Cell Biology Glycolysis and The Citric Acid Cycle

Cell Biology Glycolysis and The Citric Acid Cycle Alberts, Bruce. Essential Cell Biology. 4 th ed. New York, NY: Garland Science Pub. , 2013. Print. Copyright © Garland Science 2013

Three stages of cellular metabolism lead from food to waste • Stage 1 mostly occurs outside cells, although special organelles called lysosomes can digest large molecules in the cell interior. • Stage 2 occurs mainly in the cytosol, except for the final step of conversion of pyruvate to acetyl groups on acetyl Co. A, which occurs in mitochondria. • Stage 3 occurs entirely in mitochondria.

Steps of Glycolysis (1 -2)

Steps of Glycolysis (3 -5)
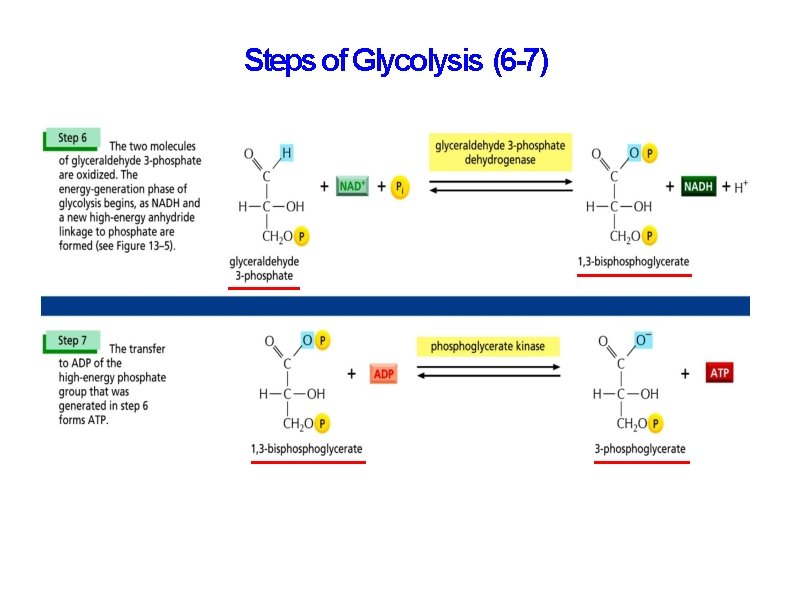
Steps of Glycolysis (6 -7)

Steps of Glycolysis (8 -10)

The oxidation of sugars begins with glycolysis The net products of glycolysis are: • 2 pyruvate • 2 ATP • 2 NADH

Net Result of Glycolysis In addition to the two molecules of pyruvate, the net product of glycolysis are two molecules of ATP and two molecules of NADH

The pyruvate is converted to lactate in the absence of oxygen When inadequate oxygen is present, for example, in a muscle cell undergoing vigorous contraction, the pyruvate produced by glycolysis is converted to lactate. The reaction restores the NAD+ consumed in glycolysis, but the whole pathway yield much less energy overall than complete oxidation. Anaerobic energy-yielding pathway like this is called fermentation.

The pyruvate is converted to ethanol and CO 2 in the absence of oxygen In some organisms that can grow anaerobically, such as yeasts, pyruvate is converted via acetaldehyde into carbon dioxide and ethanol. Again, this pathway regenerates NAD+ from NADH, as required to enable glycolysis to continue.

Fats are insoluble in water and form large lipid droplets in the specialized fat cells called adipocytes Electron micrograph of a lipid droplet in the cytoplasm The structure of fat, triacylglycerol. Three fatty acids are linked to glycerol through ester bonds

Fatty acids are converted to acetyl Co. A in mitochodria Each fatty acid in the form of fatty acyl Co. A is broken down by a cycle of reaction that trims two carbons at a time from its carboxyl end. Each turn of the cycle generates: 1 Acetyl Co. A 1 NADH 1 FADH 2

In eucaryotic cells, acetyl Co. Ais produced in the mitochondria from molecules derived from sugars and fats Most of the cell’s oxidation reactions occur in mitochondria, and most of its ATP is generated here. In eucaryotes, the citric acid cycle takes place in mitochondria, in which pyruvate and fatty acids are directed for acetyl Co. A production. Some amino acids are transported from cytosol into mitochondria, where they are also converted into acetyl Co. A or one of the other intermediates of the citric acid cycle.

The citric acid cycle catalyzes the complete oxidation of the carbon atoms in acetyl Co. A Net products: 3 NADH 1 FADH 2 1 GTP 2 CO 2

Each turn of citric acid cycle produces one molecule of GTP and GDP are close relatives of ATP and ADP, respectively, the GTP only difference being the substitution of the base guanine for adenine. The transfer of the terminal phosphate group from GTP to ADP produce one ATP molecule in each cycle. (Guanosine triphosphate)

Each turn of citric acid cycle produces one molecule of FADH 2 Oxidized form Reduced form FADH 2, like NADH, is a carrier of hydrogens and highenergy electrons. The energy that is stored in the highenergy electrons of NADH and FADH 2 is subsequently used to produce ATP through the process of oxidative phosphorylation.

