Cell Membranes Transport Communication 1 Cell Membrane Most

Cell Membranes, Transport & Communication 1
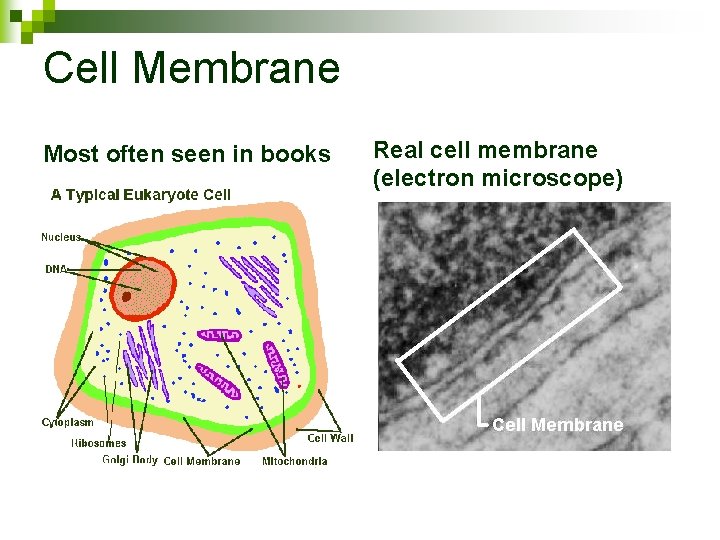
Cell Membrane Most often seen in books Real cell membrane (electron microscope)
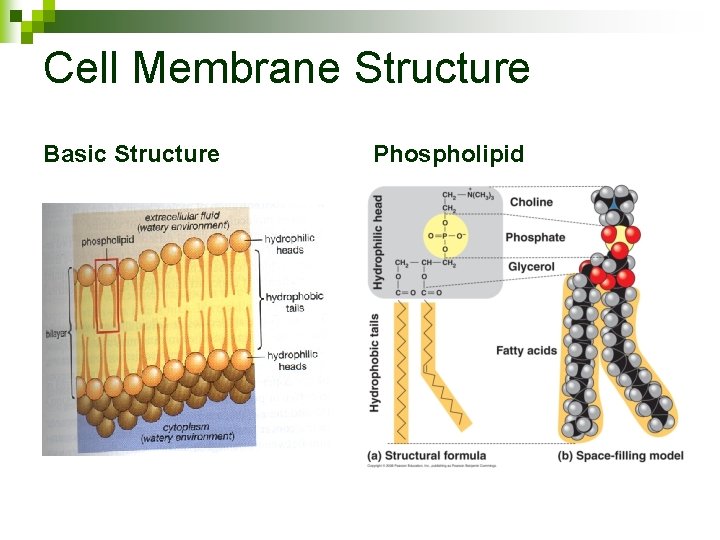
Cell Membrane Structure Basic Structure Phospholipid
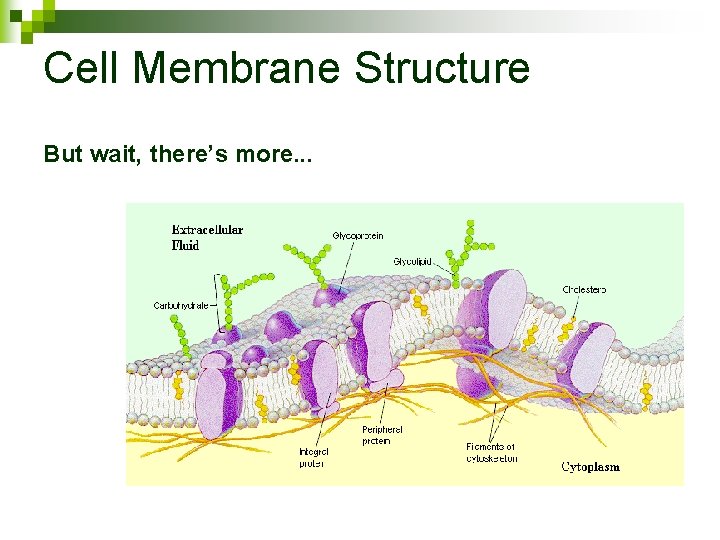
Cell Membrane Structure But wait, there’s more. . .
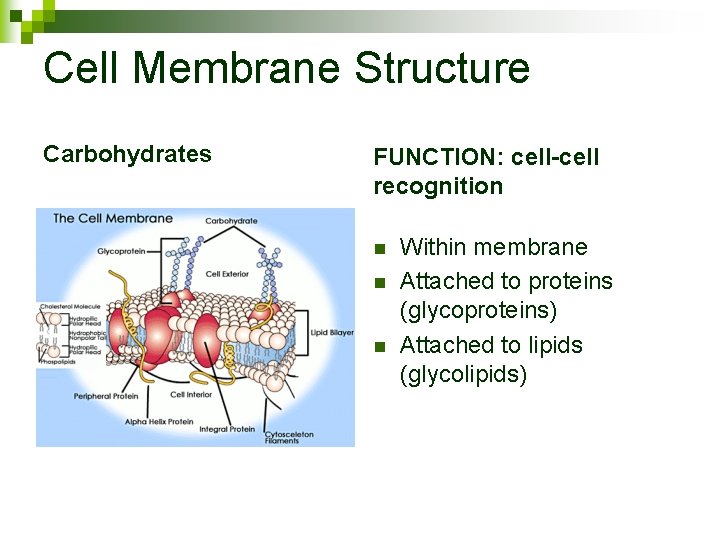
Cell Membrane Structure Carbohydrates FUNCTION: cell-cell recognition n Within membrane Attached to proteins (glycoproteins) Attached to lipids (glycolipids)
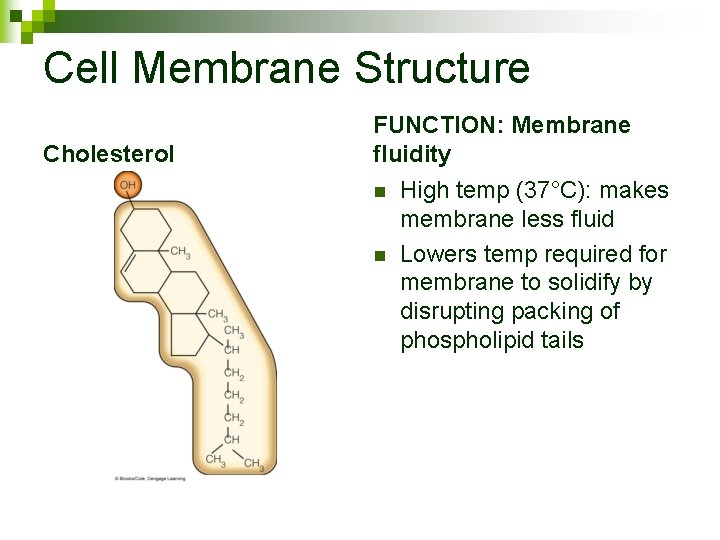
Cell Membrane Structure Cholesterol FUNCTION: Membrane fluidity n n High temp (37°C): makes membrane less fluid Lowers temp required for membrane to solidify by disrupting packing of phospholipid tails

Cholesterol and membrane fluidity n Remember lipid structure ¨ Saturated fats = no double bonds, straight tails, pack tightly ¨ Unsaturated fats = double bonds, kinked tails, pack loosely
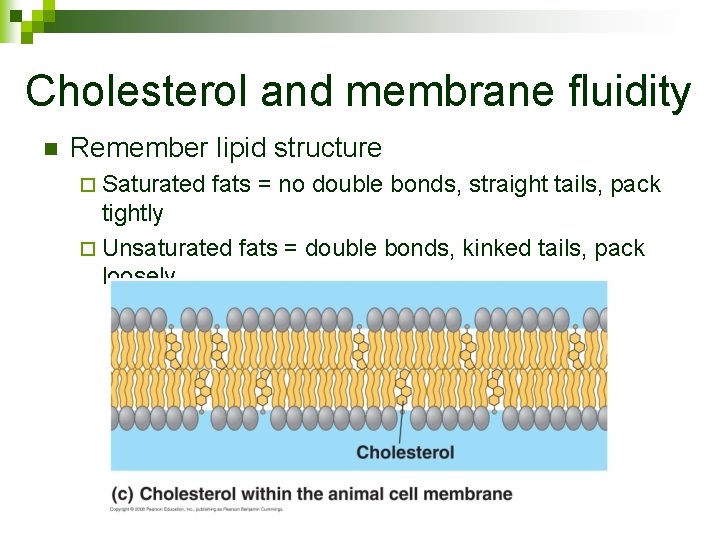
Cholesterol and membrane fluidity n Remember lipid structure ¨ Saturated fats = no double bonds, straight tails, pack tightly ¨ Unsaturated fats = double bonds, kinked tails, pack loosely

Membrane Fluidity n n Membranes must be fluid to function Different organisms have different compositions in their membranes ¨ Types of proteins ¨ Types of phospholipids ¨ Amount of cholesterol
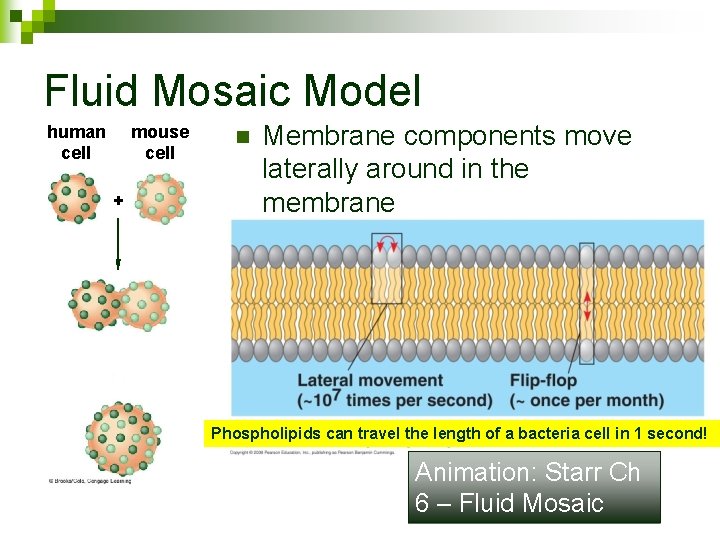
Fluid Mosaic Model human cell mouse cell + n Membrane components move laterally around in the membrane Phospholipids can travel the length of a bacteria cell in 1 second! Animation: Starr Ch 6 – Fluid Mosaic
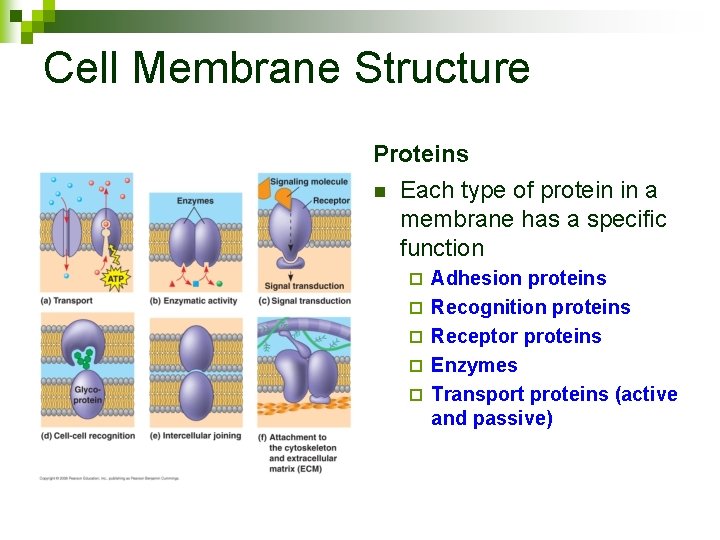
Cell Membrane Structure Proteins n Each type of protein in a membrane has a specific function ¨ ¨ ¨ Adhesion proteins Recognition proteins Receptor proteins Enzymes Transport proteins (active and passive)

Cell Membrane Structure Adhesion Protein Enzyme Receptor Protein Recognition Protein Passive Transporter Active Transporter
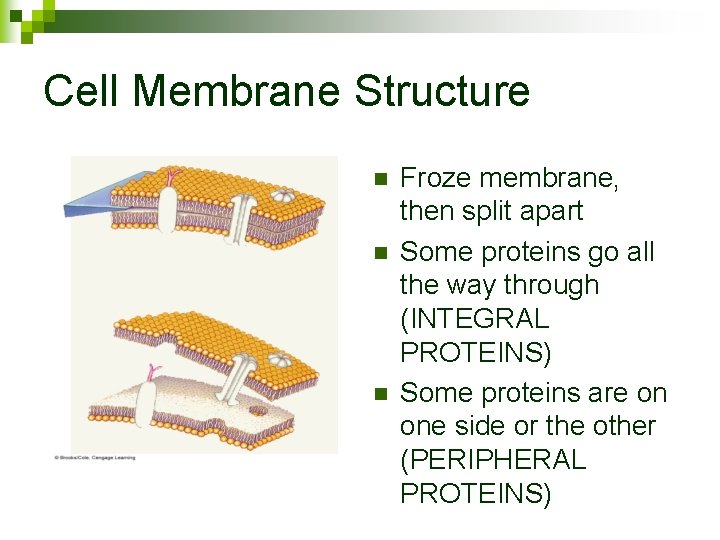
Cell Membrane Structure n n n Froze membrane, then split apart Some proteins go all the way through (INTEGRAL PROTEINS) Some proteins are on one side or the other (PERIPHERAL PROTEINS)
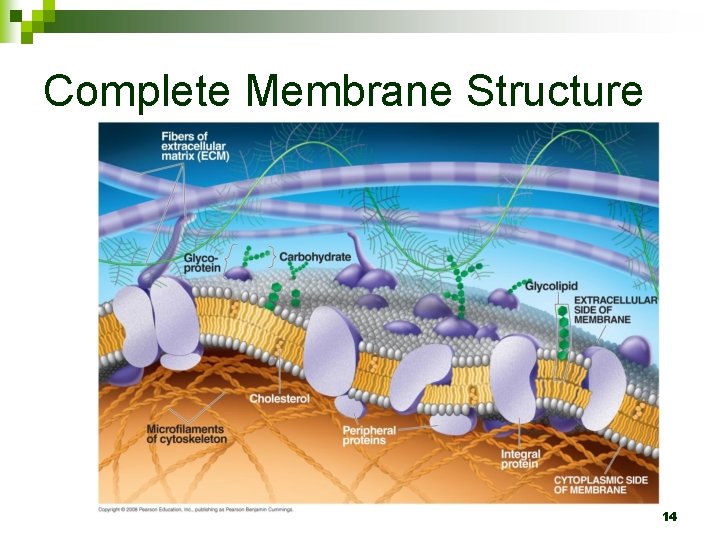
Complete Membrane Structure 14
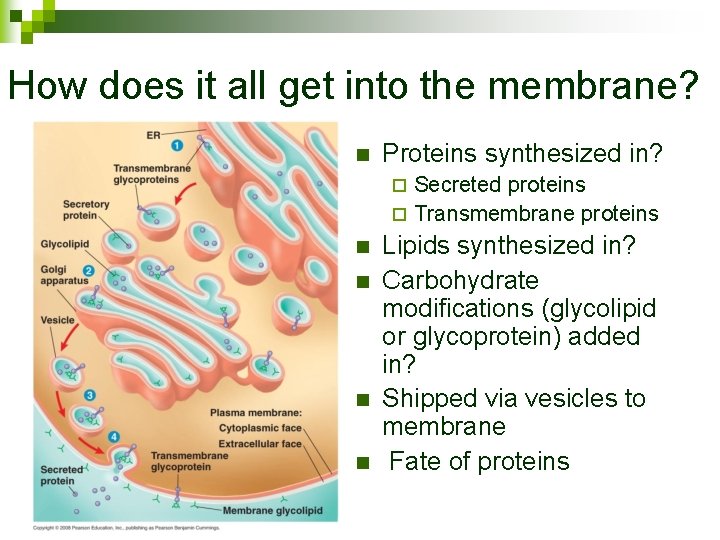
How does it all get into the membrane? n Proteins synthesized in? Secreted proteins ¨ Transmembrane proteins ¨ n n Lipids synthesized in? Carbohydrate modifications (glycolipid or glycoprotein) added in? Shipped via vesicles to membrane Fate of proteins

