Ce Po LMC Central Polymer Lab Molecular Characteristics

Ce. Po. L/MC - Central Polymer Lab / Molecular Characteristics @ If. C - Inst. f. Chemie, KFU - Karl-Franzens-Univ. Graz / Austria / Europe Lab Course Renewable Resources: Starch-based nano-particles Anton HUBER, Shazia JUNA European Polysaccharide Network of Excellence wwww. epnoe. org Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at
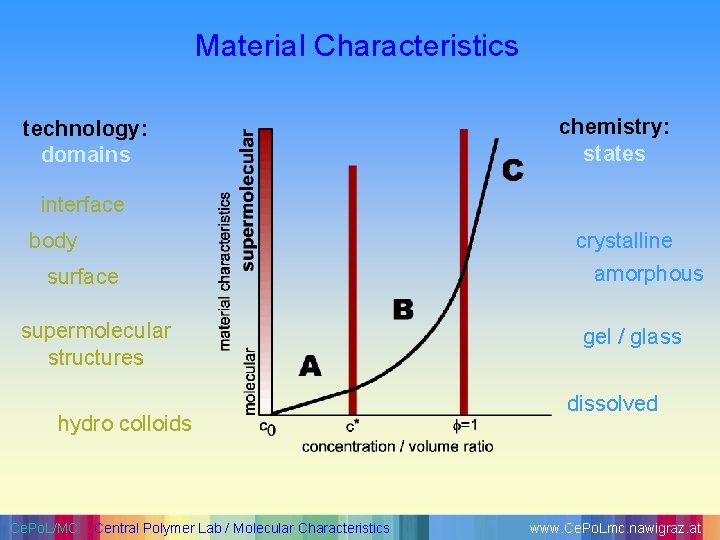
Material Characteristics technology: domains chemistry: states interface crystalline body surface supermolecular structures hydro colloids Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics amorphous gel / glass dissolved www. Ce. Po. Lmc. nawigraz. at
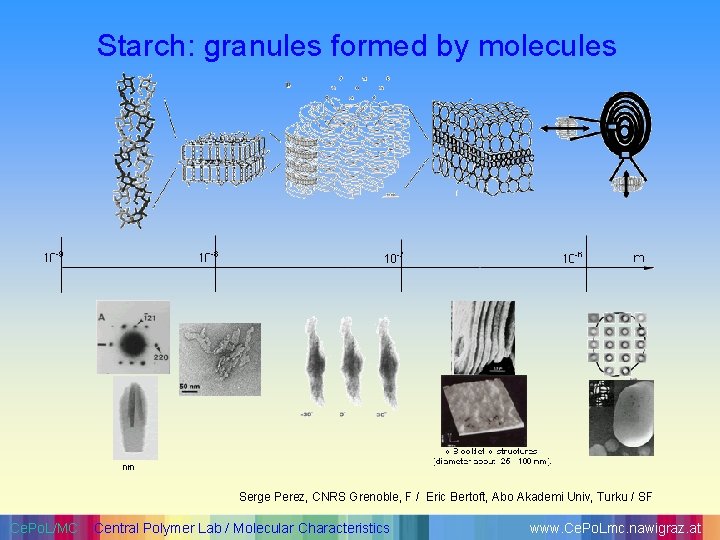
Starch: granules formed by molecules Serge Perez, CNRS Grenoble, F / Eric Bertoft, Abo Akademi Univ, Turku / SF Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at

