CELL SIGNALING AND MOTILITY BIOL 3373 ABERRANT CELL

CELL SIGNALING AND MOTILITY (BIOL 3373) ABERRANT CELL SIGNALING IN CANCER Lecture 11 1
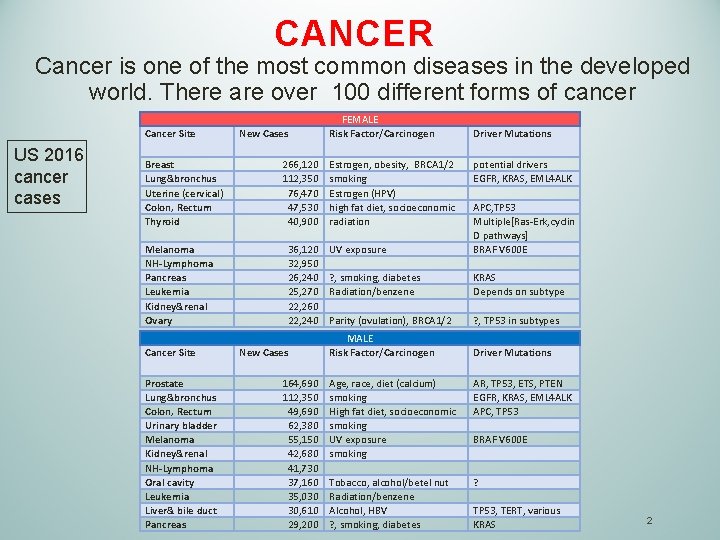
CANCER Cancer is one of the most common diseases in the developed world. There are over 100 different forms of cancer Cancer Site US 2016 cancer cases Breast Lung&bronchus Uterine (cervical) Colon, Rectum Thyroid Melanoma NH-Lymphoma Pancreas Leukemia Kidney&renal Ovary Cancer Site Prostate Lung&bronchus Colon, Rectum Urinary bladder Melanoma Kidney&renal NH-Lymphoma Oral cavity Leukemia Liver& bile duct Pancreas New Cases FEMALE Risk Factor/Carcinogen Driver Mutations 266, 120 112, 350 76, 470 47, 530 40, 900 Estrogen, obesity, BRCA 1/2 smoking Estrogen (HPV) high fat diet, socioeconomic radiation 36, 120 32, 950 26, 240 25, 270 22, 260 22, 240 UV exposure ? , smoking, diabetes Radiation/benzene Parity (ovulation), BRCA 1/2 potential drivers EGFR, KRAS, EML 4 ALK APC, TP 53 Multiple[Ras-Erk, cyclin D pathways] BRAF V 600 E KRAS Depends on subtype ? , TP 53 in subtypes MALE Risk Factor/Carcinogen Driver Mutations Age, race, diet (calcium) smoking High fat diet, socioeconomic smoking UV exposure smoking Tobacco, alcohol/betel nut Radiation/benzene Alcohol, HBV ? , smoking, diabetes AR, TP 53, ETS, PTEN EGFR, KRAS, EML 4 ALK APC, TP 53 BRAF V 600 E ? TP 53, TERT, various KRAS New Cases 164, 690 112, 350 49, 690 62, 380 55, 150 42, 680 41, 730 37, 160 35, 030 30, 610 29, 200 2

CARGINOGENESIS §Carcinogenesis is a multistep process that drives normal cells to evolve progressively towards a malignant, neoplastic state, and ultimately to acquire metastatic features. §During this process non-cancer cells develop stepwise various biological capabilities that enable them to acquire their tumorigenic potential. Note: sometimes the word carcinogenesis and oncogenesis are used interchangea Tumorigenesis: formation of a tumor 3
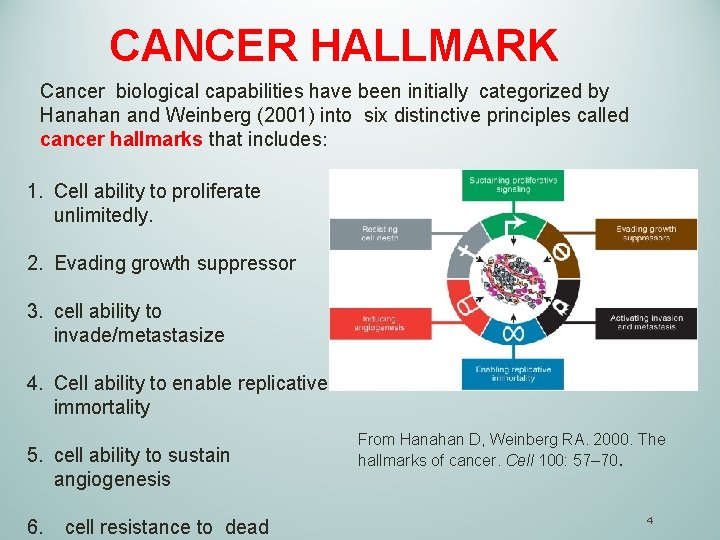
CANCER HALLMARK Cancer biological capabilities have been initially categorized by Hanahan and Weinberg (2001) into six distinctive principles called cancer hallmarks that includes: 1. Cell ability to proliferate unlimitedly. 2. Evading growth suppressor 3. cell ability to invade/metastasize 4. Cell ability to enable replicative immortality 5. cell ability to sustain angiogenesis 6. cell resistance to dead From Hanahan D, Weinberg RA. 2000. The hallmarks of cancer. Cell 100: 57– 70. 4

EMERGING CANCER HALLMARK AND ENABLING CHARACTERISTICS Cancer research field is an extremely developing field. An increasing body of research suggests that two additional hallmarks of cancer are involved in the pathogenesis of cancers: § the capability to reprogram cellular metabolism: In order to support neoplastic proliferation, cancer cells switch their mitochondrial oxidative phosphorylation process towards an (an)aerobic metabolism that consists in a robust induction of glycolysis (the Warburgeffect) § The capability of cancer cells to evade immunological destruction, in particular by T and B lymphocytes, macrophages, and natural killer cells From Hanahan D, Weinberg RA. 2011. Hallmarks of cancer: the next generation. Cell 144: 646– 74. Two characteristics facilitate acquisition of Cancer hallmarks: Genomic instability and Inflammation 5

GENETIC INSTABILITY AND CANCER q. Cancer arises from the mutation of a normal gene. q. It is thought that several mutations need to occur to give rise to cancer q. These mutations allow cancerous cells do not self destruct and continue to divide rapidly producing millions of new cancerous cells. q. These mutations “fix” the cell in the transformed state q. Additional factors such as growth factors, together with aberrant cell signalling because of additional mutations lead to enhanced proliferation, angiogenesis, lack of immune regulation, invasion and metastasis. This is usually a result of: INCORRECT SIGNAL REGULATION & FAULTY SIGNAL TERMINATION 6
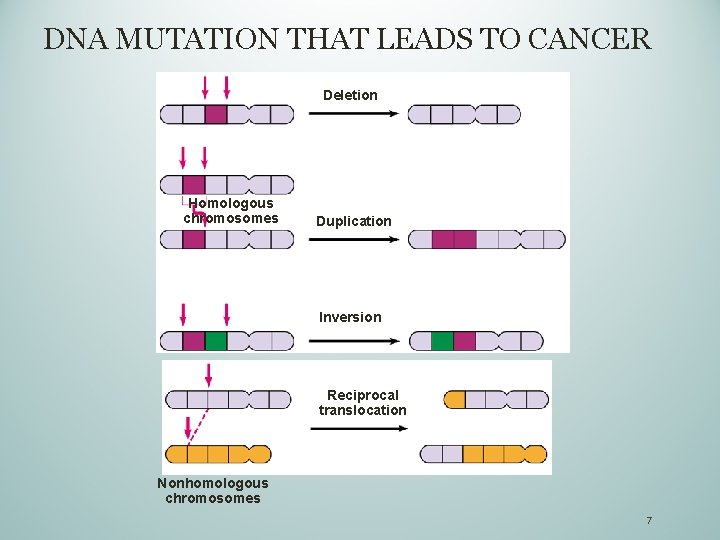
DNA MUTATION THAT LEADS TO CANCER Deletion Homologous chromosomes Duplication Inversion Reciprocal translocation Nonhomologous chromosomes 7
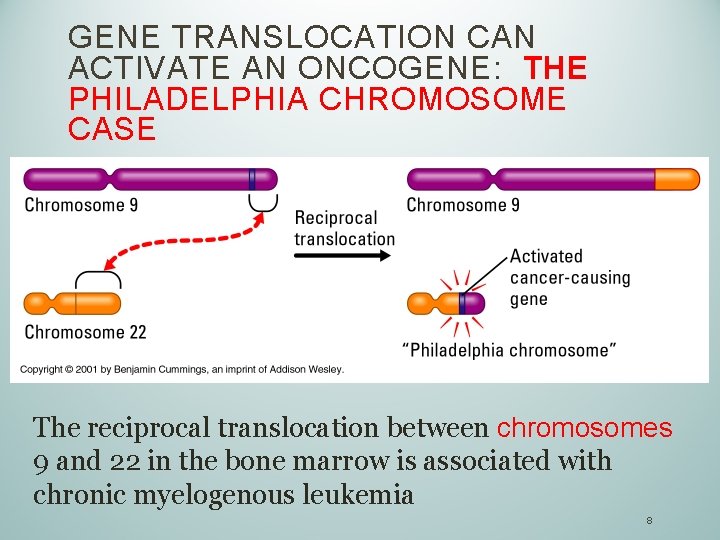
GENE TRANSLOCATION CAN ACTIVATE AN ONCOGENE: THE PHILADELPHIA CHROMOSOME CASE The reciprocal translocation between chromosomes 9 and 22 in the bone marrow is associated with chronic myelogenous leukemia 8

