CC1 BenefitRisk Assessment Murat Emre MD Professor of

CC-1 Benefit-Risk Assessment Murat Emre, MD Professor of Neurology Istanbul Faculty of Medicine Department of Neurology Behavioral Neurology and Movement Disorders Unit Istanbul University

CC-2 1 -17 DV Parkinson’s Disease Dementia u Readily diagnosable clinical condition – Develops in the context of established PD • Cognitive decline with a typical profile • Frequent neuropsychiatric symptoms • Functional disability – Exclusion of symptomatic dementias u No symptomatic treatments available for PDD – Increased burden for patients and families – Reason for nursing home placement – Considerable unmet need

CC-3 1 -17 DV Statement of Need in PDD u Treatment to benefit all symptom domains – Cognition, behavior, function u Tolerability without adverse impact on – Motor symptoms – Autonomic and cardiovascular functions

CC-4 1 -17 DV Benefits of Exelon in PDD u Statistically significant benefits were seen in – Primary efficacy measures for cognition and overall status (ADAS-cog and ADCS-CGIC) – All secondary efficacy outcome measures • Attention • Executive function • Behavioral symptoms • Activities of daily living

CC-5 1 -17 DV Benefits of Exelon in PDD u Moderate but consistent benefits in – Cognition • Overall cognition: ADAS-cog • Executive function: Verbal fluency, clock-drawing test • Attention: Composite measure of attention – Behavioral symptoms: Total NPI score – Function: ADCS-ADL – Clinical global outcome: ADCS-CGIC
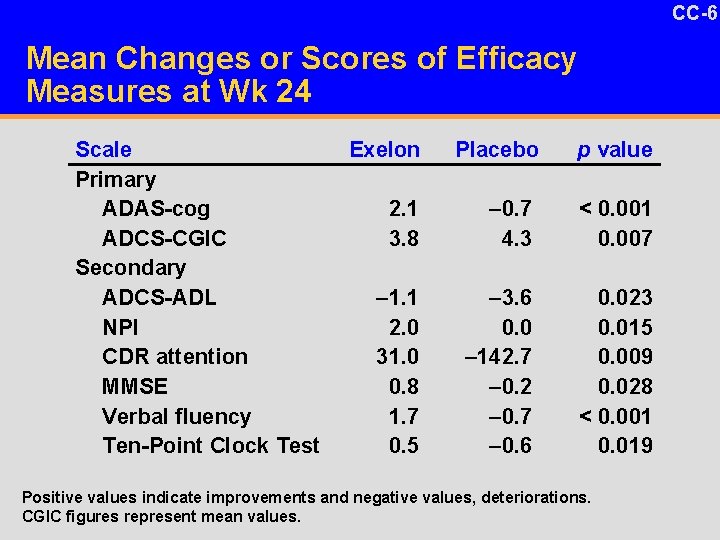
CC-6 29 Originals/Slides/04 -21 -06 Emre Slide Requests/EXPRESSsummary. Siide. ppt Mean Changes or Scores of Efficacy Measures at Wk 24 Scale Primary ADAS-cog ADCS-CGIC Secondary ADCS-ADL NPI CDR attention MMSE Verbal fluency Ten-Point Clock Test Exelon Placebo p value 2. 1 3. 8 – 0. 7 4. 3 < 0. 001 0. 007 – 1. 1 2. 0 31. 0 0. 8 1. 7 0. 5 – 3. 6 0. 0 – 142. 7 – 0. 2 – 0. 7 – 0. 6 0. 023 0. 015 0. 009 0. 028 < 0. 001 0. 019 Positive values indicate improvements and negative values, deteriorations. CGIC figures represent mean values.

