Cell Respiration GLYCOLYSIS KREBS CYCLE ELECTRON TRANSPORT FERMENTATION

Cell Respiration GLYCOLYSIS, KREBS CYCLE, ELECTRON TRANSPORT + FERMENTATION

Cell respiration • Catabolic pathway • Most prevalent and efficient • Need oxygen to function • Fermentation Partially breaks down sugars • Occurs without oxygen • • Mostly occurs in the mitochondria in Eukaryotes • Generally in the form : • Organic compounds + O₂→ CO₂ + H₂O + Energy (heat or ATP) • Carbohydrates, fats and protein

Cell respiration • Glucose (C₆H₁₂O₆) • C₆H₁₂O₆ + O₂→ CO₂ + H₂O + 686 kcal (heat or ATP) Exergonic • Negative ∆G •

ATP • Adenosine Triphosphate • 3 closely packed phosphate groups, negatively charged • Unstable Relaxes by rejecting 3 rd phosphate group • Enzymes help phosphorylate other compounds • Undergo transition to do work • • ATP → ADP + inorganic phosphate • Store less energy than ATP • Each molecules of glucose yields at most 38 ATP molecules

Redox • Oxidation • Losing electrons • Reducing agent for the reduced compound • Reduction • Gaining electrons • Oxidizing agent for the oxidized compound • Covalent products change polarity from their original compounds Electrons shared more or less evenly • Methane combustion • • Potential energy released when electrons are transferred to a more electronegative atom

Beginning of energy production • Oxidation of organic compounds • Electrons fall from hydrogen to oxygen Convert electrons’ potential energy to kinetic • Available for ATP synthesis • Carbohydrates and fats are good sources of fuel • • Activation energy Energy needed to kickstart the process • Glucose cannot spontaneously combust at body temp • Enzymes lower activation energy • Allows for the slow release of energy •

NADH • Organic fuels broken down in a series of steps • Dehydrogenases strip hydrogen atoms from organic molecules • • Removes 2 hydrogen molecules at a time • 2 protons and 2 electrons H and e- delivered to NAD+ Coenzyme • Remaining H+ released into ECM • NAD+ → NADH • NADH neutral • Electrons lose little potential energy in this process • Stepping stone to make ATP • Electrons need to be delivered to oxygen •

Electron transport chain • Several energy releasing steps • “breaks the fall of electrons to oxygen” • • Transport is an exergonic reaction • Electrons lose a small controlled amount of energy at each step • Electronegativity of the carrier molecules “downhill” are higher • Move the electron through the chain Organic hydrogen in NADH + Oxygen by the end • Molecules on the inner membrane of the mitochondrion • Mostly proteins

Glycolysis • Occurs in cytosol • Degrades glucose into pyruvate Some steps are redox reactions • Produces NADH • • Substrate level phosphorylation Small amounts of ATP directly synthesized • Phosphate group given to an ADP •
• “splitting of sugar” • Six carbon sugar > 2 three carbon sugars • Oxidized and the rearranged into 2 pyruvate molecules • Ionized 3 pyruvic acid (3 carbon acid) • Ten steps Specific enzyme for each • First half • • • Energy investment Second half • Energy payoff
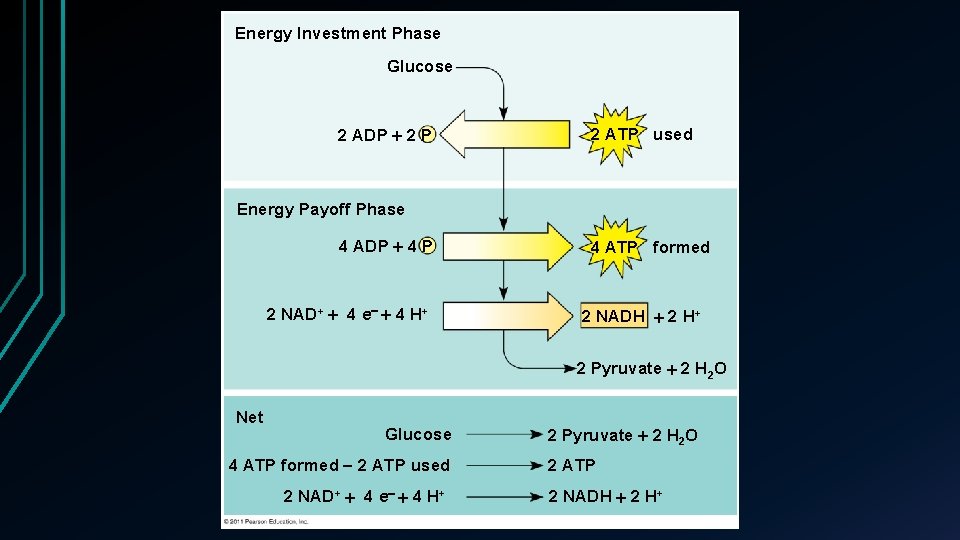
Energy Investment Phase Glucose 2 ADP 2 P 2 ATP used Energy Payoff Phase 4 ADP 4 P 2 NAD+ 4 e 4 H+ 4 ATP formed 2 NADH 2 H+ 2 Pyruvate 2 H 2 O Net Glucose 4 ATP formed 2 ATP used 2 NAD+ 4 e 4 H+ 2 Pyruvate 2 H 2 O 2 ATP 2 NADH 2 H+

