Cell MembranePlasma Membrane plasma membrane is the boundary

Cell Membrane/Plasma Membrane • plasma membrane is the boundary that separates the living cell from its surroundings • functions: • 1. maintains integrity of the cell – size and shape • 2. controls transport = “selectively permeable” • 3. maintains the ionic concentration of the cell & osmotic pressure of the cytosol • 4. forms contacts with neighboring cells • 5. sensitivity
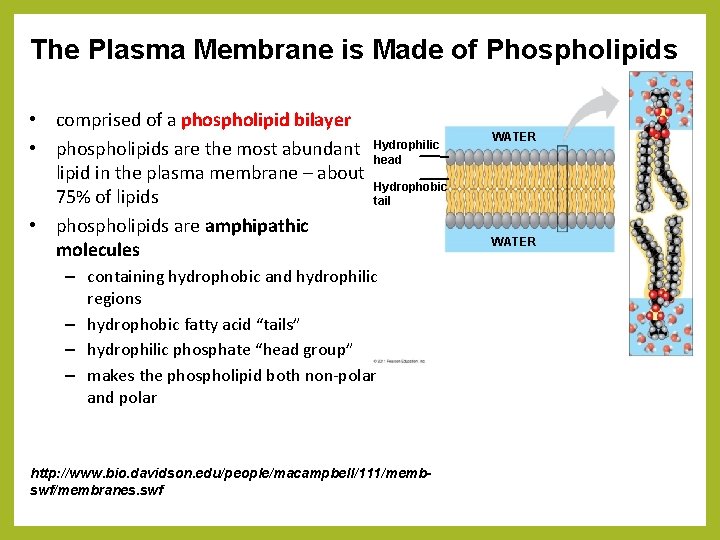
The Plasma Membrane is Made of Phospholipids • comprised of a phospholipid bilayer • phospholipids are the most abundant lipid in the plasma membrane – about 75% of lipids • phospholipids are amphipathic molecules Hydrophilic head WATER Hydrophobic tail – containing hydrophobic and hydrophilic regions – hydrophobic fatty acid “tails” – hydrophilic phosphate “head group” – makes the phospholipid both non-polar and polar http: //www. bio. davidson. edu/people/macampbell/111/membswf/membranes. swf WATER

The Phospholipid Bilayer • amphipathic nature means the phospholipids will self-assemble into a bilayer when placed into an aqueous environment

Plasma Membrane Composition cholesterol • the majority of the plasma membrane are phospholipids • inserted into the membrane will also be cholesterol - which is also amphipathic (polar OH group and non-polar steroid rings) -affects the membrane’s fluidity • cholesterol levels are high in the plasma membrane • organelle membranes contain very little
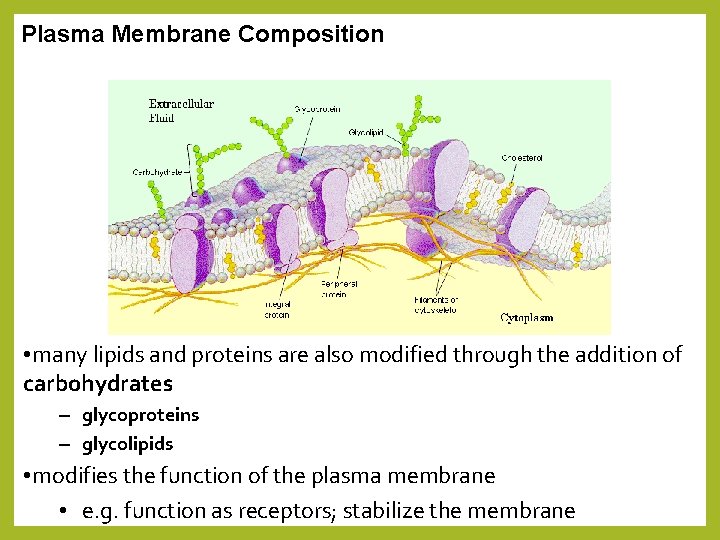
Plasma Membrane Composition • many lipids and proteins are also modified through the addition of carbohydrates – glycoproteins – glycolipids • modifies the function of the plasma membrane • e. g. function as receptors; stabilize the membrane

