Cell Biology Membrane Transport Alberts Bruce Essential Cell

Cell Biology Membrane Transport Alberts, Bruce. Essential Cell Biology. 4 th ed. New York, NY: Garland Science Pub. , 2013. Print. Copyright © Garland Science 2013
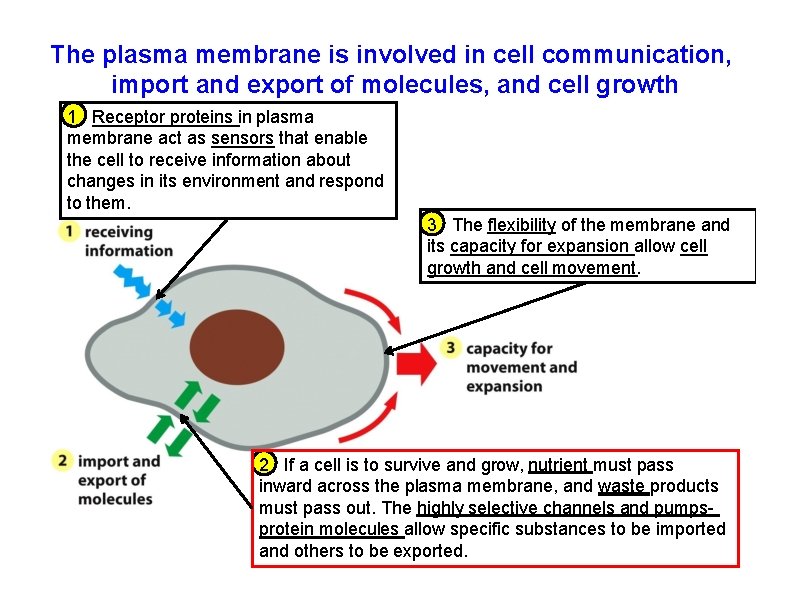
The plasma membrane is involved in cell communication, import and export of molecules, and cell growth 1 Receptor proteins in plasma membrane act as sensors that enable the cell to receive information about changes in its environment and respond to them. 3 The flexibility of the membrane and its capacity for expansion allow cell growth and cell movement. 2 If a cell is to survive and grow, nutrient must pass inward across the plasma membrane, and waste products must pass out. The highly selective channels and pumpsprotein molecules allow specific substances to be imported and others to be exported.
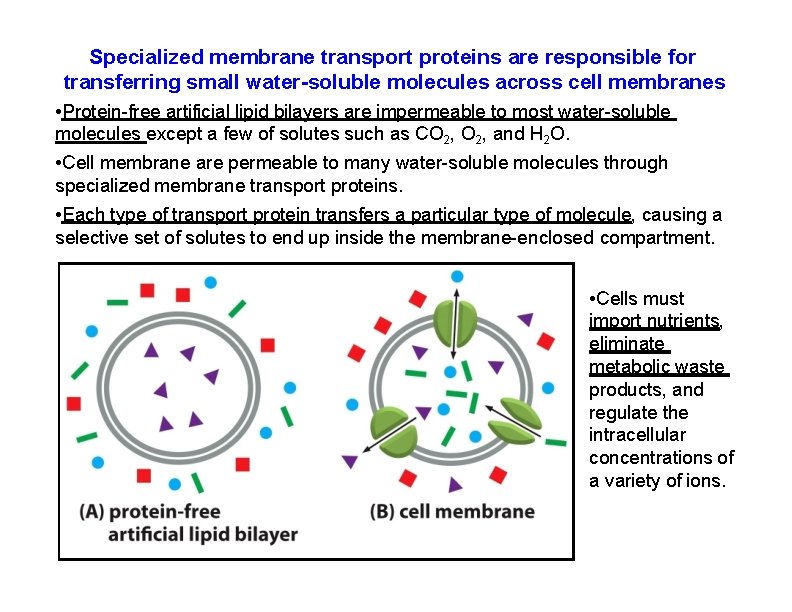
Specialized membrane transport proteins are responsible for transferring small water-soluble molecules across cell membranes • Protein-free artificial lipid bilayers are impermeable to most water-soluble molecules except a few of solutes such as CO 2, and H 2 O. • Cell membrane are permeable to many water-soluble molecules through specialized membrane transport proteins. • Each type of transport protein transfers a particular type of molecule, causing a selective set of solutes to end up inside the membrane-enclosed compartment. • Cells must import nutrients, eliminate metabolic waste products, and regulate the intracellular concentrations of a variety of ions.

