol 3 -kinase (PI 3 K) Phosphorylates Inositol Phospholipids Rather than Proteins PI-3’-Kinase catalyzes the phosphorylation ( add phosphate group) of inositol phospholipids at the 3 position of the inositol ring 39
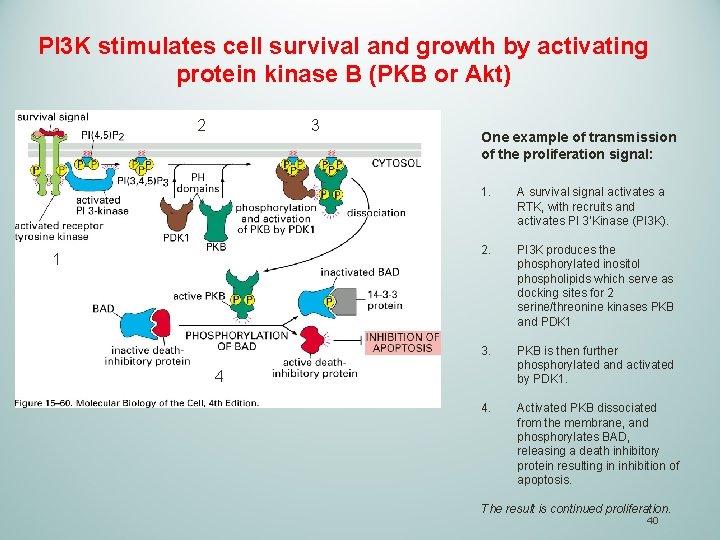
PI 3 K stimulates cell survival and growth by activating protein kinase B (PKB or Akt) 2 3 1 One example of transmission of the proliferation signal: 1. A survival signal activates a RTK, with recruits and activates PI 3’Kinase (PI 3 K). 2. PI 3 K produces the phosphorylated inositol phospholipids which serve as docking sites for 2 serine/threonine kinases PKB and PDK 1 3. PKB is then further phosphorylated and activated by PDK 1. 4. Activated PKB dissociated from the membrane, and phosphorylates BAD, releasing a death inhibitory protein resulting in inhibition of apoptosis. 4 The result is continued proliferation. 40
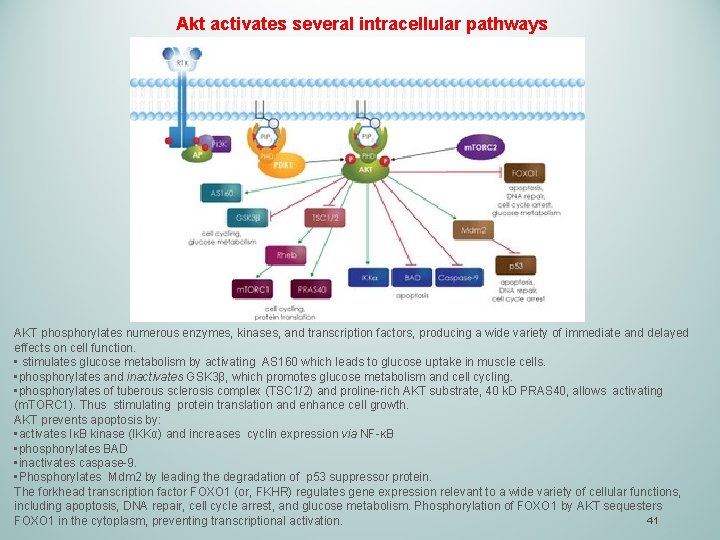
Akt activates several intracellular pathways AKT phosphorylates numerous enzymes, kinases, and transcription factors, producing a wide variety of immediate and delayed effects on cell function. • stimulates glucose metabolism by activating AS 160 which leads to glucose uptake in muscle cells. • phosphorylates and inactivates GSK 3β, which promotes glucose metabolism and cell cycling. • phosphorylates of tuberous sclerosis complex (TSC 1/2) and proline-rich AKT substrate, 40 k. D PRAS 40, allows activating (m. TORC 1). Thus stimulating protein translation and enhance cell growth. AKT prevents apoptosis by: • activates IκB kinase (IKKα) and increases cyclin expression via NF-κB • phosphorylates BAD • inactivates caspase-9. • Phosphorylates Mdm 2 by leading the degradation of p 53 suppressor protein. The forkhead transcription factor FOXO 1 (or, FKHR) regulates gene expression relevant to a wide variety of cellular functions, including apoptosis, DNA repair, cell cycle arrest, and glucose metabolism. Phosphorylation of FOXO 1 by AKT sequesters 41 FOXO 1 in the cytoplasm, preventing transcriptional activation.

