CDA Clinical Document Architecture Charlie Bishop http www

CDA Clinical Document Architecture Charlie Bishop http: //www. hl 7. org. uk

Agenda • What is CDA ? • CDA overview • Technical insight • CDA and the Mi. M

Overview • Clinical Document Architecture (CDA) • CDA = International Standard – a development of the HL 7 standards developing organization • CDA is – a document markup standard for the structure and semantics of exchanged "clinical documents" – a defined and complete information object • for storage and exchange • can include text, images, sounds and other multimedia content – documentation of observations and other services

Characteristics • A Clinical Document has the following characteristics: – – – Persistence Stewardship Authentication Context Wholeness Human Readability • Conformance with these characteristics is an integral part of the standard

Brief History • Level 1 – Generic document structure (section, paragraphs, list, tables…) • Level 2 – domain-specific document types – document ontology – constrainable for (domain-) compliant content • Level 3 – fine-grained definitions / markup as defined in the RIM
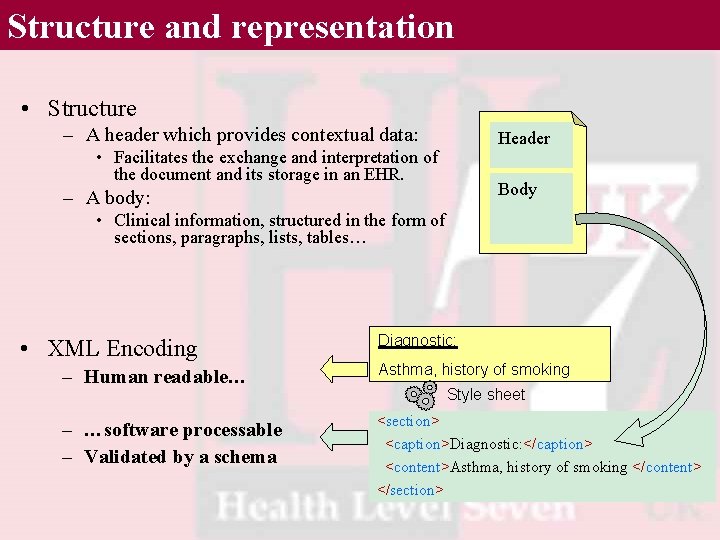
Structure and representation • Structure – A header which provides contextual data: Header • Facilitates the exchange and interpretation of the document and its storage in an EHR. Body – A body: • Clinical information, structured in the form of sections, paragraphs, lists, tables… • XML Encoding Diagnostic: – Human readable… Asthma, history of smoking – …software processable – Validated by a schema <section> <caption>Diagnostic: </caption> <content>Asthma, history of smoking </content> </section> Style sheet
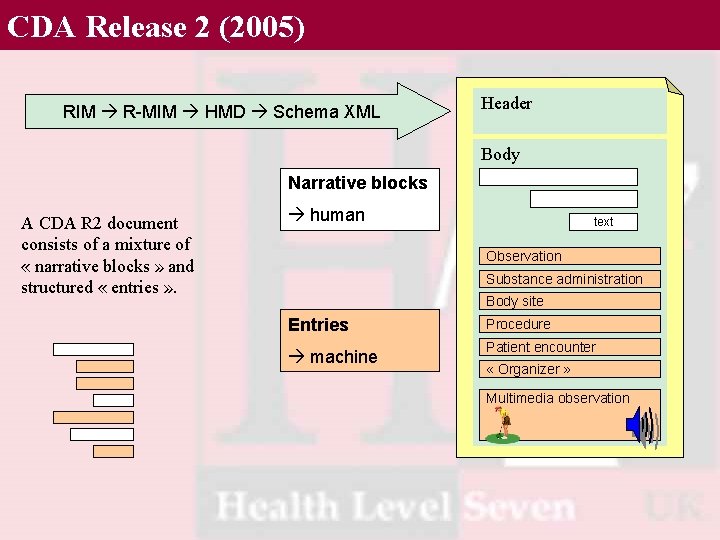
CDA Release 2 (2005) RIM R-MIM HMD Schema XML • HL 7 v 3 Standard Header Body Narrative blocks A CDA R 2 document consists of a mixture of « narrative blocks » and structured « entries » . human text Observation Substance administration Body site Entries machine Procedure Patient encounter « Organizer » Multimedia observation
