Cell Biology and Physiology The Eukaryotic Cell Cycle

Cell Biology and Physiology The Eukaryotic Cell Cycle Chapter 19 Dr. Capers Molecular and Cell Biology, Lodish, 8 th edition

• Watch this video on cell cycle checkpoints: • https: //www. youtube. com/watch? v=Yb 4 d. Qu PLgt 8

• Proper control of all cell division is vital to all organisms • Cell division is controlled by surveillance mechanisms known as checkpoint pathways • Master controllers of the cell cycle are small number of protein kinases • Regulatory subunit – cyclin • Catalytic subunit – cyclin-dependent kinase (CDK) • When CDK binds cyclin, becomes active
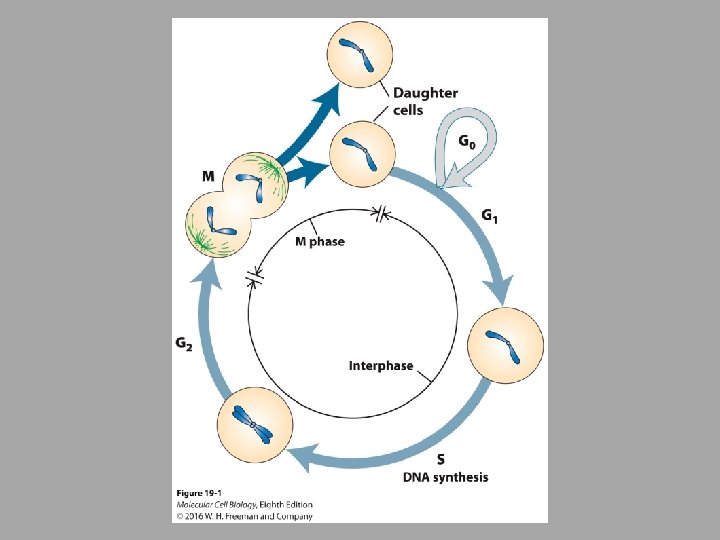

• Cell cycle divided into 4 major phases: • G 1 – cell grows in size, synthesize RNAs and proteins required for DNA replication • S – DNA synthesis • G 2 – cell gets bigger • M – mitotic phase
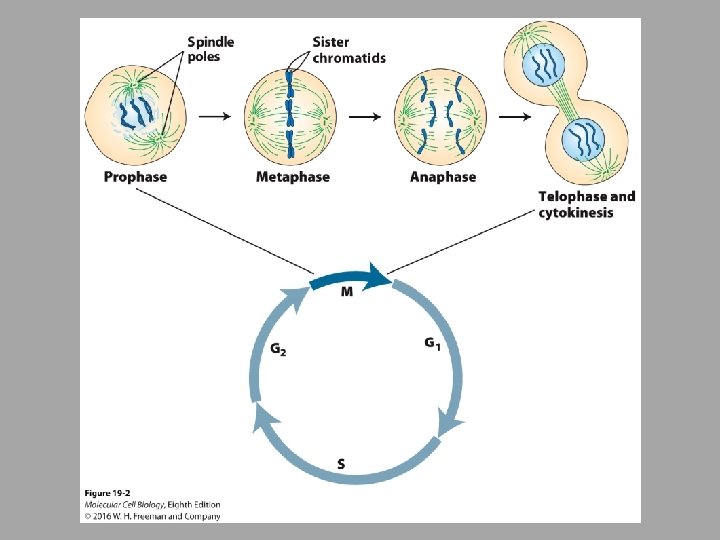
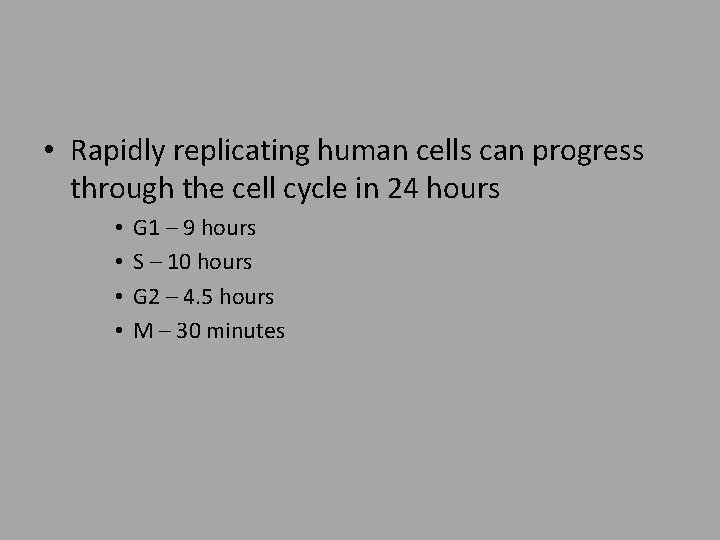
• Rapidly replicating human cells can progress through the cell cycle in 24 hours • • G 1 – 9 hours S – 10 hours G 2 – 4. 5 hours M – 30 minutes