CC-7 Risks Associated With Exelon in PDD u Gastrointestinal AEs – Occurred mostly during Exelon dose titration – Most of mild or moderate severity and not leading to discontinuation (nausea: 29% incidence, 4% discontinuation) – Lower incidence and discontinuation rates than in Exelon AD studies

CC-8 Risks Associated With Exelon in PDD u AEs ‘potentially associated with PD’ – 11% more in the Exelon group – Single episodes of mild or moderate severity; decreased incidence after completion of the dose-titration periods – Tremor was the most frequent individual PD symptom (10% vs 4%), discontinuation 1. 7% – No difference from placebo in the total UPDRS scores – Exposure over 48 wk not associated with a worsening compared with those exposed over 24 wk

CC-9 Risks Associated With Exelon in PDD u Cardiovascular safety – No cardiovascular or autonomic safety issues identified with the use of Exelon in PDD – Less frequent orthostatic hypotension and syncope with Exelon – Fewer deaths and SAEs among Exelon-treated patients u Exelon in PDD is not associated with a risk beyond that described in the product label for patients with AD

CC-10 Clinical Relevance of Benefits u Based on previous dementia trials with Ch. EIs, the treatment effect on ADAS-cog ranges from 2 to 4 points – 2. 9 points for Exelon in the core study – 2. 1 points for Exelon in AD study u In PDD, larger improvement above baseline with Exelon and less decline with placebo as compared with AD studies Corey-Bloom J, et al. Int J Geriatr Psychopharm. 1998; 1: 55 -65. Rogers SL, et al. Arch Int Med. 1998; 158: 1021 -1031. Rosler M, et al. BMJ. 1999; 318: 633 -640. Burns A, et al. Dem Geriatr Cog Disord. 1999; 10: 237 -244. Geldmacher D. Clin Geriatr Med. 2004; 20: 27 -43.
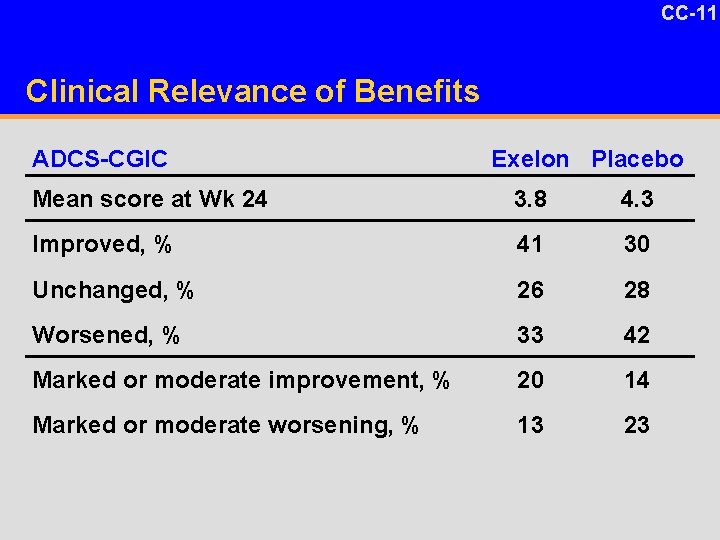
CC-11 Clinical Relevance of Benefits ADCS-CGIC Exelon Placebo Mean score at Wk 24 3. 8 4. 3 Improved, % 41 30 Unchanged, % 26 28 Worsened, % 33 42 Marked or moderate improvement, % 20 14 Marked or moderate worsening, % 13 23

CC-12 1 Summary of Benefit-Risk Assessment u Exelon provided moderate but consistent benefits across all primary and secondary measures in all symptom domains u AEs consistent with the established safety profile for Exelon; risk of worsening tremor in 10% of patients u No additional safety concerns beyond those described in current label

CC-13 Conclusion u There is currently no approved treatment for PDD u Exelon treatment associated with benefits in cognition, behavior, and activities of daily living u Tolerability risks, such as nausea and tremor, are easy to monitor, clinically recognizable, and manageable u The benefits of Exelon treatment in PD patients with dementia outweigh the risks
- Slides: 13