The difference in ion composition between a cell’s interior and its environment Inorganic ions such as Na+, K+, Ca 2+, Mg 2+ , Cl-, and H+ (protons) are most plentiful of all the solutes in a cell’s environment. The movement of ions across cell membranes plays an essential role in many biological processes, including the activity of nerve cells and ATP production by all cells. Na+ is the most plentiful positively charged ion (cation) outside the cell, while K+ is the most plentiful inside. The high Na+ outside is balanced mainly by extracellular high Cl- , while high K+ inside is balanced by a variety of negatively charged intracellular ions (anions).
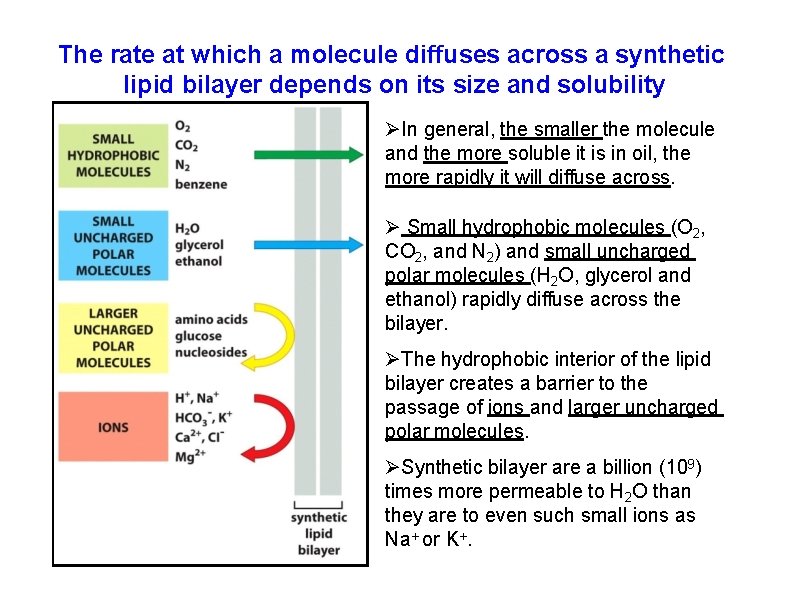
The rate at which a molecule diffuses across a synthetic lipid bilayer depends on its size and solubility In general, the smaller the molecule and the more soluble it is in oil, the more rapidly it will diffuse across. Small hydrophobic molecules (O 2, CO 2, and N 2) and small uncharged polar molecules (H 2 O, glycerol and ethanol) rapidly diffuse across the bilayer. The hydrophobic interior of the lipid bilayer creates a barrier to the passage of ions and larger uncharged polar molecules. Synthetic bilayer are a billion (109) times more permeable to H 2 O than they are to even such small ions as Na+ or K+.

