CBP Nephrology Diseases of the kidneys CBP Nephrology

CBP: Nephrology – Diseases of the kidneys!

CBP: Nephrology • A 48 -year-old man, otherwise healthy, presents with severe unspecific abdominal pain and vomiting of 2 days duration. • He is a stable bipolar personality disorder on lithium. • an X smoker, has history of alcohol use,

CBP: Nephrology • On physical examination, restless, dehydrated HR 130 and BP 90/60 (Supine), RR 28, T 37. 4. Chest and heart exams are unremarkable. Abd. exam revealed mild-moderate epigastric abdominal tenderness without peritoneal signs. Rest of the exam is unremarkable. • Lab: WBC is 16, 500, and the HCT is 49. Cr 188 (67 base line), BUN 12. 3 , K 5. 5 and the rest of electrolyte values are normal.

CBP: Nephrology • Intubated on admission due to altered LOC and inability to protect his airway as well as impending hypoxemic respiratory failure, remains on multiple vasoactive agents, and is in oliguric-to-anuric renal failure • Admitted to ICU and adequately resuscitated • Patient remains anuric despite the adequate fluid resuscitation

Question 1 • Any role for increasing doses of Lasix in an anuric patient? Any harm? Any benefit? (Eric)

Diuretics in AKI

Diuretics in AKI • Three part question: 1. Electrolyte management 2. Fluid management 3. Conversion of oliguric to non-oliguric RF

Electrolytes & Fluid* • Paucity of data answering these specific questions • Remains clinical decision and therapeutic option * Differentiate fluid management from urine output

Conversion of oliguric to non-oliguric RF Ravindra LM et al. , Diuretics, Mortality, and Nonrecovery of Renal Function in Acute Renal Failure, JAMA. 2002; 288(20): 2547 -2553

Confounders • Diuretic use at the time of consultation was significantly associated with older age, presumed nephrotoxic (rather than ischemic or multifactorial) ARF origin, a lower BUN level, acute respiratory failure, and a history of congestive heart failure.

Cause vs Correlate • After adjusting for covariates associated with the risk of death, diuretic use was significantly associated with in-hospital mortality an nonrecovery of renal function, even after adjustment for nonrandom treatment assignment using propensity scores.

Diuretics in AKI • Three part question: 1. Electrolyte management ✓ / ✗ 2. Fluid management ✓ / ✗ 3. Conversion of oliguric to non-oliguric RF ✗

Question 2 • Define Acute Kidney Injury (Eric)

Definitions of AKI

The dilemma “More than 35 definitions of AKI currently exist in the literature”
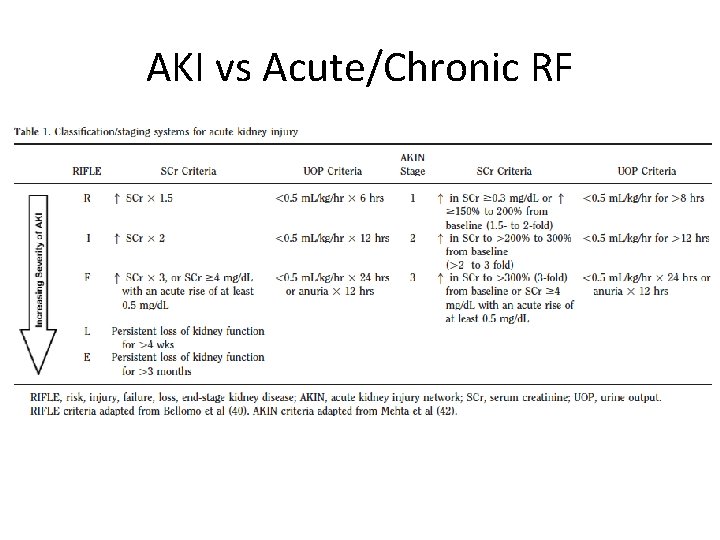
AKI vs Acute/Chronic RF Crit Care Med 2010; 38: 261– 275

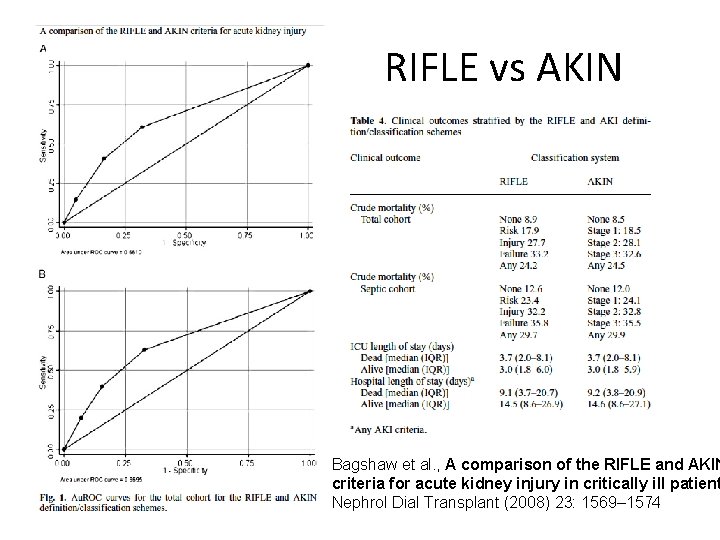
RIFLE vs AKIN Bagshaw et al. , A comparison of the RIFLE and AKIN criteria for acute kidney injury in critically ill patient Nephrol Dial Transplant (2008) 23: 1569– 1574

Bottom Line • Both the RIFLE and AKIN criteria were developed to facilitate clinical investigation and comparison across study populations. • To date, most interventional studies (e. g. NAC, Na. HCO 3, etc. ) to prevent or mitigate AKI have not used these definitions.