• All carbon accounted for in pyruvate • Occurs with and without O₂ Without = fermentation • With = production of NADH and pyruvate • • Through Krebs and Electron transport turn into ATP
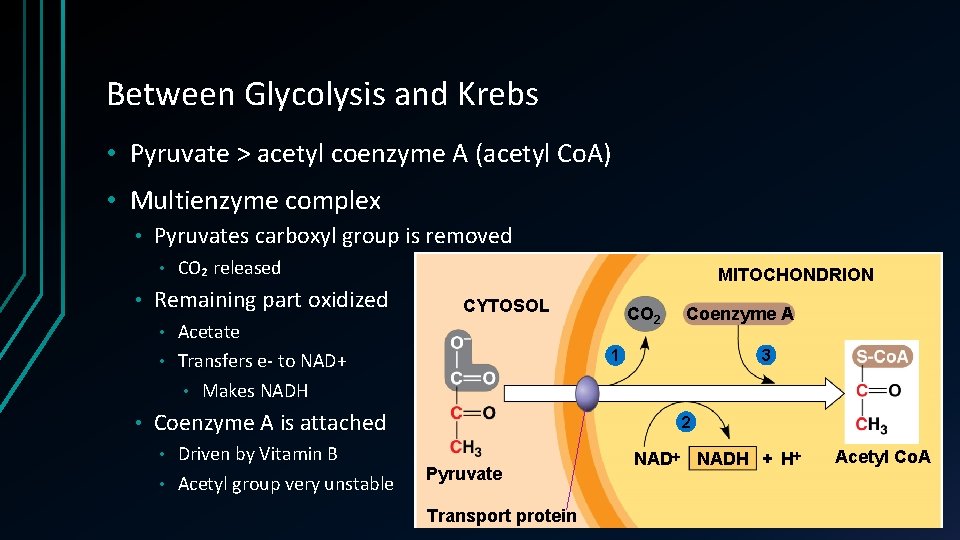
Between Glycolysis and Krebs • Pyruvate > acetyl coenzyme A (acetyl Co. A) • Multienzyme complex • Pyruvates carboxyl group is removed • • CO₂ released Remaining part oxidized MITOCHONDRION CYTOSOL Acetate • Transfers e- to NAD+ • Makes NADH CO 2 • • Driven by Vitamin B • Acetyl group very unstable 3 1 Coenzyme A is attached • Coenzyme A 2 Pyruvate Transport protein NADH + H Acetyl Co. A

Krebs Cycle • Occurs in the mitochondrial matrix • Need O₂ to enter the mitochondria • Decomposes pyruvate into carbon dioxide • Most energy from glucose stored in pyruvate • Substrate-level phosphorylation