Small molecules and ions can enter the cell through a transporter or a channel • Each protein provides a highly selective passageway across the membrane for a particular class molecules, such as ions, sugars, or amino acids. • Membrane transport proteins can be divided into two main classes: transporters and channels. • A transporter is a membrane protein that undergoes a series of conformational changes to transfer small water-soluble molecules across the lipid bilayer. • A channel is a hydrophilic pore across the lipid bilayer, with walls made of protein, through which specific ions or small molecules can diffuse. • Channels transfer molecules at a much greater rate than transporters. • Ion channels can exist in either an open or a closed conformation and they transport only in the open conformation.
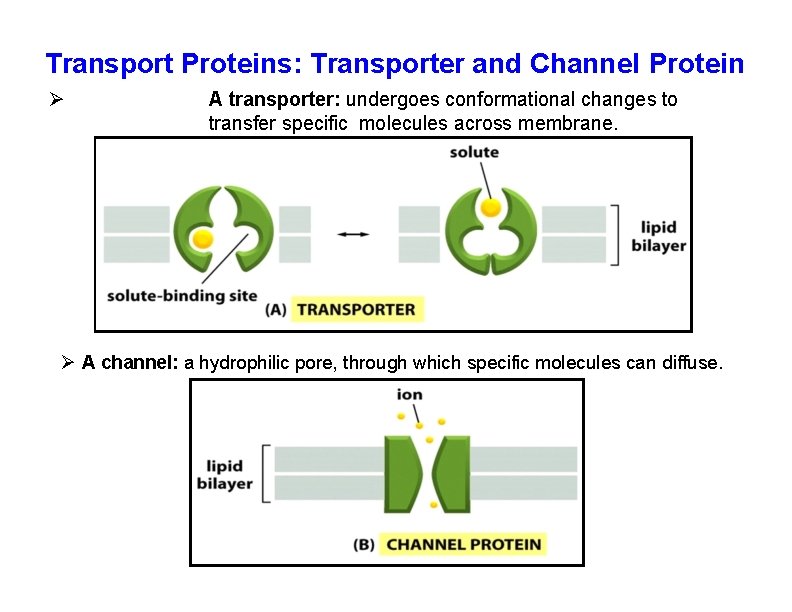
Transport Proteins: Transporter and Channel Protein A transporter: undergoes conformational changes to transfer specific molecules across membrane. A channel: a hydrophilic pore, through which specific molecules can diffuse.
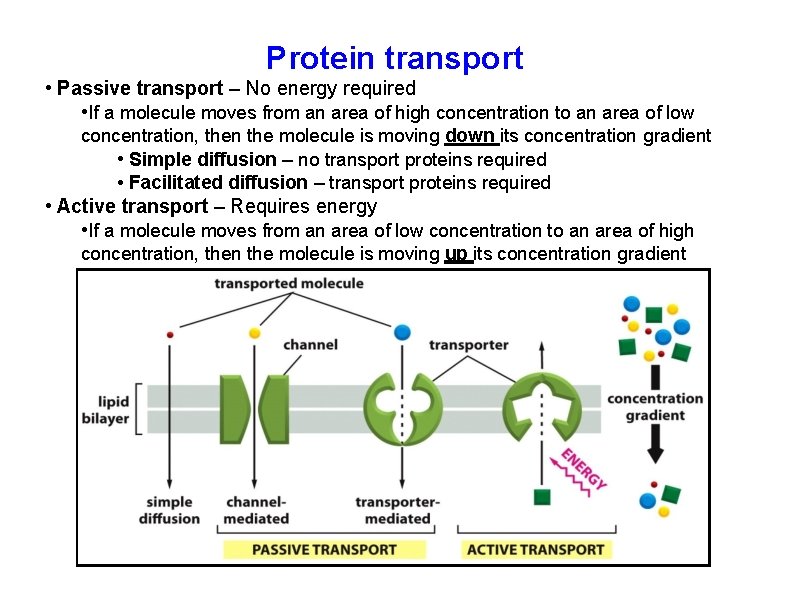
Protein transport • Passive transport – No energy required • If a molecule moves from an area of high concentration to an area of low concentration, then the molecule is moving down its concentration gradient • Simple diffusion – no transport proteins required • Facilitated diffusion – transport proteins required • Active transport – Requires energy • If a molecule moves from an area of low concentration to an area of high concentration, then the molecule is moving up its concentration gradient
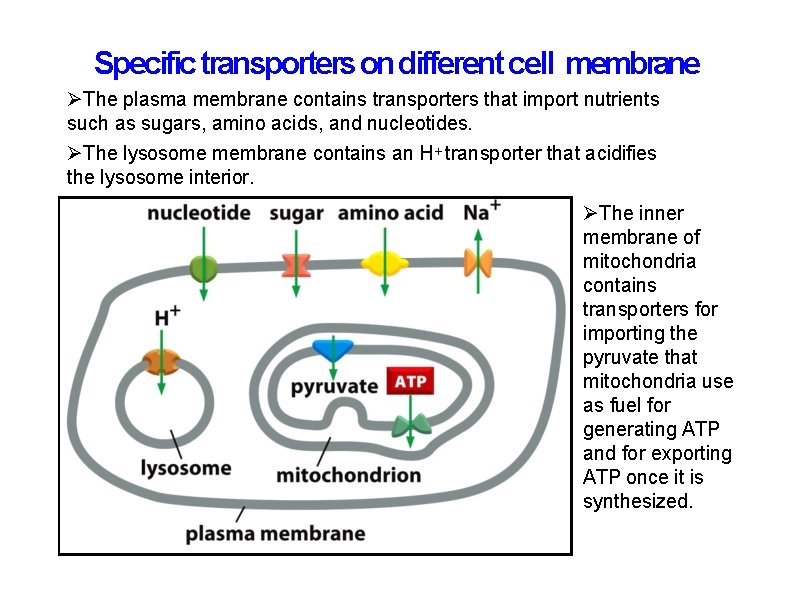
Specific transporters on different cell membrane The plasma membrane contains transporters that import nutrients such as sugars, amino acids, and nucleotides. The lysosome membrane contains an H+ transporter that acidifies the lysosome interior. The inner membrane of mitochondria contains transporters for importing the pyruvate that mitochondria use as fuel for generating ATP and for exporting ATP once it is synthesized.