Question 3 1. What is the incidence of AKI in the ICU and how does it affect patient outcomes? ( yahya)

Incidence of AKI in the ICU • AKI occurs in ~ 7% of all hospitalized patients, whereas it occurs in 36% – 67% of critically ill patients. • On average, 5 % of ICU patients with AKI require renal replacement therapy. Dennen P, Douglas IS, Anderson R. Acute kidney injury in the intensive care unit: an update and primer for the intensivist. Crit Care Med. 2010 Jan; 38(1): 261 -75

AKI and mortality • In most studies, mortality rates rise proportionally with severity of AKI. • Even small increases in serum creatinine have been associated with increasing mortality in various ICU populations despite adjusting for severity of illness and comorbidities. • In patients with AKI requiring RRT, mortality rates reach 50% to 70%. Dennen P, Douglas IS, Anderson R. Acute kidney injury in the intensive care unit: an update and primer for the intensivist. Crit Care Med. 2010 Jan; 38(1): 261 -75

AKI and other outcomes • AKI is also associated with: – Increased length of stay – Increased incidence of CKD and end-stage kidney disease – Increased cost • For example, an increase in SCr of 0. 5 mg/dl (38 mmol/L)was associated with a: – 6. 5 -fold increase in the odds of death – 3. 5 day increase in LOS – nearly $7500 in excess hospital costs Dennen P, Douglas IS, Anderson R. Acute kidney injury in the intensive care unit: an update and primer for the intensivist. Crit Care Med. 2010 Jan; 38(1): 261 -75 Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005 Nov; 16(11): 3365 -70

Question 4 • What are the methods for detecting acute kidney injury? ( yahya)

Traditional methods for detecting AKI • Currently available measures do not detect actual kidney injury the way troponin detects myocardial injury: – Creatinine – Urea – Urine output • Rather they are markers of abnormal renal function, that can be used to presume kidney inury has occurred. Bagshaw SM, Bellomo R. Early diagnosis of acute kidney injury. Curr Opin Crit Care. 2007 Dec; 13(6): 638 -44.

Serum creatinine • Used to estimate GFR • Pros – Produced at a relatively constant rate – Freely filtered by glomerulus – Not reabsorbed or metabolized by the kidney. Bagshaw SM, Bellomo R. Early diagnosis of acute kidney injury. Curr Opin Crit Care. 2007 Dec; 13(6): 638 -44.

Serum creatinine • Used to estimate GFR • Cons – 10 -40% is secreted by the tubules – Relatively insensitive (may need a 50% reduction in function before a detectable rise in SCr is seen) – Creatinine production varies based on age/sex/muscle mass/diet – Certain disease states can increase production (rhabdo) – Certain drugs can decrease secretion (cimetidine, trimethoprim) – Certain factorsmay affect assay (ketoacidosis, cefoxitin, flucytosine) – Does not reflect real-time changes in GFR Bagshaw SM, Bellomo R. Early diagnosis of acute kidney injury. Curr Opin Crit Care. 2007 Dec; 13(6): 638 -44.

Urea • Rate of production is not constant – Increases with protein intake – Increases in critical illness (burns/sepsis/trauma) – GI Bleed – Steroids • 40% - 50 % of urea is reabsorbed by the kidney (even more when dry) Bagshaw SM, Bellomo R. Early diagnosis of acute kidney injury. Curr Opin Crit Care. 2007 Dec; 13(6): 638 -44.
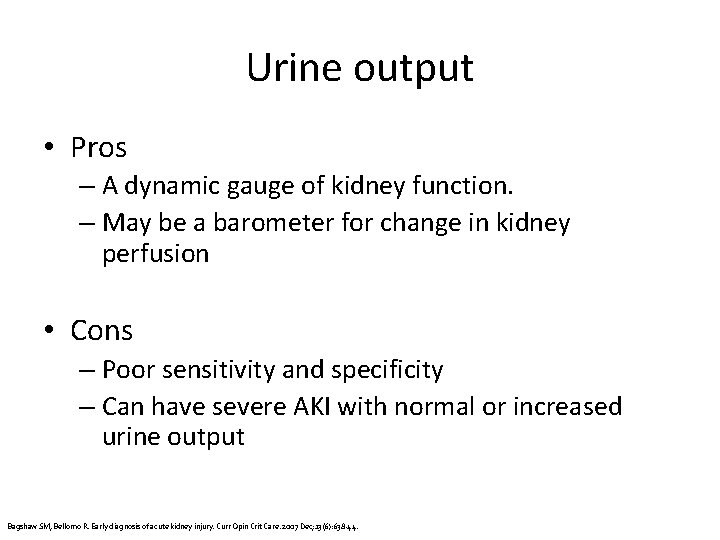
Urine output • Pros – A dynamic gauge of kidney function. – May be a barometer for change in kidney perfusion • Cons – Poor sensitivity and specificity – Can have severe AKI with normal or increased urine output Bagshaw SM, Bellomo R. Early diagnosis of acute kidney injury. Curr Opin Crit Care. 2007 Dec; 13(6): 638 -44.
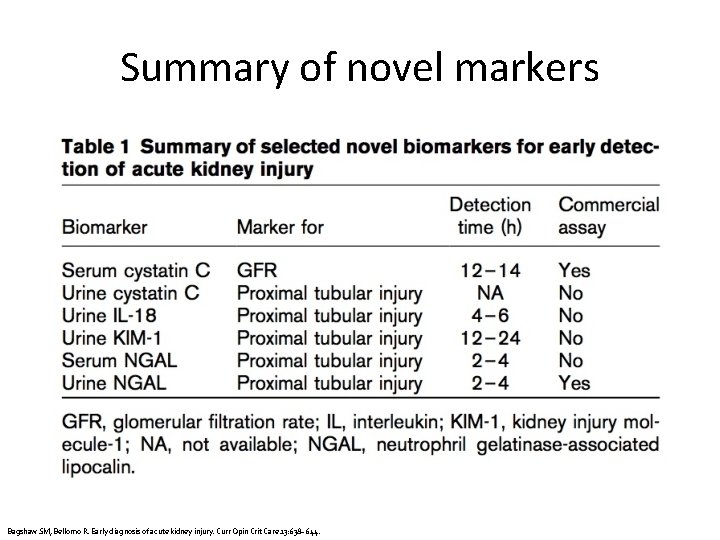
Summary of novel markers Bagshaw SM, Bellomo R. Early diagnosis of acute kidney injury. Curr Opin Crit Care 13: 638– 644.