Cell Membrane: Gate and Gatekeeper Structure allows cell to control what can pass through the membrane n Selectively permeable barrier n Allows some things through but not others 16
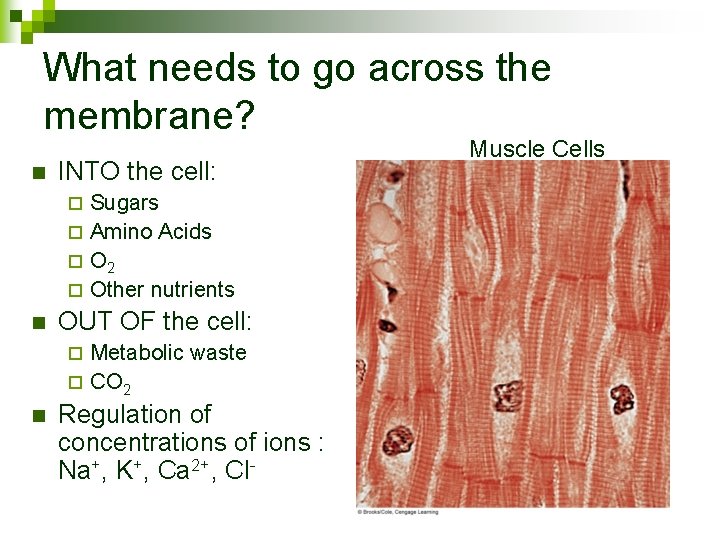
What needs to go across the membrane? n INTO the cell: Sugars ¨ Amino Acids ¨ O 2 ¨ Other nutrients ¨ n OUT OF the cell: Metabolic waste ¨ CO 2 ¨ n Regulation of concentrations of ions : Na+, K+, Ca 2+, Cl- Muscle Cells
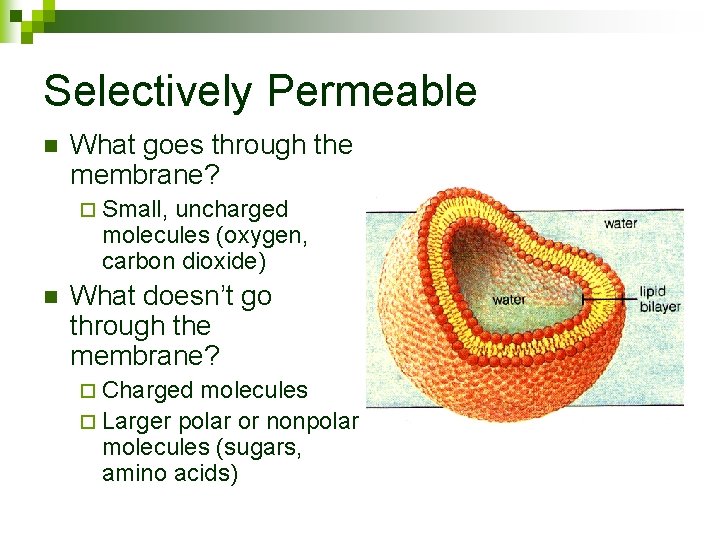
Selectively Permeable n What goes through the membrane? ¨ Small, uncharged molecules (oxygen, carbon dioxide) n What doesn’t go through the membrane? ¨ Charged molecules ¨ Larger polar or nonpolar molecules (sugars, amino acids)
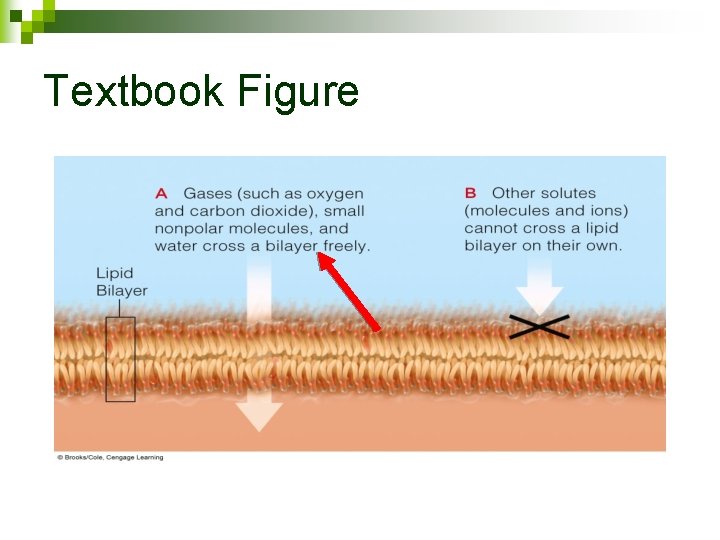
Textbook Figure

Aquaporins – “water channels” Allows 3 x 109 water molecules to pass through membrane per second

Concentration Gradients Concentration n The amount of something in a certain space Concentration Gradient

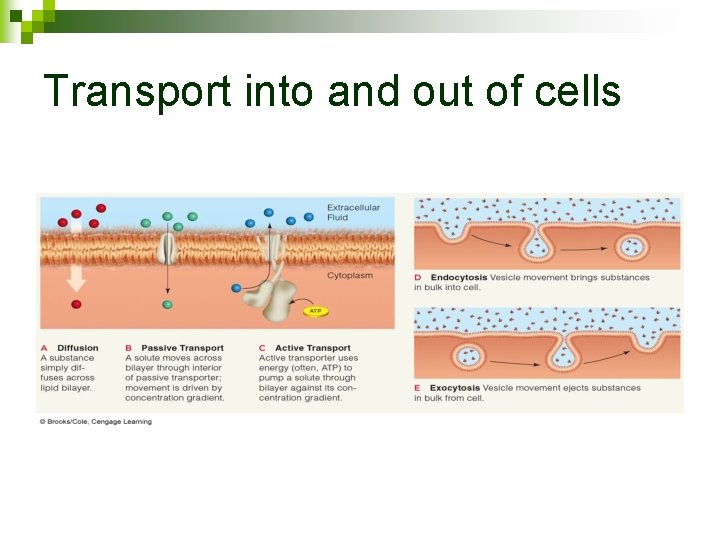
Movement of substances into and out of cells GENERAL RULE #1: n Movement from HIGH concentration to LOW concentration does not require energy 22

Passive Transport

Passive Transport

What can be Passively Transported? Membrane is selectively permeable to small molecules (oxygen, carbon dioxide) n Larger molecules and ions enter through protein carriers n Small Molecules Ion channels Carrier proteins 25

What can be Passively Transported? n Membrane is selectively permeable to small molecules (oxygen, carbon dioxide) ¨ DIFFUSION – movement from high concentration to low concentration (passive) © 2006 W. W. Norton & Company, Inc. DISCOVER BIOLOGY 3/e 26
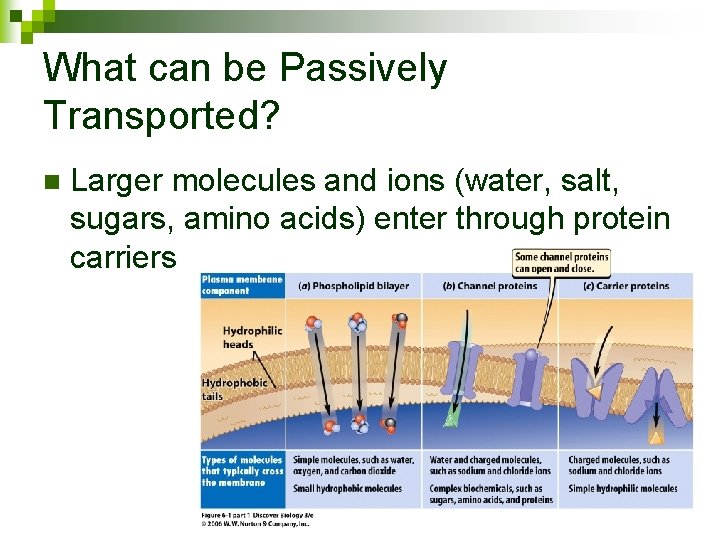
What can be Passively Transported? n Larger molecules and ions (water, salt, sugars, amino acids) enter through protein carriers

Osmosis n Water moving passively across the membrane 2% sucrose 10% sucrose water 28
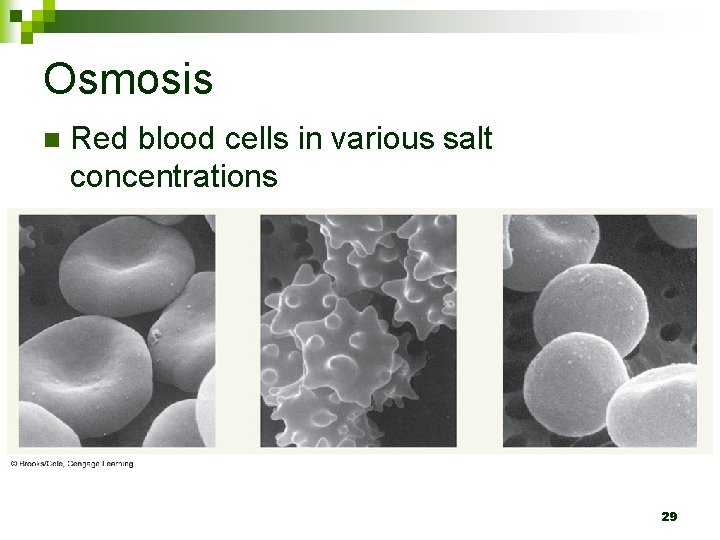
Osmosis n Red blood cells in various salt concentrations 29

Tonicity Ability of a solution to cause a change in water content via osmosis n TONIC refers to the SOLUTE n Same concentration of solute inside and outside the cell Salt concentration outside HIGHER than inside the cell Salt concentration outside LOWER than inside the cell
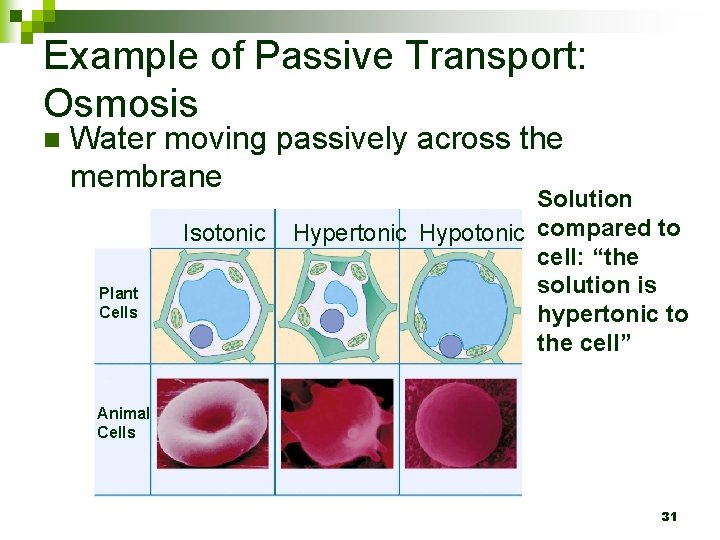
Example of Passive Transport: Osmosis n Water moving passively across the membrane Isotonic Plant Cells Solution Hypertonic Hypotonic compared to cell: “the solution is hypertonic to the cell” Animal Cells 31

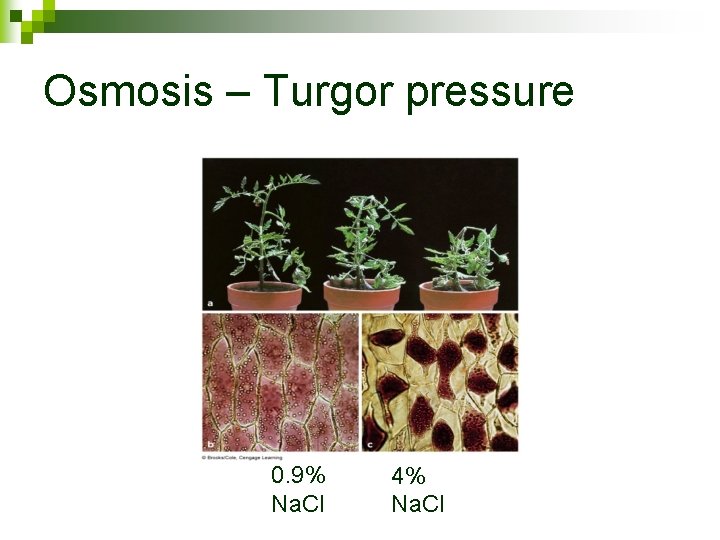
Osmosis – Turgor pressure

Factors that affect passive transport Size n Temperature n Steepness of the concentration gradient n Charge n Pressure n

Movement of substances into and out of cells GENERAL RULE #2: n Movement from LOW concentration to HIGH concentration requires added energy 34

Active Transport

What must be Actively Transported? n n n Anything moving against the concentration gradient (low to high) Active carriers use energy from ATP Energy changes the shape of the carrier Energy 36

Example of Active Transport: Endocytosis and Exocytosis n n 37 EXOCYTOSIS: Outward budding of the membrane Forms a vesicle Remove waste, release molecules the cell has made Exocytosis Animation

Example of Active Transport: Endocytosis and Exocytosis n n n 38 ENDOCYTOSIS: Inward budding of the membrane Forms a vesicle Take in food, remove cholesterol from blood, immune cells “eat” invaders ANIMATION: Endocytosis Cain Ch 6 2 b
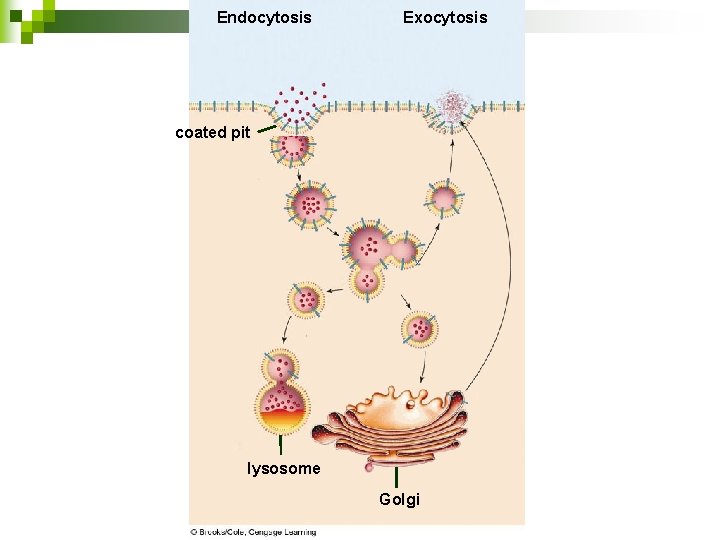
Endocytosis Exocytosis coated pit lysosome Golgi
Transport into and out of cells

Example in Real Life n Paramecium are a single-celled organism that is found in freshwater ponds. Which direction would osmosis (passive transport) cause water to move with respect to the Paramecium? WATER water

Example in Real Life n What would happen to the Paramecium if this continued? >BOOM<

Example in Real Life n The contractile vacuole of a Paramecium constantly pumps water out of the cell. Is this active or passive transport?