Membrane Proteins and Their Functions • the plasma membrane has proteins associated with it • membrane proteins determine most of the membrane’s specific functions • proteins link on the extracellular side to an extracellular matrix of proteins – support the cells within a tissue • proteins link on the cytoplasmic side to the cytoskeleton Fibers of extracellular matrix (ECM) Glycoprotein Carbohydrate Glycolipid EXTRACELLULAR SIDE OF MEMBRANE Cholesterol Microfilaments of cytoskeleton Peripheral proteins Integral protein CYTOPLASMIC SIDE OF MEMBRANE
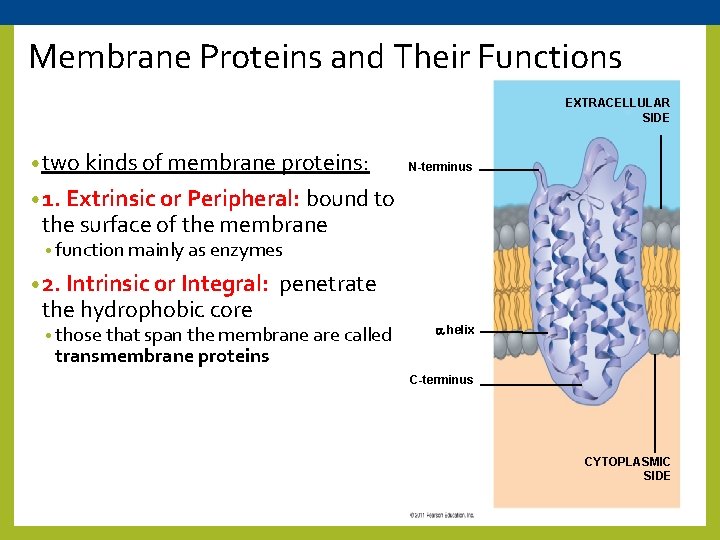
Membrane Proteins and Their Functions EXTRACELLULAR SIDE • two kinds of membrane proteins: N-terminus • 1. Extrinsic or Peripheral: bound to the surface of the membrane • function mainly as enzymes • 2. Intrinsic or Integral: the hydrophobic core penetrate • those that span the membrane are called helix transmembrane proteins C-terminus CYTOPLASMIC SIDE

Integral Membrane Protein Functions 6 major functions of integral membrane proteins: ◦ 1. transport – channel proteins & transporters ◦ 2. signal transduction – receptor proteins

Integral Membrane Protein Functions 6 major functions of integral membrane proteins: ◦ ◦ 3. enzymatic activity 4. cell-cell recognition – cell identity markers 5. intercellular joining - linkers 6. attachment- to the cytoskeleton and extracellular matrix (ECM) Enzymes Glycoprotein Enzymatic activity Cell-cell recognition Intercellular joining Attachment to the cytoskeleton and extracellular matrix (ECM)

Medical Application: Membrane Proteins • ion channels = “gates” for transport of ions across the plasma membrane • open in response to: 1. changes in voltage 2. binding of a ligand • e. g. calcium sodium chloride potassium • affected by drugs: e. g. anti-hypertensives - calcium, potassium channels -local anesthetics – sodium channels -diuretics – sodium channels -muscle relaxants – chloride channels -anti-diabetics – potassium channels • disease states affect channel function e. g. cystic fibrosis

Plasma Membrane Fluidity • fluid mosaic model: a the plasma membrane is a fluid structure with a “mosaic” of various proteins embedded in it • membrane proteins must be able to change shape to function • some of them even laterally diffuse through the PM • so the membrane must be fluid

Plasma Membrane Fluidity • the plasma membrane must have fluidity to allow membrane proteins to function • membrane fluidity is due to several factors – – temperature - lipids move around more with increased temp lipid packing – lipids with shorter fatty acid tails are less stiff saturation of fatty acids – more C=C bonds (more unsaturated) increase fluidity cholesterol – decreases fluidity at warmer temps; increases fluidity at lower temps Lateral movement occurs 107 times per second. Flip-flopping across the membrane is rare ( once per month).