PI 3 K Activation and Signaling Events Please Double Click Below for Video https: //www. youtube. com/watch? v=60_L_zo. FLa. U 42
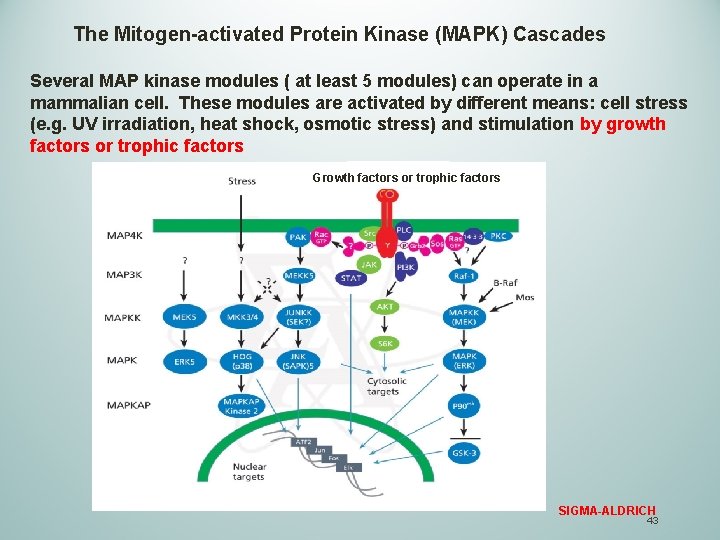
The Mitogen-activated Protein Kinase (MAPK) Cascades Several MAP kinase modules ( at least 5 modules) can operate in a mammalian cell. These modules are activated by different means: cell stress (e. g. UV irradiation, heat shock, osmotic stress) and stimulation by growth factors or trophic factors Growth factors or trophic factors SIGMA-ALDRICH 43

Growth Factors and Trophic factors o small peptides, or proteins o interact specifically with a receptor on the cell surface o secreted by multiple cell types o most are secreted as inactive precursors that need to be cleaved or fragmented before they can act o some are plasma membrane-bound or found in the extracellular matrix o transported by blood and lymphatic systems by several different routes (Endocrine, Paracrine, Autocrine) 44

Growth Factors and Trophic factors Growth factors include substances that stimulate cells to divide (hyperplasia) or increase in size (hypertrophy). Trophic factors include those substances that have effects on: • cell differentiation, • cell survival, • expression of a specific cellular phenotype (e. g. a cell becomes an inhibitory or an excitatory neuron), • cellular morphological plasticity, for example, the induction of neurite extension Importantly, some growth factors may also act as trophic factors and vice versa each growth/trophic factor may have a specific combination of cellular effects. 45

Growth Factors o Epidermal growth factor (EGF) o insulin-like growth factor (IGF) oplatelet-derived growth factor (PDGF) ohepatocyte growth factor (HGF) ofibroblast growth factor (FGF) o vascular endothelial growth factor (VEGF) o Tumor necrosis Factors (TNF) o Trasformin growth factor (TGF) 46

GROWTH FACTOR FUNCTIONS IN VITRO 1. Proliferation 2. Differentiation 3. Chemo-attraction 4. Cell death 5. Cell migration . Aaronson, Growth factor and receptor tyrosine kinases. Sci. STKE 2005, tr 6 (2005). 47
48

