Celecoxib Therapy For RRP Dr Bettie Steinberg Dr

Celecoxib Therapy For RRP Dr. Bettie Steinberg, Dr. Allan Abramson, Dr. Mark Shikowitz LIJ Medical Center Dr. Richard Smith, Dr. Harry Hoffman - Univ. Iowa Dr. Mark Courey – Univ. California, San Francisco Dr. Craig Derkay, Dr. John Sinacori – Eastern Virginia Medical School Dr. Glenn Peters, Dr. Paul Castellanos – Univ. Alabama, Birmingham Dr. Clark Rosen, Univ. Pittsburgh

Cyclooxygenase (COX-2) • Enzyme elevated in inflammation • Also elevated in many premalignant and malignant tumors – – – – Colorectal polyps Respiratory papillomas Head and neck cancer Breast cancer Lung cancer Cervical cancer Prostate cancer • Inhibiting COX-2 helps other types of tumors
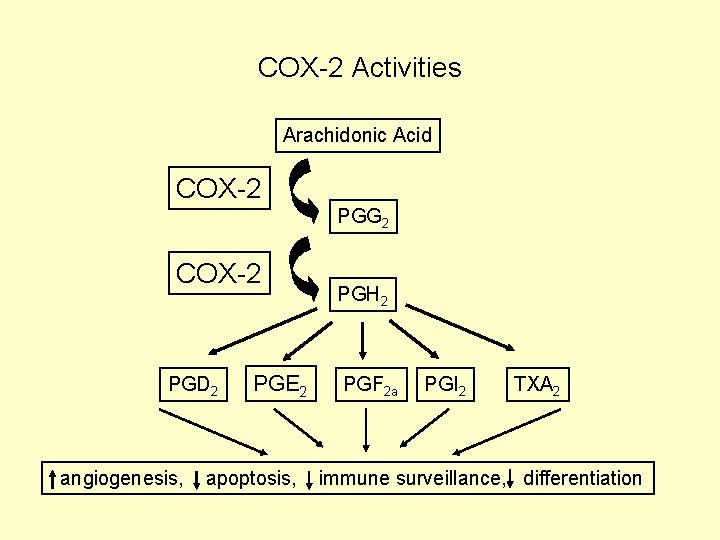
COX-2 Activities Arachidonic Acid COX-2 PGG 2 COX-2 PGD 2 angiogenesis, PGE 2 apoptosis, PGH 2 PGF 2 a PGI 2 TXA 2 immune surveillance, differentiation
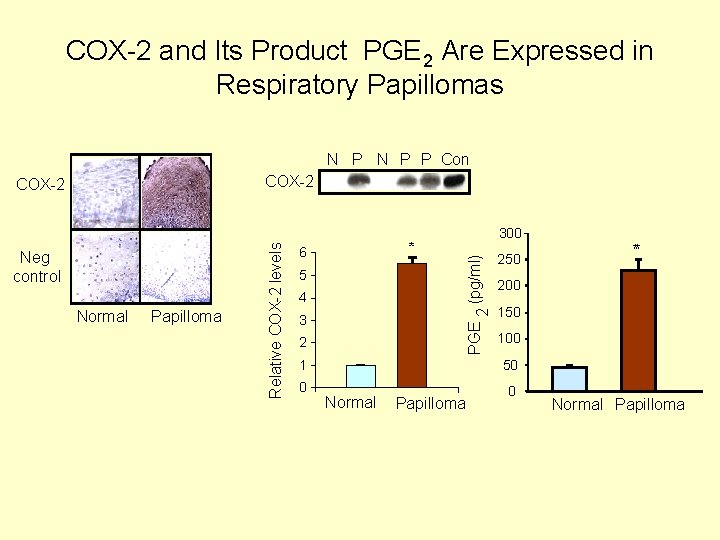
COX-2 and Its Product PGE 2 Are Expressed in Respiratory Papillomas N P P Con Normal Papilloma * 6 5 4 3 2 300 PGE 2 (pg/ml) Neg control Relative COX-2 levels COX-2 250 200 150 100 1 50 0 0 Normal Papilloma * Normal Papilloma
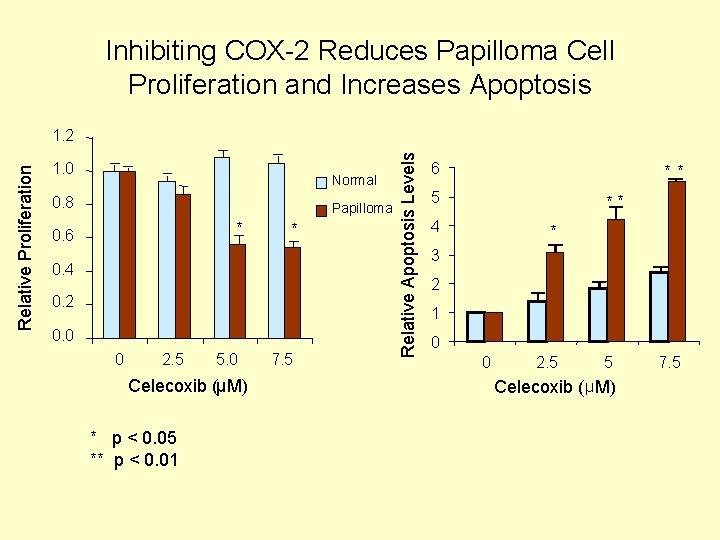
Inhibiting COX-2 Reduces Papilloma Cell Proliferation and Increases Apoptosis 1. 0 Normal 0. 8 Papilloma * 0. 6 * 0. 4 0. 2 0. 0 0 2. 5 5. 0 Celecoxib (µM) * p < 0. 05 ** p < 0. 01 7. 5 Relative Apoptosis Levels Relative Proliferation 1. 2 6 ** 5 ** 4 * 3 2 1 0 0 2. 5 5 Celecoxib ( µ M) 7. 5
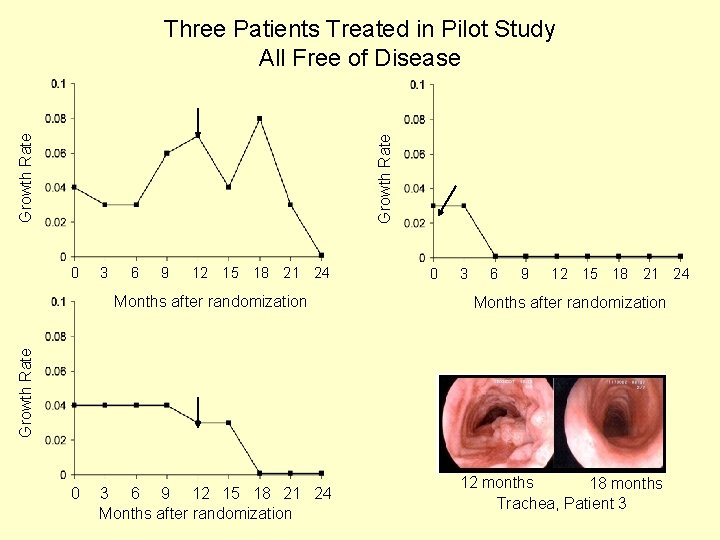
Growth Rate Three Patients Treated in Pilot Study All Free of Disease 0 3 6 9 12 15 18 21 24 Months after randomization Growth Rate Months after randomization 0 0 3 6 9 12 15 18 21 24 Months after randomization 12 months 18 months Trachea, Patient 3