Selective Permeability Øa cell must exchange materials with its surroundings ◦ controlled by the plasma membrane Øplasma membranes are selectively permeable ◦ regulating the cell’s molecular traffic Øpermeability = property that determines the effectiveness of the PM as a barrier

Selective Permeability Øpermeability varies - depending on the organization and characterization of the membrane lipids and proteins Øhydrophobic (nonpolar) molecules dissolve in the phospholipid bilayer and pass through the membrane rapidly Øionic and polar molecules do not cross the membrane easily ◦ require transport mechanisms ◦ provided by transport proteins

Membrane Gradients Øselective permeability of the PM allows the cells to control the concentration of ions within the cell (in the cytosol) and outside the cell (in the ECF) Øthis results in a distinct distribution of positive and negative ions inside and outside the cell ◦ typically the inside of the cell is more negatively charged Øthis difference in electrical charge between inside and outside = electrical gradient Ca 2+

Membrane Gradients Øbecause it occurs across the PM – we call this difference in charge = membrane potential Øcan be measured with tiny glass electrodes Øvaries from cell to cell Øvery important in the functioning of neurons and muscle cells

Transport Proteins Øtransport proteins allow passage of hydrophilic substances across the membrane Ønumerous types: 1. channel proteins – have a hydrophilic channel that certain molecules or ions can use as a tunnel to move across the membrane 2. carrier proteins - bind to molecules and change shape to shuttle them across the membrane 3. pumps – carrier proteins that require the hydrolysis of ATP (or GTP) to move substances

What determines the direction of transport? ? Øtwo basic things 1. concentration of what is being moved 2. available energy for movement

Transport Mechanisms across the Plasma Membrane passive transport diffusion osmosis facilitated active transport primary AT secondary AT endocytosis exocytosis http: //programs. northlandcollege. edu/biology/Biology 1111/animations/transport 1. html
![A. Diffusion = movement of materials from [high] to [low] • random movement, no A. Diffusion = movement of materials from [high] to [low] • random movement, no](http://slidetodoc.com/presentation_image/1c5827503a2b3376e70596de2ad68345/image-20.jpg)
A. Diffusion = movement of materials from [high] to [low] • random movement, no energy needs to be synthesized • the movement is driven by the inherent kinetic energy of the particles moving down their concentration gradient