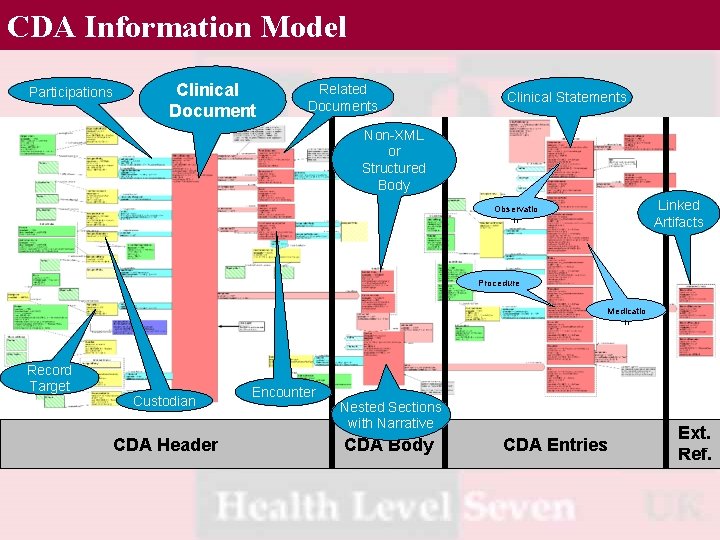
CDA Information Model Participations Clinical Document Related Documents Clinical Statements Non-XML or Structured Body Linked Artifacts Observatio n Procedure Medicatio n Record Target Custodian CDA Header Encounter Nested Sections with Narrative CDA Body CDA Entries Ext. Ref.
Body Structure
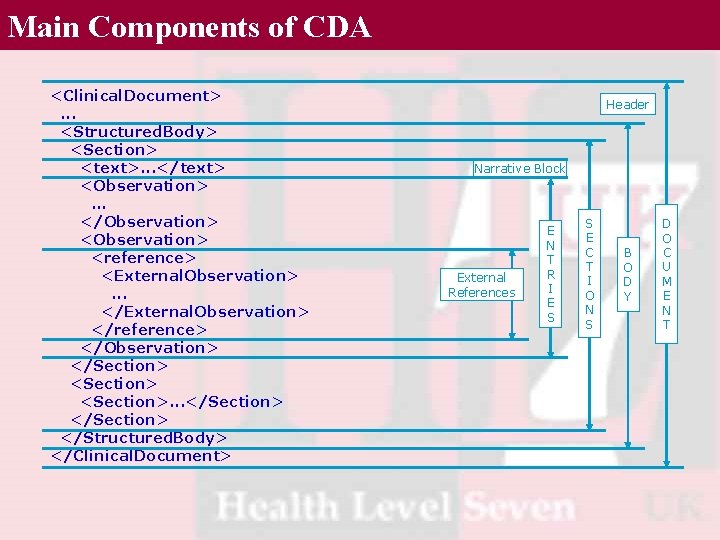
Main Components of CDA <Clinical. Document>. . . <Structured. Body> <Section> <text>. . . </text> <Observation>. . . </Observation> <reference> <External. Observation>. . . </External. Observation> </reference> </Observation> </Section> <Section>. . . </Section> </Structured. Body> </Clinical. Document> Header Narrative Block External References E N T R I E S S E C T I O N S B O D Y D O C U M E N T
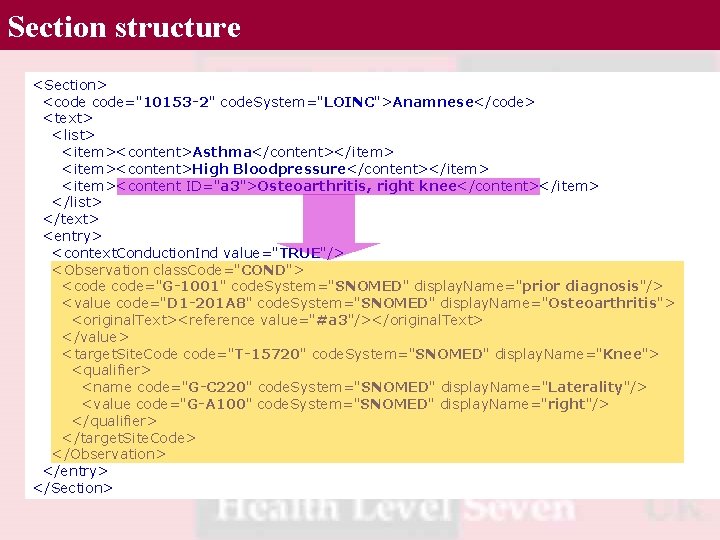
Section structure <Section> <code="10153 -2" code. System="LOINC">Anamnese</code> <text> <list> <item><content>Asthma</content></item> <item><content>High Bloodpressure</content></item> <item><content ID="a 3">Osteoarthritis, right knee</content></item> </list> </text> <entry> <context. Conduction. Ind value="TRUE"/> <Observation class. Code="COND"> <code="G-1001" code. System="SNOMED" display. Name="prior diagnosis"/> <value code="D 1 -201 A 8" code. System="SNOMED" display. Name="Osteoarthritis"> <original. Text><reference value="#a 3"/></original. Text> </value> <target. Site. Code code="T-15720" code. System="SNOMED" display. Name="Knee"> <qualifier> <name code="G-C 220" code. System="SNOMED" display. Name="Laterality"/> <value code="G-A 100" code. System="SNOMED" display. Name="right"/> </qualifier> </target. Site. Code> </Observation> </entry> </Section>

Clinical Statement • HL 7 v 3 model for structured clinical information • Allows consistent representation across domains • Used by CDA

Questions ?
- Slides: 13