GROWTH FACTOR FUNCTIONS IN VIVO 1. Early development 2. Tissue differentiation 3. Wound healing and tissue repair 4. Immune responses 5. Stromal mediators of sex and other hormones . Aaronson, Growth factor and receptor tyrosine kinases. Sci. STKE 2005, tr 6 (2005). 49

50

EGF binds to EGFR (tyrosine Kinase receptors) and activates several pathways depending of target cells EGF 51

Neurotrophic factors are endogenous soluble proteins regulating survival, growth, morphological and synaptic plasticity, or synthesis of proteins for differentiated functions of neurons. • NGF: Nerve growth factor • BDNF: Brain –derived Neurotrophic factor • NT-3 and NT-4/5: Neurotrophins -3 and 4/5 52
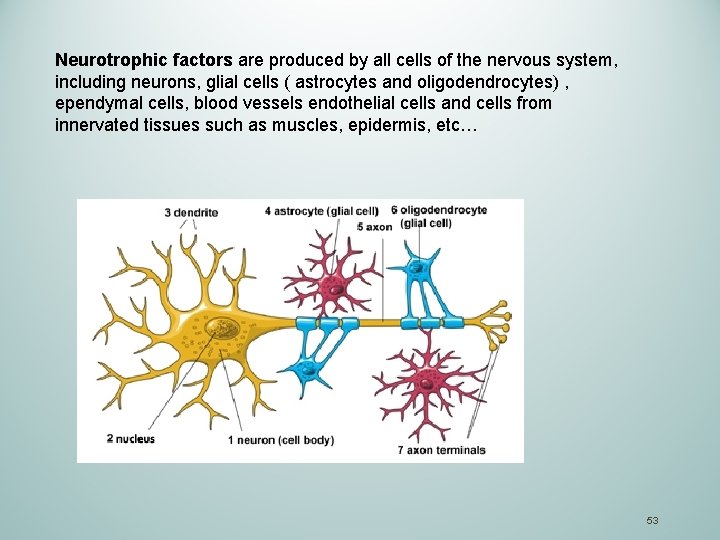
Neurotrophic factors are produced by all cells of the nervous system, including neurons, glial cells ( astrocytes and oligodendrocytes) , ependymal cells, blood vessels endothelial cells and cells from innervated tissues such as muscles, epidermis, etc… 53