A. Diffusion -three ways to diffuse: 1. through the lipid bilayer: 2. through a channel: 3. facilitated diffusion:
![B. Osmosis = diffusion of water from [high] to [low] OR movement of water B. Osmosis = diffusion of water from [high] to [low] OR movement of water](http://slidetodoc.com/presentation_image/1c5827503a2b3376e70596de2ad68345/image-22.jpg)
B. Osmosis = diffusion of water from [high] to [low] OR movement of water from [low solute] to [high solute]
![B. Osmosis = diffusion of water from [high] to [low] OR movement of water B. Osmosis = diffusion of water from [high] to [low] OR movement of water](http://slidetodoc.com/presentation_image/1c5827503a2b3376e70596de2ad68345/image-23.jpg)
B. Osmosis = diffusion of water from [high] to [low] OR movement of water from [low solute] to [high solute] Lower concentration of solute (sugar) Ø in osmosis – the membrane is permeable to water and NOT to the solutes Ø it is the concentration of solutes that causes the water to move Ø the solutes become surrounded by a hydration shell of water molecules Ø this decreases the amount of free water molecules available to move Ø so increased solute concentration decreases the concentration of free water molecules Ø water movement is affected by this drop in free water molecule concentration Higher concentration of solute Sugar molecule H 2 O Selectively permeable membrane Osmosis Same concentration of solute
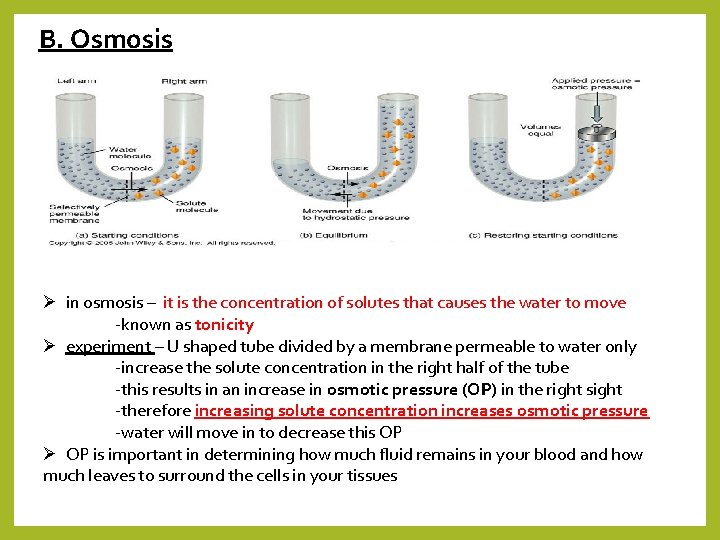
B. Osmosis Ø in osmosis – it is the concentration of solutes that causes the water to move -known as tonicity Ø experiment – U shaped tube divided by a membrane permeable to water only -increase the solute concentration in the right half of the tube -this results in an increase in osmotic pressure (OP) in the right sight -therefore increasing solute concentration increases osmotic pressure -water will move in to decrease this OP Ø OP is important in determining how much fluid remains in your blood and how much leaves to surround the cells in your tissues

Osmosis is controlled by tonicity = degree to which a the concentration of a specific solute surrounding a cell causes water to enter or leave the cell hypertonic e. g. isotonic = [S]in = [S]out hypotonic = [S]in > [S]out hypertonic = [S]in < [S]out water enters cell water exits cell no water movement

Medical Application: Osmotic pressure and IV solutions Ø medical uses of solutions requires careful consideration of osmolarity e. g. can cause destruction of red blood cells if these cells are placed in hypotonic or hypertonic solutions Ø typical saline solutions are 0. 9% Na. Cl = isotonic saline Ø other IV solutions are also isotonic e. g. D 5 W – 5% dextrose in water Ø but hypertonic and hypotonic solutions can be used in specific situations e. g. cerebral edema = water is forced out of the blood and into the brain tissue -treatment with hypertonic saline - causes water to leave the brain tissue and enter the blood plasma where it is removed by the kidneys e. g. dehydration – treatment with hypotonic solutions to increase water of the tissue fluids
![C. Facilitated transport = molecules move by a carrier protein from [high] to [low] C. Facilitated transport = molecules move by a carrier protein from [high] to [low]](http://slidetodoc.com/presentation_image/1c5827503a2b3376e70596de2ad68345/image-27.jpg)
C. Facilitated transport = molecules move by a carrier protein from [high] to [low] • binds to a carrier protein on the plasma membrane • transported by the carrier protein • no energy required – transported down the concentration gradient by the carrier protein • -molecules that are insoluble, too polar or too large e. g. glucose amino acids

Medical application: Facilitated Transport and Diabetes • increased blood sugar – production of insulin by the pancreas • insulin causes cells (e. g. adipose cells, liver cells, muscle cells) to increase their expression of a glucose transporter (GLUT protein) on the surface • this increases the uptake of sugar from the blood tissues (glycogen) • failure to produce enough insulin or failure of cells to express GLUT transporters in response to insulin = diabetes mellitus • type II – failure of GLUT transporter production or activity