GENES THAT BECOME MUTATED IN CANCER: Two classes of genes are frequently mutated in cancer: • Proto-oncogenes ( oncogenes) Ø Oncogenes result from mutation which turns on (activates) protooncogene to drive cancer • Tumor suppressor genes Ø Tumor suppressors help cause cancer when they are turned off (inactivated) by mutation 9
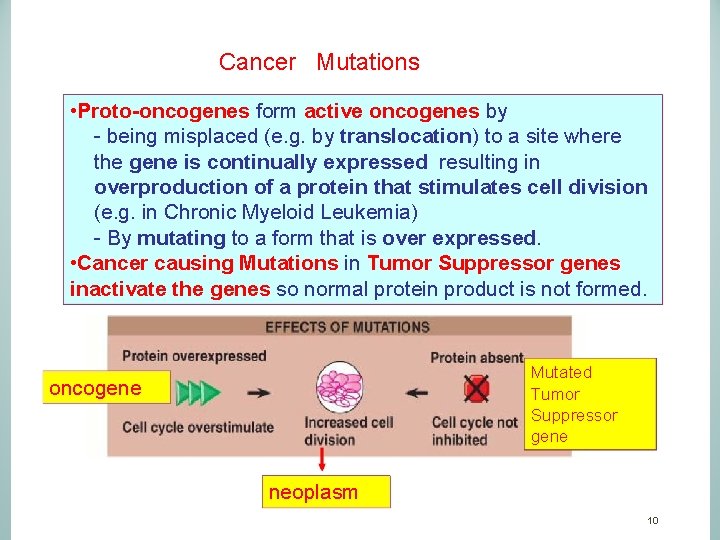
Cancer Mutations • Proto-oncogenes form active oncogenes by - being misplaced (e. g. by translocation) to a site where the gene is continually expressed resulting in overproduction of a protein that stimulates cell division (e. g. in Chronic Myeloid Leukemia) - By mutating to a form that is over expressed. • Cancer causing Mutations in Tumor Suppressor genes inactivate the genes so normal protein product is not formed. Mutated Tumor Suppressor gene oncogene neoplasm 10

Gene Mutations That Cause Cancer Mutations in 4 types of genes cause Cancer • Proto - oncogenes: genes that code for normal proteins used in cell division –Growth factors –Membrane Receptors for Growth Factors –Signaling Proteins (e. g. ras proto- oncogene mutates in 30% of cancers). • Tumor Suppressor genes: gene that code for proteins that help prevent uncontrolled cell division by blocking key steps (e. g. DNA replication). - Retinoblastoma susceptibilty (RB) gene - p 53 gene mutates in >50% of cancers. • DNA Repair genes • Genes for Apoptosis 11

Proto-oncogenes oncogenes: Proto-oncogenes • Proto-oncgenes are genes that possess normal gene products and stimulate normal cell development. Oncogenes • Oncogenes arise from mutant proto-oncogenes. • Oncogenes are more active than normal or active at inappropriate times and stimulate unregulated cell proliferation. 12

Oncogenes Are Mutated Proto-oncogenes A cell can acquire a cancer - causing oncogene from • A virus • A mutation in a proto-oncogene. Oncogenes still code for the proteins needed for cell division but they cause cancer by producing – Too much of the protein – An abnormally active protein, e. g. protein that activates division by itself – Protein that is made when it is not needed – Protein that should be made by a different (i. e. dividing) cell. 13
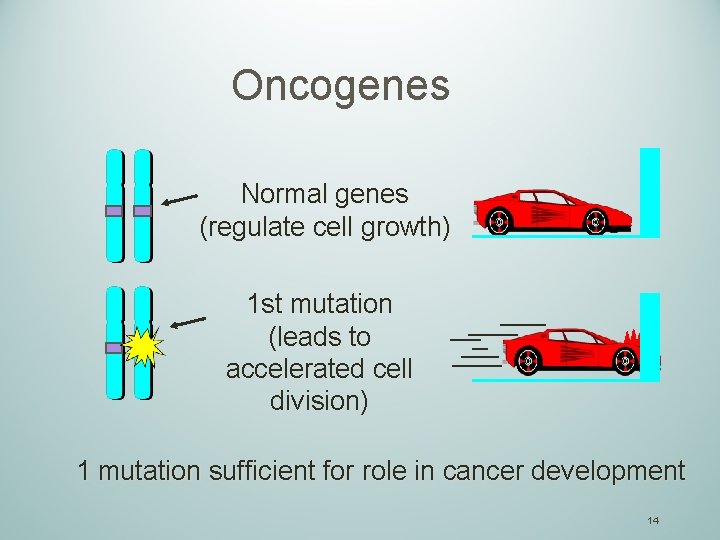
Oncogenes Normal genes (regulate cell growth) 1 st mutation (leads to accelerated cell division) 1 mutation sufficient for role in cancer development 14