Neurotrophic factors play essential roles in the developing and mature nervous system. Neurotrophic factors influence q cell proliferation, q survival, q differentiation, q migration, q axon and dendrite growth, q synaptic plasticity and q the interactions of neuronal and glial cells. 54
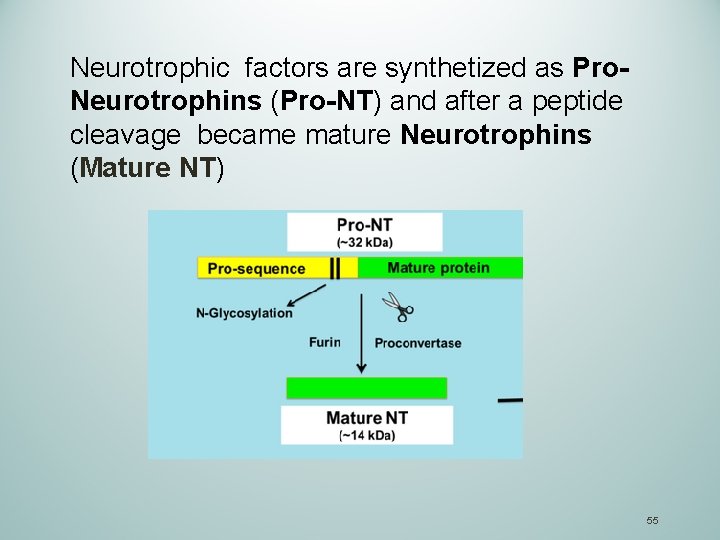
Neurotrophic factors are synthetized as Pro. Neurotrophins (Pro-NT) and after a peptide cleavage became mature Neurotrophins (Mature NT) 55
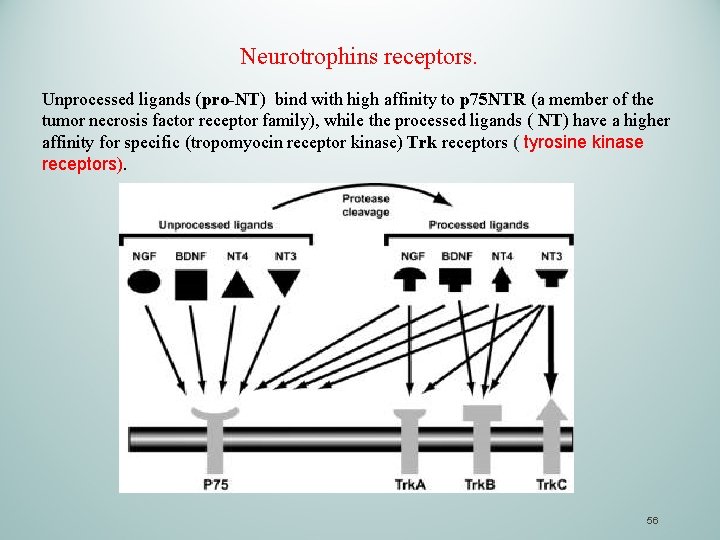
Neurotrophins receptors. Unprocessed ligands (pro-NT) bind with high affinity to p 75 NTR (a member of the tumor necrosis factor receptor family), while the processed ligands ( NT) have a higher affinity for specific (tropomyocin receptor kinase) Trk receptors ( tyrosine kinase receptors). 56
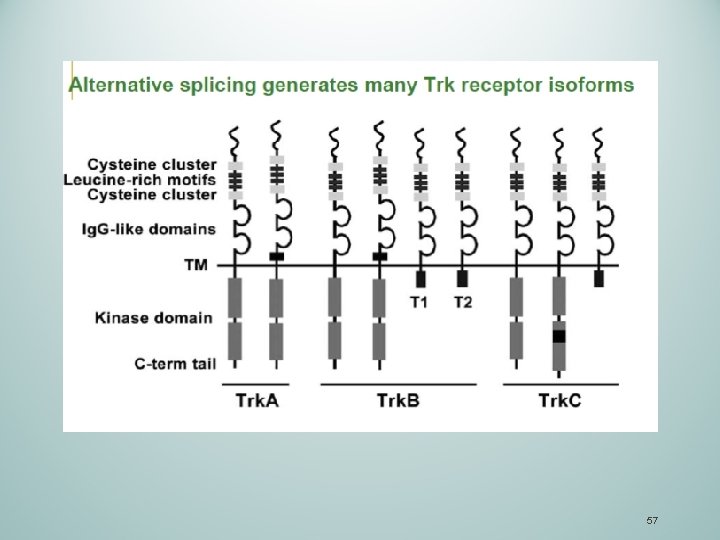