A conformational change in a transporter mediates the passive transport • The transporter has two conformational states: A: The binding sites for the solute are exposed on the outside of the membrane. B: The same sites are exposed on the other side of the membrane.

Both concentration gradients and electrical forces drive passive transport high driving force low driving force • Electrochemical gradient is determined by two factors: • concentration gradient of an ion • distribution of positive and negative charges on either side of the membrane called membrane potential

Three different types of Active Transport Coupled transport • The movement of one molecule down its concentration gradient is couple to the movement of another molecule up its concentration gradient. ATP-driven pump • The movement of one molecule up its concentration gradient is coupled to the hydrolysis of ATP to ADP. Light-driven pump (most in bacteria) • The movement of one molecule up its concentration gradient is coupled the light energy.

The Na+-K+ pump plays a key role in membrane transport in animal cells It is important for a cell to maintain a high concentration of Na+ on the extracelllular side of the membrane, as the movement of Na+ into the cell is coupled to the movement of other molecules across the membrane. It is also important for a cell to maintain a high concentration of K+ on the cytoplasmic side of the membrane for proper functioning of the nervous system. Both of these goals are achieved by the Na+ - K+ pump

The Na+-K+ pump transport ions in a cyclic manner Pumps Na+ out of the cell Pumps K+ into the cell Requires ATPase: enzyme that converts ATP to ADP Component of Na+-K+ pump

The diffusion of water is known as osmosis If the concentration of solutes inside a cell is higher than that outside, water will move in by osmosis, causing the cell to swell. If a cell is placed in a high-salt solution, however, water will rush out.

Another ion transporter: the Ca 2+ Pump Intracellular signaling often involves an increase of intracellular (cytosolic) Ca 2+, therefore, the intracellular concentration of Ca 2+ must be kept low. Ca 2+ pump • Similar to Na+/K+ pump: requires ATP • Contains ATPase within pump • Unlike Na+/K+ pump, no coupled transport

Coupled Transport vs. Uniport Transport proteins perform two different types of coupled transport: Symport • The two molecules are transported in the same direction across the membrane Antiport • The two molecules are transport in the opposite direction across the membrane These vary from uniport, in which only one molecule is transferred across the membrane

The glucose-Na+ symport protein uses the electrochemical Na+ gradient to drive the import of glucose The high extracellular concentration of Na+ can be used for symport, such as glucose/Na+ pump.