Example in Real Life n You have probably heard that eating a lot of salt will make your body retain water so you gain weight. When you eat a lot of salt, it is transported inside your cells. Why would eating a lot of salt make your body retain water? WATER SALT water

Example in Real Life n A shipwrecked sailor is stranded on a small desert island with no fresh water to drink. She knows she could last without food for up to a month, but if she didn't have water to drink she would be dead within a week. Hoping to postpone the inevitable, her thirst drove her to drink the salty seawater. She was dead in two days. Why did drinking seawater kill the sailor faster than not drinking any water at all?

Example in Real Life n Describe why oxygen would diffuse into cells inside your lungs.

Example in Real Life n Why does putting salt on a slug kill it?

Scientific Method Practice OBSERVATION: Cells gain or lose water via osmosis when placed in different concentrations of salt. n Design an experiment to determine the concentration of salt inside a human red blood cell. n Hypothesis, Prediction, Independent variable, control, constants

Scientific Method Practice n n n Hypothesis: RBCs contain 5% salt. Prediction: HYP TRUE - RBCs in <5% salt will swell, RBCs in >5% salt will shrink. HYP FALSE – other observation about cell size Independent variable: % salt in water Dependent variable: size of cell (shrink, grow) Control: RBC in blood serum (shouldn’t change) Constants: source of RBCs, amount of liquid added, time after addition, # of RBCs observed
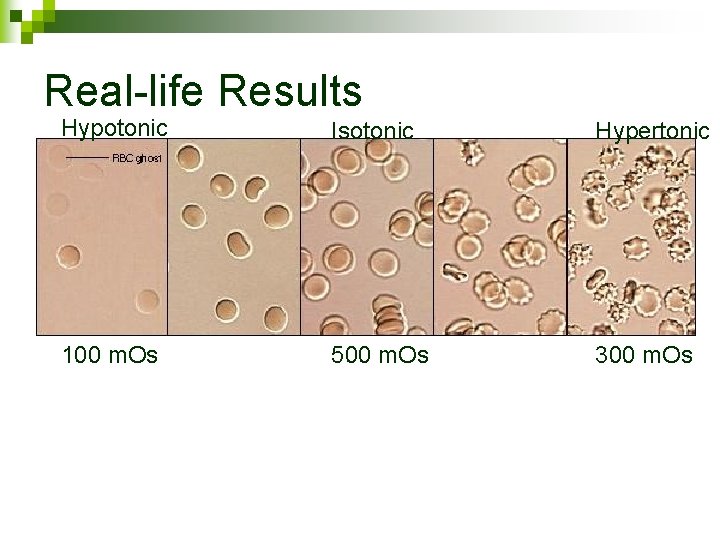
Real-life Results Hypotonic Isotonic Hypertonic 100 m. Os 500 m. Os 300 m. Os

Cell Communication in Multicellular Organisms Cells are specialized for different jobs, but cooperate so organism functions n Cells need to coordinate their activities n ¨ Neighboring cells communicate via direct connections ¨ Distant cells communicate via chemicals: hormones 51
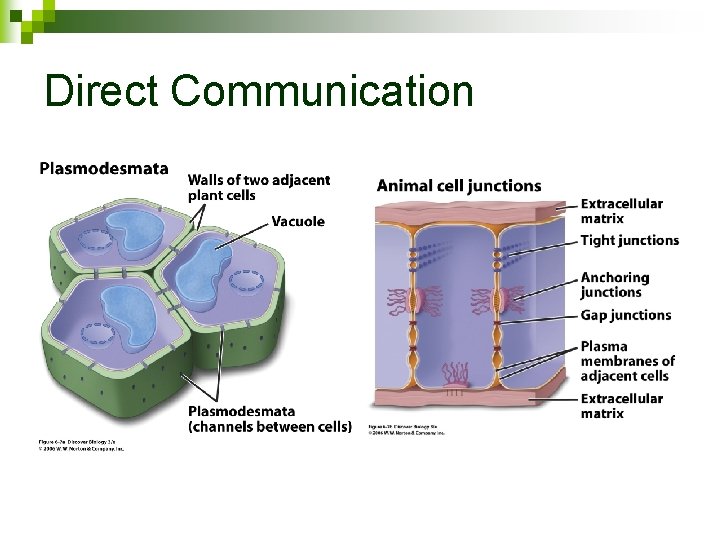
Direct Communication

Indirect Communication n Cells release small proteins or molecules ¨ Hormones, Pheromones, Steroids, Neurotransmitters Other cells have protein receptors n Signaling molecules produce changes in the receiving cell ANIMATION: Campbell n Ch 11 – Signaling Overview 53
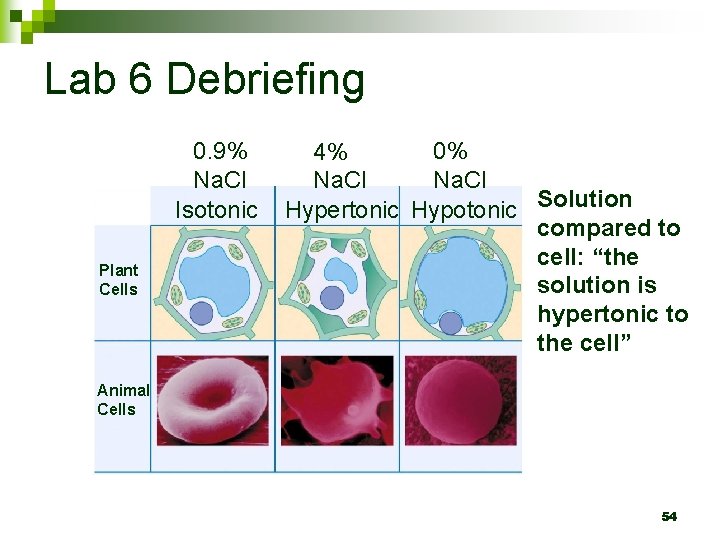
Lab 6 Debriefing 0. 9% Na. Cl Isotonic Plant Cells 0% 4% Na. Cl Hypertonic Hypotonic Solution compared to cell: “the solution is hypertonic to the cell” Animal Cells 54
Osmosis – Turgor pressure 0. 9% Na. Cl 4% Na. Cl
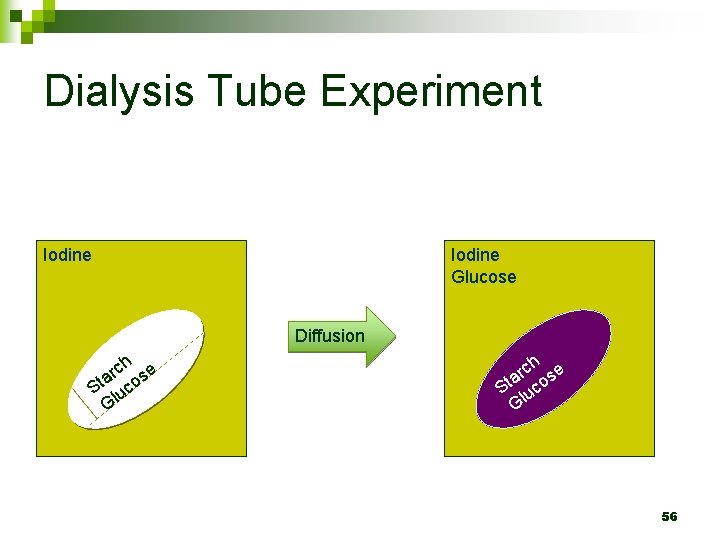
Dialysis Tube Experiment Iodine Glucose Diffusion ch se r a St luco G 56

Photosynthesis (briefly) and Cellular Respiration 57
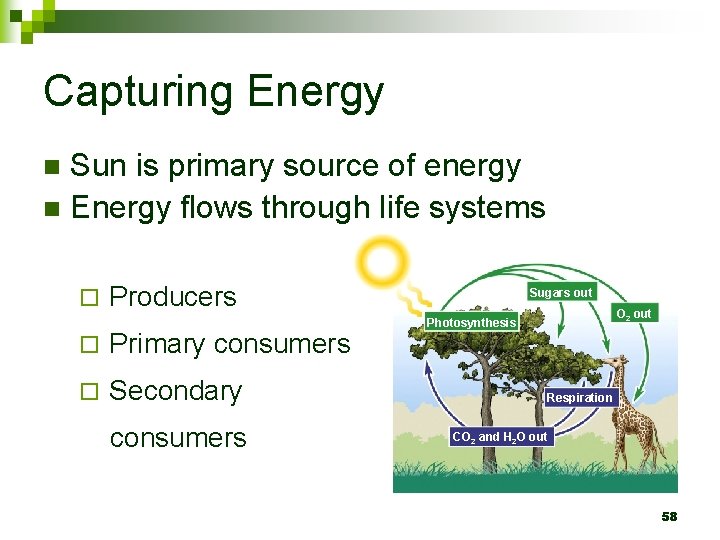
Capturing Energy Sun is primary source of energy n Energy flows through life systems n ¨ Producers ¨ Primary consumers ¨ Secondary consumers Sugars out O 2 out Photosynthesis Respiration CO 2 and H 2 O out 58

What this means… All energy needs are met by the plant kingdom n All this energy originally comes from the sun n Photosynthesis converts the energy in sunlight into chemical energy that can be stored in the plant n 59
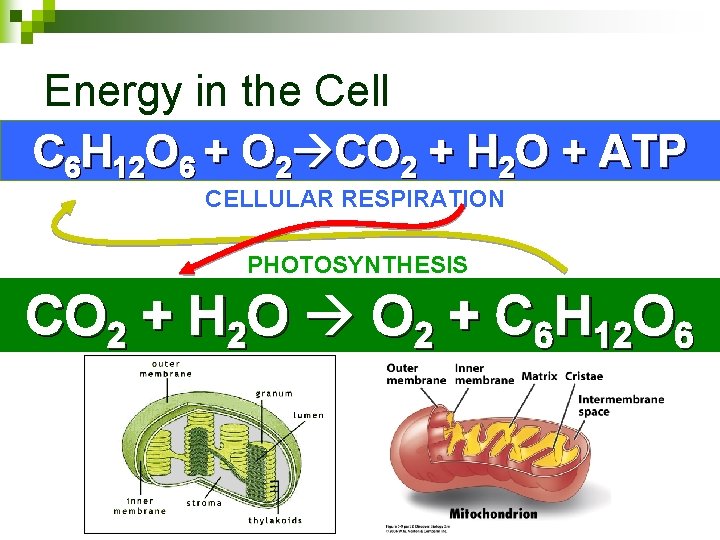
Energy in the Cell C 6 H 12 O 6 + O 2 CO 2 + H 2 O + ATP CELLULAR RESPIRATION PHOTOSYNTHESIS CO 2 + H 2 O O 2 + C 6 H 12 O 6
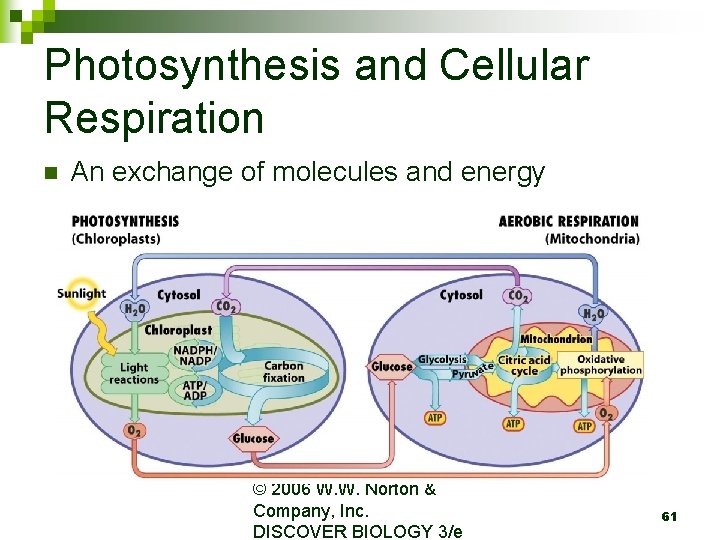
Photosynthesis and Cellular Respiration n An exchange of molecules and energy © 2006 W. W. Norton & Company, Inc. DISCOVER BIOLOGY 3/e 61
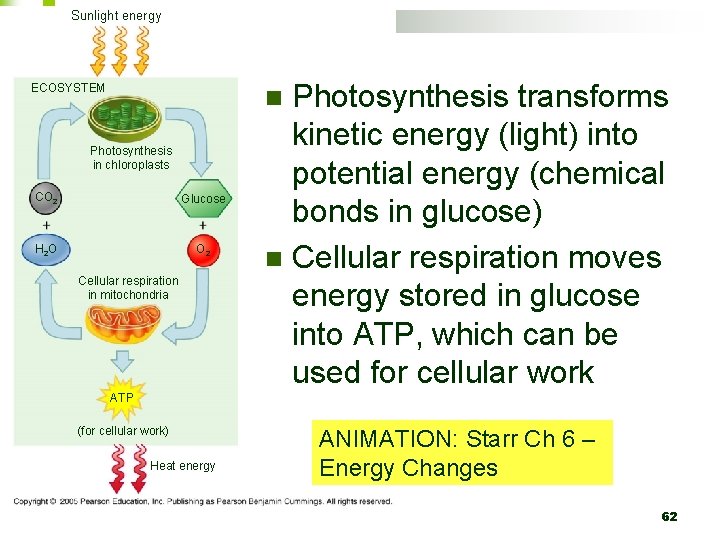
Sunlight energy ECOSYSTEM Photosynthesis transforms kinetic energy (light) into potential energy (chemical bonds in glucose) n Cellular respiration moves energy stored in glucose into ATP, which can be used for cellular work n Photosynthesis in chloroplasts CO 2 Glucose H 2 O O 2 Cellular respiration in mitochondria ATP (for cellular work) Heat energy ANIMATION: Starr Ch 6 – Energy Changes 62
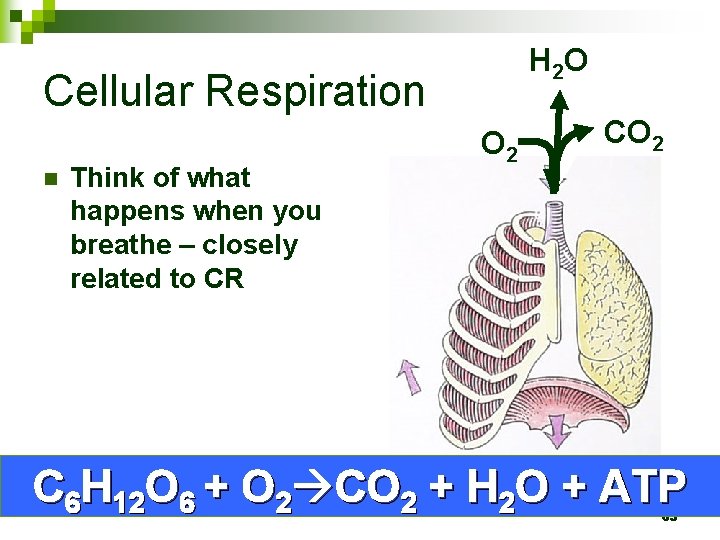
H 2 O Cellular Respiration n Think of what happens when you breathe – closely related to CR O 2 C 6 H 12 O 6 + O 2 CO 2 + H 2 O + ATP 63