57
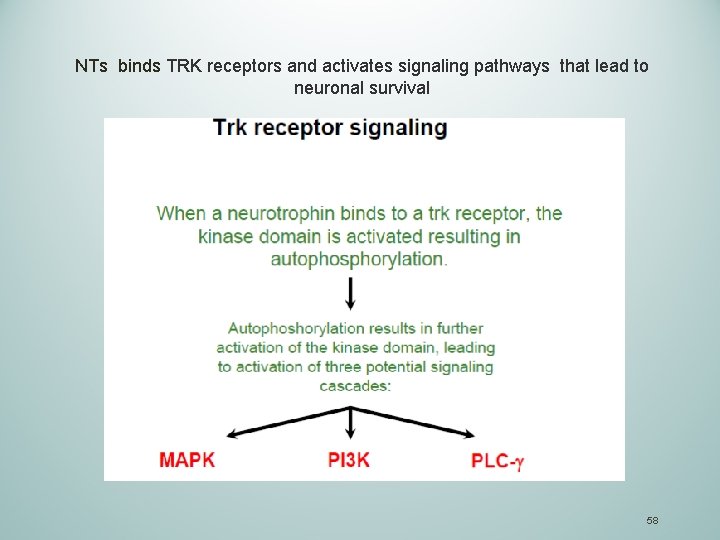
NTs binds TRK receptors and activates signaling pathways that lead to neuronal survival 58
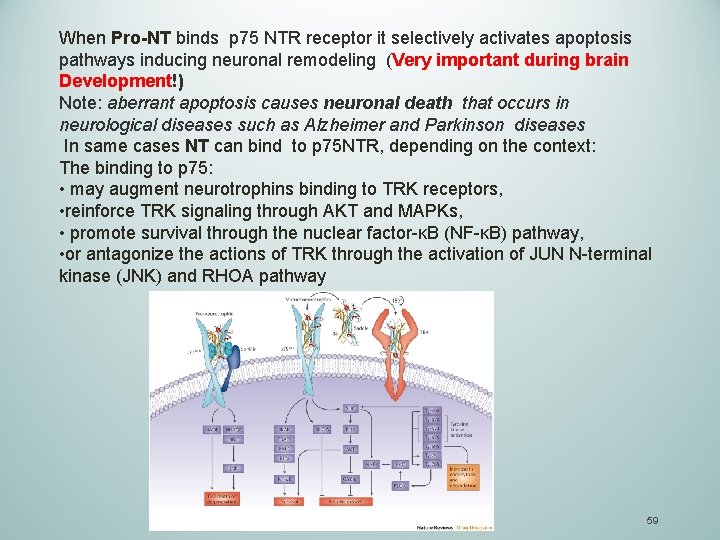
When Pro-NT binds p 75 NTR receptor it selectively activates apoptosis pathways inducing neuronal remodeling (Very important during brain Development!) Note: aberrant apoptosis causes neuronal death that occurs in neurological diseases such as Alzheimer and Parkinson diseases In same cases NT can bind to p 75 NTR, depending on the context: The binding to p 75: • may augment neurotrophins binding to TRK receptors, • reinforce TRK signaling through AKT and MAPKs, • promote survival through the nuclear factor-κB (NF-κB) pathway, • or antagonize the actions of TRK through the activation of JUN N-terminal kinase (JNK) and RHOA pathway 59