Tumor Suppressor Proteins Inhibit Cell Division & Prevent Cancer Tumor suppressor proteins are proteins that bind to checkpoint proteins to stop the cell cycle & prevent cell division if DNA is damaged. • Tumor suppressor proteins stop division of mutated cells until mistakes in DNA are repaired by enzymes. • TS proteins keep most mutations from being passed on to daughter cells & developing into cancer. • If the genes for TS proteins mutate the brake on cell division is removed cancers may result. • Two important TS proteins are the p 53 protein & the RB protein. 15
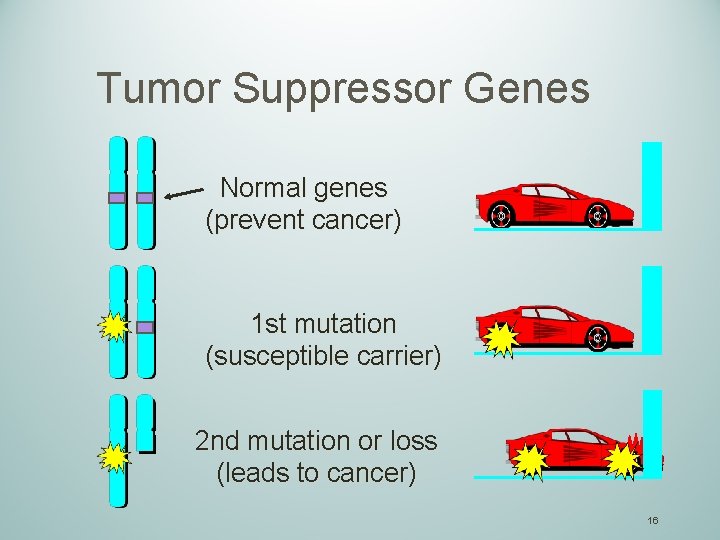
Tumor Suppressor Genes Normal genes (prevent cancer) 1 st mutation (susceptible carrier) 2 nd mutation or loss (leads to cancer) 16
CELL CYCLE CDK 17
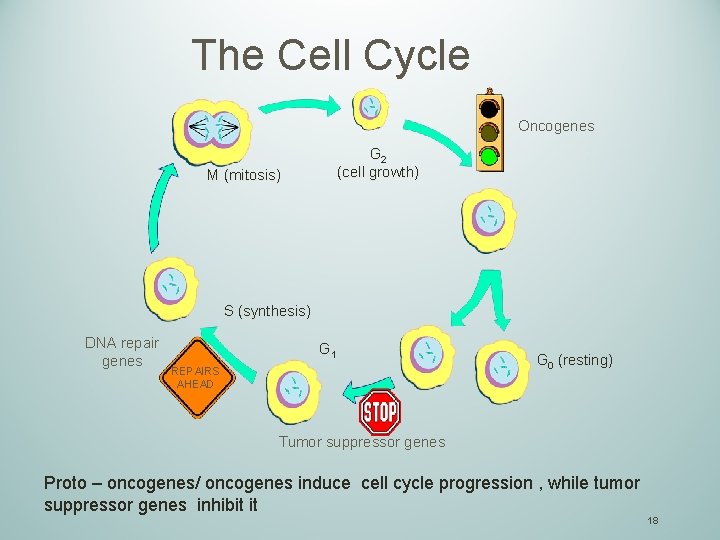
The Cell Cycle Oncogenes G 2 (cell growth) M (mitosis) S (synthesis) DNA repair genes G 1 REPAIRS AHEAD G 0 (resting) Tumor suppressor genes Proto – oncogenes/ oncogenes induce cell cycle progression , while tumor suppressor genes inhibit it 18
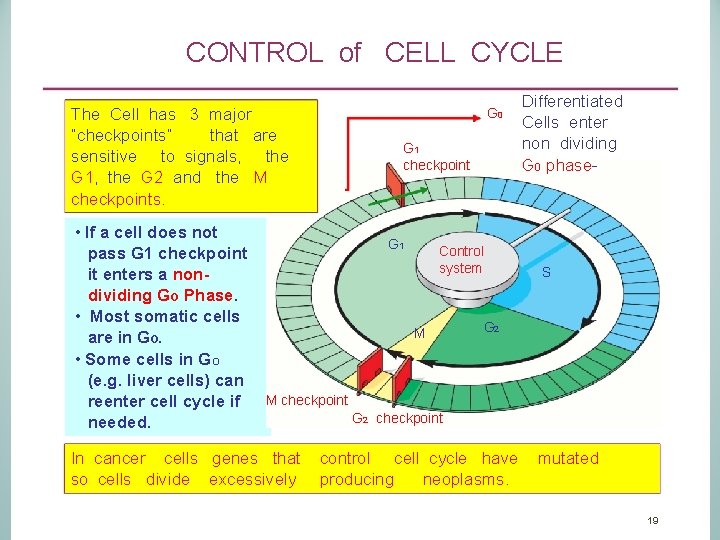
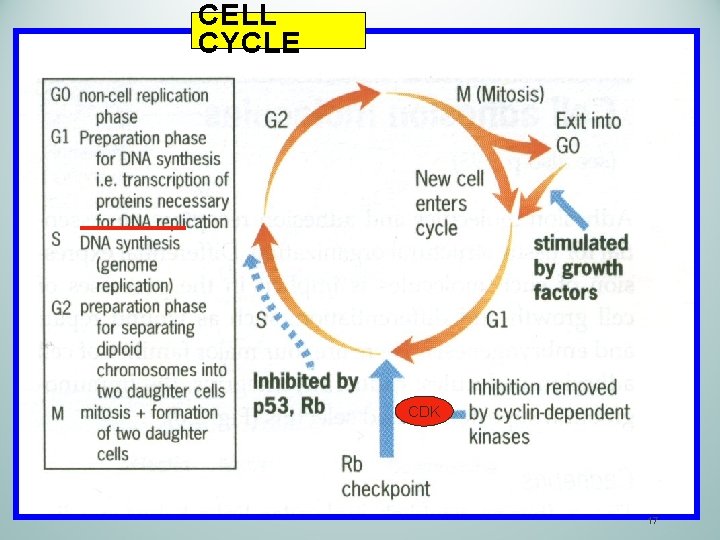
CONTROL of CELL CYCLE The Cell has 3 major “checkpoints” that are sensitive to signals, the G 1, the G 2 and the M checkpoints. • If a cell does not pass G 1 checkpoint it enters a nondividing GO Phase. • Most somatic cells are in GO. • Some cells in GO (e. g. liver cells) can reenter cell cycle if needed. G 0 G 1 checkpoint G 1 Control system M Differentiated Cells enter non dividing G 0 phase- S G 2 M checkpoint G 2 checkpoint In cancer cells genes that control cell cycle have mutated so cells divide excessively producing neoplasms. 19

Normal cell cycle is controlled by signal transduction: • Growth factors bind to surface receptors (e. g tyrosine Kinase Receptors; G Protein receptors) on the cell; • Two types of growth factors: 1. Growth factors stimulate cell division. • • 2. Growth-inhibiting factors inhibit cell division. Healthy cells divide only when growth factor and growthinhibiting factor balance favors cell division. Cancer cells divide without constraint (e. g. , mutations in growth and growth-inhibiting factor genes). 20
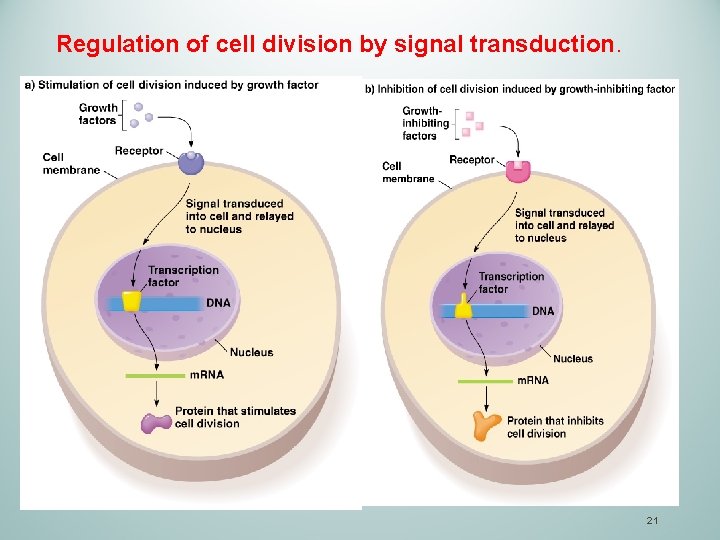
Regulation of cell division by signal transduction. 21