Replenishing the supply of any single intermediate has a dramatic effect on the rate at which the entire citric acid cycle operates

Glycolysis and the citric acid cycle provide the precursors needed to synthesize many important biological molecules Many of the intermediates formed in the glycolysis and the citric acid cycle are used for biosynthetic (anabolic) pathways, where they are converted into amino acids, nucleotides, lipids, and other small organic molecules. Each black arrow indicates a single enzyme-catalyzed reaction The red arrows generally represent pathways with many steps that are required to produce the indicated products.

NADH donate high-energy electrons for synthesis of The electron carriers NADH and FADH 2 transfer the electrons that they have gained ATP by oxidizing other molecules to the electron-transport chain. The specialized chain of electron carriers is embedded in the inner membrane of the mitochondrion in eucaryotic cells. As the electrons pass through the series of electron acceptor and donor molecules that forms the chain, they fall to successively lower energy states. The energy released is used to drive H+ ions (protons) across the membrane to the outside. This generates a transmembrane gradient of H+ ions the serves as a source of energy for phosphorylation of ADP to generate ATP. The electrons are added to O 2 molecules which combines with H+ to produce H 2 O.

Regulation of metabolism A cell is an intricate chemical machine. Glycolysis and the citric acid cycle is only tiny fraction of the many enzymatic reactions in a cell at any time. To allow the cell to survive and respond to its environment, the choice of which pathway each metabolite will follow must be carefully regulated at every branch point. To maintain order with cells, all organisms need to constantly replenish their ATP pools through sugar or fat oxidation. Since animals have only periodic access to food, and plants need to survive overnight without sunlight, one way is to synthesize food reserves in times of plenty that can be consumed when other energy sources are scarce. A cell must control whether key metabolites will be routed into anabolic or catabolic pathways – in other words, whether they will be used to build other molecules or burned to provide energy.

Glycolysis and the citric acid cycle lie at the center of The same molecule is often a metabolism part of many different pathways. Pyruvate is a substrate of half a dozen or more different enzymes. • Pyruvate dehydrogenase converts pyruvate to acetyl Co. A • Lactate dehydrogenase convert it to lactate • A third enzyme can convert it to oxaloacetate • A fourth enzyme can changes it to the amino acid alanine. All these different pathways compete for the same pyruvate molecule, and similar competition for thousands of other small molecules go on at the same time. The metabolic balance of a cell is amazingly stable and regulated by an elaborate net work of control mechanisms that act on enzymes to regulate and coordinate the rates of the many metabolic reactions in a cell.

Gluconeogenesis ‘reverse” reactions that occur during A set of four bypass reactions (labeled A, B, C, and D) is glycolysis needed to get around steps 1, 3, and 10 in glycolysis, which are essentially irreversible. Gluconeogenesis is a process in which blood glucose is synthesized from small non-carbohydrate organic molecules such as lactate, pyruvate, or amino acids. Synthetic reactions carried out in gluconeogenesis require an input of energy, whereas glycolysis as a whole is an energetically favorable set of reactions. Gluconeogenesis occurs mainly in liver cells to keep the blood supplied with glucose by using different molecules. The balance between glycolysis and gluconeogenesis must be highly regulated, so that glucose is broken down rapidly when energy reserves run low, but is synthesized and exported to other tissues when the liver cell has sufficient energy reserves in the form of pyruvate, citrate, or ATP. One of the key control point is the breakdown of glucose lies in step 3 of glycolysis, the production of fructose 1, 6 -bisphosphate by the enzyme phosphofructokinase. It is activated by AMP, ADP and inorganic phosphate – the byproducts of ATP hydrolysis, and inhibited by ATP, citrate, and fatty acid. The other enzyme that catalyzes the reverse reaction (B), fructose 1, 6 -bisphosphatase, is regulated in the opposite direction.

Animal cells store glycogen to provide energy in times of fasting The structure of glycogen in animal cells is very similar to starch in plants as branched polymer of glucose. Starch has many fewer branch points than glycogen. An electron micrography showing glycogen granules in the cytoplasm of a liver cell.

Animal cells store glycogen to provide energy in times of When more ATP is needed than can fasting be generated from food molecules taken from the bloodstream, cell break down glycogen. The glycogen phosphorylase catalyzes the break-down reaction of glycogen to glucose 1 -phospate, which is then converted to the glucose 6 -phosphate that feeds into the glycolytic pathway. The glycogen synthetic and degradation pathways are regulated by glucose 6 -phosphate, but in opposite directions. Glycogen synthase in the synthetic pathway is activated by glucose 6 phosphate, whereas the glycogen phosphorylase that catalyzes the breakdown of glycogen is inhibited by both glucose 6 -phosphate and ATP.

Fats are stored in the form of fat droplets in animal cells Fat is far more important storage material than glycogen, in part because the oxidation of a gram of fat releases about twice as much energy as the oxidation of a gram of glycogen. Glycogen binds a great deal of water, producing a six fold difference in the actual mass of glycogen required to store the same amount of energy as fat. An average of adult human stores enough glycogen for only about a day of normal activity, but enough fat to last nearly a month. Fat droplets (stained red) accumulate in developing adipose cells.
- Slides: 25