• Goal of each cell division is to generate 2 daughter cells of identical make-up – Cell cycle events must always happen in order – For instance, DNA replication must always precede chromosome segregation – CDKs that promote S phase are active during S phase but inactive during M phase

• Model organisms and methods of studying the cell cycle • Yeasts – Saccharomyces cerevisiae and Schizosaccharomyces pombe • Early frog embryos, Xenopus laevis • Fruit fly, Drosophila melanogaster • Mammalian tissue culture cells

• Regulation of CDK Activity – 3 key features of the kinases: • Cyclin-dependent kinases (CDKs) are active only when bound to a regulatory cyclin subunit • Different types of cyclin-CDK complexes initiate different events. » G 1 CDKs and G 1/S CDKs promote entry into cell cycle » S CDKs trigger S phase » Mitotic CDKs initiate mitosis • Multiple mechanisms are in place to ensure that the different CDKs are active only in the stages of the cell cycle trigger

• Cyclin-dependent kinases (CDKs) are a family of small serine/threonine kinases

• Cyclin levels are primarily regulated by protein degradation • Timely activation of CDKs depends on the presence of the appropriate cyclins in the cell • Transcriptional control of the cyclins is one way to manage this • Ubiquitin-mediated, proteasome-dependent protein degradation is another way to control

• Commitment to the cell cycle and DNA replication

E 2 F is transcription factor When Rb is bound to E 2 F, E 2 F is inactive Growth factors detected by cell stimulate G 1 CDKs phosphorylate Rb causing it to Rrelease E 2 F

• Whether or not cells enter the cell cycle is influenced by extracellular as well as intracellular signals • Yeasts, for example, only enter into the cell cycle when they have reached the critical cell size • When nutrients are abundant • In multicellular organisms, control comes from growth factors » Mitogens – promote growth » Anti-mitogens – inhibit growth

Control of S phase onset in yeast by regulated proteolysis of the S phase inhibitor Sic 1

DNA replication is initiated from helicase loading sites known as replication origins

Cohesions establish linkages between the replicated DNA (sister chromatids) which are essential for their accurate segregation later in the cell cycle

• Entry into Mitosis • Once the S phase is finished, the cell can enter into mitosis • Sister chromatids are separated • Mitotic spindle must form • Cell remodels as far as location of organelles, etc • All triggered by mitotic CDKs

• Mitotic cyclins synthesized during S and G 2 activate mitotic CDKs. • The dual-specificity Wee 1 kinase inhibits mitotic cyclin-CDK activity by phosphorylation of CDK T 14 and Y 15 until DNA replication is completed. • When DNA replication is completed, the dual-specificity Cdc 25 phosphatase is activated (by an unknown mechanism) and dephosphorylates CDK T 14 and Y 15 , reversing the inhibitory effect of Wee 1 and activating cyclin-CDK activity.