PRO-ONCOGENES AND CELL CYCLE Pro-oncogenes code for proteins that trigger Cell Cycle progression: • Intracellular Cyclins and Cyclin Dependent Kinases (CDKs) control the checkpoints. • Hormones or extracellular proteins from other cells (called Growth Factors) signal target cell to divide. - Hormones (e. g. Growth Hormone) or Growth Factors bind to receptor proteins of target cell membrane. - This triggers a molecular signaling pathway. - A series of linked proteins activate Cyclin- CDKs which Allows Cells to Pass Cell Cycle Checkpoints & divide. 22
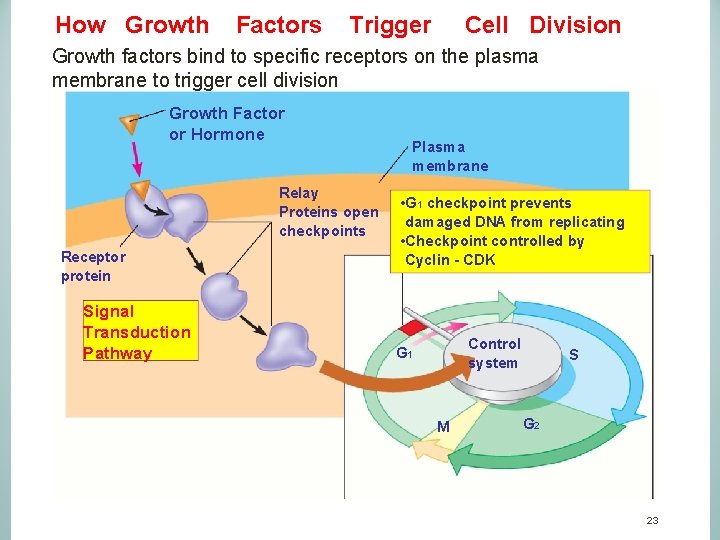
How Growth Factors Trigger Cell Division Growth factors bind to specific receptors on the plasma membrane to trigger cell division Growth Factor or Hormone Relay Proteins open checkpoints Receptor protein Signal Transduction Pathway Plasma membrane • G 1 checkpoint prevents damaged DNA from replicating • Checkpoint controlled by Cyclin - CDK Control system G 1 M S G 2 23
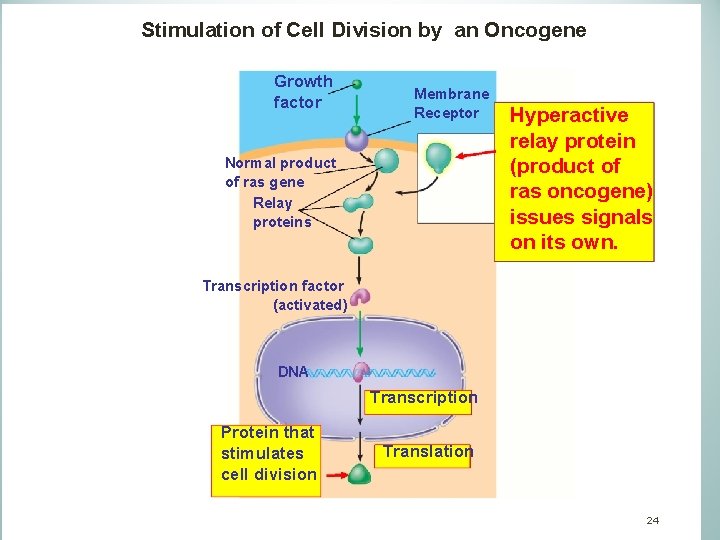
Stimulation of Cell Division by an Oncogene Over Growth factor Membrane Receptor Normal product of ras gene Relay proteins Hyperactive relay protein (product of ras oncogene) issues signals on its own. Transcription factor (activated) DNA Transcription Protein that stimulates cell division Translation 24
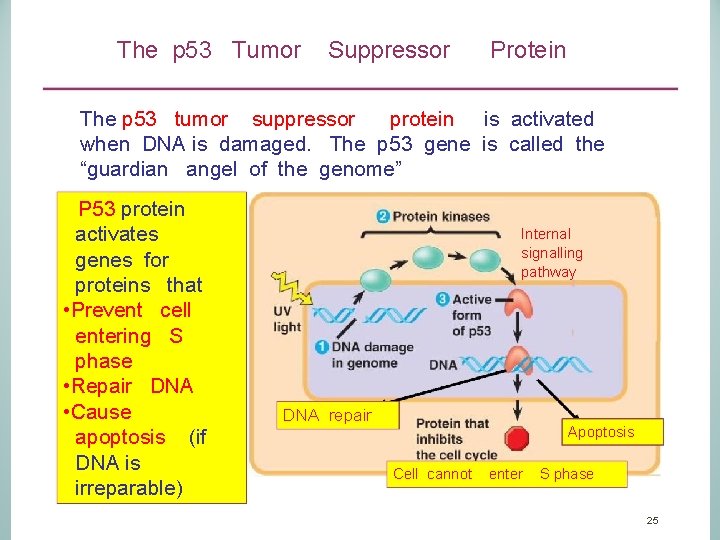
The p 53 Tumor Suppressor Protein The p 53 tumor suppressor protein is activated when DNA is damaged. The p 53 gene is called the “guardian angel of the genome” P 53 protein activates genes for proteins that • Prevent cell entering S phase • Repair DNA • Cause apoptosis (if DNA is irreparable) Internal signalling pathway DNA repair Apoptosis Cell cannot enter S phase 25
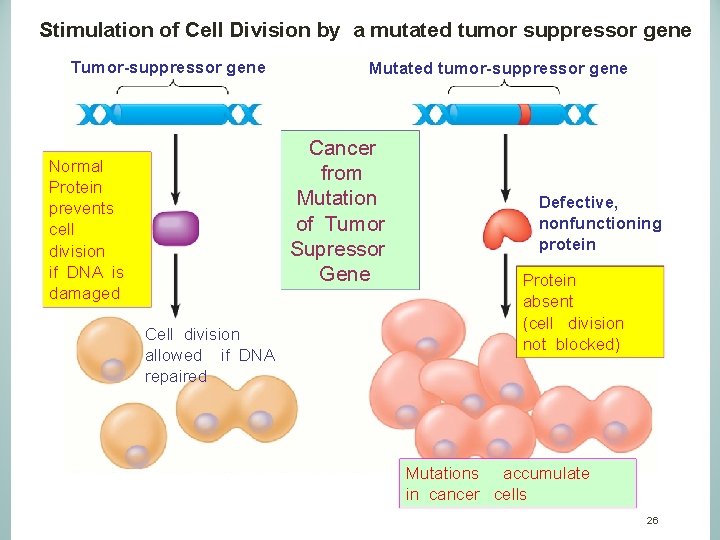
Stimulation of Cell Division by a mutated tumor suppressor gene Tumor-suppressor gene Mutated tumor-suppressor gene Cancer from Mutation of Tumor Supressor Gene Normal Protein prevents cell division if DNA is damaged Cell division allowed if DNA repaired Defective, nonfunctioning protein Protein absent (cell division not blocked) Mutations accumulate in cancer cells 26
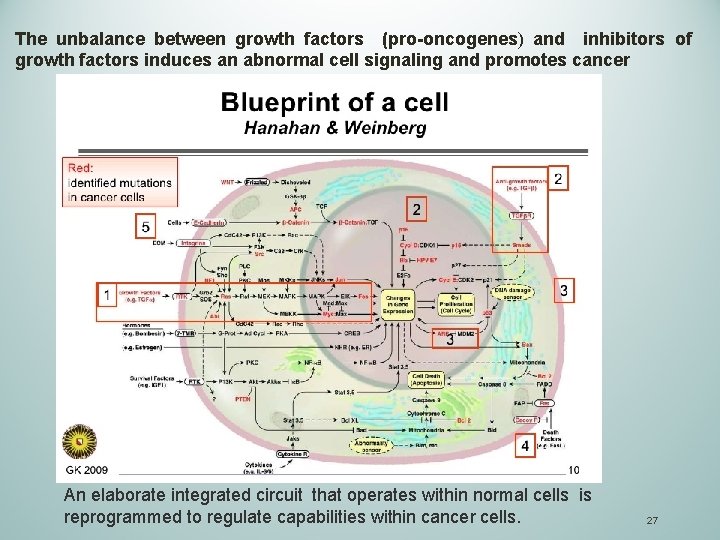
The unbalance between growth factors (pro-oncogenes) and inhibitors of growth factors induces an abnormal cell signaling and promotes cancer An elaborate integrated circuit that operates within normal cells is reprogrammed to regulate capabilities within cancer cells. 27