CBP: Nephrology • Patient continues to have increasing ventilation support requirments and is now on. 85 Fi. O 2. His K+ is now 5. 6. He is given routine hyper. K+ therapy. He has been started on vasopressors because of declining MAP

Question 6 • When should RRT be started? (Indication and timing) (Brian)

Historical aspects • Use of HD in ARF started in the years immediately following WWII (1947 -1950) • Initial indications: advanced symptoms of renal failure – clinical uremia, severe hyperkalemia, pulmonary edema • Reduction in mortality could not be demonstrated, with high complication rates • Teschan et al reported improved survival with “prophylactic dialysis” in 1960

“Indisputable” indications 1. 2. 3. 4. Volume overload Hyperkalemia Metabolic acidosis Uremic signs or symptoms • Refractory to medical management • No specific objective criteria

Other Indications 1. Progressive azotemia in the absence of uremia (no consensus) 2. Other electrolyte disturbances (Na, Mg, PO 4, Uric acid)

Timing of initiation of RRT • Competing risks – Risk of delay in therapy – Potential harm of therapy, including complications of therapy and the potential that dialysis may prolong the course of ARF

Teschan et al. Prophylactic hemodialysis in the treatment of acute renal failure. Ann Int Med 1960. • Paul Teschan of US Army Medical Corps after the Korean War introduced the concept of “prophylactic dialysis”, applied before overt uremic symptoms appeared • N=15, uncontrolled trial, initiation of dialysis before serum Urea Nitrogen reached 100 mg/dl • Twin coil cellulosic dialyzers at BF 75 -250 ml/min to maintain BUN less than 75 mg/dl • All cause mortality 33%, mortality due to hemorrhage or sepsis 20% • No control group. However, investigators reported that the result represented “dramatic” increase in survival cf their past experience in pt in whom dialysis was not initiated until “conventional” indications were present
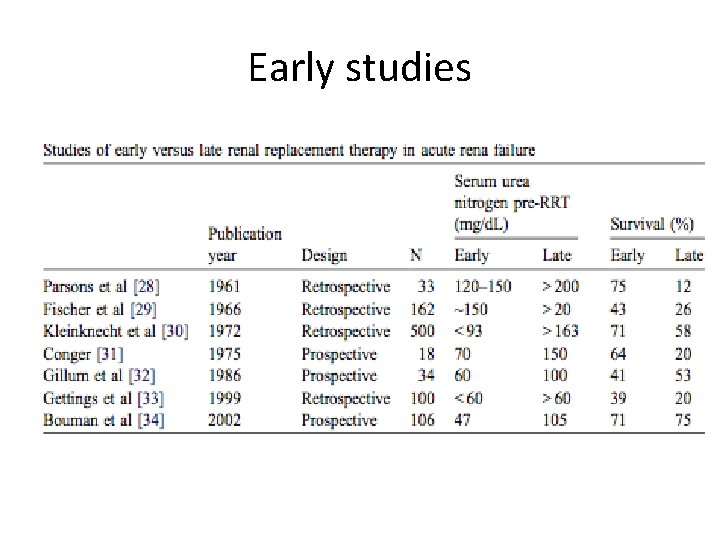
Early studies

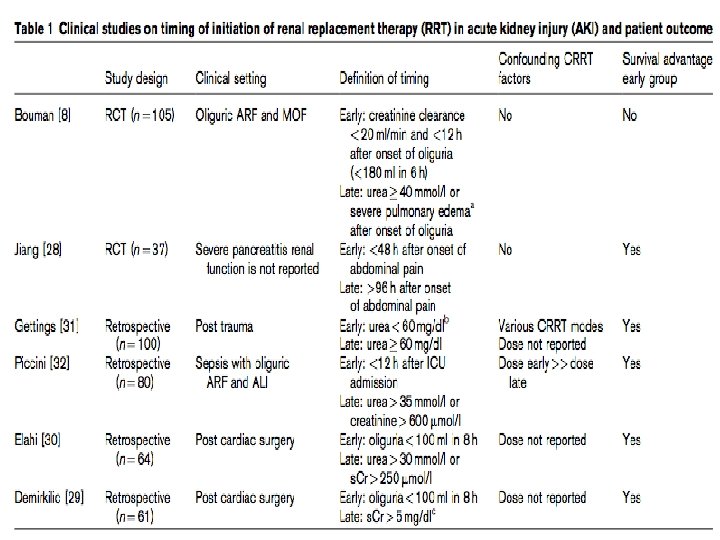

Bouman et al (CCM 2002) • 2 center RCT (n=106). ICU pts on MV with vasopressor dependent circulation and oliguric ARF • ARF: Cr. Cl<20, UO<180 ml/6 h • Early: CVVH within 12 h after onset of liguria • Late: urea>40 mmol/l, pulmonary edema with Pa. O 2/Fi. O 2<150 despite PEEP 10 • Many issues

Jiang, et al (2005) • RCT (n=37) in severe pancreatitis WITHOUT documented evidence of ARF • Early: CVVH within 48 hours onset of abdo pain • Late: within 96 hours • Improved hemodynamics and 14 d survival