Active Transport Mechanisms • • • transport requires the expenditure of energy usually provided through the hydrolysis of ATP ADP+Pi cell is moving a substance against its concentration gradient cell is forming transport vesicles cell is internalizing something

Active Transport Mechanisms Several kinds: 1. primary active transport = molecules are moved against its concentration gradient -i. e. from [low] to [high] -directly uses the energy of ATP hydrolysis for this 2. secondary active transport = molecules are moved against its concentration gradient -i. e. from [low] to [high] -movement is dependent upon another ion’s concentration gradient 3. Exocytosis – cell secretion 4. Endocytosis – cell internalization a. pinocytosis b. phagocytosis Exocytosis and Endocytosis c. receptor-mediated endocytosis are referred to as Bulk Transport (Active process)

A. Primary Active Transport • requires a protein carrier called a pump - pump that hydrolyzes ATP (ATPase) • e. g. sodium/potassium ATPse pump : maintains a specific concentration of Na+ and K+ in and out of the cell – three Na+ are pumped out of a cell and 2 K+ are pumped into the cell https: //www. youtube. com/watch? v=M 6_NCd. V 7 YO 8

A. Primary Active Transport • • 3 Na+ ions bind to the pump ATP binds to the pump and is hydrolyzed a phosphate group remains bound to the pump = phosphorylation changes the activity of the pump by altering its shape Na+ is expelled out of the cell – against its concentration gradient 2 K+ ions then bind the pump and causes the release of the PO 4 group another shape change by the pump - releases the K+ into the cell

B. Secondary Active Transport • the energy stored in a concentration gradient is used to drive the transport of other materials • primary active transport establishes high [Na+] outside the cell - creates a Na+ gradient • diffusion of Na back into the cell allows the movement of a second ion through a carrier protein - against its concentration gradient

B. Secondary Active Transport • if the second ion moves in the same direction as the Na+ = symport • if the second ion moves in the opposite direction = antiport diffusion Na pump diffusion https: //www. youtube. com/watch? v=Ghw. Jz. Vq-3 GY

Bulk transport across the plasma membrane occurs by exocytosis and endocytosis • large molecules (e. g. polysaccharides and proteins) cross the membrane in bulk - via vesicles • this bulk transport requires energy • two kinds of bulk transport: • 1. Exocytosis – known as secretion • 2. Endocytosis – known as internalization

A. Exocytosis = secretion of a substance outside the cell • products made within the cell, packaged by the Golgi into secretory vesicles fusion with the plasma membrane and release outside the cell e. g. nerve cells - neurotransmitter release http: //highered. mcgrawhill. com/sites/0072437316/student_view 0/chapter 6/animations. html#

B. Endocytosis = reverse of exocytosis, internalization of substances -3 forms: 1. pinocytosis = “cell drinking”

2. phagocytosis = “cell eating”

3. receptor-mediated endocytosis (RME) = internalization of specific substances • requires binding of a ligand with a receptor internalization into the cell • occurs at specific sites in the plasma membrane called clathrin-coated pits • proteins accumulate at these clathrin-coated pits • internalization clathrin-coated vesicle http: //sumanasinc. com/webcontent/animations/content/endocyt osis. html

3. receptor-mediated endocytosis (RME) - the clathrin-coated vesicle fuses with a small organelle called an early endosome – clathrin is recycled back to the PM - the early endosomes deliver their cargo to another organelle called a late endosome - the late endosomes fuse with small immature lysosomes for digestion http: //sumanasinc. com/webcontent/animatio ns/content/endocytosis. html https: //www. youtube. com/watch? v=Qa. VNU TL 7 w. RA

Medical application ØHIV and receptor-mediated endocytosis Øbinding of HIV virus to the CD 4 protein on the surface of T helper cells and macrophages results in the RME of the HIV virus Ønew HIV viral particles are made by the host cell machinery and assembled at the host’s PM – released from the cell = exocytosis Øthe infected T cells are killed leading to low T cell counts in infected people
- Slides: 41