STEPS OF CANCER PROGRESSION 28
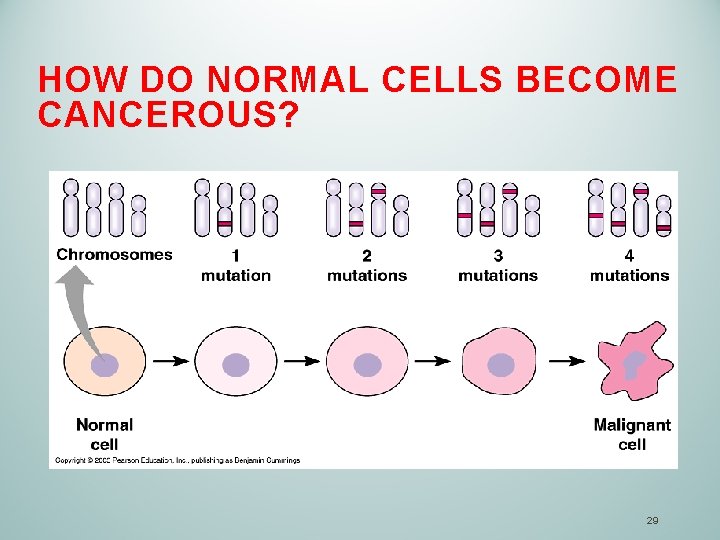
HOW DO NORMAL CELLS BECOME CANCEROUS? 29
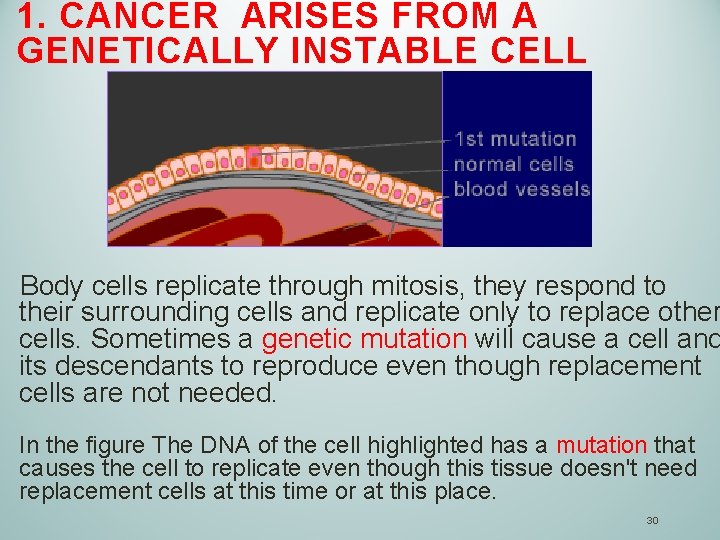
1. CANCER ARISES FROM A GENETICALLY INSTABLE CELL Body cells replicate through mitosis, they respond to their surrounding cells and replicate only to replace other cells. Sometimes a genetic mutation will cause a cell and its descendants to reproduce even though replacement cells are not needed. In the figure The DNA of the cell highlighted has a mutation that causes the cell to replicate even though this tissue doesn't need replacement cells at this time or at this place. 30
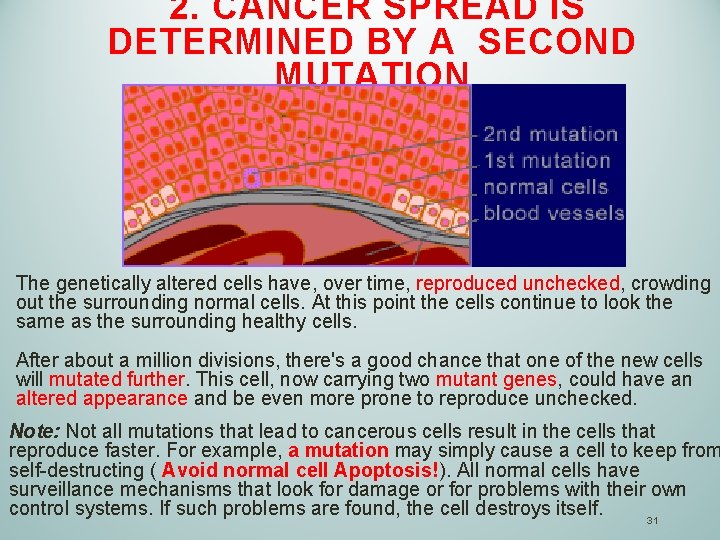
2. CANCER SPREAD IS DETERMINED BY A SECOND MUTATION The genetically altered cells have, over time, reproduced unchecked, crowding out the surrounding normal cells. At this point the cells continue to look the same as the surrounding healthy cells. After about a million divisions, there's a good chance that one of the new cells will mutated further. This cell, now carrying two mutant genes, could have an altered appearance and be even more prone to reproduce unchecked. Note: Not all mutations that lead to cancerous cells result in the cells that reproduce faster. For example, a mutation may simply cause a cell to keep from self-destructing ( Avoid normal cell Apoptosis!). All normal cells have surveillance mechanisms that look for damage or for problems with their own control systems. If such problems are found, the cell destroys itself. 31
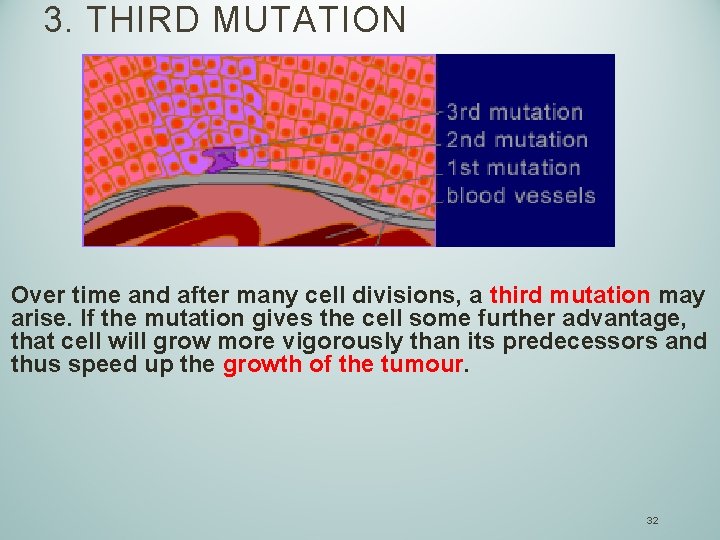
3. THIRD MUTATION Over time and after many cell divisions, a third mutation may arise. If the mutation gives the cell some further advantage, that cell will grow more vigorously than its predecessors and thus speed up the growth of the tumour. 32
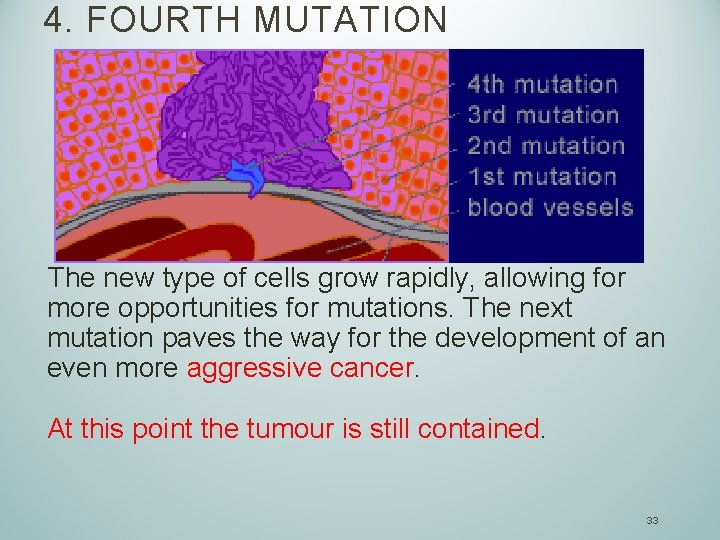
4. FOURTH MUTATION The new type of cells grow rapidly, allowing for more opportunities for mutations. The next mutation paves the way for the development of an even more aggressive cancer. At this point the tumour is still contained. 33
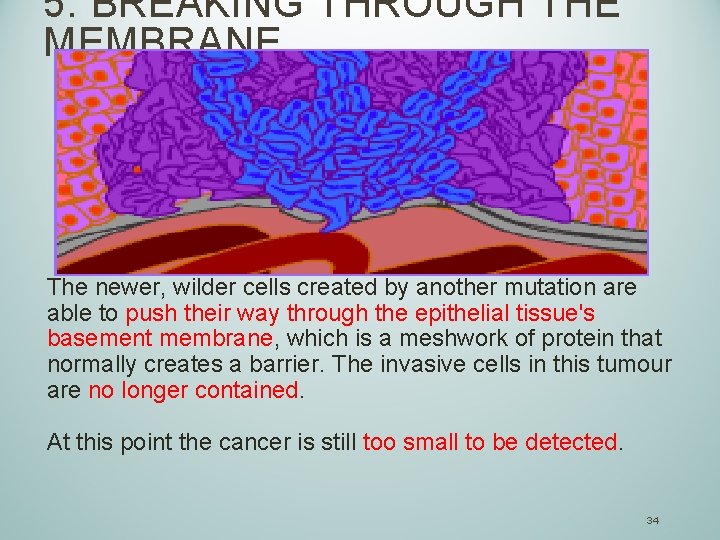
5. BREAKING THROUGH THE MEMBRANE The newer, wilder cells created by another mutation are able to push their way through the epithelial tissue's basement membrane, which is a meshwork of protein that normally creates a barrier. The invasive cells in this tumour are no longer contained. At this point the cancer is still too small to be detected. 34

6. ANGIOGENESIS Often during the development of earlier stages of the tumour, or perhaps by the time the tumour has broken through the basement membrane (as pictured above), angiogenesis takes place. Angiogenesis is the recruitment of blood vessels from the network of neighbouring vessels. Without blood and the nutrients it carries, a tumour would be unable to continue growing. With the new blood supply, however, the growth of the tumour accelerates; it soon contains thousand million cells and, now the size of a small grape, is large enough to be detected as a lump 35
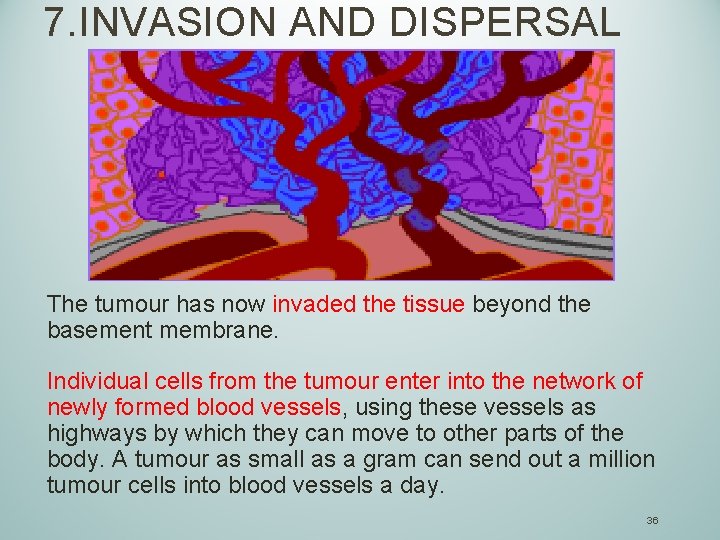
7. INVASION AND DISPERSAL The tumour has now invaded the tissue beyond the basement membrane. Individual cells from the tumour enter into the network of newly formed blood vessels, using these vessels as highways by which they can move to other parts of the body. A tumour as small as a gram can send out a million tumour cells into blood vessels a day. 36
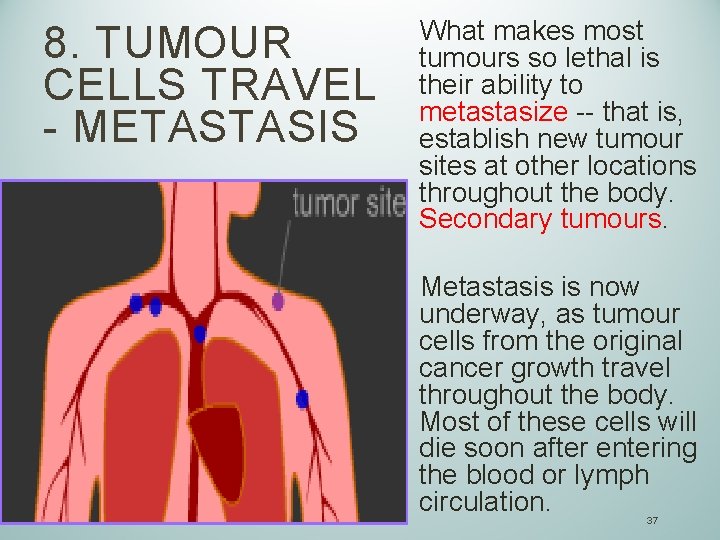
8. TUMOUR CELLS TRAVEL - METASTASIS What makes most tumours so lethal is their ability to metastasize -- that is, establish new tumour sites at other locations throughout the body. Secondary tumours. Metastasis is now underway, as tumour cells from the original cancer growth travel throughout the body. Most of these cells will die soon after entering the blood or lymph circulation. 37
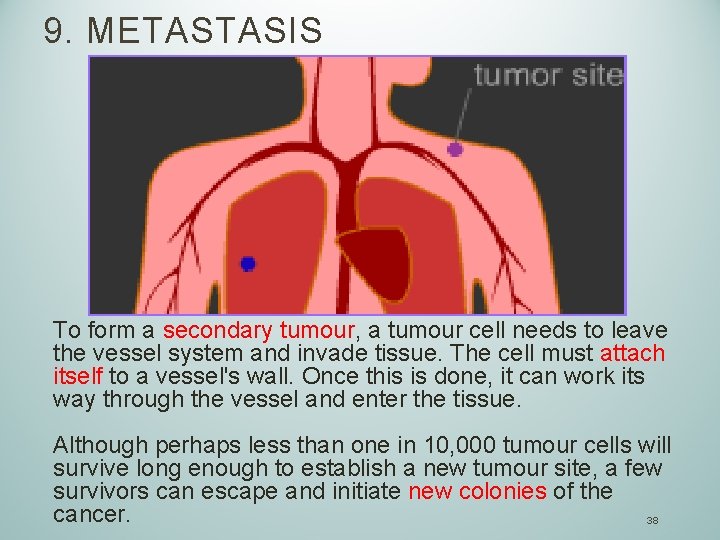
9. METASTASIS To form a secondary tumour, a tumour cell needs to leave the vessel system and invade tissue. The cell must attach itself to a vessel's wall. Once this is done, it can work its way through the vessel and enter the tissue. Although perhaps less than one in 10, 000 tumour cells will survive long enough to establish a new tumour site, a few survivors can escape and initiate new colonies of the cancer. 38