Cellular Respiration and Gasses Breathing brings O 2 into the body from the environment n O 2 is distributed to cells in the bloodstream n In cellular respiration, mitochondria use O 2 to harvest energy and generate ATP n Breathing disposes of the CO 2 produced as a waste product of cellular respiration n 64

Summary Equation n Remember laws of thermodynamics! ¨ First law? ¨ Second law? HEAT Glucose Oxygen gas Carbon dioxide Water Energy 65
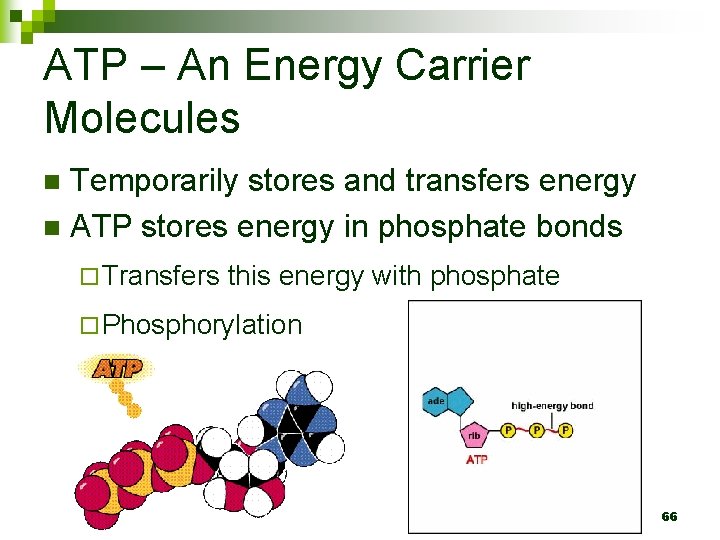
ATP – An Energy Carrier Molecules Temporarily stores and transfers energy n ATP stores energy in phosphate bonds n ¨ Transfers this energy with phosphate ¨ Phosphorylation 66
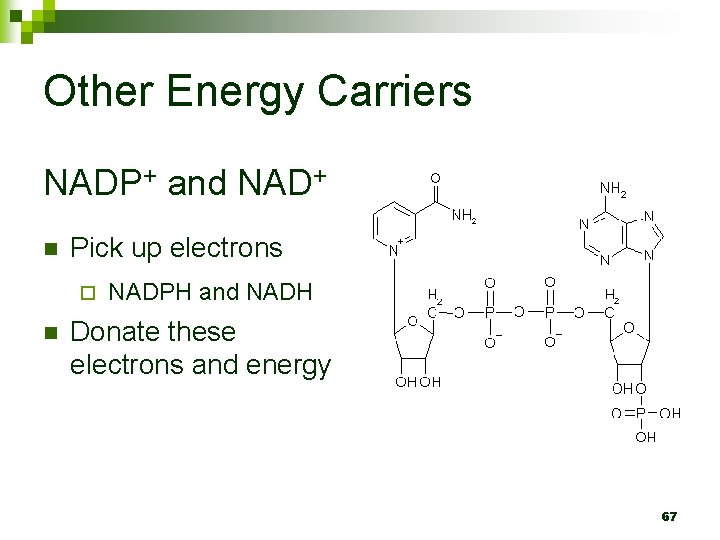
Other Energy Carriers NADP+ and NAD+ n Pick up electrons ¨ n NADPH and NADH Donate these electrons and energy 67
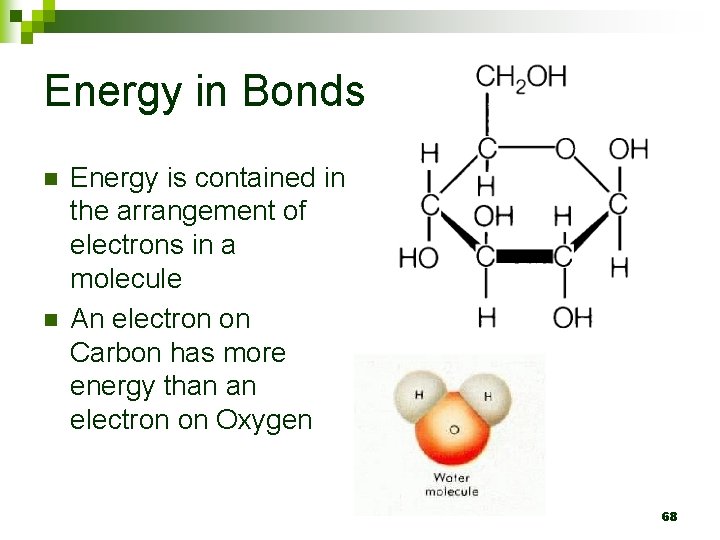
Energy in Bonds n n Energy is contained in the arrangement of electrons in a molecule An electron on Carbon has more energy than an electron on Oxygen 68

Burning sugar n n Electrons “fall” from carbon in glucose to oxygen in water Energy released rapidly as light and heat 69
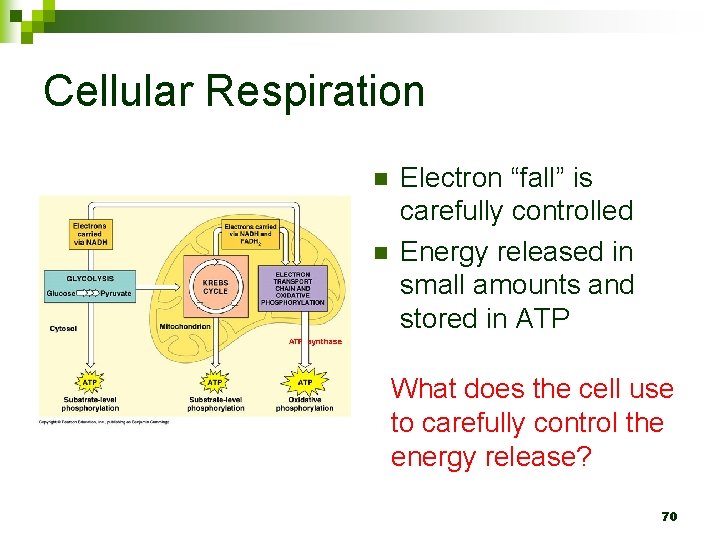
Cellular Respiration n n Electron “fall” is carefully controlled Energy released in small amounts and stored in ATP What does the cell use to carefully control the energy release? 70

C 6 H 12 O 6 + O 2 CO 2 + H 2 O + ATP Where are these electrons that are moving? n What is moving from a carbon to an oxygen? n 71

Oxidation – Reduction Reactions Oxidation: loss of electrons from an atom (loss of a H atom) n Reduction: addition of electrons to an atom (gain of a H atom) n ¨ Think: n reduction in CHARGE due to more e- Always paired (one loses an e-, one gains) 72

C 6 H 12 O 6 + O 2 CO 2 + H 2 O + ATP What molecule gets OXIDIZED (loses e-)? n What molecule gets REDUCED (gains e-)? n 73
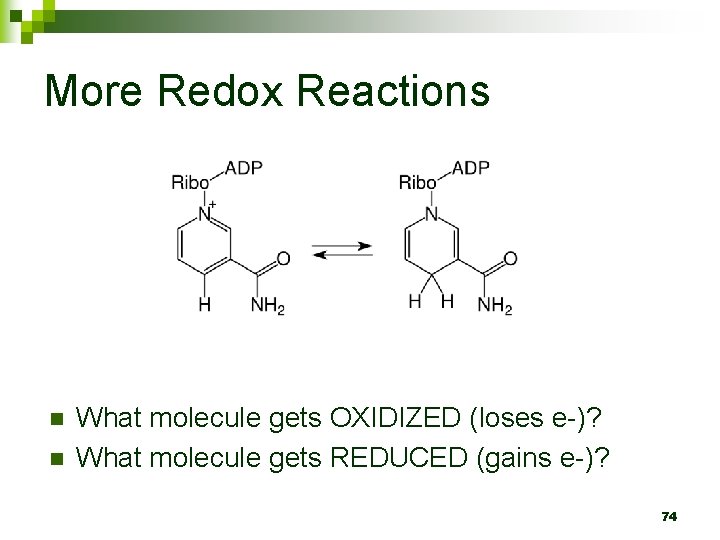
More Redox Reactions n n What molecule gets OXIDIZED (loses e-)? What molecule gets REDUCED (gains e-)? 74

Redox Reactions in Cellular Respiration n Glucose loses electrons (in H atoms) and becomes oxidized 75
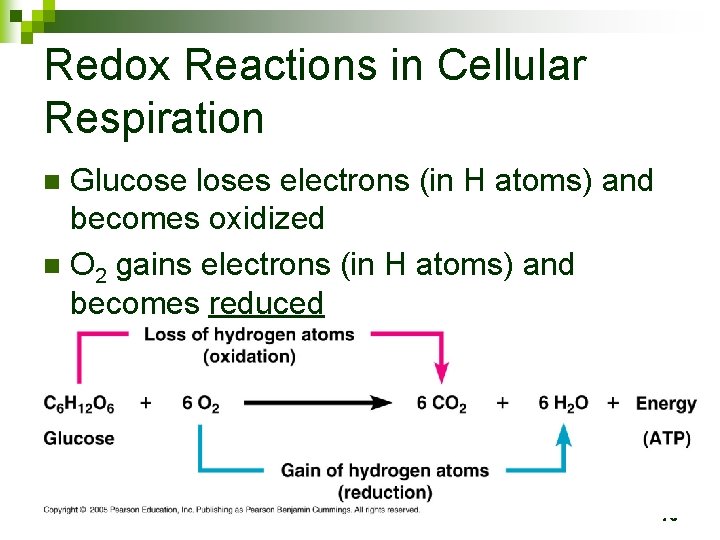
Redox Reactions in Cellular Respiration Glucose loses electrons (in H atoms) and becomes oxidized n O 2 gains electrons (in H atoms) and becomes reduced n Along the way, the electrons lose potential energy, and energy is released n 76

Redox Reactions in Cellular Respiration Glucose loses electrons (in H atoms) and becomes oxidized n O 2 gains electrons (in H atoms) and becomes reduced n Electrons lose potential energy, and energy is released n 77

Important Players n Dehydrogenase removes electrons from glucose ¨ What type of molecule is dehydrogenase? ¨ How are the electrons removed? 78
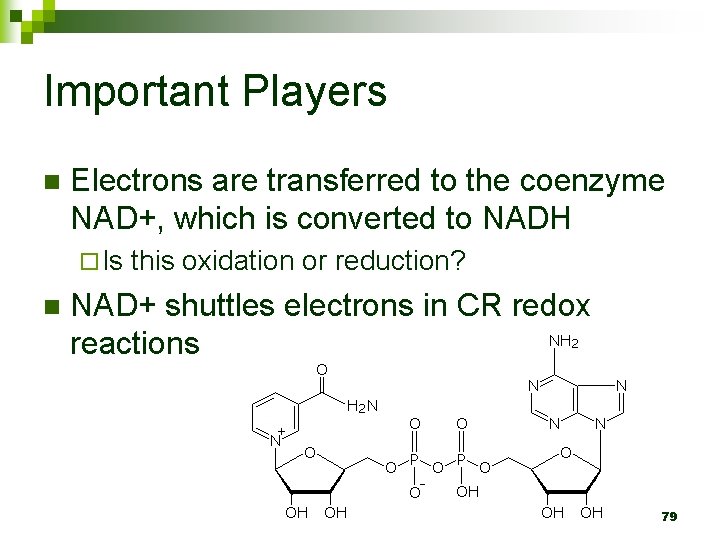
Important Players n Electrons are transferred to the coenzyme NAD+, which is converted to NADH ¨ Is n this oxidation or reduction? NAD+ shuttles electrons in CR redox reactions 79
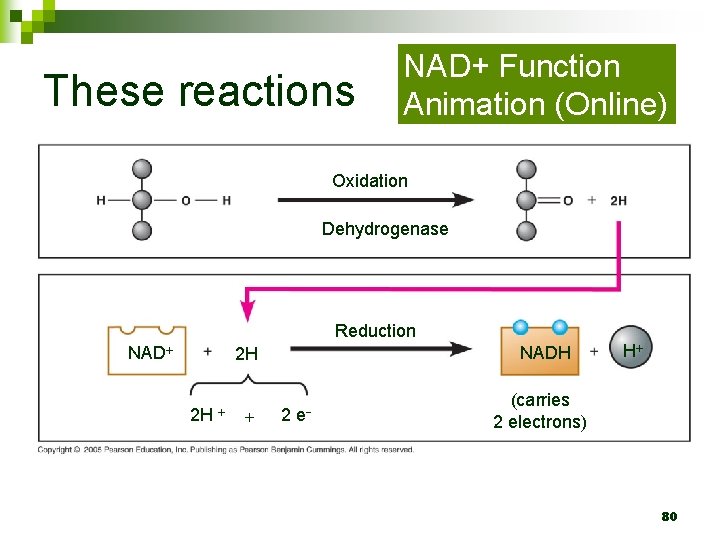
These reactions NAD+ Function Animation (Online) Oxidation Dehydrogenase Reduction NADH 2 H 2 H 2 e H (carries 2 electrons) 80
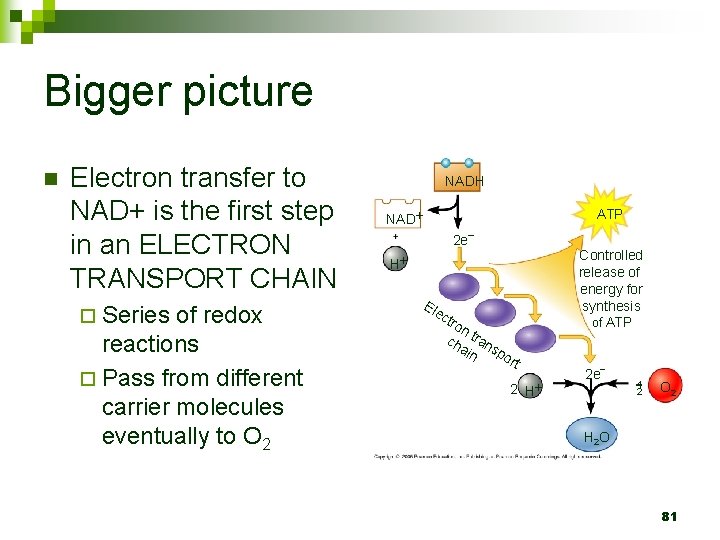
Bigger picture n Electron transfer to NAD+ is the first step in an ELECTRON TRANSPORT CHAIN ¨ Series of redox reactions ¨ Pass from different carrier molecules eventually to O 2 NADH ATP NAD 2 e Controlled release of energy for synthesis H Ele c tro n ch tran ain sp or of ATP t 2 H 2 e 1 2 O 2 H 2 O 81