Two types of glucose transporters in gut epithelial cells Apical surface Basal surface

Comparison of transporter-mediated membrane transport • H+ pumps are important in plants, animals, and bacteria • H+ gradient serves a similar function in bacteria and plants as the Na+ gradient serves in animals • electrochemical gradient is coupled to transport of other molecules • H+ is pumped into lysosomes in animal cells and vacuoles in plant cells to control the p. H and function of the organelles

A K+ channel possesses a selectivity filter to control ion transport Ion selectivity: permitting some inorganic ions to pass but not others. Depends on the diameter and shape of the ion channel and on the distribution of the charged amino acids that line it. Ion channel is narrow enough to force ions into contact with the channel wall. Only those of appropriate size and charge are able to pass. Opposite charges work the best. Channels with a negatively charged lining will deter negative ions from entering because of the mutual electrostatic repulsion. Each ion in aqueous solution is surrounded by a shell of water molecules, and the ions have to shed most of their associated water molecules in order to pass through.

Gated ion channels respond to different types of stimuli Most ion channels are also gated (switch between open and closed conformation) Gated ion channels vary in the conditions that influence their opening and closing (A) Voltagegated channels Controlled by membrane potential Major player in transmission of electrical signals between nerve cells Controlled by extremely sensitive voltage sensors (specialized charged protein domains on ion channel proteins) (B) and (C) Ligand-gated channels Controlled by binding of a ligand (such as neurotransmitters) to the channel protein (receptor) (D) Stress-activated channels Controlled by mechanical force Example: sounds trigger the opening of ion channel in ear hair cells Changes in the membrane potential above a certain threshold value exert sufficient electrical force on voltage sensors to encourage channel to open or close.

Stress-gated ion channels allow us to hear (A) Auditory portion of the inner ear (B) Sound vibration cause the basilar membrane to vibrate up and down, causing the stereocilia to tilt. • Each stereocilium on each hair cell is attached to the next shorter stereocilium by fine filament. • The tilting stretches the filaments, which pull open stressgated ion channels in the stereocilium membrane, allowing positively charged ions to enter from the surrounding fluid. • The influx of ions activates the hair cells, which stimulate underlying endings of the auditory nerve fibers that convery the auditory signal to the brain. (A) The basilar membrane below and the tectorial membrane above are not lipid bilayer membranes but sheets of extracellular matrix. (B)

Ions on each side of membrane decide the membrane potential Extracellular space Cytosol Membrane potential: Voltage difference across a membrane due to a slight excess of positive ions on one side and of negative ions on the other. A typical membrane potential for an animal cell plasma membrane is about -60 m. V (inside negative), measured relative to the surrounding fluid.

K+leak channels is important for the membrane potential The high intracellular concentration of K+ is in part generated by the Na+-K+ pump, which actively pumps of K+ into the cells. In a resting cell, K+ leak channels are the main ion channels open in the plasma membrane.

Aneuron has a cell body, a single axon, and multiple dendrites Gated ion channels are extremely important in transmitting signals between nerve cells (neurons). Neurons consist of: • Cell body • Axon (carries signals away from cell) • Dendrites (receive signals from the axons of other neurons) • Nerve terminals • Branch ends of axon • Pass the neuron’s message simultaneously to many target cells, other neurons or muscle or gland cells.