Krebs Cycle • Citric Acid Cycle • 8 step process • Specific enzyme for each All enzymes in the mitochondrial matrix • Except Succinate in the inner mitochondrial wall • • Acetyl Co. A > 2 CO₂ molecules + 2 ATP
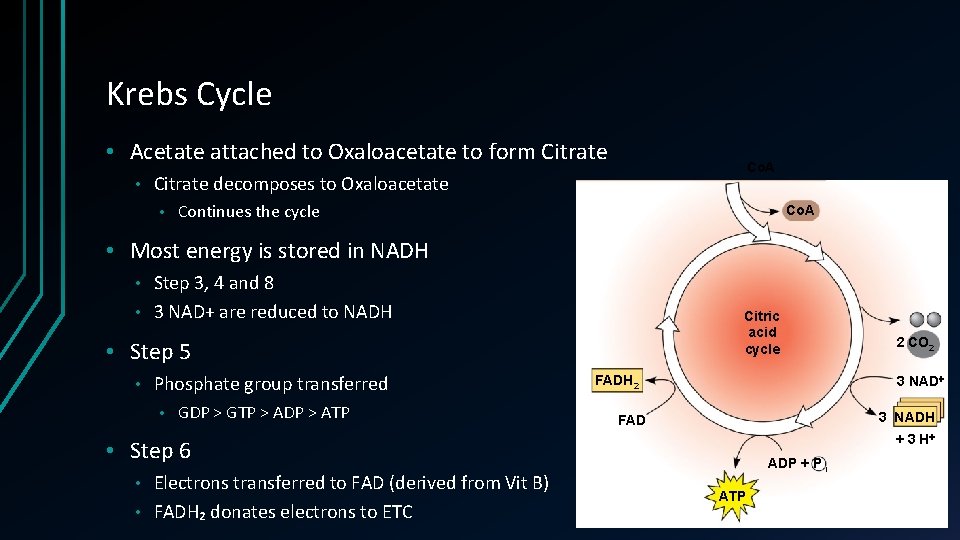
Krebs Cycle • Acetate attached to Oxaloacetate to form Citrate • Co. A Citrate decomposes to Oxaloacetate • Continues the cycle Co. A • Most energy is stored in NADH Step 3, 4 and 8 • 3 NAD+ are reduced to NADH • Citric acid cycle • Step 5 • Phosphate group transferred • GDP > GTP > ADP > ATP 3 NAD FADH 2 3 NADH FAD + 3 H • Step 6 Electrons transferred to FAD (derived from Vit B) • FADH₂ donates electrons to ETC • 2 CO 2 ADP + P i ATP
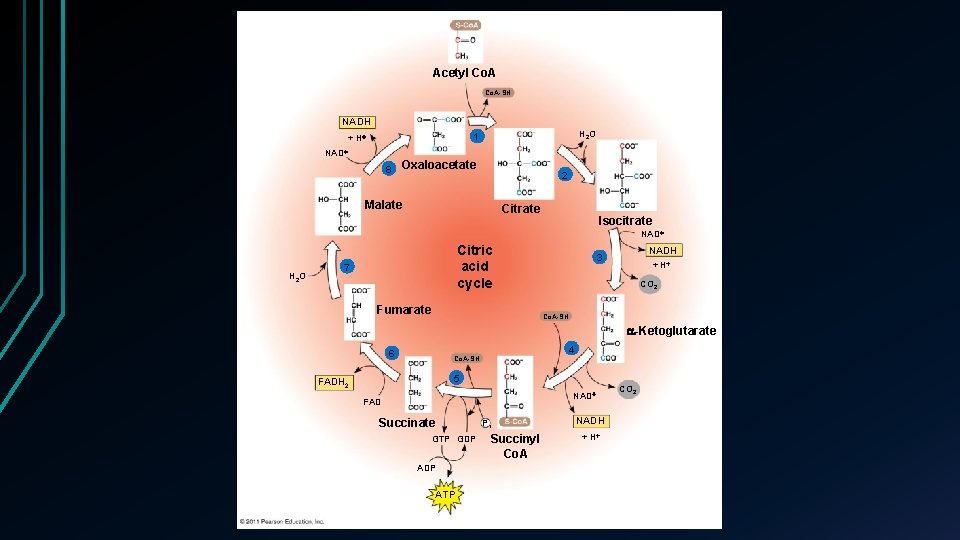
Acetyl Co. A-SH NADH H 2 O 1 + H NAD 8 Oxaloacetate 2 Malate Citrate Isocitrate NAD H 2 O Citric acid cycle 7 Fumarate NADH 3 + H CO 2 Co. A-SH -Ketoglutarate 6 4 Co. A-SH 5 FADH 2 NAD FAD Succinate GTP GDP ATP Pi Succinyl Co. A NADH + H CO 2

Electron transport chain • Oxidative phosphorylation ATP synthesized through redox reactions of the ETC • 90% of all ATP synthesis • 4 ATP produced by Glycolysis and Krebs together • 34 produced • • Occurs in the inner membrane of the mitochondria Folds of the cristae • Most components are proteins + prosthetic groups • • Non protein groups that aid in enzyme function