Gettings et al (ICM 1999) • Retrospective nonrandomized cohort study (n=100) • Trauma patients • “Timing” defined by BUN level • Early: RRT started at a mean BUN 15 mmol/l • Late: at BUN 34 mmol/L • Survival: 39% Early, 20% Late

Piccini et al. (ICM 2006) • • • Retrospective study (n=80) Patients with septic shock and oliguric AKI Historical control Early: <12 h after ICU admission Late: Urea>35 mmol/l or Cr>600 Improved hemodynamics, gas exchange, 28 d survival

Elahi et al. (2004) Retrospective cohort study (n=80) Cardiac surgery patients Early: CVVH when UO<100 ml/8 h despite lasix Late: Urea>30 mmol/l, Cr>250, or K>6 regardless of UO • Survival 44% early, 22% late, p<0. 05 • •

Demirkilic et al. (2004) • • • Retrospective study (n=61) ARF following cardiac surgery Historical control Early: CVVHDF if UO<100 ml/8 h Late: Cr>444 Hospital mortality 23. 5% Early 55% late p=0. 02

Summary • Trend towards better outcome with earlier timing of RRT • Methodology poor • Nonuniform definition of timing • Heterogeneity of population • Heterogeneity of RRT

Summary • Nonuniform and arbitrary definition of ARF prevents direct comparison of trials • But how about using RIFLE criteria and AKIN definition?


Shiao et al. (2009) • Multicenter prospective observational study • N=98 who underwent RRT according to local indications for post-major abdo surgery AKI • Early: s. RIFLE – 0 or Risk • Late: s. RIFLE – I or F

RIFLE/AKIN
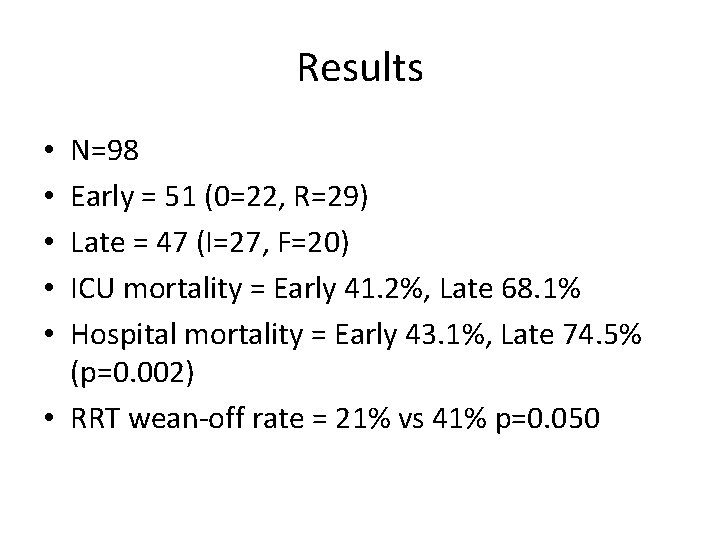
Results N=98 Early = 51 (0=22, R=29) Late = 47 (I=27, F=20) ICU mortality = Early 41. 2%, Late 68. 1% Hospital mortality = Early 43. 1%, Late 74. 5% (p=0. 002) • RRT wean-off rate = 21% vs 41% p=0. 050 • • •
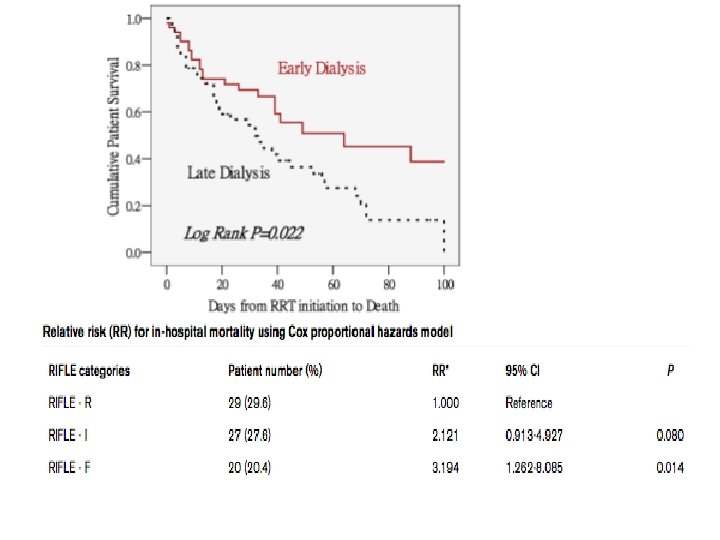

Conclusions/Limitations • Late dialysis defined by s. RIFLE-I or s. RIFLE-F is an independent predictor for inhospital mortality • Support earlier initiation of RRT • Small N • Only GFR criterion of RIFLE used (s. RIFLE)