Cellular Respiration n STEP 0: Eat to get glucose ¨ Glucose is absorbed by cells in small intestine ¨ Glucose enters the bloodstream and is transported to all the cells in your body ¨ How is this different in a plant? 82
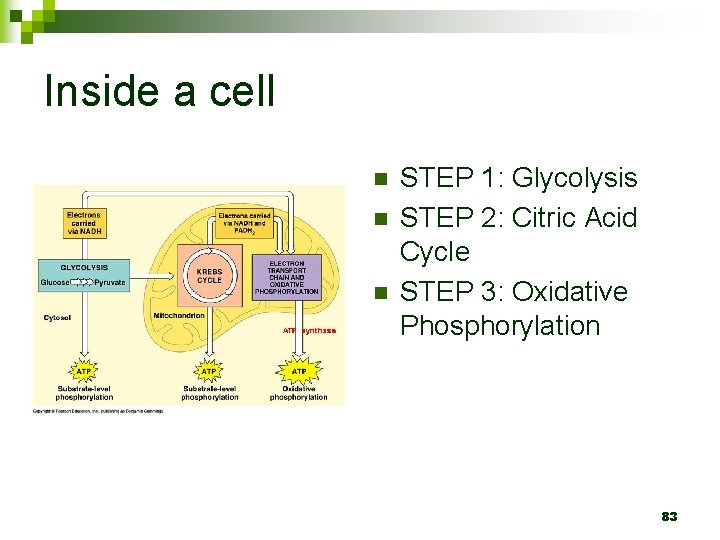
Inside a cell n n n STEP 1: Glycolysis STEP 2: Citric Acid Cycle STEP 3: Oxidative Phosphorylation 83
Cellular Respiration GLUCOSE 84
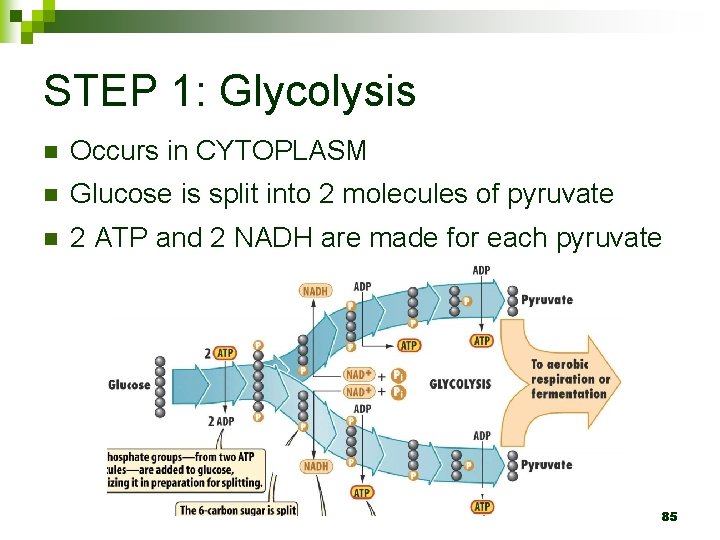
STEP 1: Glycolysis n Occurs in CYTOPLASM n Glucose is split into 2 molecules of pyruvate n 2 ATP and 2 NADH are made for each pyruvate 85

STEP 1: Glycolysis Animation: Campbell Ch 6 – Glycolysis GLYCOLYSIS ANIMATION (online) 86

STEP 1: Glycolysis Summary n Preparatory phase: ¨ Glucose is energized using 2 ATPs ¨ Splits into 2 three-carbon intermediates 87

STEP 1: Glycolysis Summary n Energy Payoff phase: ¨ Redox reaction generates NADH ¨ 2 ATP and pyruvate produced per intermediate 88
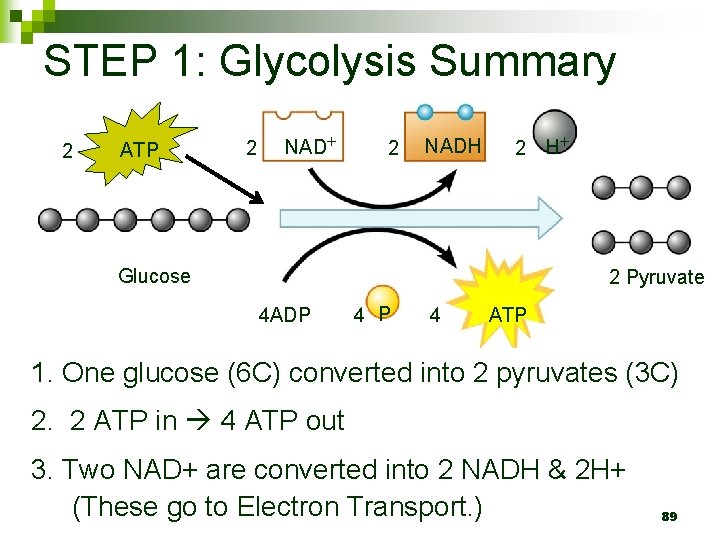
STEP 1: Glycolysis Summary 2 ATP 2 NADH 2 H Glucose 2 Pyruvate 4 ADP 4 ATP 1. One glucose (6 C) converted into 2 pyruvates (3 C) 2. 2 ATP in 4 ATP out 3. Two NAD+ are converted into 2 NADH & 2 H+ (These go to Electron Transport. ) 89
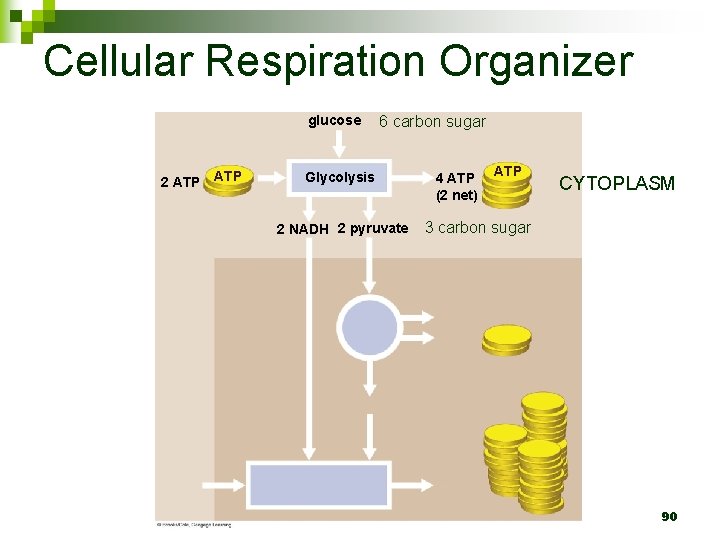
Cellular Respiration Organizer glucose 2 ATP 6 carbon sugar Glycolysis 2 NADH 2 pyruvate 4 ATP (2 net) ATP CYTOPLASM 3 carbon sugar 90
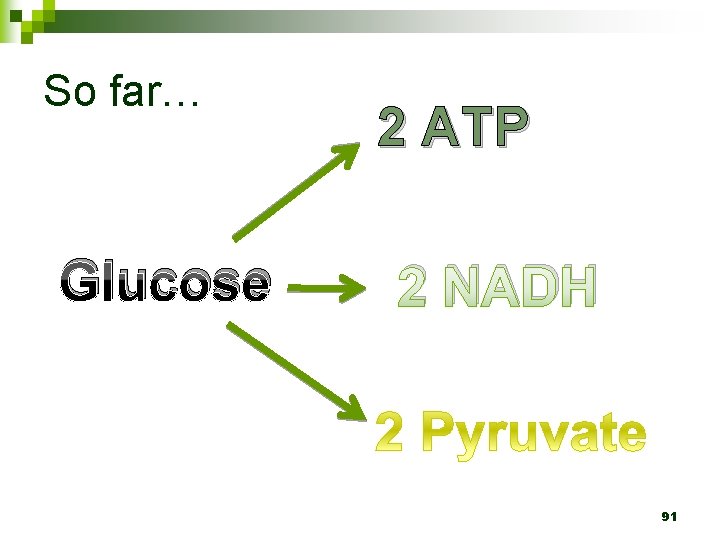
So far… Glucose 2 ATP 2 NADH 91
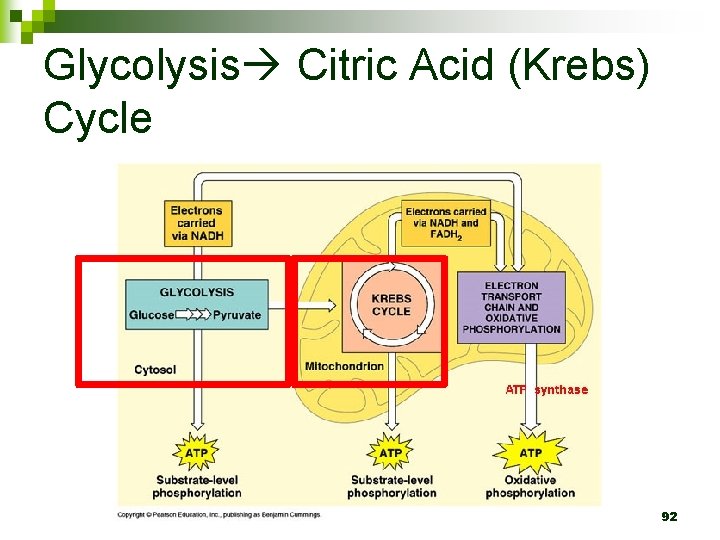
Glycolysis Citric Acid (Krebs) Cycle 92
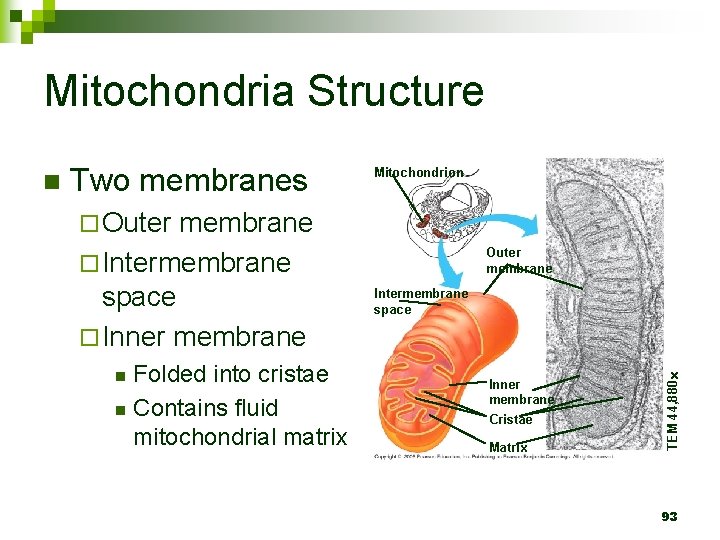
Mitochondria Structure Two membranes membrane ¨ Intermembrane space ¨ Inner membrane Mitochondrion ¨ Outer Folded into cristae n Contains fluid mitochondrial matrix n Outer membrane Intermembrane space Inner membrane Cristae Matrix TEM 44, 880 n 93
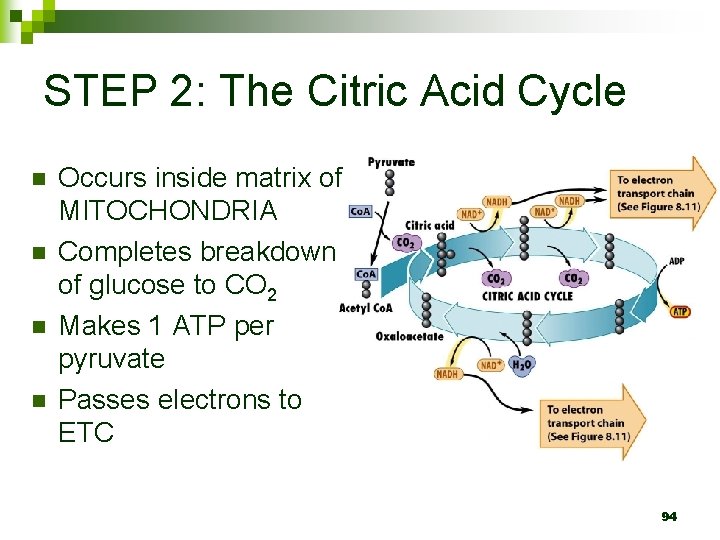
STEP 2: The Citric Acid Cycle n n Occurs inside matrix of MITOCHONDRIA Completes breakdown of glucose to CO 2 Makes 1 ATP per pyruvate Passes electrons to ETC 94

STEP 2: Citric Acid Cycle Animation: Campbell Ch 6 – TCA n Also called TCA (The Citric Acid cycle) n Also called the Krebs Cycle ONLINE: TCA ANIMATION n REMEMBER: There’s TWO pyruvates!! 95