ETC pathway • Electrons transferred via NADH to a flavoprotein • Flavin mononucleotide attached • Flavoprotein passes electrons down to iron – sulfur protein • Iron – sulfur passes electrons down to ubiquione • Lipid • Remaining proteins are cytochromes • Heme group attached • 4 organic rings surrounding a iron atom

• Cyt a passes on electrons to an O₂ molecule • Picks up hydrogens from the surrounding environment • FADH₂ adds electrons at a lower energy level than NADH • Produces less energy
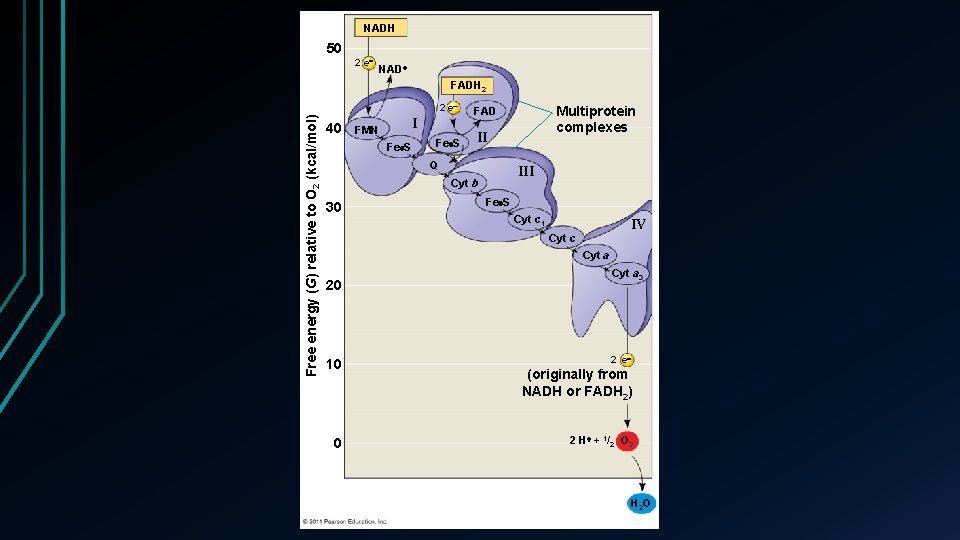
NADH 50 2 e NAD FADH 2 Free energy (G) relative to O 2 (kcal/mol) 2 e 40 FMN I Fe S II Q III Cyt b 30 Multiprotein complexes FAD Fe S Cyt c 1 IV Cyt c Cyt a 20 10 0 Cyt a 3 2 e (originally from NADH or FADH 2) 2 H + 1/2 O 2 H 2 O