Tyrosine-Kinase-associated Receptors • These receptors lack intrinsic tyrosine kinase activity and rely on cytoplasmic tyrosine kinases. • The largest family of receptors is the Src family (Src, Yes, Fgr, Fyn, Lck, Lyn, Hck, Blk). • These proteins contain SH 2 and SH 3 domains which are located on the cytoplasmic side of the plasma membrane. • Cytokines functions are correlated with tyrosine kinase associated receptors 60

Cytokines • Cytokines are secreted, low-molecular-weight proteins that regulate the nature, intensity and duration of the immune response by exerting a variety of effects on lymphocytes and/or other cells. • Cytokines bind to specific receptors on target cells. 61

Cytokines Families • Hematopoietin Family also known as Interleukins (IL-2, IL-4) • Interferon Family (IFN-α, β, γ) • Chemokine Family • Tumor necrosis family 62

Based on structural homology, there are six major cytokine receptor families: - Ig superfamily receptors - Interferon receptors - TNF receptor superfamily - Chemokine receptors - TGF receptor family - Hematopoietin receptors (Cytokine receptor superfamily 63
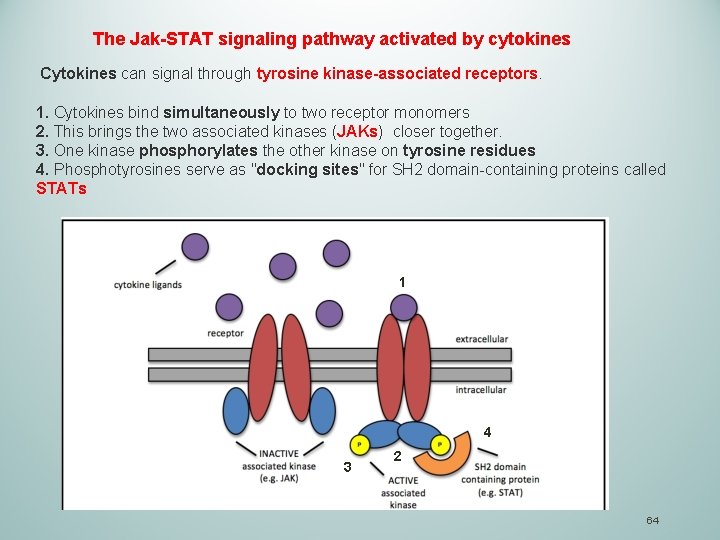
The Jak-STAT signaling pathway activated by cytokines Cytokines can signal through tyrosine kinase-associated receptors. 1. Cytokines bind simultaneously to two receptor monomers 2. This brings the two associated kinases (JAKs) closer together. 3. One kinase phosphorylates the other kinase on tyrosine residues 4. Phosphotyrosines serve as "docking sites" for SH 2 domain-containing proteins called STATs 1 4 3 2 64
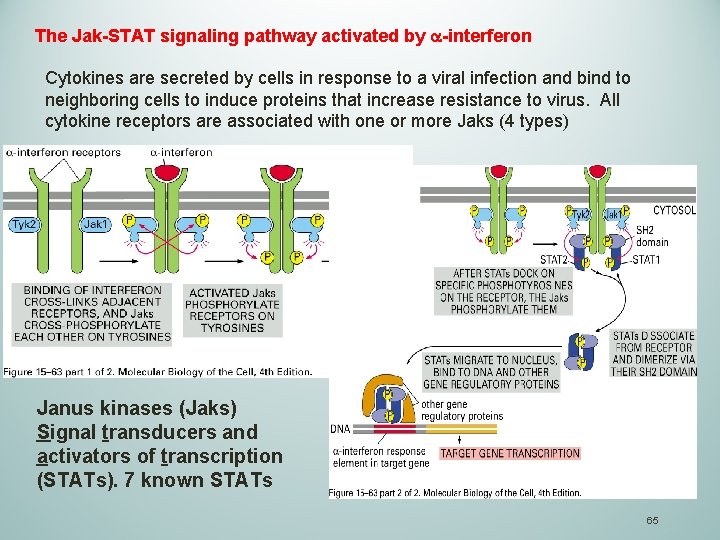
The Jak-STAT signaling pathway activated by a-interferon Cytokines are secreted by cells in response to a viral infection and bind to neighboring cells to induce proteins that increase resistance to virus. All cytokine receptors are associated with one or more Jaks (4 types) Janus kinases (Jaks) Signal transducers and activators of transcription (STATs). 7 known STATs 65
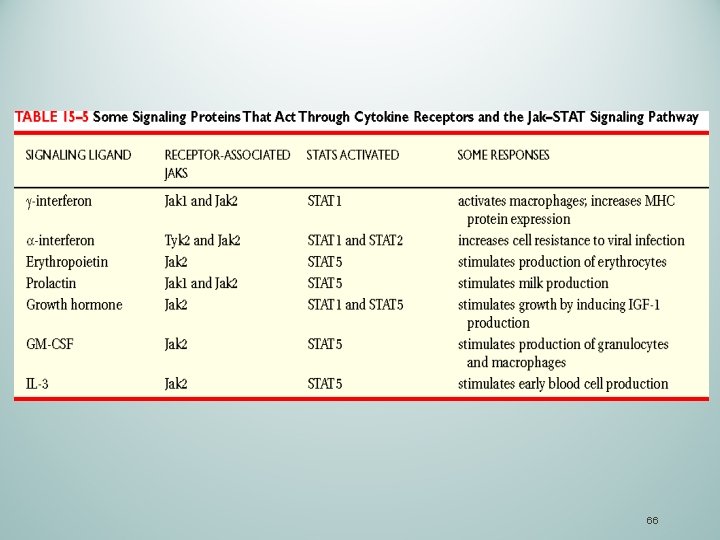
66