STEP 2: TCA Summary n Pyruvate (x 2) is groomed for TCA cycle: ¨ Carbon atom released as CO 2 ¨ 2 C compound oxidized; NAD+ reduced to NADH ¨ Coenzyme A joins 2 C 96
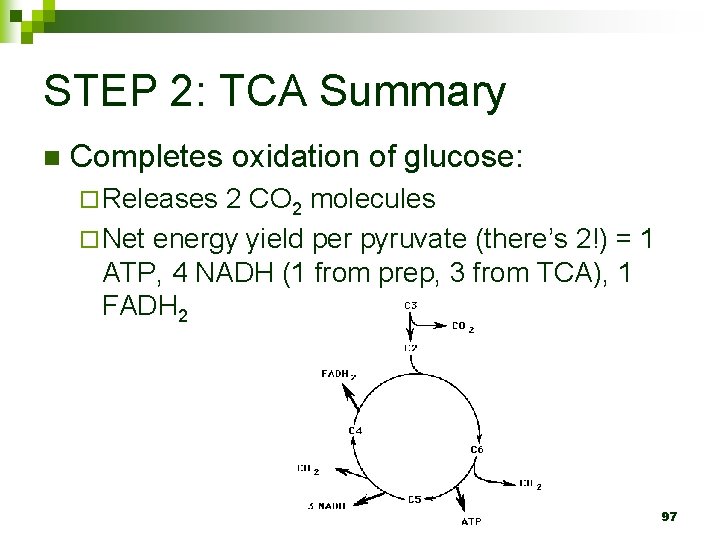
STEP 2: TCA Summary n Completes oxidation of glucose: ¨ Releases 2 CO 2 molecules ¨ Net energy yield per pyruvate (there’s 2!) = 1 ATP, 4 NADH (1 from prep, 3 from TCA), 1 FADH 2 97
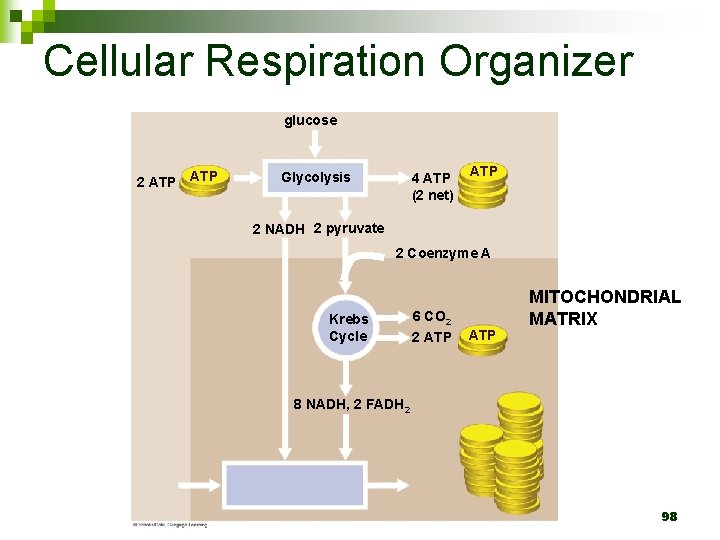
Cellular Respiration Organizer glucose 2 ATP Glycolysis 4 ATP (2 net) ATP 2 NADH 2 pyruvate 2 Coenzyme A Krebs Cycle 6 CO 2 2 ATP MITOCHONDRIAL MATRIX 8 NADH, 2 FADH 2 98
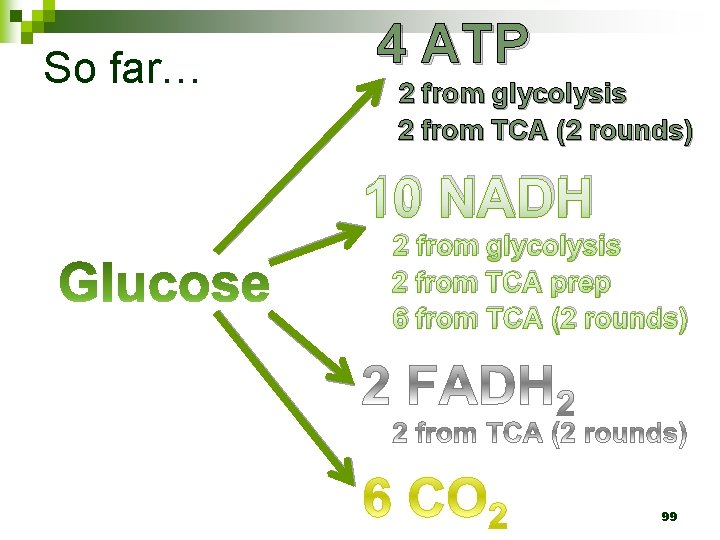
So far… 4 ATP 2 from glycolysis 2 from TCA (2 rounds) 10 NADH 2 from glycolysis 2 from TCA prep 6 from TCA (2 rounds) 99
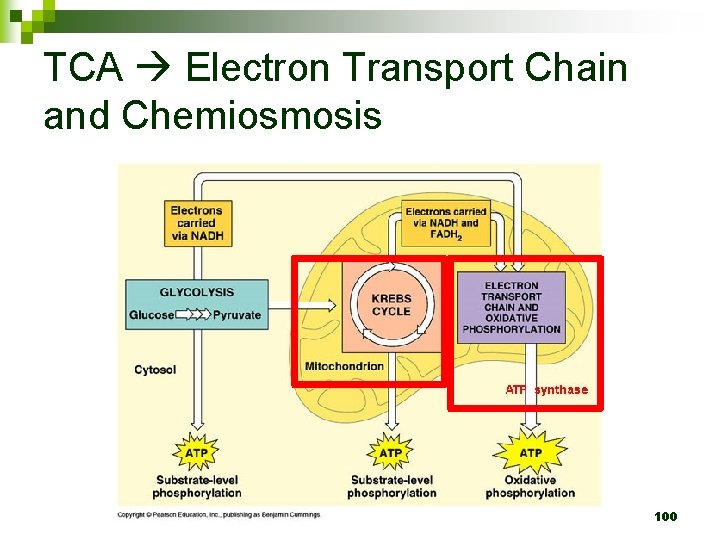
TCA Electron Transport Chain and Chemiosmosis 100
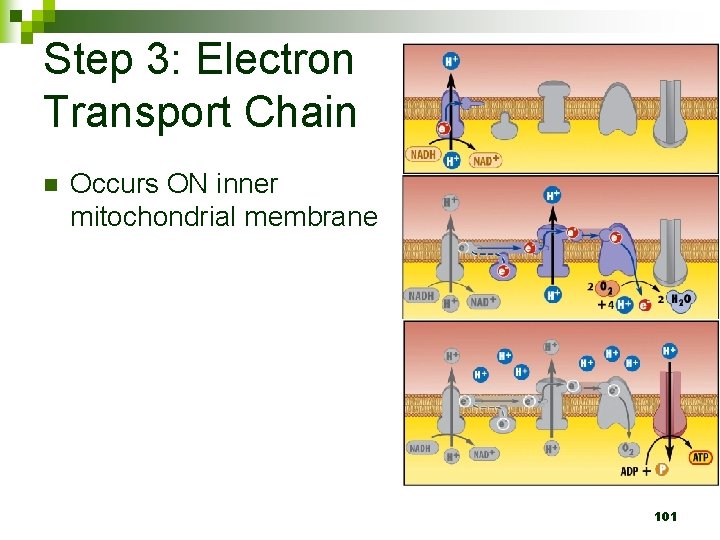
Step 3: Electron Transport Chain n Occurs ON inner mitochondrial membrane 101
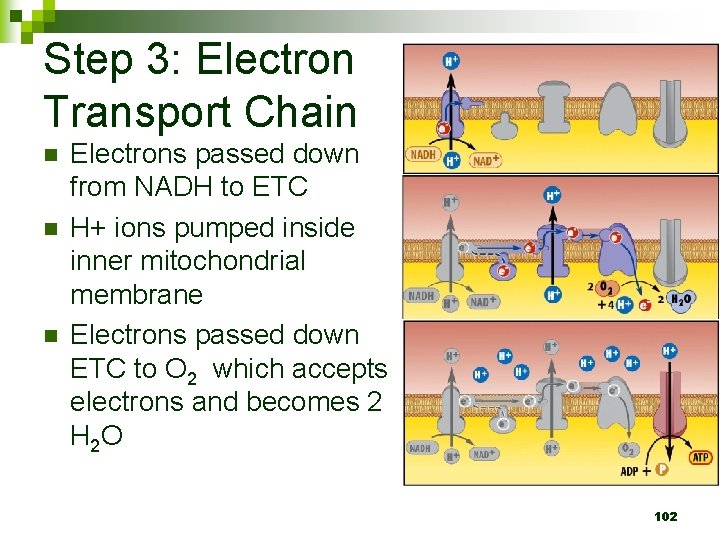
Step 3: Electron Transport Chain n Electrons passed down from NADH to ETC H+ ions pumped inside inner mitochondrial membrane Electrons passed down ETC to O 2 which accepts electrons and becomes 2 H 2 O 102

STEP 3: Electron Transport Chain Animation: Campbell Ch 6 – ETC 103
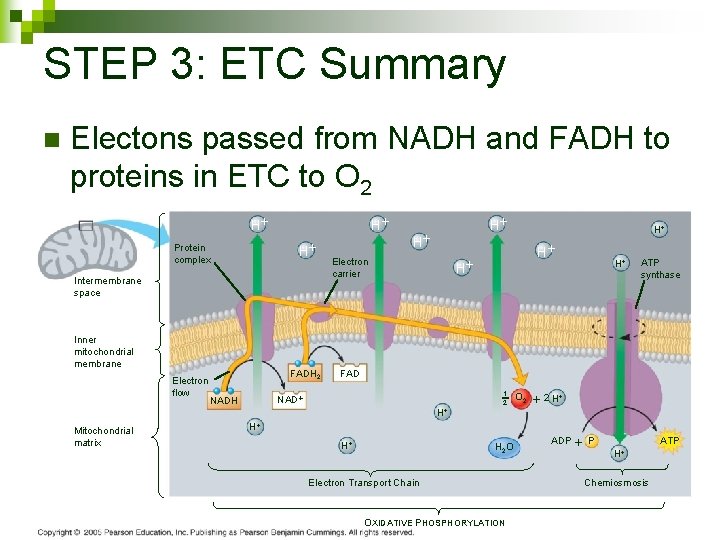
STEP 3: ETC Summary n Electons passed from NADH and FADH to proteins in ETC to O 2 H H H Protein complex Intermembrane space Inner mitochondrial membrane Electron flow Mitochondrial matrix FADH 2 H Electron carrier H H ATP synthase FAD NADH H H 1 2 O 2 2 H H 2 O Electron Transport Chain OXIDATIVE PHOSPHORYLATION ADP P H ATP Chemiosmosis 104
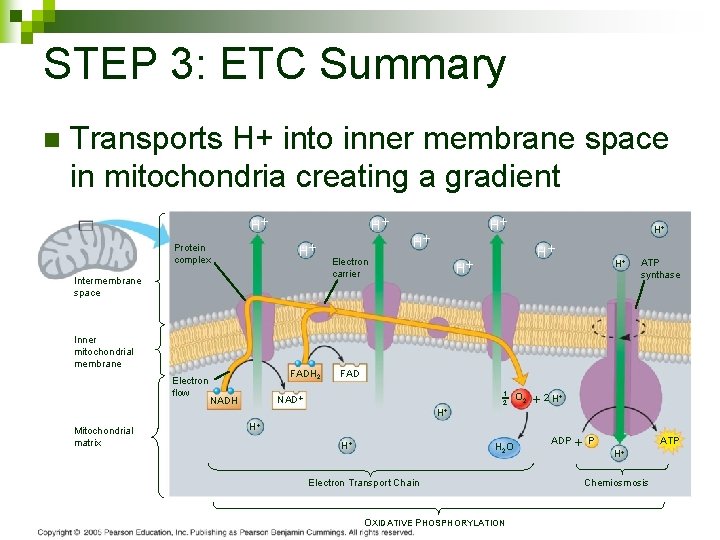
STEP 3: ETC Summary n Transports H+ into inner membrane space in mitochondria creating a gradient H H H Protein complex Intermembrane space Inner mitochondrial membrane Electron flow Mitochondrial matrix FADH 2 H Electron carrier H H ATP synthase FAD NADH H H 1 2 O 2 2 H H 2 O Electron Transport Chain OXIDATIVE PHOSPHORYLATION ADP P H ATP Chemiosmosis 105
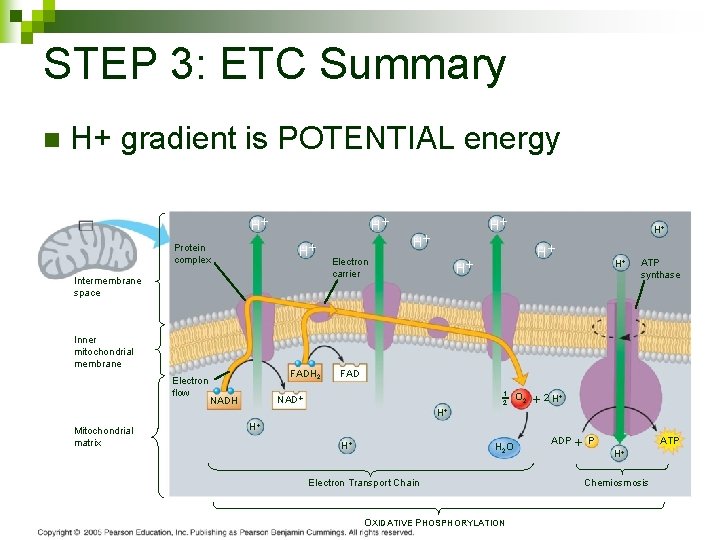
STEP 3: ETC Summary n H+ gradient is POTENTIAL energy H H H Protein complex Intermembrane space Inner mitochondrial membrane Electron flow Mitochondrial matrix FADH 2 H Electron carrier H H ATP synthase FAD NADH H H 1 2 O 2 2 H H 2 O Electron Transport Chain OXIDATIVE PHOSPHORYLATION ADP P H ATP Chemiosmosis 106
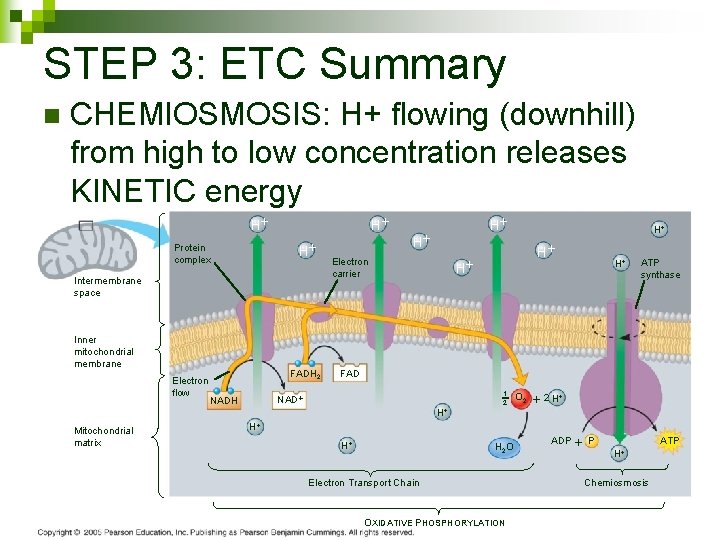
STEP 3: ETC Summary n CHEMIOSMOSIS: H+ flowing (downhill) from high to low concentration releases KINETIC energy H H H Protein complex Intermembrane space Inner mitochondrial membrane Electron flow Mitochondrial matrix FADH 2 H Electron carrier H H ATP synthase FAD NADH H H 1 2 O 2 2 H H 2 O Electron Transport Chain OXIDATIVE PHOSPHORYLATION ADP P H ATP Chemiosmosis 107
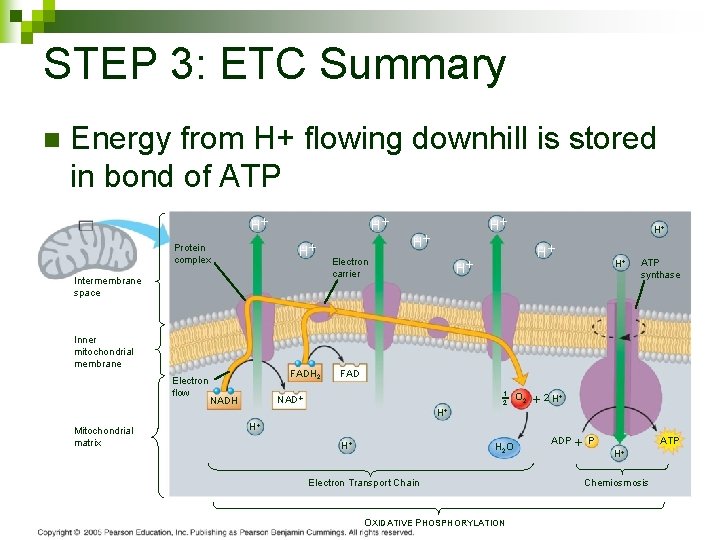
STEP 3: ETC Summary n Energy from H+ flowing downhill is stored in bond of ATP H H H Protein complex Intermembrane space Inner mitochondrial membrane Electron flow Mitochondrial matrix FADH 2 H Electron carrier H H ATP synthase FAD NADH H H 1 2 O 2 2 H H 2 O Electron Transport Chain OXIDATIVE PHOSPHORYLATION ADP P H ATP Chemiosmosis 108
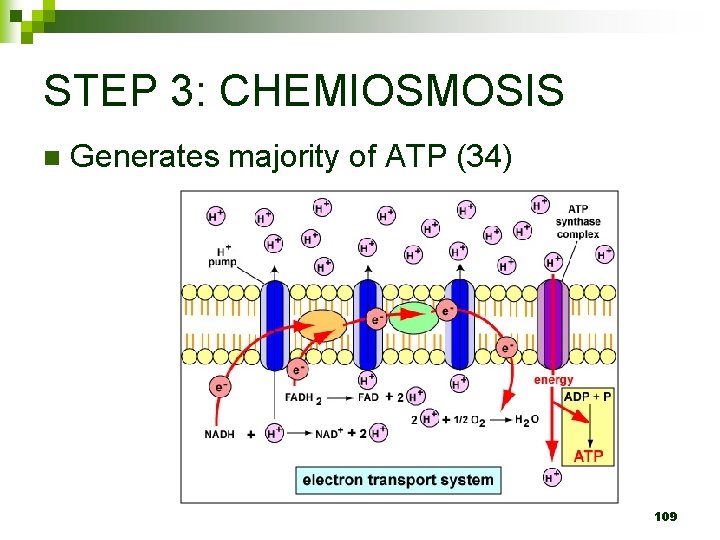
STEP 3: CHEMIOSMOSIS n Generates majority of ATP (34) 109
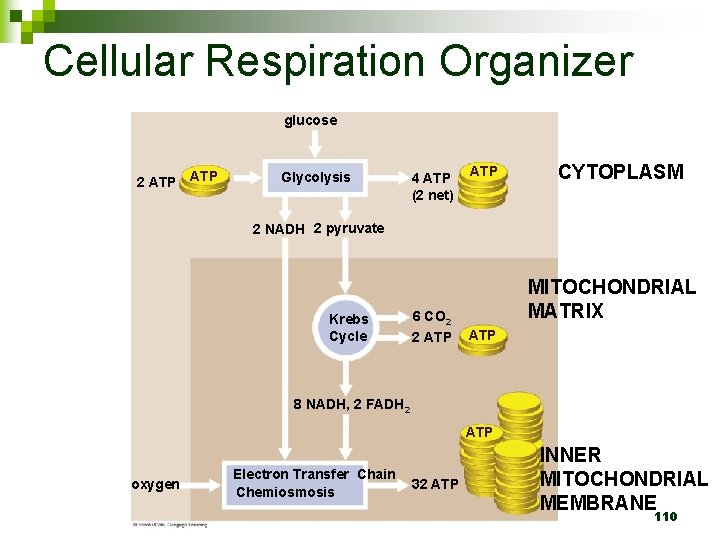
Cellular Respiration Organizer glucose 2 ATP Glycolysis 4 ATP (2 net) ATP CYTOPLASM 2 NADH 2 pyruvate Krebs Cycle MITOCHONDRIAL MATRIX 6 CO 2 2 ATP 8 NADH, 2 FADH 2 ATP oxygen Electron Transfer Chain Chemiosmosis 32 ATP INNER MITOCHONDRIAL MEMBRANE 110
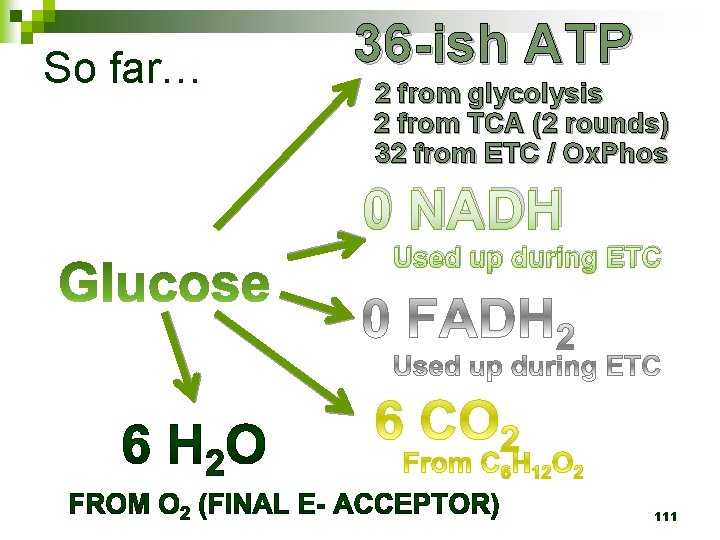
So far… 36 -ish ATP 2 from glycolysis 2 from TCA (2 rounds) 32 from ETC / Ox. Phos 0 NADH Used up during ETC 111