New NIH Grant To Study Efficacy of Celebrex • 5 year grant • Enrolling patients beginning of year • Grant will pay for patient travel to one of participating centers • Celebrex provided by Pfizer at no cost • Grant will permit us to determine whether Celebrex is an effective therapy for RRP, and if some patients respond and others do not, why

Eligibility • Age 4 years or older • 3 or more surgeries in past year or tracheal/ broncheal involvement • No history of heart disease or current high blood pressure • No significant kidney or liver disease • Not allergic to Celebrex or sulfa drugs
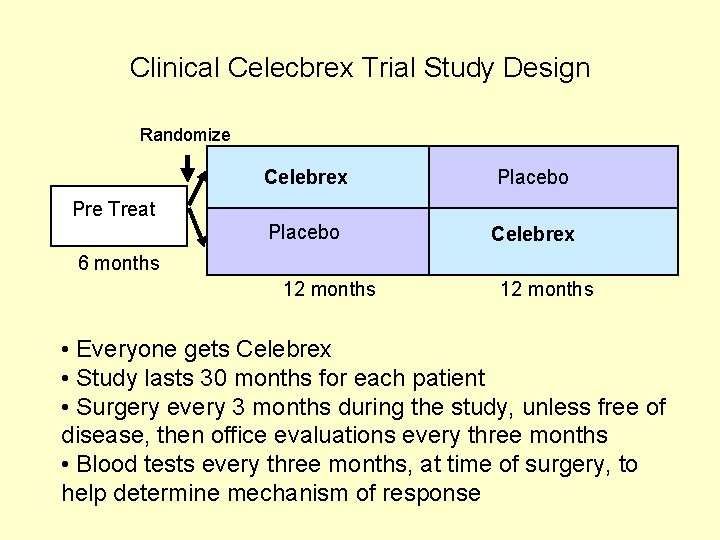
Clinical Celecbrex Trial Study Design Randomize Celebrex Placebo Celebrex Pre Treat 6 months 12 months • Everyone gets Celebrex • Study lasts 30 months for each patient • Surgery every 3 months during the study, unless free of disease, then office evaluations every three months • Blood tests every three months, at time of surgery, to help determine mechanism of response

If Interested in Participating Send patient records to: Dr. Allan Abramson or Dr. Mark Shikowitz Department of Otolaryngology Long Island Jewish Medical Center 270 -05 76 th Ave New Hyde Park, NY 11040 Tel: 718 -470 -7550
- Slides: 10