Carbohydrates Aldoses Ketoses aldo-triose, aldo-tetraoses, aldo-pentoses, aldo-hexoses keto-triose, keto-tetraoses, keto-pentoses, keto-hexoses http: //en. wikipedia. org/wiki/Monosaccharide cyclic form: hemi-acetals cyclic form: hemi-ketals polymerization: formation of (full)acetals / (full)ketals by glycosidic linkages Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at
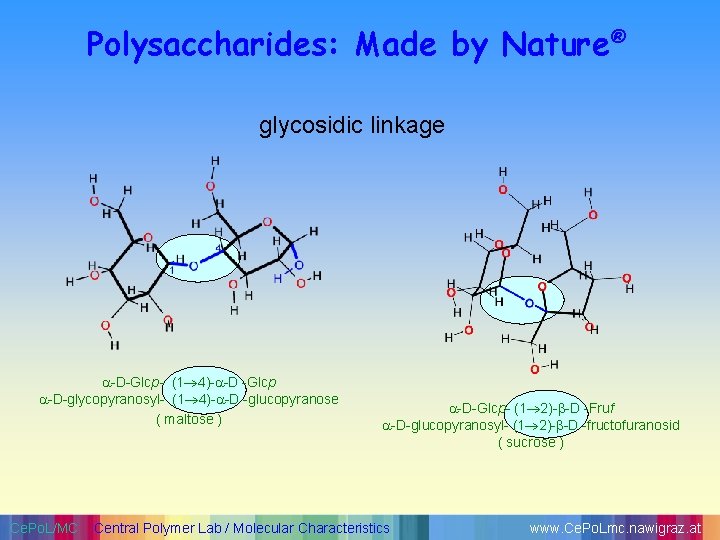
Polysaccharides: Made by Nature® glycosidic linkage -D-Glcp- (1 4)- -D -Glcp -D-glycopyranosyl- (1 4)- -D -glucopyranose ( maltose ) Ce. Po. L/MC -D-Glcp- (1 2)- -D -Fruf -D-glucopyranosyl- (1 2)- -D -fructofuranosid ( sucrose ) Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at
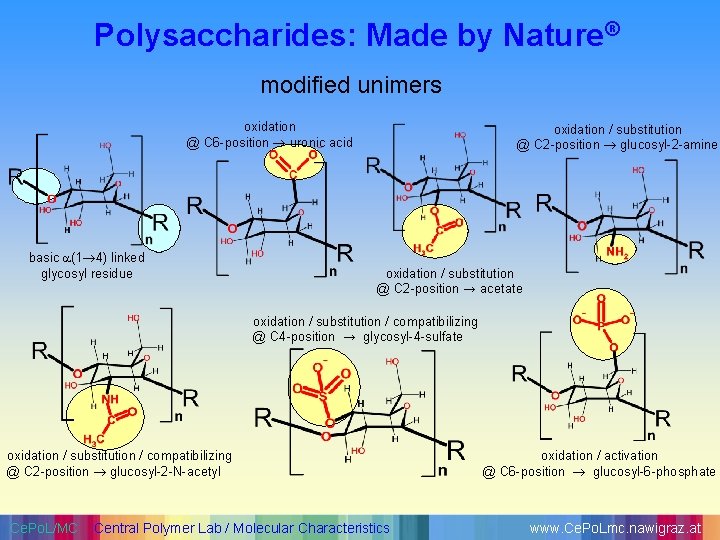
Polysaccharides: Made by Nature® modified unimers oxidation @ C 6 -position uronic acid basic (1 4) linked glycosyl residue oxidation / substitution @ C 2 -position glucosyl-2 -amine oxidation / substitution @ C 2 -position → acetate oxidation / substitution / compatibilizing @ C 4 -position → glycosyl-4 -sulfate oxidation / substitution / compatibilizing @ C 2 -position glucosyl-2 -N-acetyl Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics oxidation / activation @ C 6 -position glucosyl-6 -phosphate www. Ce. Po. Lmc. nawigraz. at
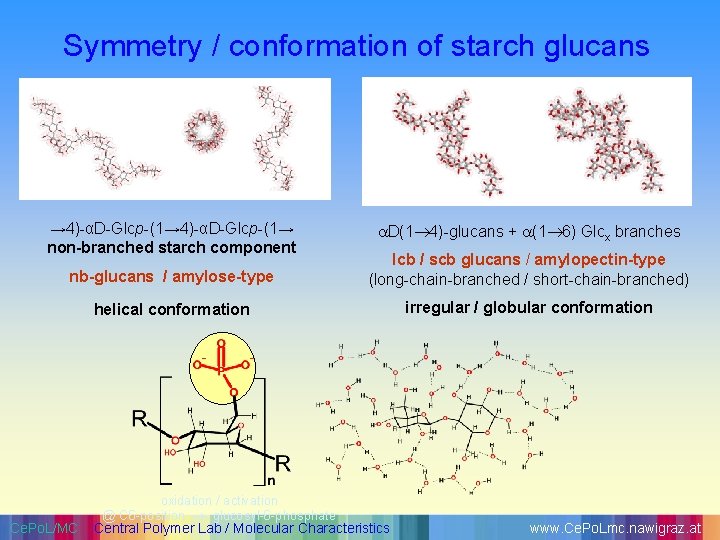
Symmetry / conformation of starch glucans → 4)-αD-Glcp-(1→ non-branched starch component D(1 4)-glucans + (1 6) Glcx branches nb-glucans / amylose-type lcb / scb glucans / amylopectin-type (long-chain-branched / short-chain-branched) helical conformation irregular / globular conformation Ce. Po. L/MC oxidation / activation @ C 6 -position glucosyl-6 -phosphate Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at
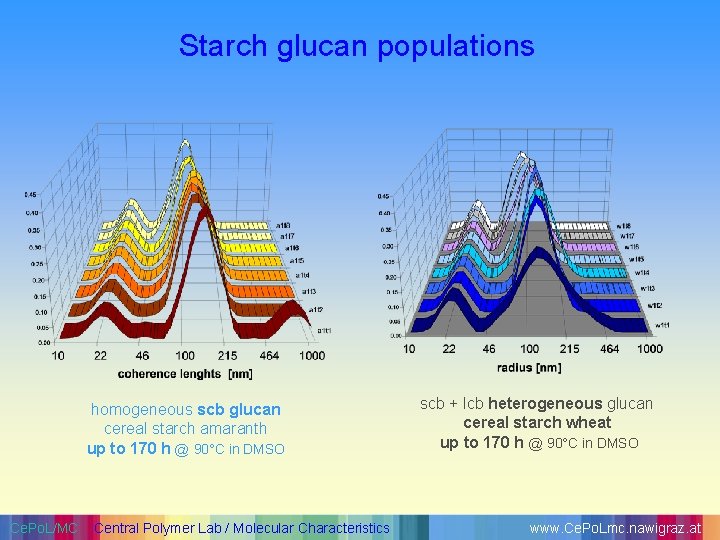
Starch glucan populations homogeneous scb glucan cereal starch amaranth up to 170 h @ 90°C in DMSO Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics scb + lcb heterogeneous glucan cereal starch wheat up to 170 h @ 90°C in DMSO www. Ce. Po. Lmc. nawigraz. at