Cellular Respiration Overview Animation n NML_Cain 3_CD 3/Student_Animations/Full /Macintosh/cain_ch 08 a 02. app 112

Cellular Respiration n Aerobic metabolism: three steps ¨ Glycolysis, Citric Acid Cycle, & Oxidative Phosphorylation ¨ Releases LOTS of energy – typically 36 ATP per molecule of glucose 113

“Aerobic” Respiration? n Requires OXYGEN – WHY and WHERE? ¨ Last e- acceptor in ETC 114
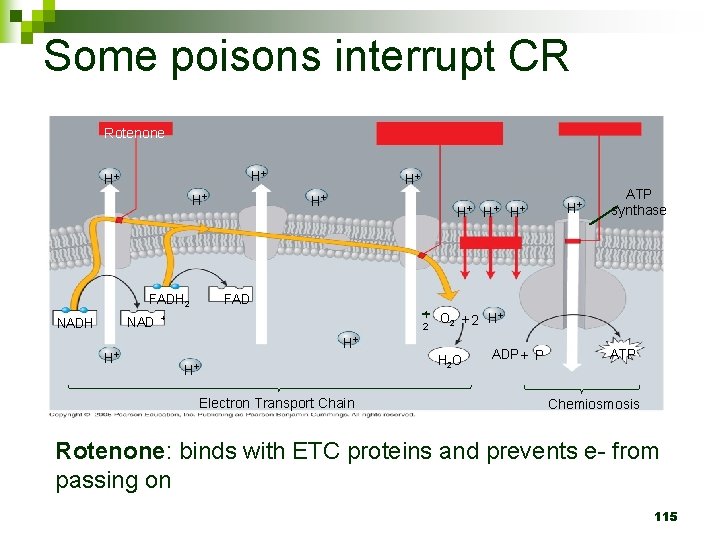
Some poisons interrupt CR Rotenone H+ H+ H+ NAD H+ H+ H+ ATP synthase FADH 2 NADH H+ 1 2 + O 2 + 2 H+ H+ H+ Electron Transport Chain H 2 O ADP + P ATP Chemiosmosis Rotenone: binds with ETC proteins and prevents e- from passing on 115
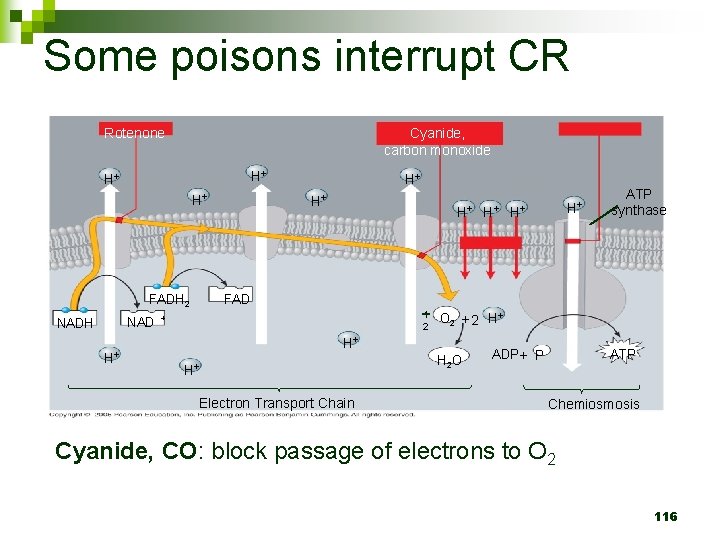
Some poisons interrupt CR Cyanide, carbon monoxide Rotenone H+ H+ H+ NAD H+ H+ H+ ATP synthase FADH 2 NADH H+ 1 2 + O 2 + 2 H+ H+ H+ Electron Transport Chain H 2 O ATP ADP + P Chemiosmosis Cyanide, CO: block passage of electrons to O 2 116
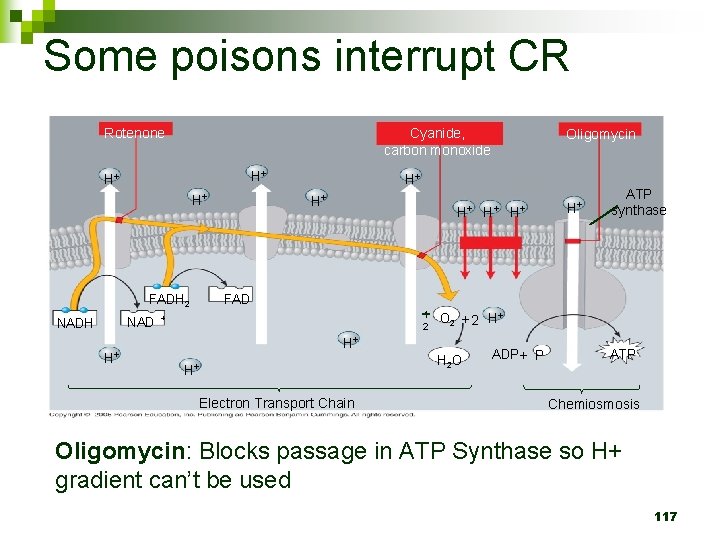
Some poisons interrupt CR Cyanide, carbon monoxide Rotenone H+ H+ H+ NAD H+ H+ ATP synthase FADH 2 NADH Oligomycin 1 2 + O 2 + 2 H+ H+ H+ Electron Transport Chain H 2 O ADP + P ATP Chemiosmosis Oligomycin: Blocks passage in ATP Synthase so H+ gradient can’t be used 117
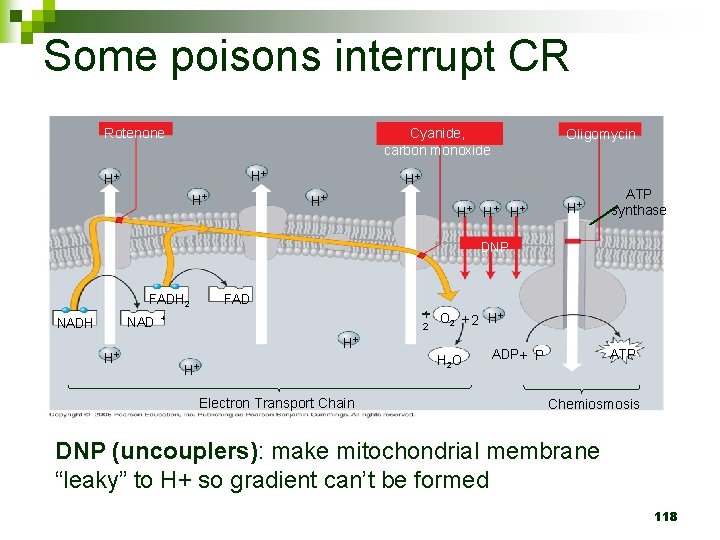
Some poisons interrupt CR Cyanide, carbon monoxide Rotenone H+ H+ H+ Oligomycin H+ H+ H+ ATP synthase DNP FADH 2 NADH H+ 1 2 + O 2 + 2 H+ H+ H+ Electron Transport Chain H 2 O ATP ADP + P Chemiosmosis DNP (uncouplers): make mitochondrial membrane “leaky” to H+ so gradient can’t be formed 118

Atkins Diet n n n Use fats for glycolysis in the absence of sugars Fats are broken down and enter as Actyl. Co. A Produce ketones when metabolized and change p. H of blood 119

Respiration Practice n Complete Respiration Practice Worksheet 120

Cellular Respiration Review Occurs in all eukaryotes n Generates ATP n Involves oxidation – reduction reactions n ¨ Oxidation = loss of electron / H atom; gain of charge ¨ Reduction = gain of electron / H atom; loss of charge C 6 H 12 O 6 + 6 O 2 6 CO 2+ 6 H 2 O + ATP
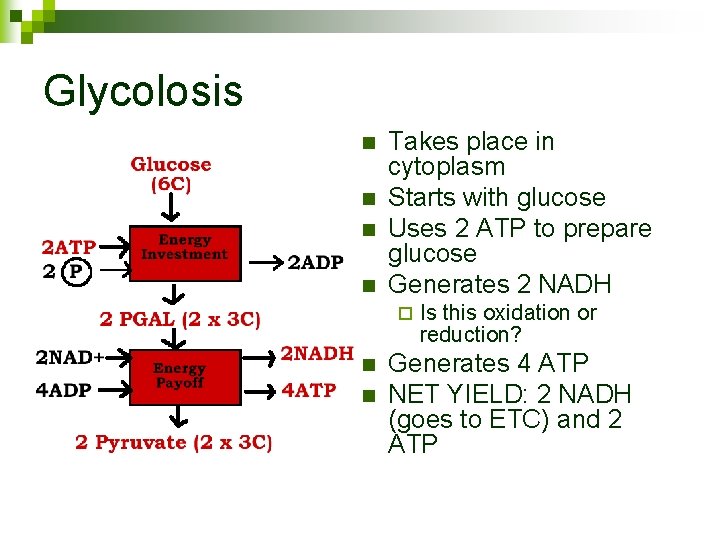
Glycolosis n n Takes place in cytoplasm Starts with glucose Uses 2 ATP to prepare glucose Generates 2 NADH ¨ n n Is this oxidation or reduction? Generates 4 ATP NET YIELD: 2 NADH (goes to ETC) and 2 ATP
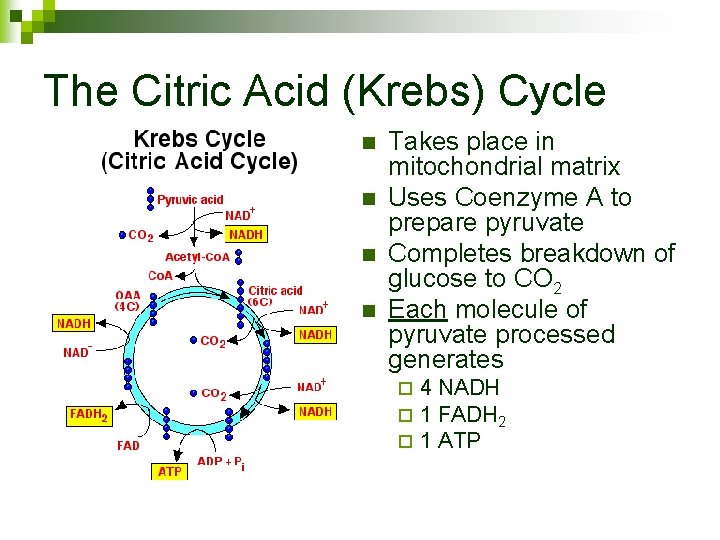
The Citric Acid (Krebs) Cycle n n Takes place in mitochondrial matrix Uses Coenzyme A to prepare pyruvate Completes breakdown of glucose to CO 2 Each molecule of pyruvate processed generates ¨ ¨ ¨ 4 NADH 1 FADH 2 1 ATP
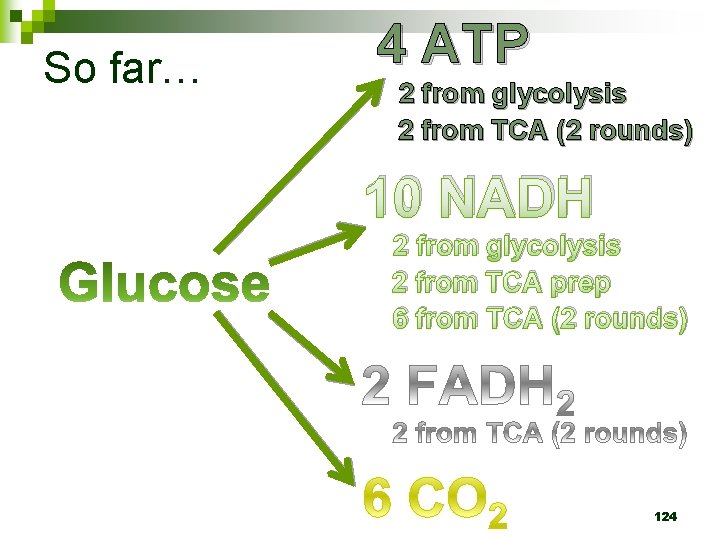
So far… 4 ATP 2 from glycolysis 2 from TCA (2 rounds) 10 NADH 2 from glycolysis 2 from TCA prep 6 from TCA (2 rounds) 124
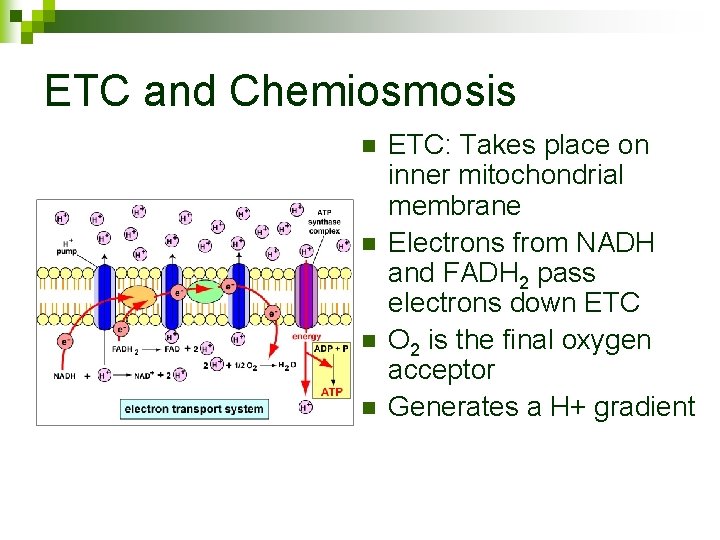
ETC and Chemiosmosis n n ETC: Takes place on inner mitochondrial membrane Electrons from NADH and FADH 2 pass electrons down ETC O 2 is the final oxygen acceptor Generates a H+ gradient