Keeping the signal strong Signals in the nervous system are transmitted over long-distances, stimulating changes in membrane potentials as they go • How do the signals keep from fading? Action Potentials • A large change in the membrane potential is caused by a local stimulus • Provides a strong enough signal to travel the length of the nerve cell, then starts all over again at next neuron Stimulating the action potential: • Ligand-gated channels open temporarily and cause depolarization of the plasma membrane (change in membrane potential) at that particular site • Membrane potential becomes less negative • Depolarization stimulates opening of voltage-gated Na+ channels • Leads to further depolarization
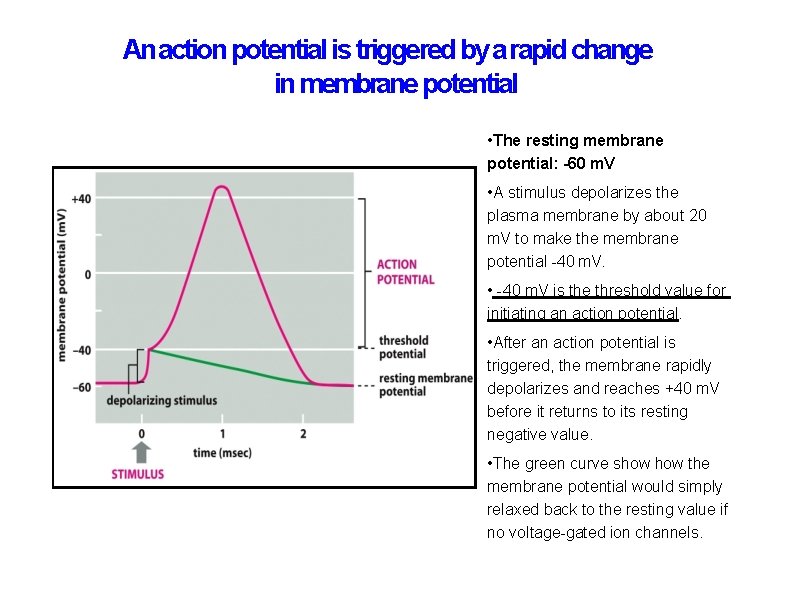
An action potential is triggered by a rapid change in membrane potential • The resting membrane potential: -60 m. V • A stimulus depolarizes the plasma membrane by about 20 m. V to make the membrane potential -40 m. V. • -40 m. V is the threshold value for initiating an action potential. • After an action potential is triggered, the membrane rapidly depolarizes and reaches +40 m. V before it returns to its resting negative value. • The green curve show the membrane potential would simply relaxed back to the resting value if no voltage-gated ion channels.
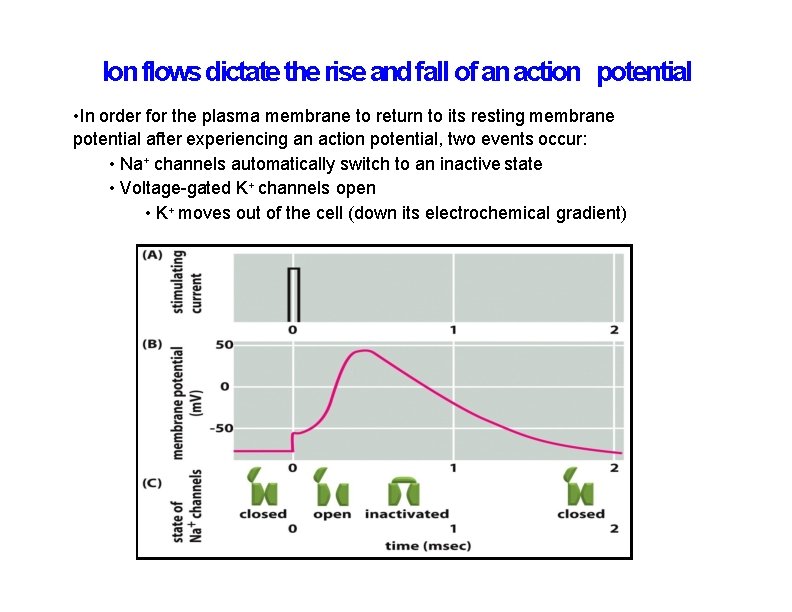
Ion flows dictate the rise and fall of an action potential • In order for the plasma membrane to return to its resting membrane potential after experiencing an action potential, two events occur: • Na+ channels automatically switch to an inactive state • Voltage-gated K+ channels open • K+ moves out of the cell (down its electrochemical gradient)
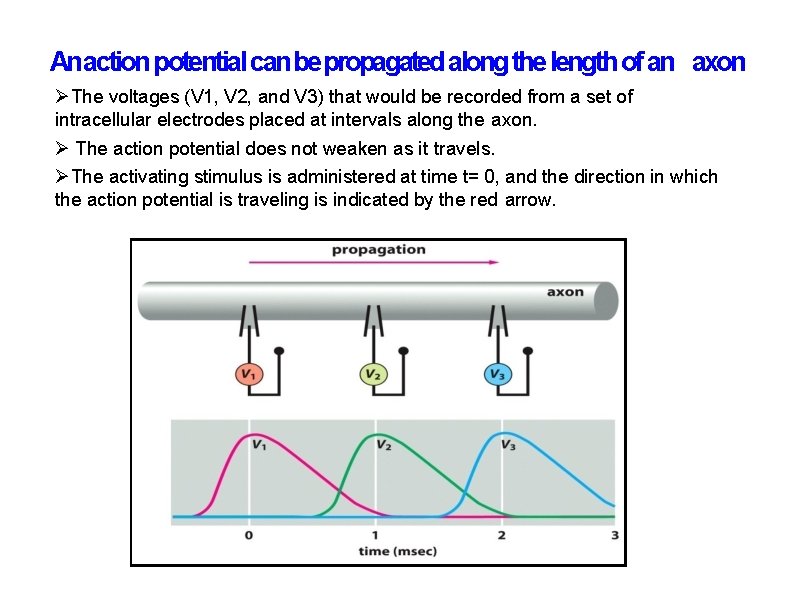
An action potential can be propagated along the length of an axon The voltages (V 1, V 2, and V 3) that would be recorded from a set of intracellular electrodes placed at intervals along the axon. The action potential does not weaken as it travels. The activating stimulus is administered at time t= 0, and the direction in which the action potential is traveling is indicated by the red arrow.

