Celiac Disease Ben Thomas D O Gastroenterology Specialty

Celiac Disease Ben Thomas, D. O. Gastroenterology Specialty Medicine Care, Beavercreek, OH

• 38 y. o. female new patient with biopsy and serology confirmed celiac disease. She has been on a strict gluten free diet for 5 years and doing well. She visits your office for a recent increase in symptoms. What is most likely causing her symptoms? • A- Small Bowel Lymphoma • B- Inadvertent Gluten Ingestion*** • C- Irritable Bowel Syndrome • D- Type 2 Refractory Celiac Disease
38 y. o. female new patient with biopsy and serology confirmed celiac disease. She has been on a strict gluten free diet for 5 years and doing well. She visits your office for a recent increase in symptoms. What is most likely causing her symptoms? 25% 25% A. B. C. D. A. Small Bowel Lymphoma B. Inadvertent Gluten Ingestion C. Irritable Bowel Syndrome D. Type 2 Refractory Celiac Disease 10
21 y. o. male with Type 1 diabetes. New bloating and diarrhea. Mild iron deficiency anemia. You suspect celiac disease. What is the best initial evaluation? A. Initiate gluten free diet and assess for response 25% 25% A. B. C. D. B. Order upper endoscopy with gastric biopsies C. HLA testing D. TTG Ig. A antibody testing with serum Ig. A level 10
You’ve been asked to evaluate a 35 y. o. female who is concerned that she may have celiac disease. Her friend told her to start a gluten free diet. She feels a little better on the diet. How would you test her today to rule out celiac disease? 25% 25% A. Upper endoscopy with biopsy B. Anti-TTG Ig. A C. HLA testing D. D-xylose testing A. B. C. D. 10

• First described by Samuel Gee in 1888, “On the Coeliac Affection” • Cause was unexplained until Dutch physician during WWII noticed patient symptoms change during food shortages/rationing • • wheat, barley, rye, and oats Lesions in the small bowel were not identified until 1954

• One of the most common causes of chronic malabsorption • injury to the small intestine with loss of absorptive surface area, reduction of digestive enzymes, and impaired absorption • net secretion of fluid

• Epidemiology • primarily in whites of northern European ancestry • • 1: 70 to 1: 300 individuals affected strong family history • 1: 22 first degree relatives

• Underdiagnosed in the United States • prevalence in dyspepsia (1%)- mucosal biopsies should be considered in patients with dyspepsia • first degree relative increases risk (5 -10%) • • testing asymptomatic patients? type 1 diabetes mellitus (3 -10%)

• Risk of malignancy • non-hodgkin’s lymphoma • increased risk of all-cause mortality

• Genetic factors • immune disorder triggered by an environmental agent (gliadin) • • HLA DQ 2 and/or DQ 8 Serum autoantibodies • ELISA for Ig. A antibodies to gliadin • immunofluorescence for Ig. A antibodies to endomysium • • tissue transglutaminase Ig. G antibody deaminated gliadin

Presentation • • Classic Celiac Disease • villous atrophy • symptoms of malabsorption • resolution of mucosal lesions and symptoms upon withdrawal of gluten-containing foods Typically present with diarrhea, weight loss, or malabsorption and have antibodies against gliadin (TTG)

• Atypical celiac disease- anemia, dental enamel defects, osteoporosis, arthritis, elevated liver enzymes, infertility • Silent Celiac Disease- incidental finding with minor symptoms that resolve on gluten free diet

• presents often between the ages of 10 and 40 years • diarrhea with bulky, foul-smelling, floating stools, flatulence • growth failure in children, weight loss, anemia, deficiencies in B vitamins, and osteopenia

• Subclinical disease • fatigue, mild iron deficiency, mild elevation in transaminases, or asymptomatic • still at risk for malignancy, nutritional deficiencies, low birth weights, and other autoimmune diseases

• Non-GI manifestations • Neuropsychiatric disease- headache, neuropathy, ataxia, depression • Iron deficiency- • Metabolic bone disease- malabsorption, vitamin D deficiency • Fertility