• Mitotic CDKs collaborate with other protein kinases to bring about mitotic events • Polo kinase family – critical formation of mitotic spindle • Aurora kinase – plays role in ensuring chromosome bind to mitotic spindle

• Mitotic CDKs promote nuclear envelope breakdown • Lipid bilayer of the inner nuclear membrane is associated with the nuclear lamina (intermediate filaments) • Mitotic CDKs once activated at the end of G 2 phosphorylate specific serine residues in the nuclear lamins – Causes depolymerization and disintegration

• Mitotic CDKs promote mitotic spindle formation • Made of microtubules that attach to chromosome via the kinetochore • Function of the mitotic spindle is to segregate chromosomes so that the sister chromatids separate from each other and move opposite poles of the cell – Mitotic spindle must attach so that the kinetochore of each sister chromatid pair attaches to microtubules emanating from opposite poles » Bi-oriented

• Chromosome condensation facilitates chromosome segregation • Activated by mitotic CDKs


• Inactivation of mitotic CDKs leads to dephosphorylation of the CDK substrates leading to exit from mitosis • Degradation of mitotic cyclins leads to this inactivation of mitotic CDKs

Reassembly of the nuclear Envelope during telophase

• Cytokinesis produces 2 daughter cells – In animals this happens by a contractile ring • Made of actin and actin motor myosin

• Checkpoint pathways • Surveillance mechanisms – operate to ensure that the next cell cycle event is not initiated until the previous one is done and until the proper signals are there • Checkpoint pathways consist of: – Sensors – monitor particular cellular event – Signaling cascade – initiates the response – Effector – halts cell cycle progression and activates repair when necessary


• Cell cycle events that are monitored by checkpoint pathways: • • • Growth DNA replication DNA damage Kinetochore attachment to the mitotic spindle Positioning of the spindle • If a defect cannot be corrected, apoptosis will be induced

• Nutrients and therefore cell size initiates cell division in unicellular organisms • Nutrients are not usually the limiting factor in multicellular organism • Cell growth is controlled by growth factor signaling pathways: » Ras » AMPK » TOR

• DNA has to be completely and accurately replicated for cell division to occur correctly • Enzymes that replicate DNA are very precise by inaccuracies can occur • DNA response system – repair pathways • In multicellular organisms, if the repair is too big, the cell will go through apoptosis

• DNA damage • Detected by ATM and ATR – ATM – ataxia telangiectasia mutated (mutations were found when studying ataxia telangiectasia – issues with nervous, immune system, problems in correcting DNA) – ATR – ataxia telangiectasia and Rad 3 -related protein (mutations cause Seckel syndrome – microcephaly and other issues) • Both of these proteins are recruited to areas of DNA damage » The recruit other protein kinases, Chk 1 and Chk 2 » Can activate DNA repair or apoptosis


• The spindle assembly checkpoint pathway prevents entry into anaphase until every kinetochore of every chromatid is properly attached • Aurora B and the spindle assembly checkpoint pathway collaborate during every cell cycle to accurately attach every single pair of sister chromatids to the mitotic spindle in the correct, bi-oriented manner

• Meiosis • In nearly all diploid eukaryotes, meiosis results in haploid cells • Homologous chromosomes have to find each other and one homologous chromosome will end up in the resulting gamete • DNA replication followed by 2 divisions • Specialized cues induce meiotic division



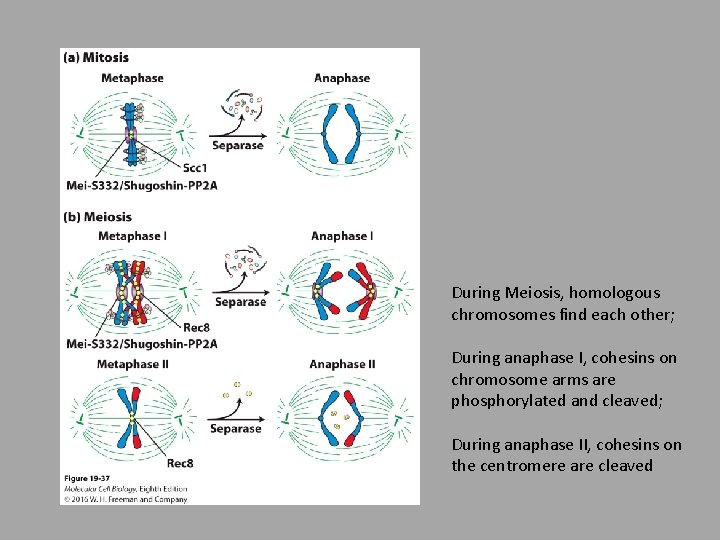
During Meiosis, homologous chromosomes find each other; During anaphase I, cohesins on chromosome arms are phosphorylated and cleaved; During anaphase II, cohesins on the centromere are cleaved
- Slides: 41