An action potential can be propagated along the length of an axon The changes in the Na+ channels and the consequent flows of electric current across the membrane (orange arrows) disturbs the membrane potential and gives rise to the traveling action potential. The region of the axon with a depolarized membrane is shaded in blue. An action potential can only travel forward from the site of depolarization. This is because Na+channel inactivation prevents the depolarization from spreading backward
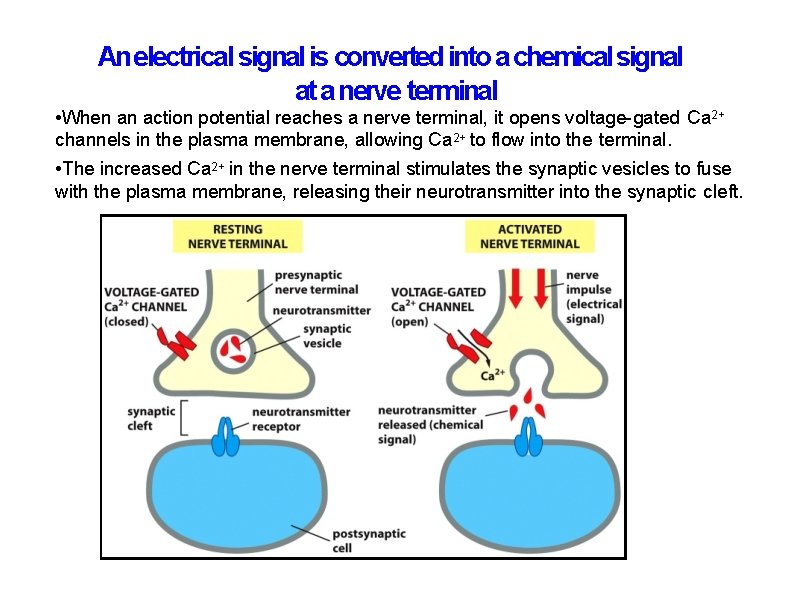
An electrical signal is converted into a chemical signal at a nerve terminal • When an action potential reaches a nerve terminal, it opens voltage-gated Ca 2+ channels in the plasma membrane, allowing Ca 2+ to flow into the terminal. • The increased Ca 2+ in the nerve terminal stimulates the synaptic vesicles to fuse with the plasma membrane, releasing their neurotransmitter into the synaptic cleft.
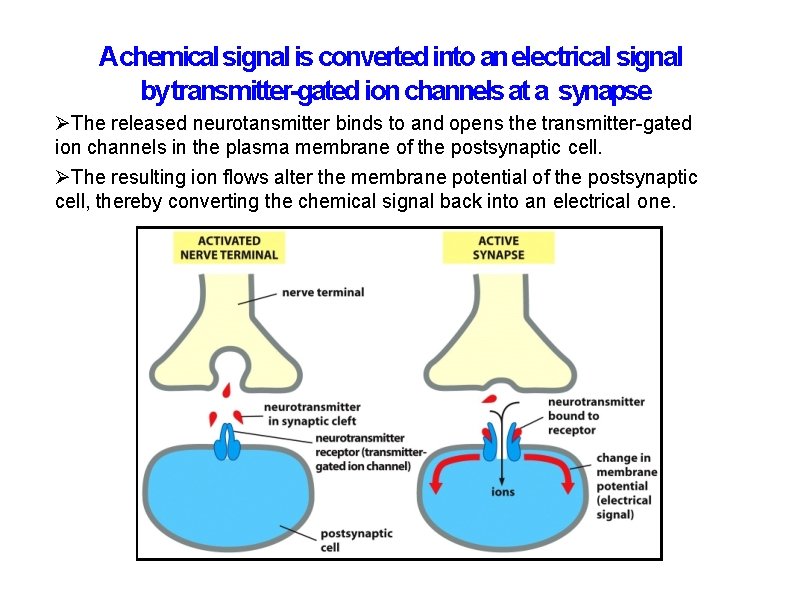
Achemical signal is converted into an electrical signal by transmitter-gated ion channels at a synapse The released neurotansmitter binds to and opens the transmitter-gated ion channels in the plasma membrane of the postsynaptic cell. The resulting ion flows alter the membrane potential of the postsynaptic cell, thereby converting the chemical signal back into an electrical one.