• Anemia • common manifestation • impaired iron or folate absorption from proximal intestine • hyposplenism, thrombocytosis, deformed erythrocytes

• Osteopenia • common manifestation in older patients • impaired calcium absorption and vitamin D deficiency • unclear whether untreated celiac disease leads to increase in fractures

• Neurologic symptoms • poorly understood • progressive gait and limb ataxia • • immunologic damage to the cerebellum, spinal cord, and peripheral nerves epilepsy and seizures

• Fertility problems • common in women with untreated celiac disease • amenorrhea • infertility • sperm abnormalities

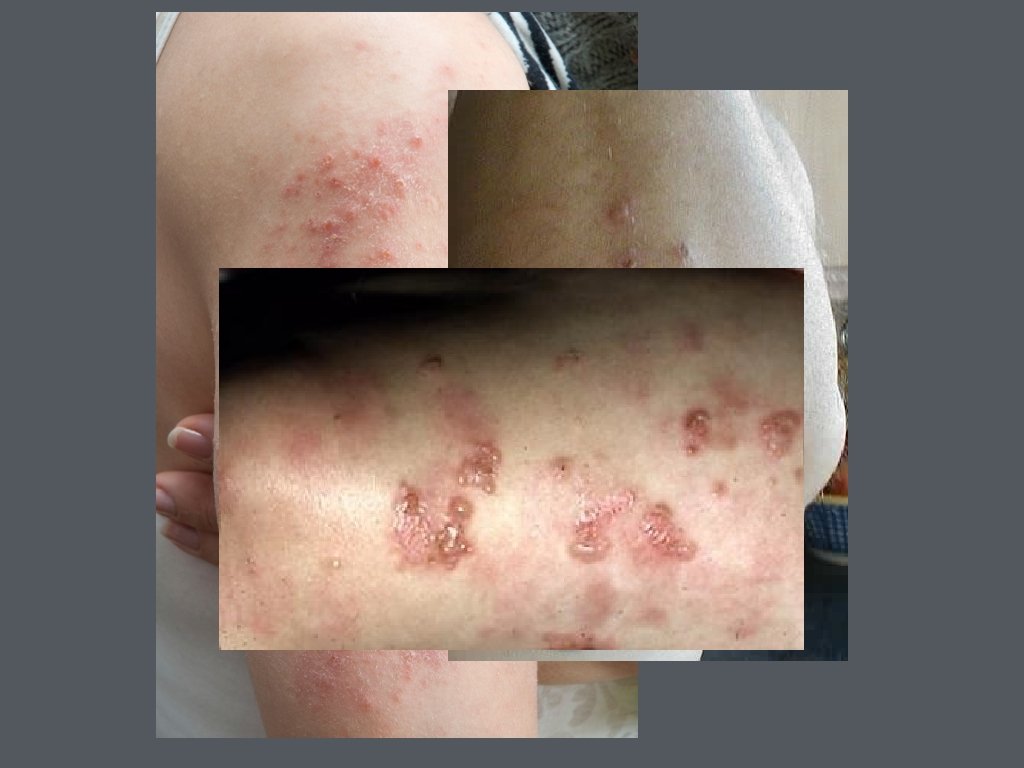
• Dermatitis herpetiformis • papulovesicular lesion that occur symmetrically over extensor surfaces of extremities and buttocks, trunk, neck, and scalp • immunofluorescence of speckled Ig. A deposits • can have patchy erythema in intestine unrelated to celiac disease • HLA-DQ 2 association

• Diabetes mellitus • closely associated with type 1 diabetes and autoimmune thyroiditis • shared genetic loci- HLA DR 3, DQ 2 (DQ 8) • Down Syndrome- 20 fold increase risk of celiac disease • Liver disease- non-specific mild chronic elevation in AST/ALT

Diagnosis • Who should be tested? • chronic or recurrent diarrhea, malabsorption, weight loss, abdominal distention or bloating • iron deficiency anemia, folate, or B 12 deficiency, elevated liver enzymes, neuropathies, or recurrent headaches • type 1 diabetes and first degree relatives with celiac disease; Down syndrome