Managing Heterogeneity by Fractionation LC af. FFF/ AF 4 Entropy controlled separation ( S/k) due to differences in excluded volume (Ve) Diffusion mobility controlled separation due to 1 st approach: differences in excluded volume (Ve) Size Exclusion Chromatography (SEC) asymmetric flow field flow fractionation (af. FFF or AF 4) Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at
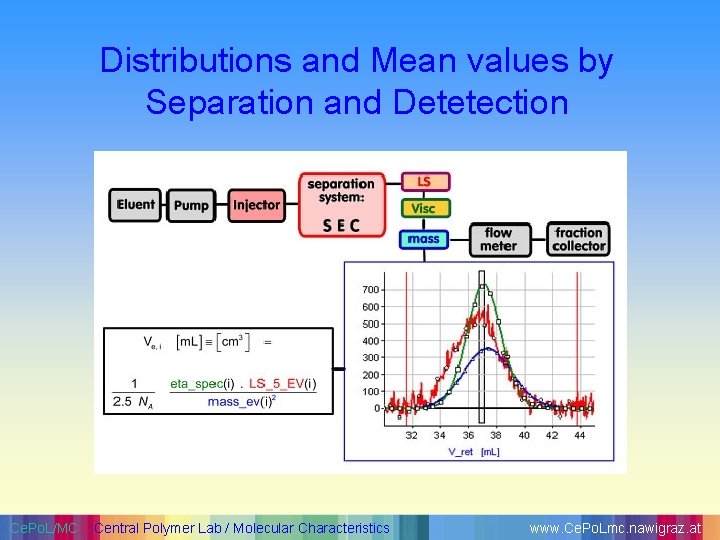
Distributions and Mean values by Separation and Detetection Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at

asym flow Field Flow Fractionation: AF 4 Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at

asymmetric Field Flow Fractionation: AF 4 objects are separated according to differences in their diffusion coefficient / difusive mobility Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at

Diffusive Mobilty DT Hydrodynamic Radius Rh • • Stokes-Einstein: DT from retention times (tr) of AF 4 t 0 (void time), Fcr (cross-flow), w (channel thickness), V 0 (void volume) and tr (retention time) T (temp), k (Boltzmann const), η (intrinsic visc) Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at

Light Scattering Detection: Scattering vs. Spectroscopy Ø probing soft matter by radiation (typically electromagnetic, but also neutrons). Ø The electromagnetic radiation introduces dipole oscillations. Part of the energy will be absorbed when close to resonance → spectroscopy Ø Any acceleration of charges leads to emission of secondary radiation → scattering field. Ø Analysis of the scattered field provides information about local structure (due to interference) and mobility (time dependence of the signal) of the sample Ø At short wavelengths (X-rays) we are far above most resonance frequencies (spectroscopy), all electrons are polarized and emit a secondary wave, i. e. the signal will depend on the electron density Ø At much larger wavelengths (visible light) we are below most resonances, but we only polarize the valence electrons. The polarizablility is a function of the wavelength and is related to the refractive index. Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at

Static and Dynamic Scattering Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at

Scattering facts Zimm plot x-axis: sin 2(Θ/2) + k c q 2 + k c y-axis: K c / RΘ intercept: 1 / Mw slope (q 2 0): Rg slope (c 0): A 2 Ce. Po. L/MC Central Polymer Lab / Molecular Characteristics www. Ce. Po. Lmc. nawigraz. at
- Slides: 16