CEA CARCINO EMBRYONIC ANTIGEN CEA maybe elevated in













CEA- CARCINO EMBRYONIC ANTIGEN CEA maybe elevated in some malignancies : Breast, colon, pancreas, ovary, bladder, HN CA. s CEA may also be raised in some non-neoplastic conditions : ulcerative colitis , pancreatitis, cirrhosis, COPD, Crohn's disease, hypothyroidism , heavy smokers So…. . The CEA blood test is not reliable for diagnosing cancer or as a screening test for early detection of cancer.

CA 19 -9 can be elevated in many types of GIs cancer, such as : Pancreas, CRC, esophageal ca. and HCC Non-malignancy- conditions : Pancreatitis, diseases of the bile ducts and cirrhosis, and obstruction of the bile ducts (cholectases).

CA 19 -9 can only be used as a tumor marker if the cancer is producing elevated amounts of it. CA 19 -9 is elevated in about : 65% of bile duct ca. (cholangiocarcinoma) 75% of pancreatic ca. ………. so it must be ordered at first : detect the secretion >>>then to monitoring
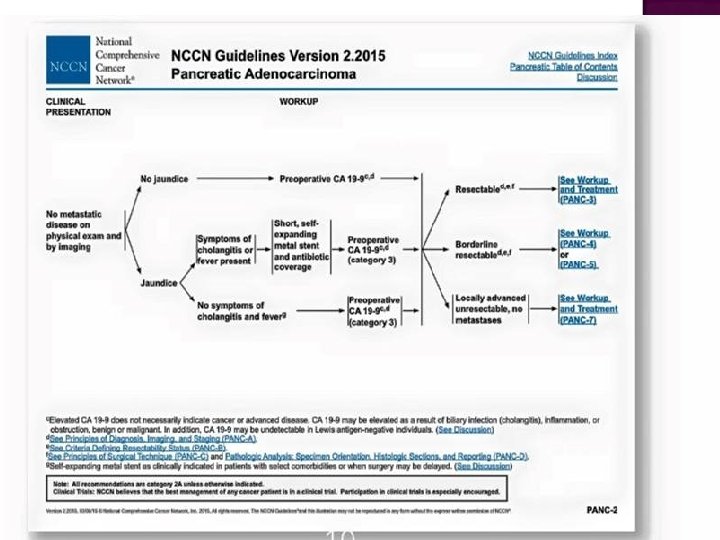



CA 19 -9 is not sensitive or specific enough to use as a screening test for cancer. Its main use is as a tumor marker: **** To monitor the response to CTx and/or progression or recurrence , ***** prognostic and predictive TM in resectable, advanced and metastatic disease