EXAMPLES OF MUTATIONS IN SIGNAL TERMINATORS RESULTING IN CANCER Cell Signaling Signal Result in Types of Pathway Termination mutation in signal terminator Cancers this is seen in PI 3 K-AKT PTEN phosphatase (tumor suppressor) PTEN can’t Breast hydrolyze PIP 3: Lung so PI 3 K and AKT activity high Wnt B-catenin Destruction complex APC (tumor suppressor) Mutant APC Colon leads to Possibly increased B ovarian catenin since no proteosomal degradation 39

WHAT CAUSE GENE MUTATION A factor which brings about a mutation is called a mutagen. A mutagen is mutagenic. Any agent that causes cancer is called a carcinogen and is described as carcinogenic. A carcinogen induces Cancer due to the ability to damage the genome or to the disruption of cellular metabolic processes. 40

FACTORS BELIEVED TO CONTRIBUTE TO GLOBAL CAUSES OF CANCER 41

REPRODUCTIVE AND HORMONAL RISKS FOR CANCER ØPregnancy and oral contraceptives increase a woman’s chances of breast cancer (increases risk 2 -4 X) ØLate menarche, early menopause, early first childbirth, having many children have been shown to reduce risk of breast cancer ØHowever, genetic factors can increase breast cancer risk >4 fold ØBRCA 1/2, TP 53, ATM increase relative risk 4 -8 fold ØFamily history of breast and early ovarian cancer increase risk 4 fold 42

OVARIAN CANCER RISK Change in relative risk Nulliparity 4 Multiparity 0. 5 Birth Control Pill Use 0. 6 Fertility Drug Use BRCA 1/2, one 1◦ relative 2 4
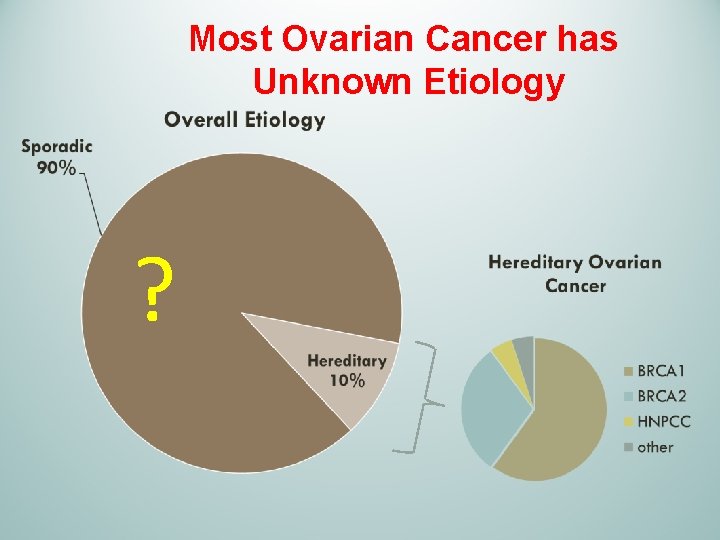
Most Ovarian Cancer has Unknown Etiology ?

OCCUPATIONAL AND ENVIRONMENTAL FACTORS ØAsbestos ØNickel ØChromate ØBenzene ØArsenic ØRadioactive substances ØCool tars ØHerbicides/pesticides Note: Each chemical or environmental carcinogen has its own unique a tumor type and usually governed by route of exposure as well 45
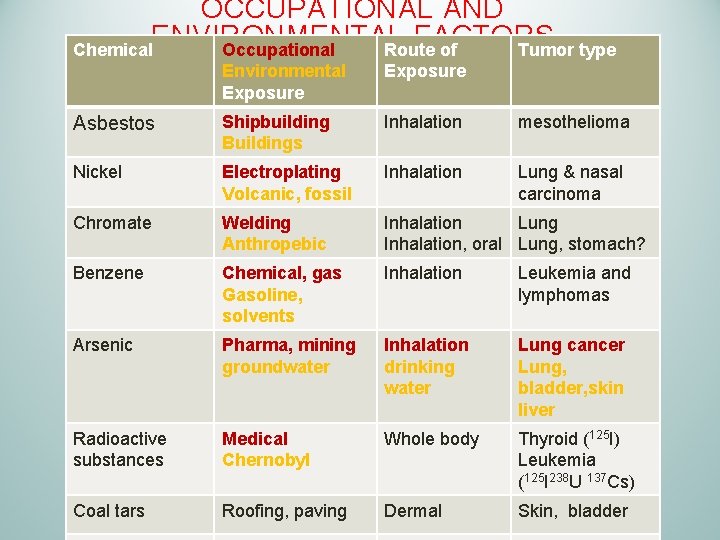
OCCUPATIONAL AND ENVIRONMENTAL FACTORS Chemical Occupational Route of Tumor type Environmental Exposure Asbestos Shipbuilding Buildings Inhalation mesothelioma Nickel Electroplating Volcanic, fossil Inhalation Lung & nasal carcinoma Chromate Welding Anthropebic Inhalation Lung Inhalation, oral Lung, stomach? Benzene Chemical, gas Gasoline, solvents Inhalation Leukemia and lymphomas Arsenic Pharma, mining groundwater Inhalation drinking water Lung cancer Lung, bladder, skin liver Radioactive substances Medical Chernobyl Whole body Thyroid (125 I) Leukemia (125 I 238 U 137 Cs) Coal tars Roofing, paving Dermal Skin, bladder 46

SOCIAL AND PSYCHOLOGICAL FACTORS Stress has been implicated in increased susceptibility to several types of cancers Sleep disturbances, diet, or a combination of factors may weaken the body’s immune system 47