Question 7 • Define different modes of RRT (Brian)
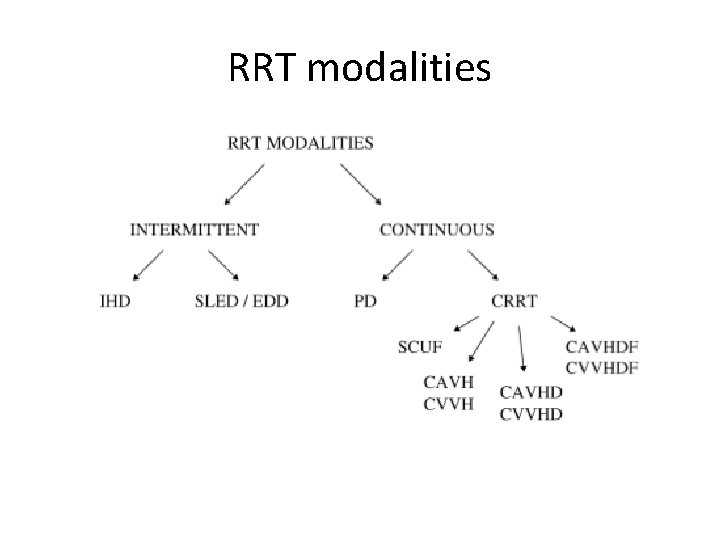
RRT modalities
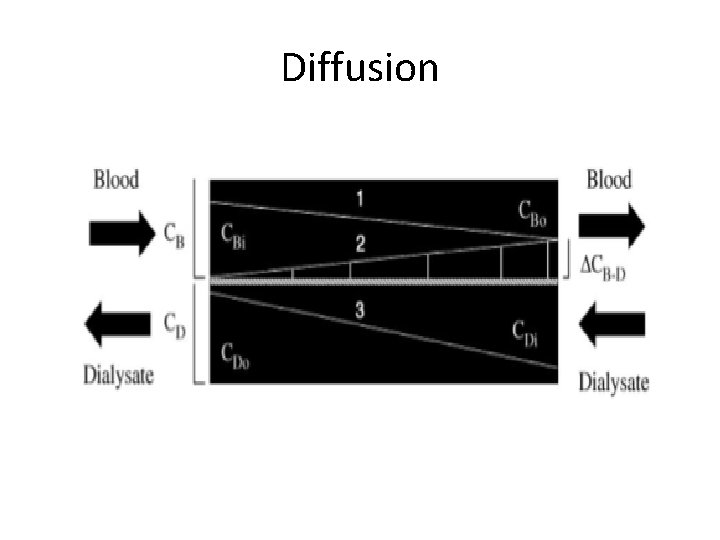
Diffusion
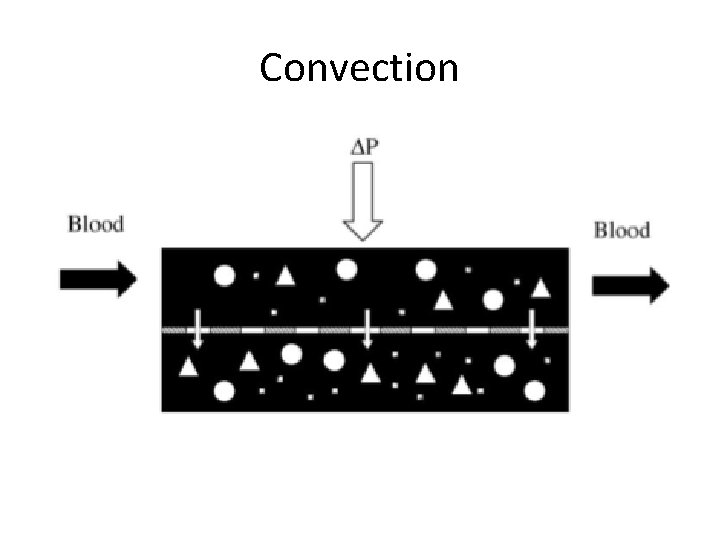
Convection
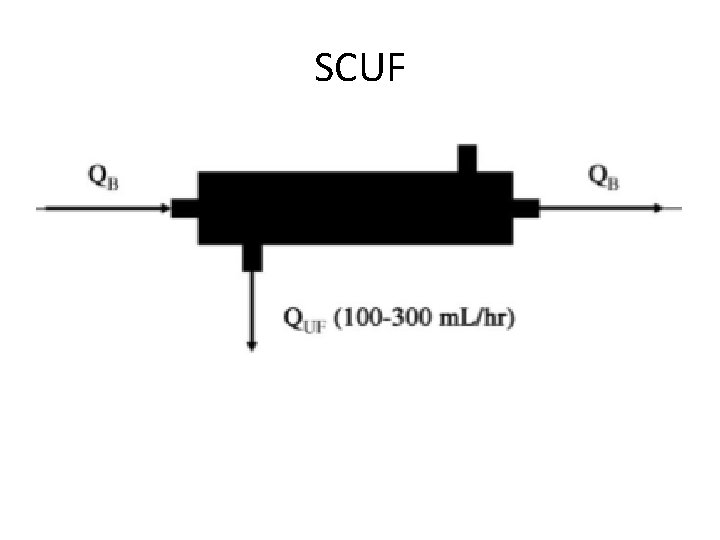
SCUF
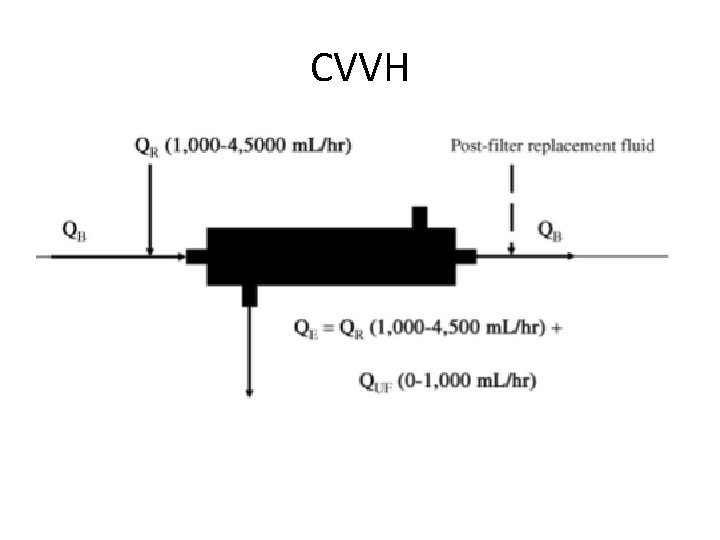
CVVH
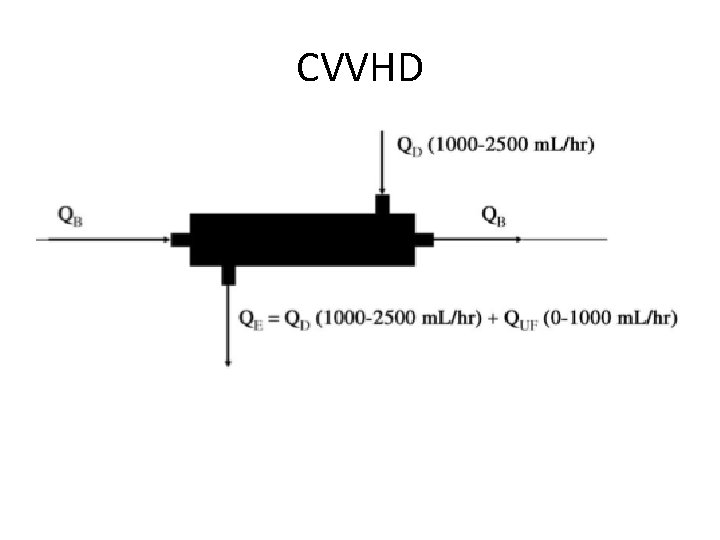
CVVHD
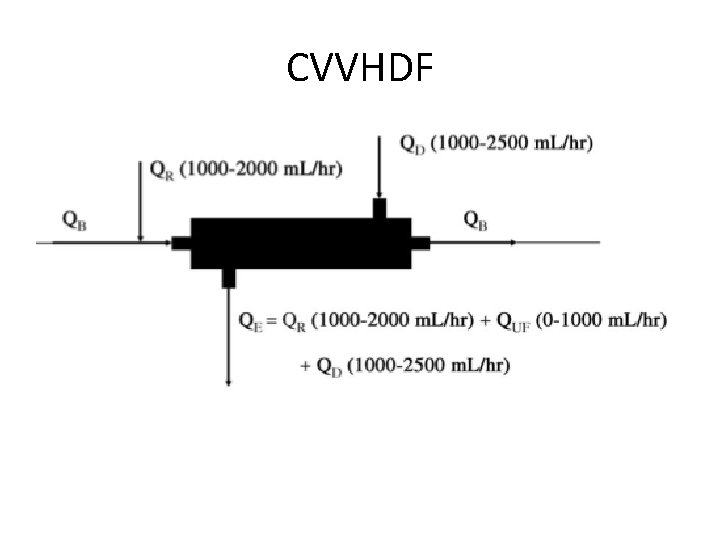
CVVHDF