Chemiosmosis n Chemiosmosis: H+ gradient powers ATP Synthase enzyme to phosphorylate ADP to make ATP ADP + P ATP n Yield = 32 -34 ATP molecules
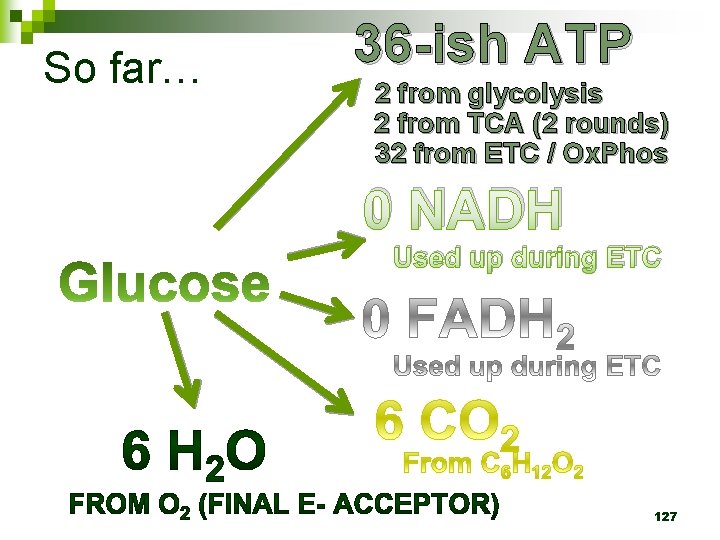
So far… 36 -ish ATP 2 from glycolysis 2 from TCA (2 rounds) 32 from ETC / Ox. Phos 0 NADH Used up during ETC 127

What if there’s no Oxygen? O 2 can’t act as final electron acceptor n ETC can’t happen n Can still get 2 ATP from glycolysis (doesn’t require O 2) n

What if there’s no Oxygen? Can generate 2 ATP n Makes 2 NADH n
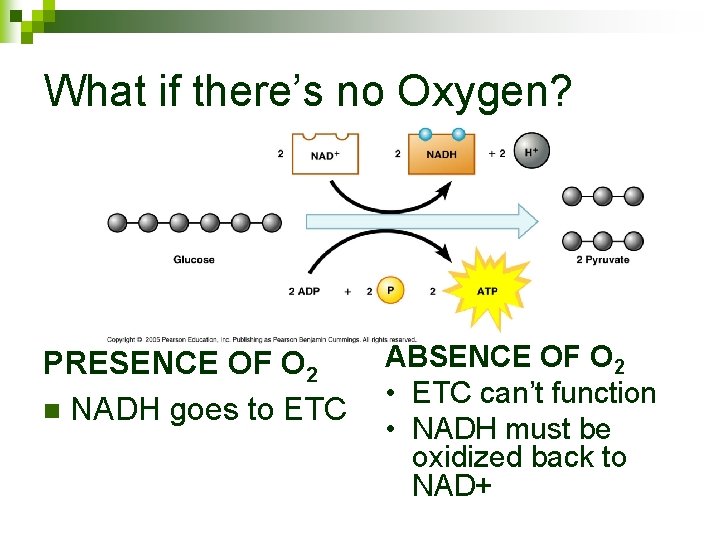
What if there’s no Oxygen? PRESENCE OF O 2 n NADH goes to ETC ABSENCE OF O 2 • ETC can’t function • NADH must be oxidized back to NAD+

Anaerobic Respiration Cellular respiration in the absence of oxygen n Oxidizes NADH to replenish NAD+ n Lactic Acid Fermentation n Ethanol Fermentation n
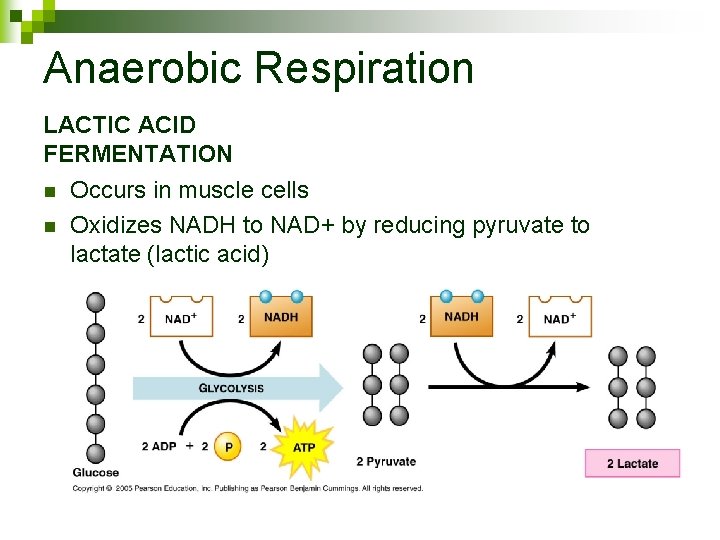
Anaerobic Respiration LACTIC ACID FERMENTATION n n Occurs in muscle cells Oxidizes NADH to NAD+ by reducing pyruvate to lactate (lactic acid)
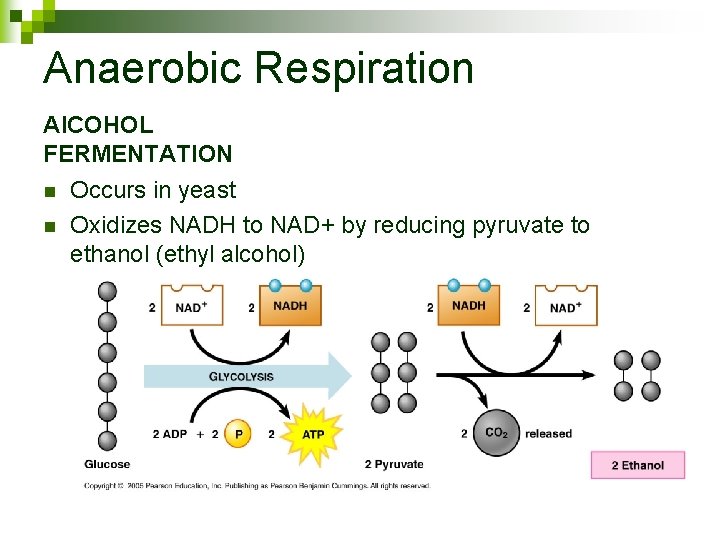
Anaerobic Respiration Al. COHOL FERMENTATION n n Occurs in yeast Oxidizes NADH to NAD+ by reducing pyruvate to ethanol (ethyl alcohol)

Why we like fermentation Often used by bacteria to make tasty foodies n n Used for thousands of years Method of preserving food
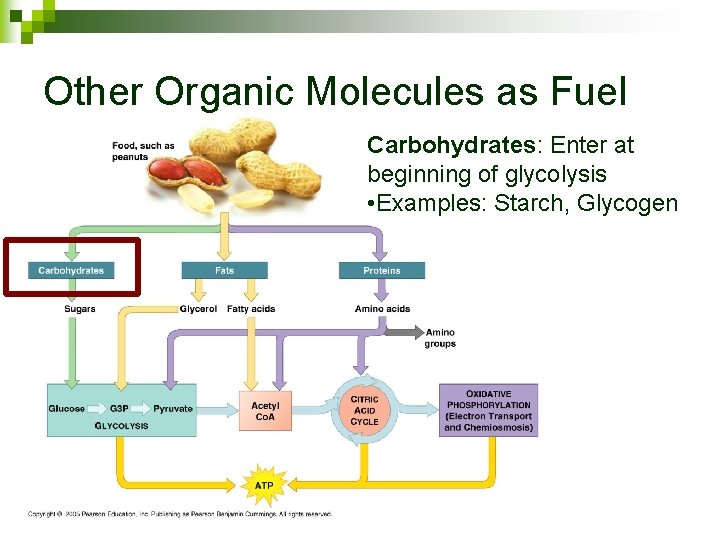
Other Organic Molecules as Fuel Carbohydrates: Enter at beginning of glycolysis • Examples: Starch, Glycogen
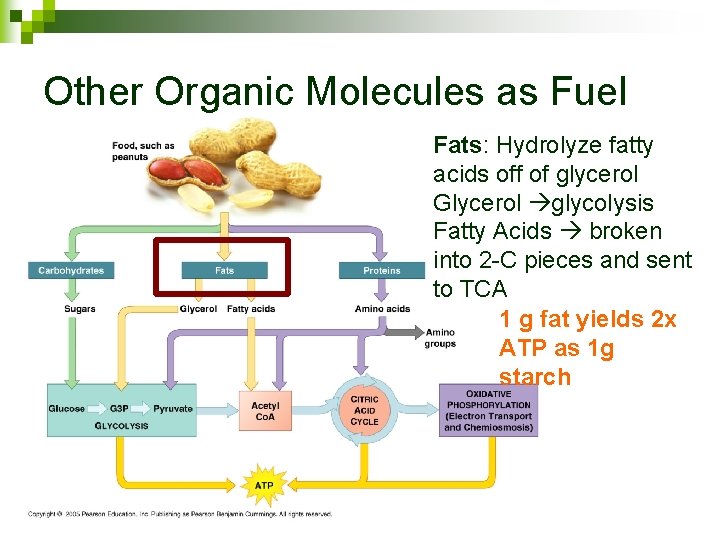
Other Organic Molecules as Fuel Fats: Hydrolyze fatty acids off of glycerol Glycerol glycolysis Fatty Acids broken into 2 -C pieces and sent to TCA 1 g fat yields 2 x ATP as 1 g starch
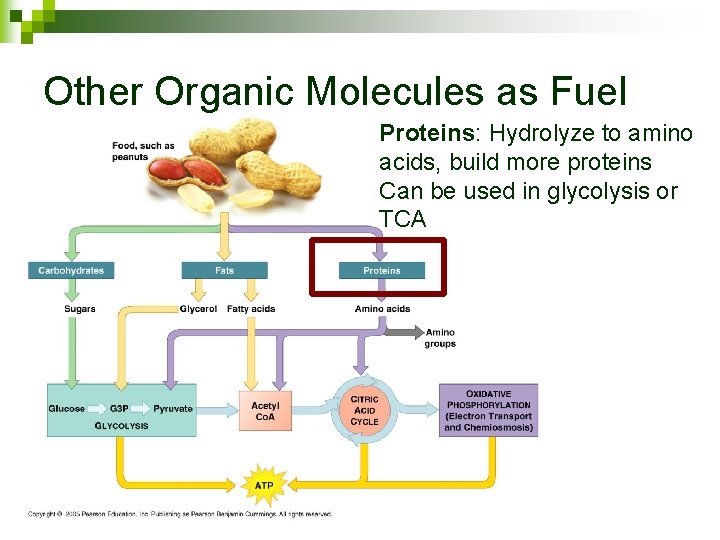
Other Organic Molecules as Fuel Proteins: Hydrolyze to amino acids, build more proteins Can be used in glycolysis or TCA
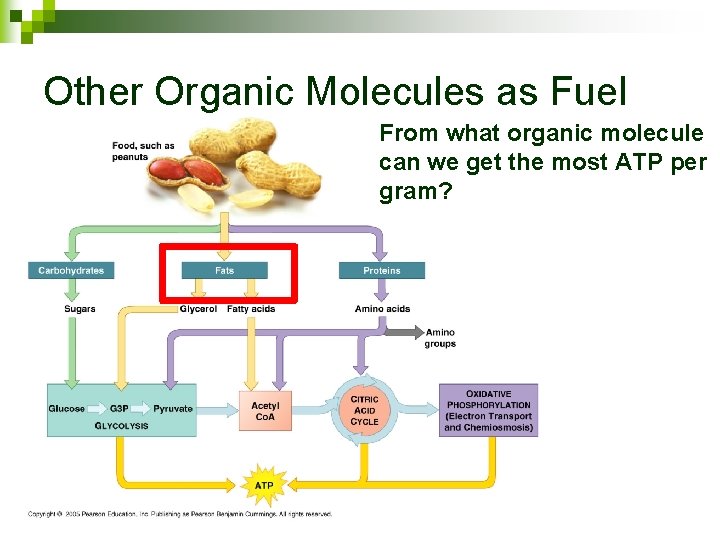
Other Organic Molecules as Fuel From what organic molecule can we get the most ATP per gram?
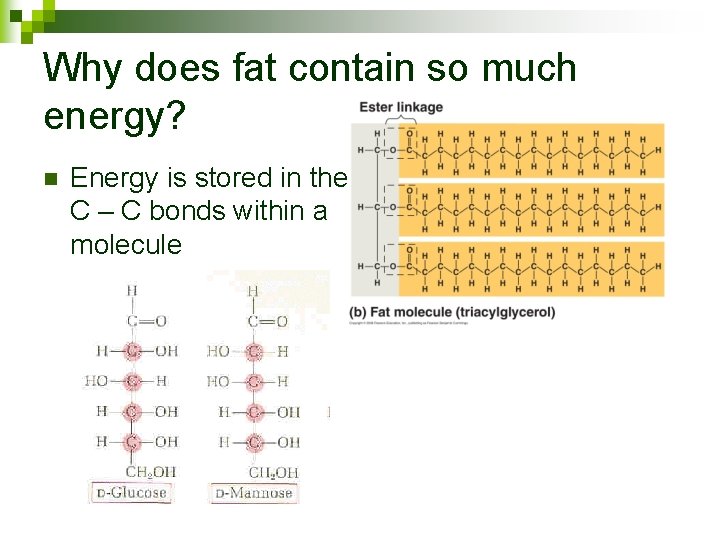
Why does fat contain so much energy? n Energy is stored in the C – C bonds within a molecule
- Slides: 139