CHEMICALS IN FOODS Sodium nitrate when ingested forms a potential carcinogen, nitrosamine Sodium nitrate is still used because it is effective in preventing botulism Pesticide and herbicide residues 48
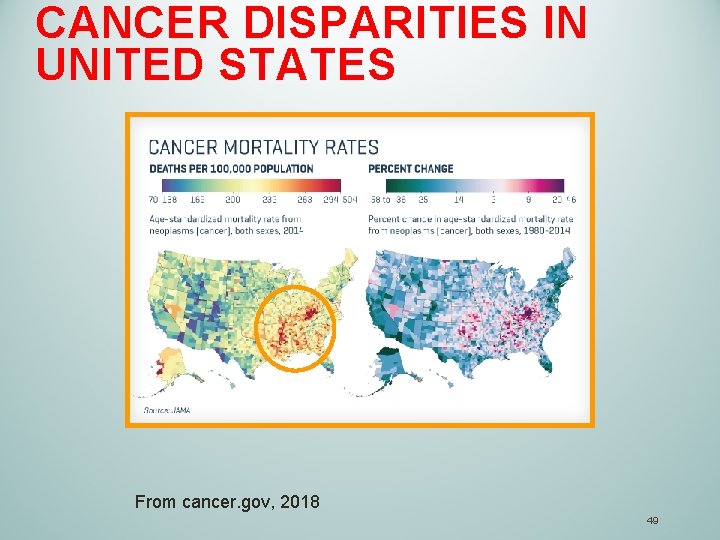
CANCER DISPARITIES IN UNITED STATES From cancer. gov, 2018 49
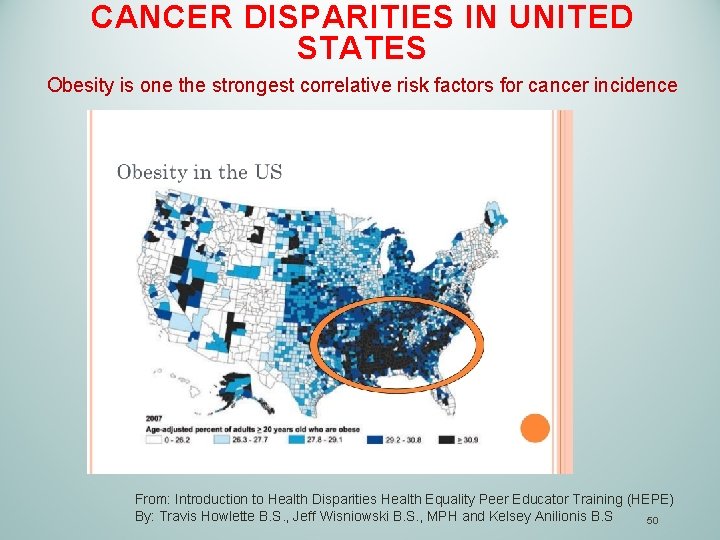
CANCER DISPARITIES IN UNITED STATES Obesity is one the strongest correlative risk factors for cancer incidence From: Introduction to Health Disparities Health Equality Peer Educator Training (HEPE) By: Travis Howlette B. S. , Jeff Wisniowski B. S. , MPH and Kelsey Anilionis B. S 50
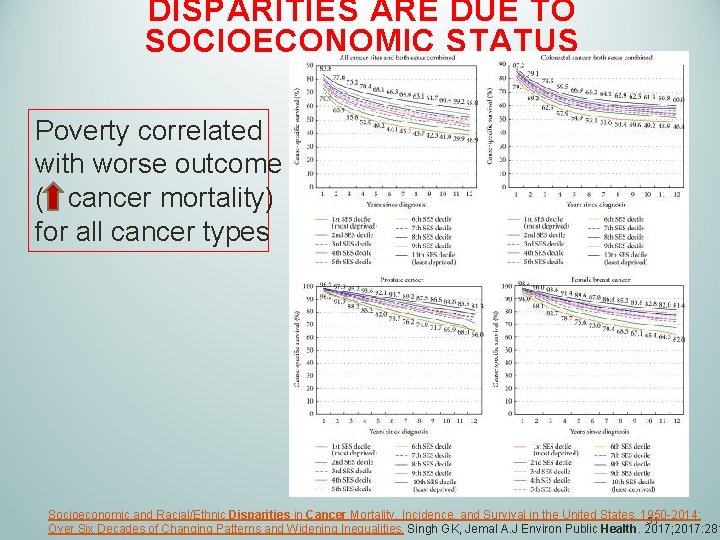
DISPARITIES ARE DUE TO SOCIOECONOMIC STATUS Poverty correlated with worse outcome ( cancer mortality) for all cancer types Socioeconomic and Racial/Ethnic Disparities in Cancer Mortality, Incidence, and Survival in the United States, 1950 -2014: 51 Over Six Decades of Changing Patterns and Widening Inequalities. Singh GK, Jemal A. J Environ Public Health. 2017; 2017: 281
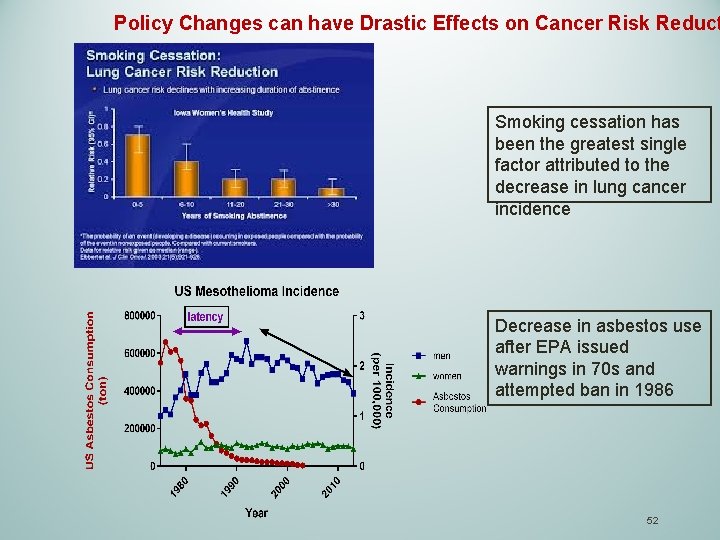
Policy Changes can have Drastic Effects on Cancer Risk Reduct Smoking cessation has been the greatest single factor attributed to the decrease in lung cancer incidence Decrease in asbestos use after EPA issued warnings in 70 s and attempted ban in 1986 52

VIRAL FACTORS ØHerpes-related viruses may be involved in the development of leukemia, Hodgkin’s disease, cervical cancer, and Burkitt’s lymphoma ØEpstein-Barr virus, associated with mononucleosis, may contribute to cancer ØHuman papillomavirus (HPV), virus that causes genital warts, has been linked to cervical cancer ØHelicobacter pylori causes ulcers which are a major factor in the development of stomach cancer 53

MEDICAL FACTORS Some medical treatments actually increase a person’s risk for cancer Diethylstilbestrol (DES) used 1940 to 1960 to control bleeding during pregnancy, the daughters of mothers that used DES were found to have an increased risk for cancers of the reproductive organs Estrogen supplementation Chemotherapy used to treat one form of cancer may increase risk for another type of cancer 54

DIAGNOSTIC METHODS FOR CANCER 1 - Karyotype analysis: It is a direct morphological analysis of chromosomes from tumor cells under the microscope. 2 - Fluorescent in situ hybridization analysis FISH analysis involves the use of fluorescent- labelled genetic probes which hybridize to specific parts of the genome. This can detect extra copies of genetic material or reveal chromosomal translocation. 3 - Southern blot analysis: It involves extraction of DNA from leukaemic cells followed by restriction enzyme digestion, gel electrophoresis and transfer by blotting to a suitable membrane. The DNA is then hybridized to a probe complementary to the gene of interest. 4 - Polymerase chain reaction: Can be performed on blood or bone marrow for a number of specific translocations such as t(9; 22) and t(15; 17). It is very sensitive and can detect one abnormal cell in one million normal cells. It is of great value to diagnose minimal residual disease. 55

DIAGNOSTIC METHODS FOR CANCER 5 - DNA microarray: Allows rapid and comprehensive analysis of cellular transcription by hybridizing labelled cellular m. RNA to DNA probes which are immobilized on a solid support. This approach can rapidly determine m. RNA expression from a large number of genes and may be used to determine the m. RNA expression pattern of different leukaemia or lymphoma subtypes. 6 - Flow cytometry: Normal cells each have a characteristic profile but malignant cells often express an aberrant phenotype that can be useful in allowing their detection. 7 - Immunohistochemistry: Antibodies can be used to stain tissue sections with fluorescent markers. Value of using these methods: a- Initial diagnosis. b- Establishing treatment protocol. c- Monitoring response to therapy. 56

THERAPY 1 -local therapy: -surgery. -radiation therapy. 2 -systemic treatment: chemotherapy. Hormonal therapy. Monoclonal antibodies. Radioactive material. 57

SURGERY ØSurgery was the first modality used successfully in the treatment of cancer. ØIt is the only curative therapy for many common solid tumors. ØThe most important determinant of a successful surgical therapy are the absence of distant metastases and no local infiltration. 58

RADIATION THERAPY: Radiation therapy: is a local modality used in the treatment of cancer. Success depend in the difference in the radio sensitivity between the tumor and normal tissue. It involves the administration of ionizing radiation in the form of x-ray or gamma rays to the tumor site. Method of delivery: External beam(teletherapy). Internal beam therapy(Brachytherapy). 59

COMPLICATION OF RADIATION: There is two types of toxicity , acute and long term toxicity. Systemic symptoms such as Fatigue, local skin reaction, GI toxicity, oropharyngeal mucositis myelosuppression. Long-term sequelae may occur many months or years after radiation therapy. Radiation therapy is known to be mutagenic, carcinogenic, and teratogen, and having increased risk of developing both secondary leukemia and solid tumor. 60

CHEMOTHERAPY: Systemic chemotherapy is the main treatment available for disseminated malignant diseases. Progress in chemotherapy resulted in cure for several tumors. Chemotherapy usually require multiple cycles. 61