IHD • Blood flow 200 -300 ml/min • Dialysate flow 500 -800 ml/min • Solute removal by diffusion, fluid removal by ultrafiltration • Solute clearance dependent on blood flow • Advantages: rapid solute and fluid removal (rapid electrolyte correction, certain toxin removal), no need for anticoagulation • Disadvantages: systemic hypotension,

Renal Replacement Therapy • All forms of RRT rely on the principle of allowing water and solute transport through a semipermeable membrane and discarding waste products • Fluid removal – ultrafiltration • Solute transport – diffusion, convection, or both

Question 8 • IHD vs CRRT (Brian)

Dialysis modality • 1999 NKF survey revealed IHD as preferred form of RRT (75%), while CRRT + PD was less than 10% • More recent survey revealed IHD as preferred by nephrologists/intensivists in 57%, while CRRT was preferred in 37% in US • Internationally: BEST Kidney study (JAMA 2005) revealed CRRT as the initial modality of choice for RRT in ICU used in 80%, followed by IHD (17%)

So: is CRRT better?

Preference of CRRT – Putative advantages • Improved hemodynamic stability • More effective control of acid/base and electrolyte status • Improved removal of uremic toxins • Removal of inflammatory mediators

Disadvantages • Need for anticoagulation • 2 -3 x more expensive than IHD

Evidence • Two 2002 metaanalyses of earlier trials comparing survival in ICU AKI assigned to IHD or CRRT and adjusted for severity of illness did not support CRRT • Several observational and prospective RCTs comparing IHD vs CRRT failed to confirm expected survival advantage of CRRT • Limitations: dose difference, high crossover rate, randomization failure, nonstandardization of protocol

Cochrane Review 2008 Intermittent vs. continuous RRT for ARF in adults

Objectives • To compare CRRT with IRRT to establish if any of these techniques is superior to each other in patients with AF

Methods • Types of studies: RCTs • Interventions – IRRT defined as any form of RRT (HD, HF, HDF, UF) prescribed for period of <24 h within any 24 h period – CRRT defined as any RRT intended to run on a continuous basis until recovery of renal function occurred

Methods • Outcome measures – Mortality (prior to ICU/hospital DC, time to ICU/hospital death/DC – Recovery of renal function – Cardiovascular stability – Complications of therapy (bleeding, sepsis)

Results – Mortality • In-hospital mortality – no difference (7 studies, N=1245): RR 1. 01 (0. 92 -1. 12), no evidence of significant heterogeneity • ICU mortality – no difference (5 studies, N=515): RR 1. 03 (0. 90 -1. 26) • Time to hospital death or discharge – no difference (1 study, N=25) • Time to ICU discharge or death – not assessed

Results – Recovery of RF • Surviving pt not requiring dialysis – No difference (3, N=161) RR 0. 99 (0. 92 -1. 07), no evidence of sig. heterogeneity • s. Cr or e. GFR at hospital discharge – no difference (1, N=129) RR 1. 13 (0. 94 -1. 36)