• Most important genetic risk factor for celiac disease • HLA-DQ 2 and DQ 8 • negative testing makes celiac disease very unlikely
• Serology • Ig. A anti-tissue transglutaminase • Ig. G-deaminated gliadin • important for testing to be performed without gluten withdrawal • DQ 2/DQ 8 testing

• Ig. A endomysial assay • bind to connective tissue surrounding smooth muscle cells • positive or negative test • target antigen is tissue transglutaminase • moderately sensitive and highly specific

• Antigliadin antibody assay • component of wheat storage protein gluten • not recommended due to low positive predictive value • second generation testing against deaminated gliadin more sensitive and specific

• Anti-tissue transglutaminase (TTG) antibody • improvement upon anti-gliadin, reticulin, and endomysium antibody based testing • Ig. A deficiency • Ig. G based TTG or Ig. G DGP

• Small bowel biopsy • upper endoscopy with small bowel biopsies • duodenal mucosa appears atrophic with loss of folds, visible fissures, nodular appearance, or scalloping of the folds • histologic features include loss of villi, increased intraepithelial lymphocytes, mucosal atrophy, and crypt hyperplasia
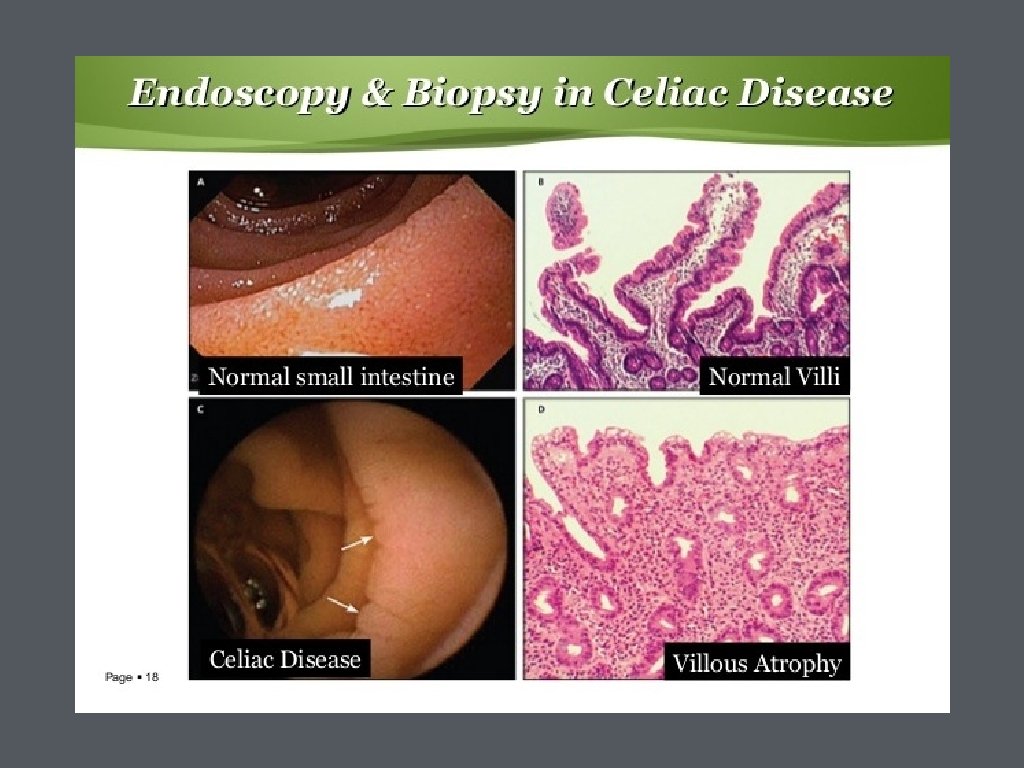
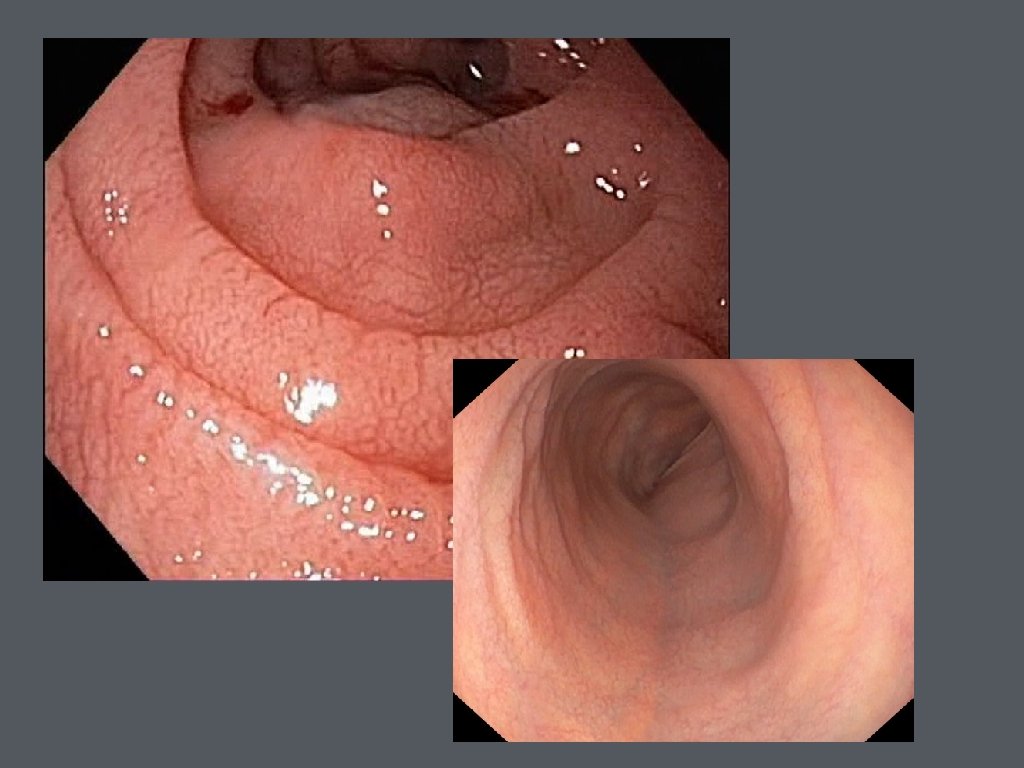