Receptor serine/threonine kinases – phosphorylate specific serines or threonines on associated gene regulatory proteins. PROTEIN SER/THR KINASES ASSOCIATED WITH RECEPTOR SERINE/THREONINE KINASES: Cyclin dependent kinases (CDKs) and CDK regulating kinases Cdc-2, CAK kinase GPCR kinases GRK 2 (βARK 1), GRK 3 (βARK 2), GRK 5, GRK 6 P 21 -activated kinases (PAK) Kinases involved in cytoskeletal organization and development ROCK/Rho kinase Transmembrane receptor protein ser/thr kinases TGFβ receptor protein kinase Casein kinases CK 1, CK 2 67

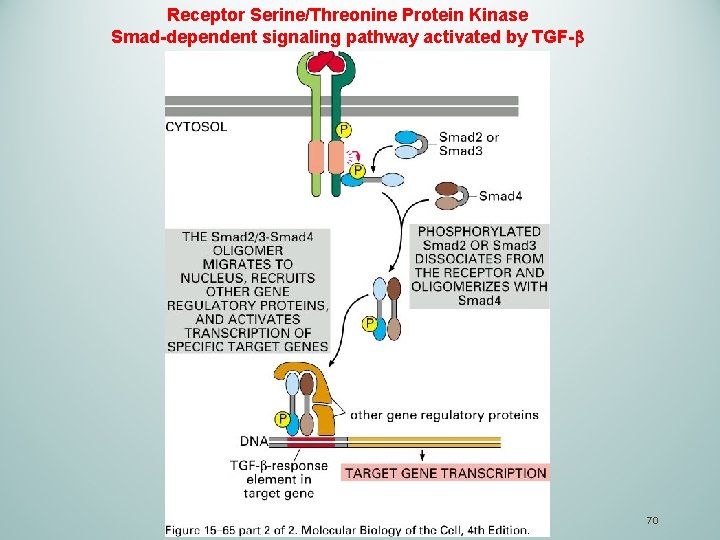
TGFβ Transforming growth factor (TGF ) is a cytokine that consists of a large number of structurally related, secreted, dimeric proteins. q. TGFβ mediates a wide range of biological functions in animals: proliferation, differentiation, ECM production, cell death, tissue repair and immune regulation. q. TGFβ act through receptor serine/threonine kinases type I and type II q. Smad family members are directly phosphorylated by the type I receptor, and moves to the nucleus to direct gene transcription 68
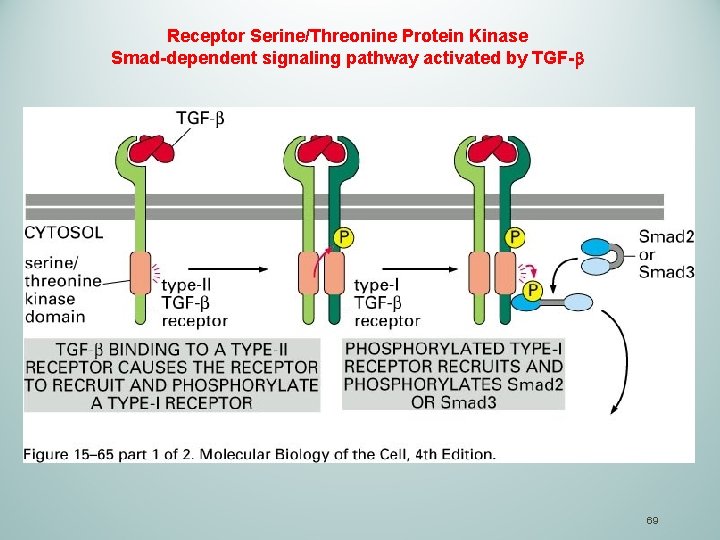
Receptor Serine/Threonine Protein Kinase Smad-dependent signaling pathway activated by TGF- 69
Receptor Serine/Threonine Protein Kinase Smad-dependent signaling pathway activated by TGF- 70