Chemiosmosis • ATP synthase Enzyme • ADP + inorganic phosphate > ATP • Reverse ion pump • Uses energy of existing proton gradient • ETC pumps H+ ions across the membrane • Mitochondrial matrix to intermembrane • • Only part permeable for H+ Exergonic reaction • Uses energy fro oxidative phosphorylation • • Coupling ETC and ATP synthesis
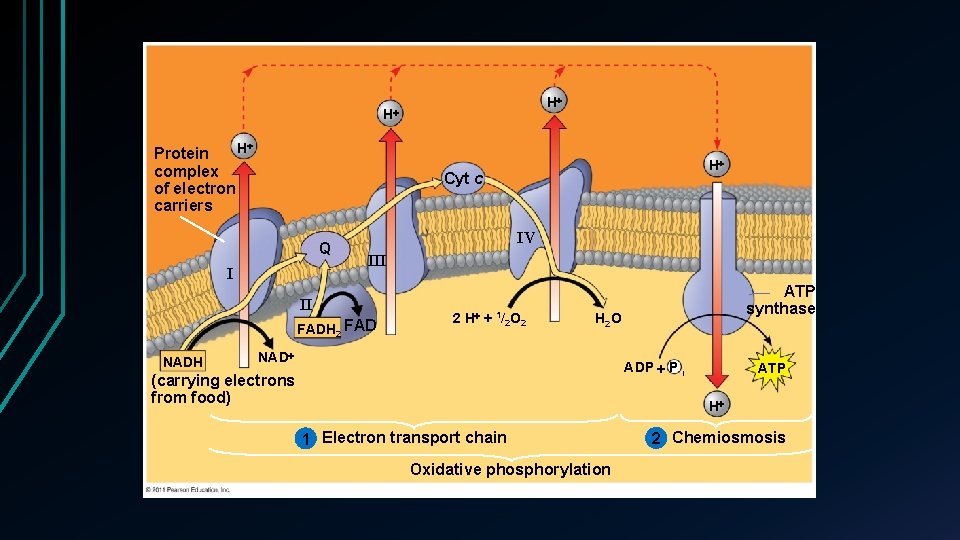
H H H Protein complex of electron carriers Cyt c Q I IV III II FADH 2 FAD NADH H 2 H + 1/2 O 2 ATP synthase H 2 O NAD ADP P i (carrying electrons from food) ATP H 1 Electron transport chain Oxidative phosphorylation 2 Chemiosmosis
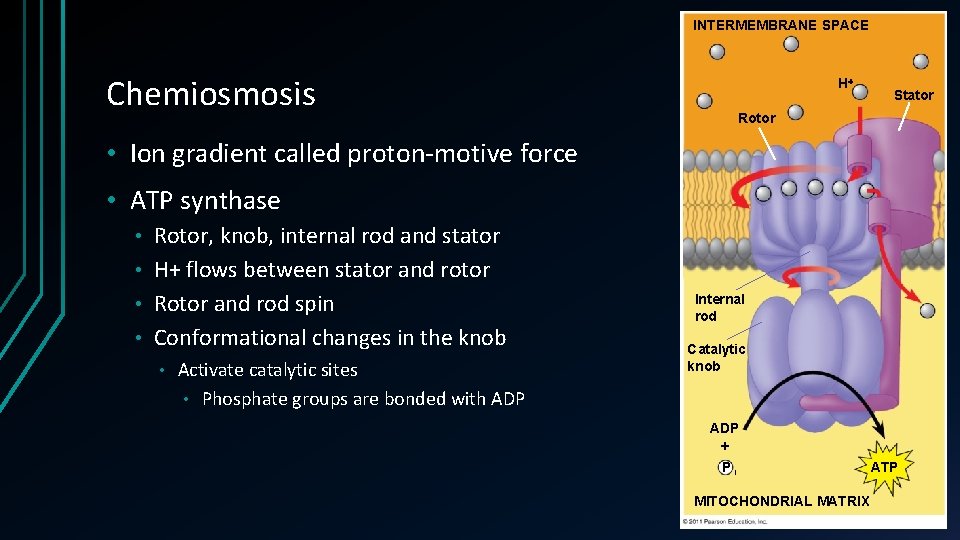
INTERMEMBRANE SPACE Chemiosmosis H Stator Rotor • Ion gradient called proton-motive force • ATP synthase Rotor, knob, internal rod and stator • H+ flows between stator and rotor • Rotor and rod spin • Conformational changes in the knob • • Activate catalytic sites • Phosphate groups are bonded with ADP Internal rod Catalytic knob ADP + Pi MITOCHONDRIAL MATRIX ATP

Chemiosmosis in other organisms • Plants Chloroplasts generate ATP during photosynthesis • Light drives ETC and H+ gradient • • Prokaryotes • H+ gradient across plasma membrane Produce ATP • Pump nutrients and waste across the membrane • Rotate flagella •
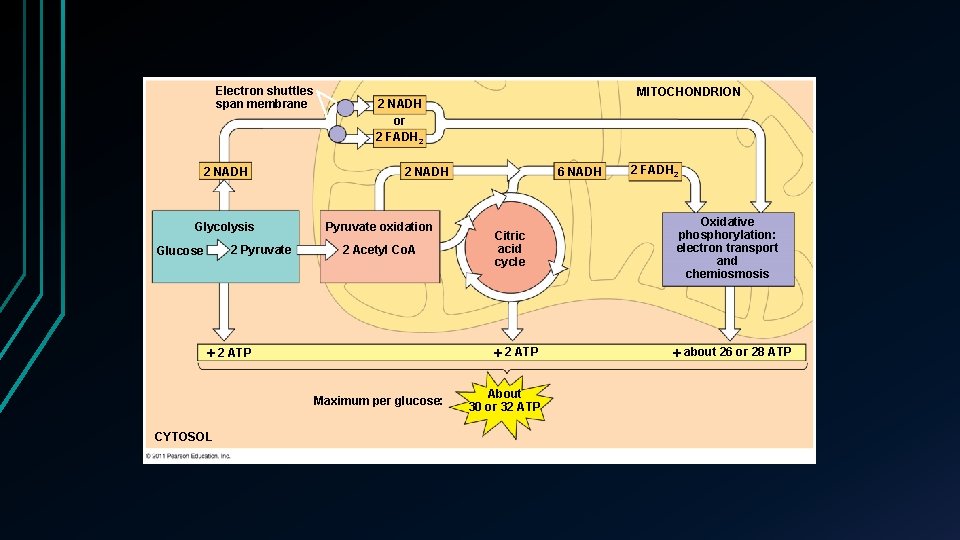
Electron shuttles span membrane 2 NADH Glycolysis 2 Pyruvate Glucose 2 NADH or 2 FADH 2 2 NADH Pyruvate oxidation 2 Acetyl Co. A 2 ATP Maximum per glucose: CYTOSOL MITOCHONDRION 6 NADH 2 FADH 2 Citric acid cycle Oxidative phosphorylation: electron transport and chemiosmosis 2 ATP about 26 or 28 ATP About 30 or 32 ATP