• Biopsies are recommended 2 from duodenal bulb and at least 4 from beyond the duodenal bulb • Marsh Classification
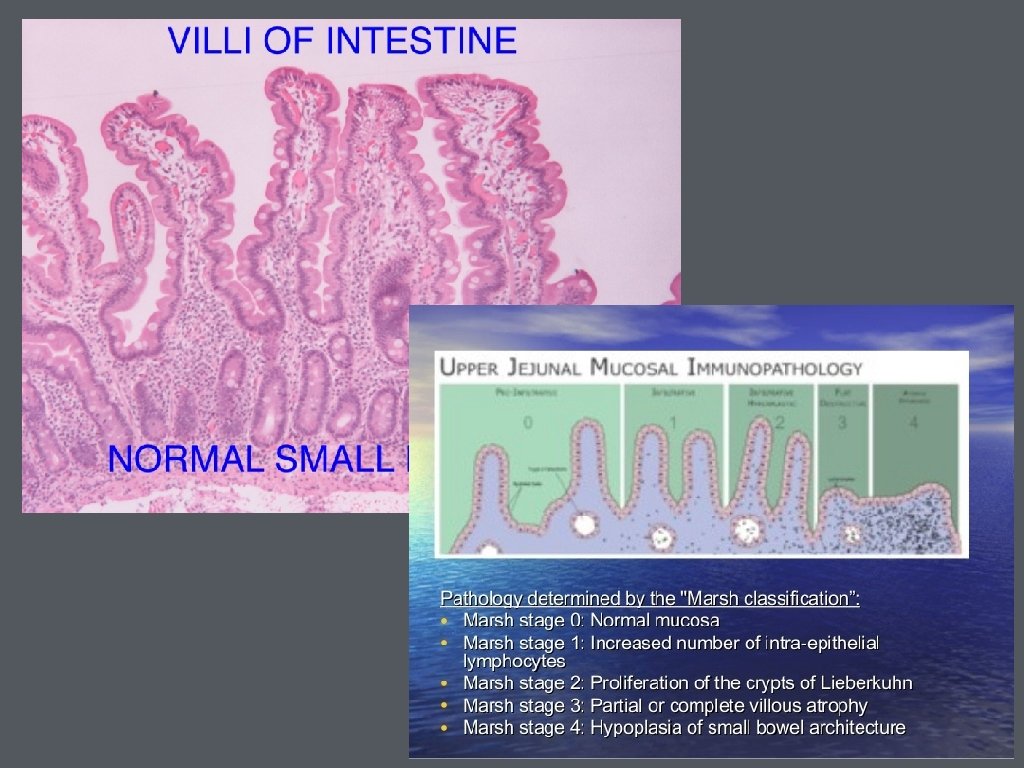
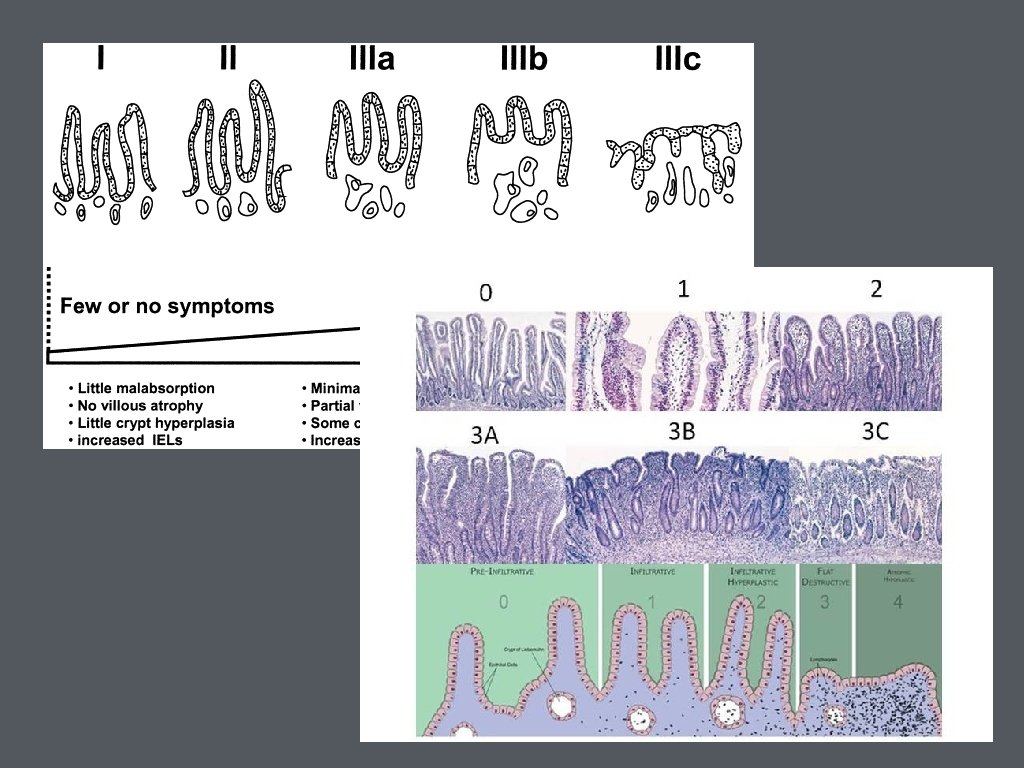
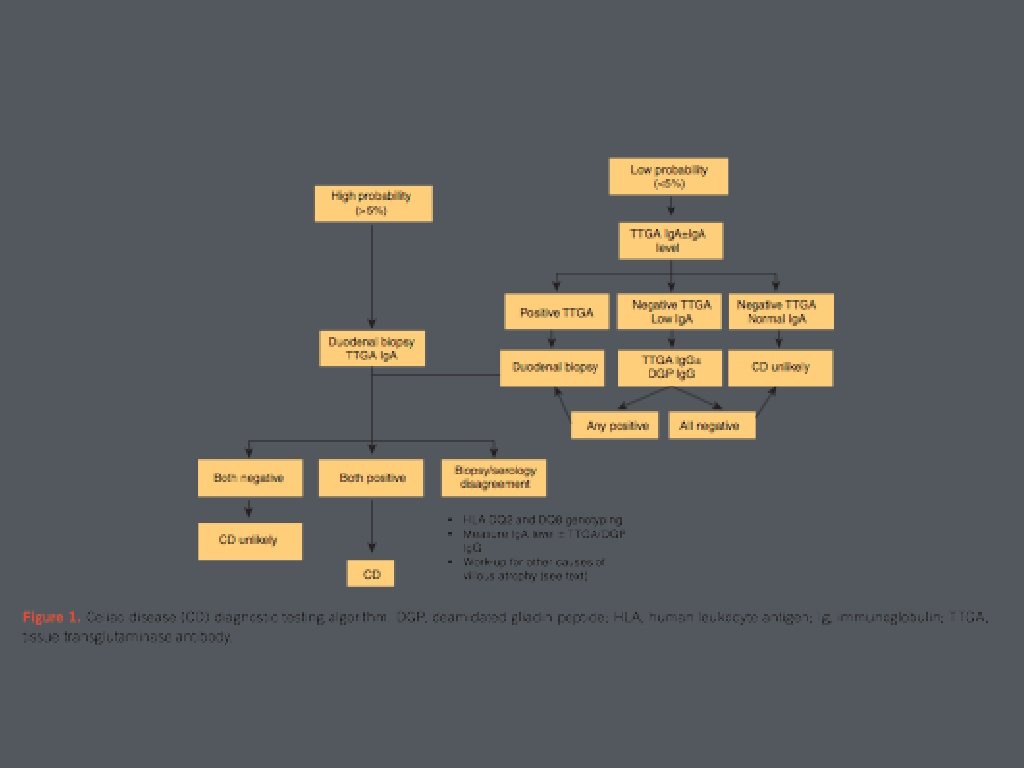