Protein Tyrosine Phosphatases Protein tyrosine phosphatases (PTPs) remove selected phosphotyrosines on a subset of tyrosine-phosphorylated proteins. Exhibit high degree of substrate selectivity. These enzyme ensure that the tyrosine phosphorylations are short-lived and are responsible for regulating the intensity of the signal. There about 30 known PTPs and occur as both transmembrane and cytoplasmic forms. 71
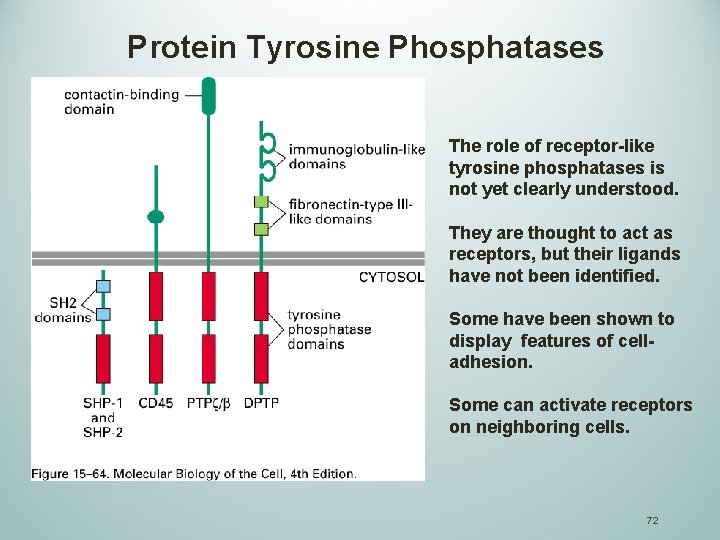
Protein Tyrosine Phosphatases The role of receptor-like tyrosine phosphatases is not yet clearly understood. They are thought to act as receptors, but their ligands have not been identified. Some have been shown to display features of celladhesion. Some can activate receptors on neighboring cells. 72
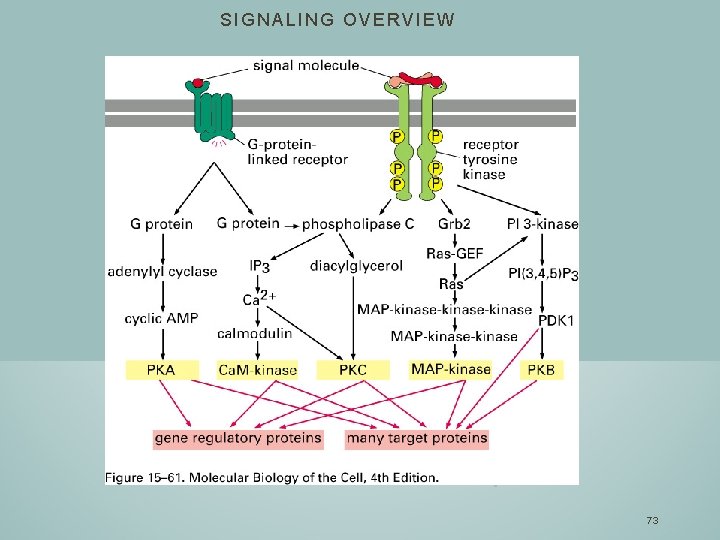
SIGNALING OVERVIEW 73

EXAM 2: IN CLASS PRESENTATION ON FEBRUARY 25, 2019 Establish 4 team groups (12 max people per group ): Each student should join a group of choice. (Each of you is free to join a group of interest but, if you prefer, I can make up the group) Each team needs to: 1. Choose 1 out 4 scientific articles I have sent for today class; 2. Read the chosen article and make a 20 min presentation to be delivered for in class presentation on February 12; 3. The presentation should be shared among team members; 4. Be prepared to answer in class questions from the professor and your peers. Please send to me (antonio. giordano@ temple. edu) the following: § Name of the students for each group and the paper chosen by February 25 §PPT presentation along with information for each group by 12 AM on February 25. 74
EXAM 2: PRESENTATION GRADING ON FEBRUARY 25, 2019 75
- Slides: 75