Cell Compounds Basic Chemistry Water Acids Bases and

Cell Compounds Basic Chemistry Water Acids, Bases and p. H

Basic Chemistry • Organic versus Inorganic Compounds • Organic compounds are the basis of all living things – those present in living organisms are grouped into four types • All compounds are made of bonded atoms • Covalent Bonds, Polar Covalent, Ionic Bonds

Water • All living things depend on water • Inorganic, Covalent, Polar Negative dipole Positive Dipole O H H
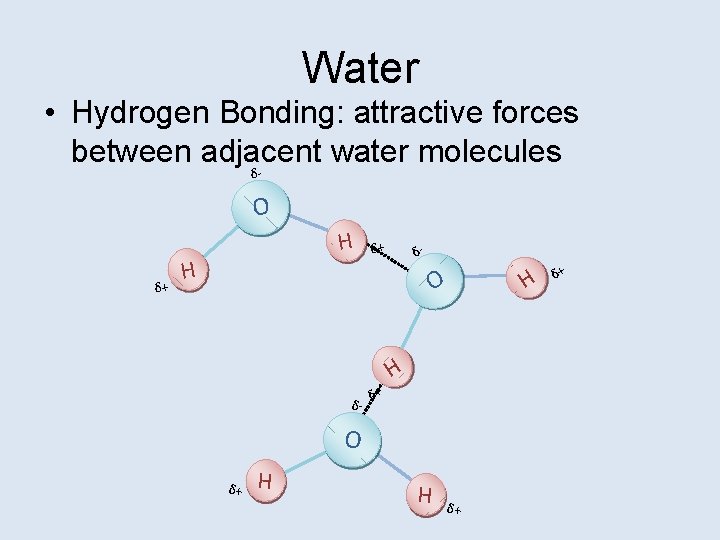
Water • Hydrogen Bonding: attractive forces between adjacent water molecules δ O δ+ H H δ+ δ- O δ- δ+ H O δ+ H H δ+

Water Hydrogen Bonding gives water some very important properties. • • • High Heat Capacity Excellent Solvent Transparent Greatest Density at 4˚C Cohesive Lubricant

ACIDS AND BASES

Acids • Acids are molecules that dissociate to release hydrogen ions (H+) • Strong acids such as hydrochloric acid dissociate easily: HCl → H+ + Cl • Weak acids dissociate to a much lesser extent H 2 O → H 1+ + OH 1 -

Bases • Bases are molecules that release hydroxide ions (OH 1 -) • Bases combine with Acids to neutralize them H 1+ + Cl 1 - + Na 1+ + OH 1 -→ H 2 O + Na. Cl

p. H • • A measure of the amount of free H 1+ -log[H 1+] Range of 1 -14 7 is neutral < 7 is acidic and >7 is basic In organisms p. H is very important Buffers help to maintain p. H by binding with excess hydrogen or hydroxide ions

IN REVIEW Organic Compounds will be examined throughout this course. Inorganic Compounds like acids, bases and water are important in living systems. Hydrogen Bonding gives water many properties that make it significant in all living things.

HOMEWORK 1) For each property of water – explain what the property is and how it is important in living systems. 2) Describe dehydration synthesis and hydrolysis 3) For each of the following compounds provide an example and draw its structure. a) Carbohydrate b) Lipid c) Protein d) Nucleic Acid
- Slides: 11