• Clinical features without positive serologic testing • Ig. A deficiency • low gluten diet • false negative testing • non-celiac disease- wheat allergy or non-celiac gluten sensitivity

• Positive Serologies but Negative Biopsies • repeat Ig. A based TTG testing • gluten challenge (6 -12 weeks)
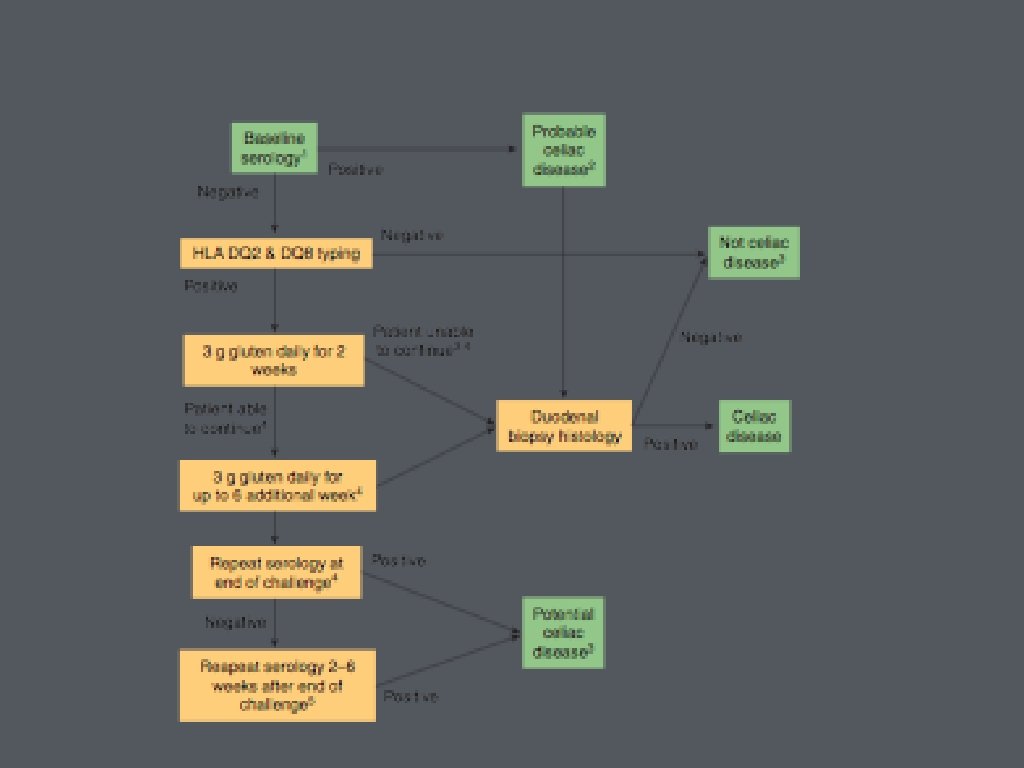