Aerobic • Glycolysis generates 2 ATP regardless of O₂ content • Oxidized by NAD+ • With oxygen • NADH > NAD+ through the ETC • Recycled

Anaerobic • Without oxygen • ETC doesn’t function Need a mechanism to convert NADH back to NAD+ • Transfers electrons from NADH to pyruvate • • Fermentation • Glycolysis + reactions needed to recycle NAD+ • NADH > pyruvate or derivatives • Substrate- level phosphorylation • Nets 2 ATP

Fermentation • Alcohol • Pyruvate > ethanol Carbon dioxide released from pyruvate • Produces acetaldehyde • Acetaldehyde reduced by NADH • NAD+ regenerated • Produces ethanol • • Yeast • • Used in brewing Bacteria use it to produce energy without oxyge

Fermentation • Lactic Acid • Pyruvate reduced by NADH • • Fungi + bacteria • • Cheese and yogurt Microbes • • Produces lactate without CO₂ Acetone and Methanol Muscle cells Sugar catabolism is faster than oxygen supply in blood • Muscle fatigue and pain • Converted back to pyruvate in the liver •

FERMENTATION RESPIRATION • Anaerobic • Aerobic • Produce ATP • Glycolysis • Oxidize glucose to pyruvate • 2 ATP substrate-level phosphorylation • • Recycling NAD+ • • • Final electron acceptor • pyruvate or acetaldehyde Energy in pyruvate unavailable to the cell • 2 ATP max • Glycolysis Recycling NAD+ • • Oxidative phosphorylation • • Through the ETC Krebs Cycle • • Final electron acceptor • Oxygen Oxidizes pyruvate 38 ATP max

Facultative anaerobes • Produce enough ATP to survive by either fermentation or respiration Bacteria and yeast • Muscle cells •

Evolutionary basis of glycolysis • ?

Versatility • Glycolysis can accept many types of carbohydrates • Starch hydrolyzed into glucose • • Through the digestive tract Glycogen hydrolyzed into glucose • Found usually in animals liver or muscle Disaccharides hydrolyzed into glucose and monosaccharides • Proteins digested into amino acids • Many used to build other proteins • Excess stripped of amino groups by enzymes • Deamination • Nitrogen groups excreted through ammonia, urea etc. •

• Fats Digested into glycerol • Converted into glyceraldehyde phosphate • Beta oxidization • • • Breaks down fatty acids into 2 carbon pieces • Enter Krebs Cycle as acetyl Co. A Each gram produces twice as much energy than a gram of Carbs

Biosynthesis • Intermediates of glycolysis and Krebs cycle • Diverted into anabolic pathways Amino acids from digested proteins • Pyruvate > glucose • Acetyl Co. A > fatty acids • • Metabolic interchanges • Carbs and proteins can be converted into fat • Too many calories

Feedback mechanisms • Feedback inhibition • End product stops the catalyst of the first step • Respiration can speed up or slow down depending on ATP demand • Phosphofructokinase • Catalyzes step 3 of glycolysis • • Controlling rate of this enzyme = controlling the rate of glycolysis • Inhibited by ATP • Stimulated by AMP • Sensitive to Citrate • Correlates rate of glycolysis to rate of Krebs cycle Commits a substrate to the pathway
- Slides: 37