Results – Cardiovascular stability • Hemodynamic instability – no difference (2, N=205) RR 0. 48 (0. 2 -2. 28). One study did not specify definition, while the other defined it as avaerage variability b/w max ad min daily MAP. No heterogeneity • Hypotension – No difference (3, N=514) RR 0. 92 (0. 721. 16). Variable definition of hypotension • MAP at end of study – CRRT significantly higher (2, N=112) mean dif 5. 35 (1. 41 -9. 29) • Systolic BP – No difference (1, N=30) • Escalation of pressor rx – No difference when analysed by random effects model • Dose of inotropic drugs – no difference

Results – Complications of RRT • Bleeding – no difference (5, N=638) • Clotting of dialysis filter – CRRT significantly more likely to clot filter (3, N=149) RR 8. 5 (1. 14 -63. 33) • Arrhythmia – no difference (2, N=439) • RRT modality switch due to complications – no difference (4, N=920)

Conclusions • CRRT offers no survival advantage cf IRRT in ARF • Pt surviving ARF who are managed with CRRT has similar recovery of RF as those treated with IRRT • CRRT is associated with sig higher MAP • CRRT is associated with sig increased filter clotting

Limitations • Each RCTs are not large enough to provide an accurate evaluation of the difference in outcome • Considerable variations in definition of ARF and hypotension, heterogeneity in dialysis x (dose, membrane) and pt characteristics

What kind of anticoagulation should be used with CRRT? UBC AHD Nephrology CBP Samuel Kohen November 18, 2010
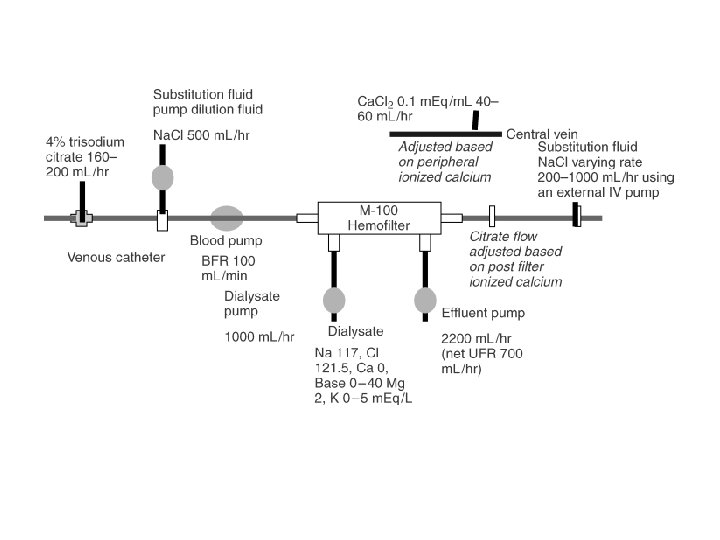

Introduction • CRRT is used in hemodynamically unstable patients with renal failure. • The most common problem with CRRT is circuit clotting. – Anticoagulation decreases this. • Heparin and Citrate are the two most common CRRT anticoagulants. • Until recently, it was not clear which was better.
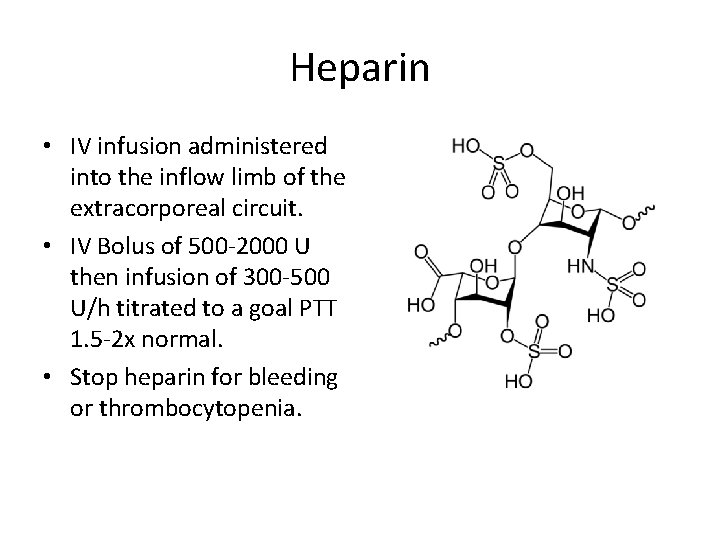
Heparin • IV infusion administered into the inflow limb of the extracorporeal circuit. • IV Bolus of 500 -2000 U then infusion of 300 -500 U/h titrated to a goal PTT 1. 5 -2 x normal. • Stop heparin for bleeding or thrombocytopenia.
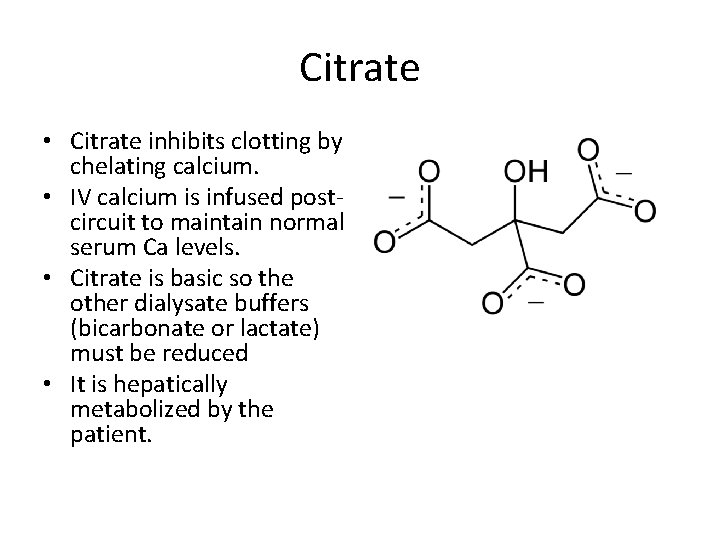
Citrate • Citrate inhibits clotting by chelating calcium. • IV calcium is infused postcircuit to maintain normal serum Ca levels. • Citrate is basic so the other dialysate buffers (bicarbonate or lactate) must be reduced • It is hepatically metabolized by the patient.