The acetylcholine receptor in the plasma membrane opens when it binds to the neurotransmitter acetylcholine released by a nerve • Negatively charged amino acid side chains at either end of the pore ensure that only positively charged ions, mainly Na+ and K+ can pass. • The acetylcholine binding to the channel causes its conformation change in which these side chains move apart and the gate opens, allowing Na+ to flow through the membrane.
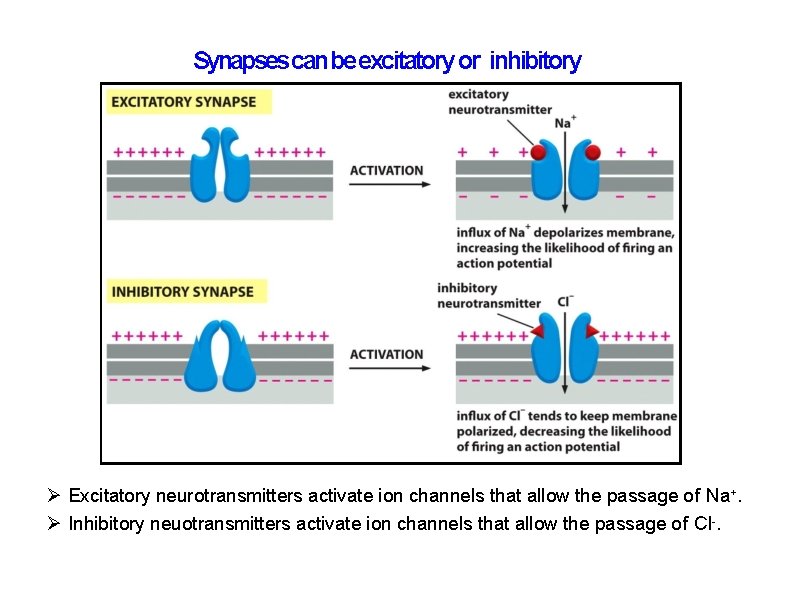
Synapses can be excitatory or inhibitory Excitatory neurotransmitters activate ion channels that allow the passage of Na+. Inhibitory neuotransmitters activate ion channels that allow the passage of Cl-.
- Slides: 35