• Confirmation of diagnosis • symptoms alone cannot differentiate • 20 -50% of patients with celiac disease met Rome criteria for IBS • ingestion of gluten cause GI symptoms regardless of celiac disease
• Other testing • capsule endoscopy • CT enterography (MR) • D-xylose measurement

Treatment • Dietary Counseling • gluten free diet recommended in patients with celiac disease • referral to a registered dietitian knowledgeable about celiac disease

• lifelong gluten free diet is the only effective treatment for celiac disease • decreases risk for malignancies associated with celiac disease • improves nutritional parameters

• • principle sources of dietary gluten • wheat • rye • barley • oats? ? read labels carefully, especially additives

• distilled alcohol and wine • dairy products may not be well tolerated • oats should be introduced with caution and intake be limited

• Response to gluten free diet • 70% have noticeable clinical improvement within 2 weeks • symptoms improve much faster than histology • repeat lab testing 4 -6 weeks after gluten free diet

• Should we stay on a gluten free diet? • micronutrient deficiencies • increased overall mortality and risk of lymphoproliferative disease and GI cancers • development of other autoimmune disorders

• Repeat serologic testing • Ig. A anti tissue transglutaminase or Ig. A deaminated gliadin peptide should be used to monitor response to gluten-free diet • • normal levels do not correlate with histology persistently high levels usually reflect continued exposure to dietary gluten

• Repeat small bowel biopsy • upper endoscopy should be repeated in patients who fail to respond to a gluten-free diet or have relapse of symptoms on a gluten-free diet

• if duration of gluten free diet less than 1 month traditional testing/serology often are still abnormal • DQ 2 and DQ 8 testing are not influenced by diet • gluten challenge

• Non-responders • persistent symptoms or serologic/histologic abnormalities after two years on a gluten free diet • Often due to: • poor compliance or inadvertent gluten ingestions • features that overlap with celiac disease • other disorders • refractory sprue • ulcerative jejunitis or intestinal lymphoma

• • Poor compliance • most common reason for lack of response • recommend meticulous dietary history should be obtained and dietary counseling Other diagnosis • lactose intolerance, irritable bowel disease, small bowel bacterial overgrowth, pancreatic insufficiency, microscopic colitis

• Attention must be paid to hidden sources of gluten in commercially prepared foods • Registered dietitians are trained to evaluate and counsel patients on how to maintain a strict gluten free diet • Academy of Nutrition and Dietetics • long term monitoring for compliance

• Non-celiac gluten sensitivity • no diagnostic features of celiac disease but develop celiac-like symptoms upon exposure to dietary gluten