Citrate versus heparin for anticoagulation in continuous venous hemofiltration: a prospective randomized study Mehran et al. Intensive Care Med (2004) 30: 260– 265

Mehran et al. • Prospective trial randomizing 20 patients receiving CVVHF to heparin or citrate by hemofilter. • 49 hemofilters used: 23 heparin and 26 citrate. • Patients requiring more than one hemofilter were crossed over. • Patients with liver dysfunction or deemed at high risk of bleeding were excluded.

Mehran et al. • There is no difference in CRRT function (urea and creatinine clearance). • The median circuit lifetime was longer with citrate than heparin (70 vs 40 hours) mostly due to clotting (74 vs 46%). • Citrate anticoagulation is associated less bleeding: – Fewer significant bleeding episodes (1 UGIB vs 0) – Fewer PRBC transfusions (1 U/d vs 0. 2 U/d) • Citrate is more often associated with metabolic derangements – Metabolic alkalosis and hypocalcemia.

Regional citrate versus systemic heparinization for continuous renal replacement in critically ill patients. Demetrios et al. Kidney international. Vol 67 (2005). P 2361 – 2367.

Demetrios et al. • 30 critically ill adult patients with acute renal failure on CRRT were randomized to either heparin (16) or citrate (14). 2 patients crossed over treatment groups. 79 hemofilters were used: Heparin (43), Citrate (36).

Demetrios et al. • No significant difference in survival to hospital discharge. • (Citrate 14%; Heparin 29%; p = 0. 69) • Filters using citrate functioned much longer than those with heparin – (124. 5 vs 38. 3 hours).

Conclusions: Citrate is better • No difference in creatinine clearance or 60 day mortality. • Citrate treated circuits clot less frequently and last longer. • Citrate anticoagulation is associated with fewer bleeding episodes. • Metabolic alkalosis and hypocalcemia are the most common complications of citrate-treated circuits but are easily detected and usually harmless.

What are we doing at VGH? • Prime the circuit with 5000 U heparin and 2 L of saline • Infuse pre-circuit dialysate (1. 2 L/h) • Run high blood flow rates (300 m. L/min) • No continuous anticoagulation

What is the ideal dialysis dose? UBC AHD Nephrology CBP Samuel Kohen November 18, 2010

Dialysis dosing Dialysis Dose = Kt / V K = Clearance constant per time t = dialysis time V =Patient Fluid volume • The ideal IHD DD is 1. 2 • CVVHF effluent flow rate correlates with solute clearance rates. – Effluent rate of 20 ml/h/kg = DD 0. 8 – Effluent rate of 35 ml/h/kg = DD 1. 4 • Initial trials showed that higher dialysis intensity improved patient outcomes. • Two recent trials investigated this further: VA/NIH and RENAL.
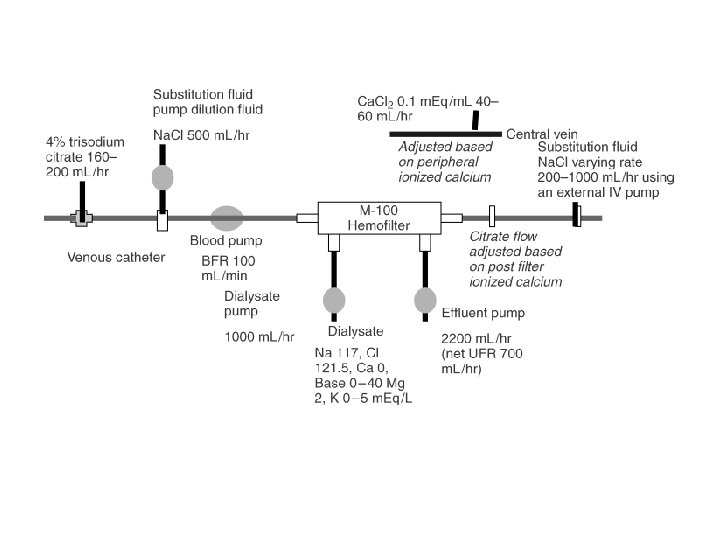

VA/NIH Palevsky et al. Intensity of renal support in critically ill patients with acute kidney injury. NEJM july 3, 2008. 359: 1 p. 7 – 20.

VA/NIH • 1124 critically ill patients with ARF were randomized to “low” (563) or “high” (561) intensity RRT. • Low intensity group – IHD 3 x/wk when HD stable (SOFA 0 -2) – CVVHDF at 20 m. L/kg/hr or SLED when unstable (SOFA 3 -4). • High intensity group – IHD 6 d/wk when stable – CVVHDF at a goal effluent rate of 35 m. L/kg/h) or SLED when unstable. • Results: – No significant difference in 60 day mortality (low 51. 5%, high 53. 6%), duration of RRT, rate of renal function recovery or evolution of nonrenal organ failure. – Higher rates of hypotension, hypophosphatemia and hypokalemia in the high-intensity RRT group.
RENAL Bellomo et al. Intensity of continuous renal replacement therapy in critically ill patients. NEJM 2009. 361: 17. 1627 – 1638.

RENAL • 1508 Critically ill patients with ARF on CVVHF were randomized to low (25 m. L/kg/hr – 747 patients) or high intensity (40 m. L/kg/hr – 761 patients) effluent rates. • There was no difference in 90 day mortality rate (44. 7%) or the need for RRT at 90 d between the two treatment groups.

Conclusion: Equal • Higher intensity CVVHF is associated with higher rates of electrolyte abnormalities without any clinical benefit. • A goal effluent flow rate of 20 ml/h/kg is adequate for patients on CVVHF.
- Slides: 102