CHEMOTHERAPEUTIC AGENTS: Alkylating agents: cisplatin, cyclophosphamide Antimetabolites: 5 -floururacil, methotrexate, 6 -thioguanine Antitumor antibiotic: doxorubicin, daunorubicin, actinomycin D Plant alkaloids: vinblastine, vincristine (prevent microtubule assembly) Other agents: taxol (prevent microtubule disassembly), topoisomerase inhibitors (etopiside, topotecan) Hormonal agent: tamoxifen, flutamide, antiandrogens Immunotherapy: Rituximab, Herceptin (trastuzumab), Yervoy, Opdivio 62

COMPLICATION OF CHEMOTHERAPY: Every chemotherapeutic will have some deleterious side effect on normal tissue. E. G; Myelosuppression, nausea &vomiting. Stomatitis, and alopecia are the most frequently observed side effects. 63

COMPLICATION OF CHEMOTHERAPY: RESISTANCE Two types of chemoresistance • Intrinsic resistance: Tumor shows no clinical response on initiation of treatment due to pre-existing factors. Also referred to as insensitivity or refractory. Mechanism not well understood. • Acquired resistance: Tumor becomes unresponsive to drug after treatment with the drug. Mechanism of resistance of a chemotherapeutic usually related to pharmacodynamic parameter (drug’s mechanism of action) or pharmacokinetic parameter (availability of drug). Usually due to acquired mutations during treatment and/or changes in gene 64

MECHANISMS OF ACQUIRED RESISTANCE: Chemotherapy Mechanism of Action Mechanism of resistance Alkylating agents Bind DNA; DNA damage Increased DNA repair Increased drug efflux Altered apoptotic gene expression Taxol Binds to tubulin and inhibits microtubule function Mutation in tubulin binding site Increased drug efflux Tyrosine kinase inhibitors Inhibits activity of oncogenic tyrosine kinases Gene amplification Kinome plasticity 65
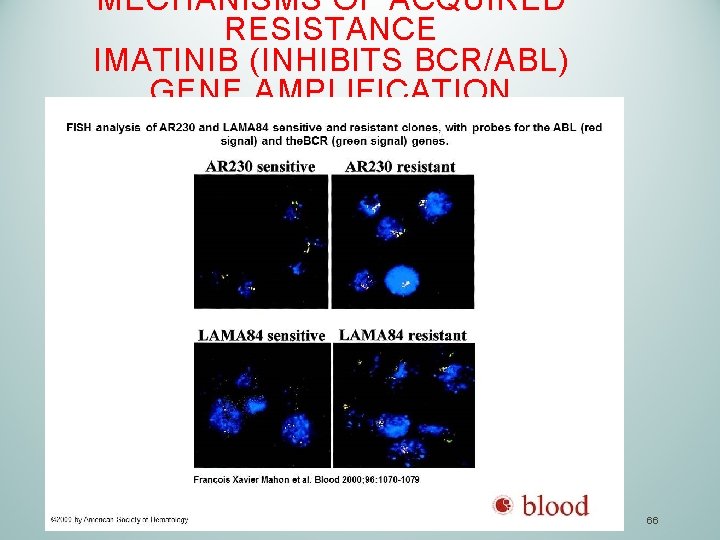
MECHANISMS OF ACQUIRED RESISTANCE IMATINIB (INHIBITS BCR/ABL) GENE AMPLIFICATION 66
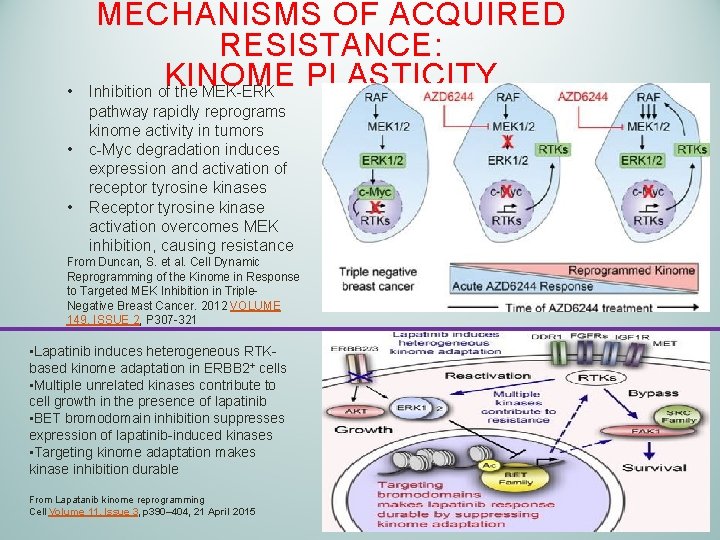
• • • MECHANISMS OF ACQUIRED RESISTANCE: KINOME PLASTICITY Inhibition of the MEK-ERK pathway rapidly reprograms kinome activity in tumors c-Myc degradation induces expression and activation of receptor tyrosine kinases Receptor tyrosine kinase activation overcomes MEK inhibition, causing resistance From Duncan, S. et al. Cell Dynamic Reprogramming of the Kinome in Response to Targeted MEK Inhibition in Triple. Negative Breast Cancer. 2012 VOLUME 149, ISSUE 2, P 307 -321 • Lapatinib induces heterogeneous RTKbased kinome adaptation in ERBB 2+ cells • Multiple unrelated kinases contribute to cell growth in the presence of lapatinib • BET bromodomain inhibition suppresses expression of lapatinib-induced kinases • Targeting kinome adaptation makes kinase inhibition durable From Lapatanib kinome reprogramming Cell Volume 11, Issue 3, p 390– 404, 21 April 2015 67
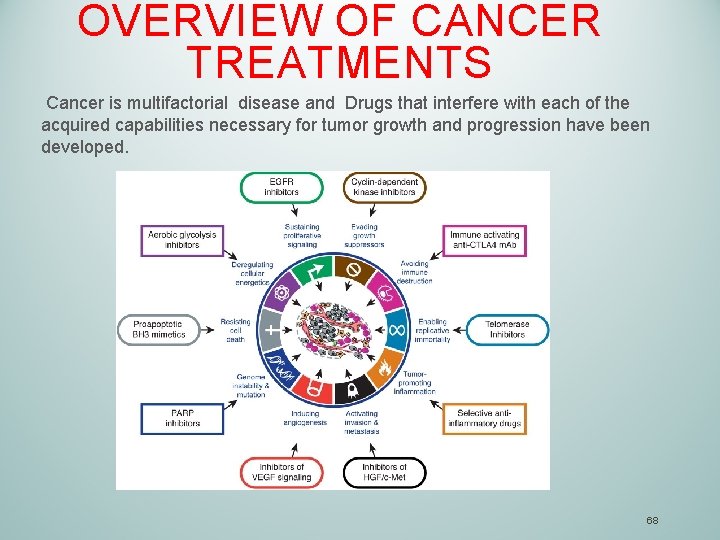
OVERVIEW OF CANCER TREATMENTS Cancer is multifactorial disease and Drugs that interfere with each of the acquired capabilities necessary for tumor growth and progression have been developed. 68

Abnormal Signal Transduction in Cancer and current Therapeutic approach https: //www. youtube. com/watch? v=jjf. YQMW_nek 69

FUTURE IN CANCER TREATMENTS § The Crosstalk the major signaling pathways provide normal cells with an ability to deal with perturbations of homeostasis. • In cancer cells these feedback and crosstalk mechanisms become a problem in the treatment of cancer, as the inhibition of one cancer-relevant signaling pathway can lead to the activation of a secondary survival pathway that interferes with cancer drug efficacy. THIS IS THE MAIN REASON WHY CANCER THERAPHIES THAT AIM TO TARGET ONE SINGLE CELL SIGNALING PATHWAY FAIL SO DISPLAYING CANCER RECURRENCIES. • Future direction in cancer treatment should allow the design of powerful synthetic lethal drug combination therapies able to target different signaling pathway and so to eliminate feedback and crosstalk mechanisms. From Opportunities and challenges provided by crosstalk between signalling pathways in cancer A Prahallad and R Bernards, 2016 70

To read: • A Prahallad and R Bernards, 2016 Opportunities and challenges provided by crosstalk between signaling pathways in cancer. Oncogene (2016) 35, 1073– 1079 • Hanahan D, Weinberg RA. 2011. Hallmarks of cancer: the next generation. Cell 144: 646– 74. 71

IN CLASS PRESENTATION ON MAY 6, 2019 Establish 4 team groups (8 -9 people per group ): Each student should join a group of choice. (Each of you is free to join a group of interest but, if you prefer, I can make up the group) Choose 1 topic from the list below and make a 25 -30 min presentation to be delivered for in class presentation on April 29: 1. Signaling via G-proteins: implication in cell physiology and disease ; 2. Tyrosine kinase receptors signaling: implication in cell physiology and disease 3. Wnt Signaling: implication in cell physiology and disease 4. Medical consequences of aberrant signaling pathways; The presentation should be shared among team members; Be prepared to answer in class questions from the professor and your peers. Please send me the PPT presentation along with students name for 72 each group by 12: 01 AM on April 29
- Slides: 72