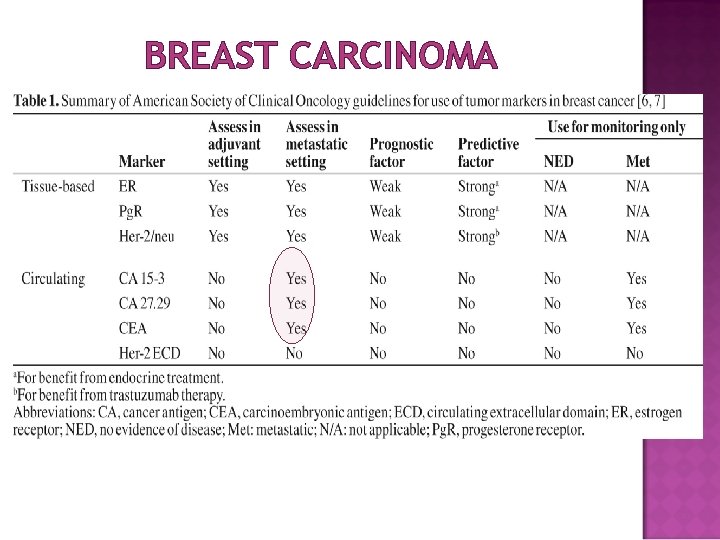
BREAST CARCINOMA

CA -125
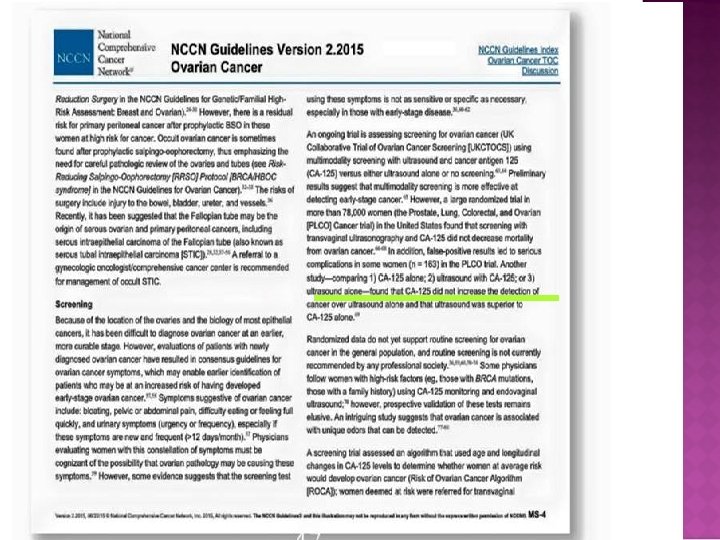


CA -125 NCCN. GL

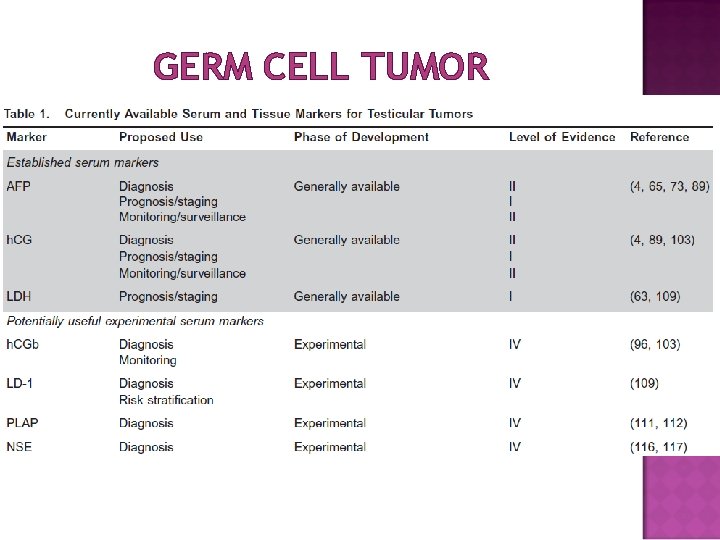
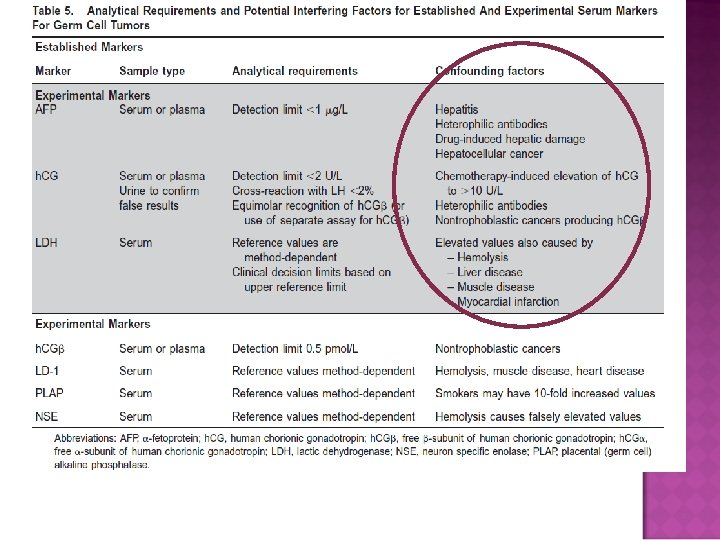
GERM CELL TUMOR
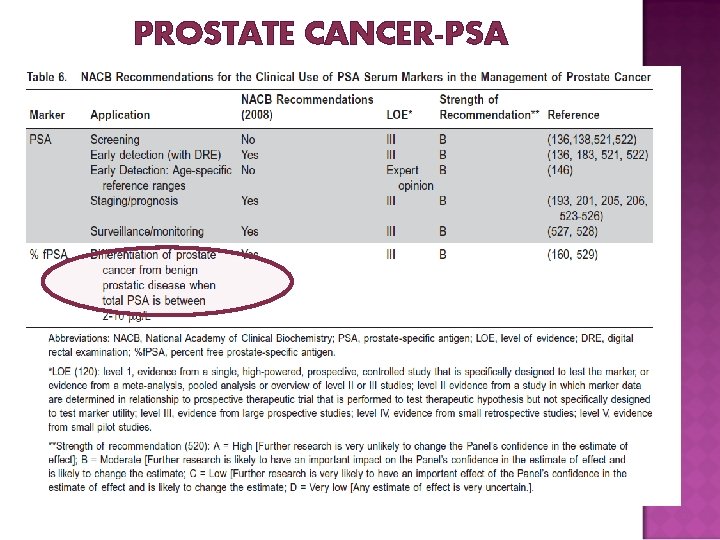
PROSTATE CANCER-PSA


TUMOR MARKERS IN (GEP-NETS) Biochemical markers are applied in (GEPNETs) for diagnostic, prognostic or predictive purposes. And in monitoring in mets-setting Chromogranin A. in every patient with a suspected NET, (Well Differentiated tumours) Neuron-Specific-Enolase. . NSE in poorly differentiated NETs. Pancreatic-Polypeptides: pancreatic-nonfunctioning NETs

TUMOR MARKERS IN DIFF-THYROID CANCER Tg is a prohormone of (T 4) and(T 3). It is synthesized only by thyroid follicular cells and released into serum along with the thyroid hormones. Given the cellular specificity of Tg, its detection in biopsy specimens provides proof of the thyroid origin of the tissue. Measurements of serum Tg provide important information about the presence or absence of residual, recurrent, or metastatic disease in patients with differentiated thyroid cancer Tg is not used for screening, nor prognosis !!!

CALCITONIN & CEA MEDULLARY THYROID CARCINOMA Serum Calcitonin and CEA concentrations should be measured in patients with MTC on the basis of cytologic evaluation of a thyroid nodule The basic value can be compared to postoperative values. Assessment of calcitonin and CEA postoperatively provide sensitive markers for progression and aggressiveness of metastatic MTC


REFERENCES Bishop, M. , Fody, E. , & Schoeff, l. (2013). Clinical Chemistry: Techniques, principles, Correlations. Baltimore: Wolters Kluwer Lippincott Williams & Wilkins Rhea, J. M. , & Molinaro, R. J. (2011, March). Cancer Biomarkers: Surviving the Journey From Bench to Bedside. MLO, 43(3), 10 -18. Sunheimer, R. , & Graves, L. (2010). Clinical Laboratory Chemistry. Upper Saddle River: Pearson. NCCN Guidelines 2015 -2016

- Slides: 36