• Long term follow up is recommended • annual follow up with serology is associated with increased rate of seroconversion of TTG antibody • gold standard for monitoring adherence to diet is consultation with dietitian • median time from onset of gluten free diet to mucosal healing was 3 years
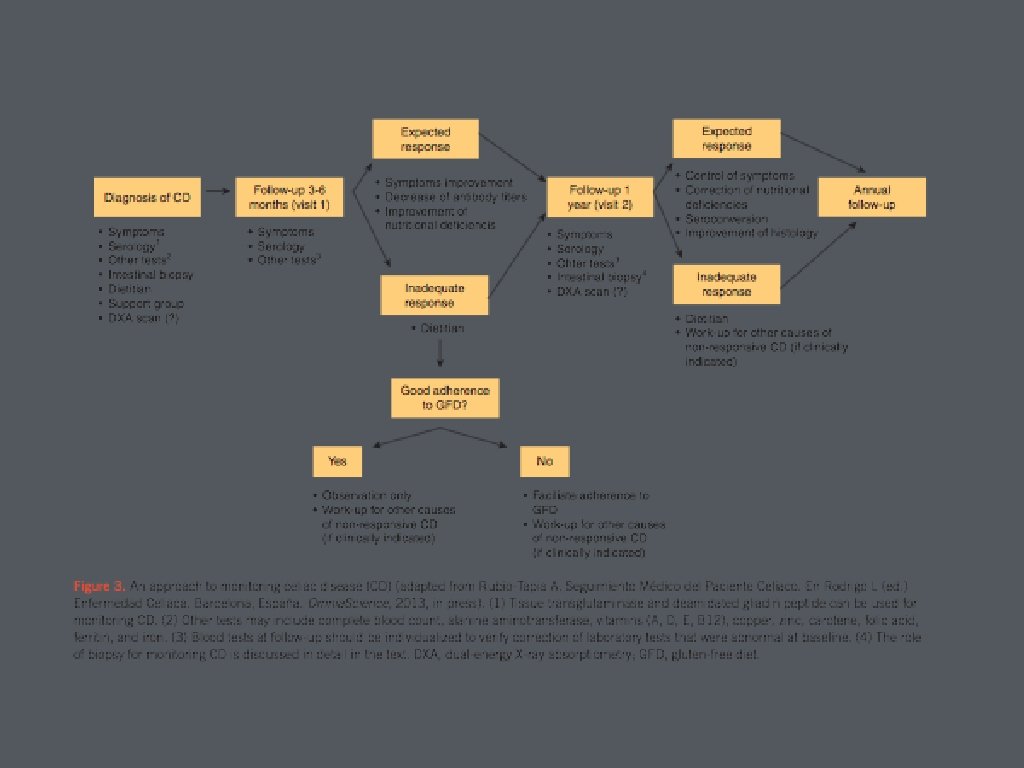

• Non-responsive or Refractory Celiac Disease • persistent symptoms, signs, and lab abnormalities typical of celiac disease despite 6 -12 months of dietary gluten avoidance • most commonly inadvertent gluten ingestion • if dietary causes have been excluded, repeat endoscopy with biopsy is recommended

• Ulcerative jujunoileitis • rare complication of celiac disease • ulceration and strictures of the small intestine • suspect in celiac disease with weight loss, abdominal pain, and diarrhea that do not respond • enteroscopy, capsule endoscopy, or small bowel studies

• Refractory celiac disease is persistent or recurrent symptoms and signs of malabsorption with small bowel villous atrophy despite strict GFD for more than 12 months • type 1: lymphocyte infiltration of small bowel mucosa • type 2: CD 3 -positive intraepithelial T cells with abnormal immunophenotype

• Type 1 RCD • excluding gluten exposure • treatment for nutrition deficiencies • symptomatic treatment • systemic steroid therapy; azathioprine

• Type II RCD • same as Type I RCD but less likely to respond • profound malnutrition requiring parenteral nutrition support • 5 year survival 44% compared to 93% with Type I • transformation to enteropathy-associated Tcell lymphoma is prominent risk

• Questions? ?

• Resources • American College of Gastroenterology Guidelines (ACG) - Diagnosis and Management of Celiac Disease • American Gastroenterological Association (AGA) Institute Technical Review on the Diagnosis and Management of Celiac Disease • Google Images • Sleisenger and Fordtran’s Gastrointestinal and Liver Disease (Ninth Edition) • Up. To. Date
- Slides: 63