CBE 417 Chemical Engineering Equilibrium Separations Lecture 8

CBE 417 “Chemical Engineering Equilibrium Separations” Lecture: 8 24 Sep 2012 1

Overview • Multicomponent Flash • Flash Unit Operation (Aspen. Plus) • Staged systems • Mc. Cabe-Thiele 2
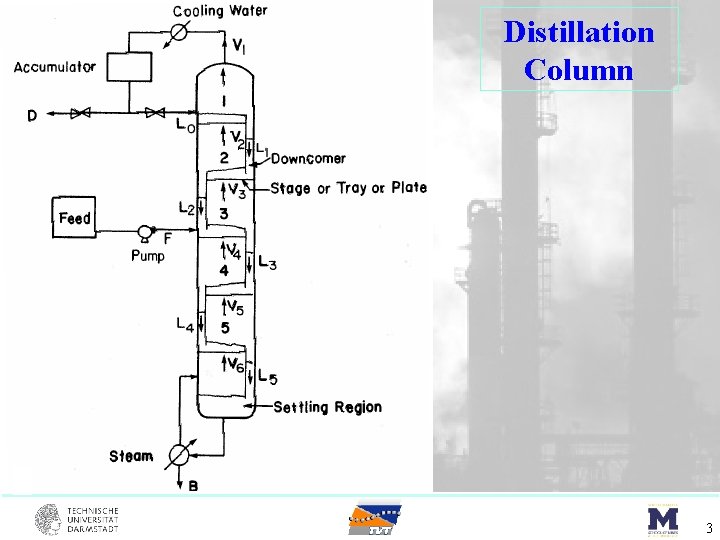
Distillation Column 3
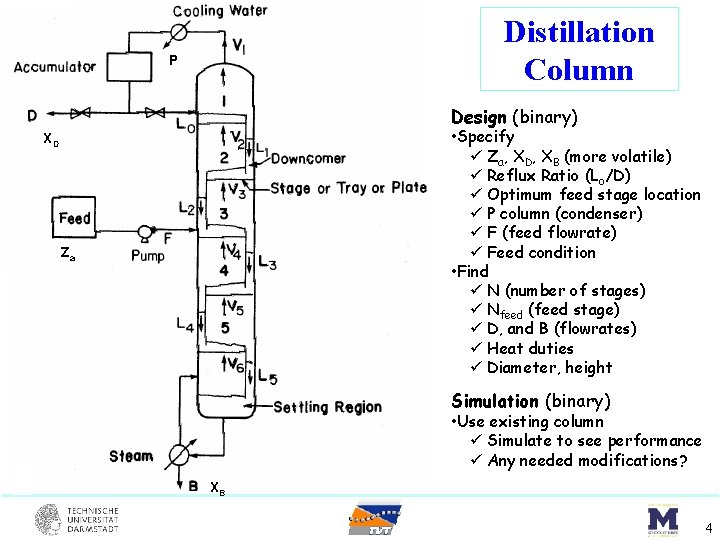
Distillation Column P Design (binary) • Specify ü Za, XD, XB (more volatile) ü Reflux Ratio (Lo/D) ü Optimum feed stage location ü P column (condenser) ü F (feed flowrate) ü Feed condition • Find ü N (number of stages) ü Nfeed (feed stage) ü D, and B (flowrates) ü Heat duties ü Diameter, height XD Za Simulation (binary) • Use existing column ü Simulate to see performance ü Any needed modifications? XB 4
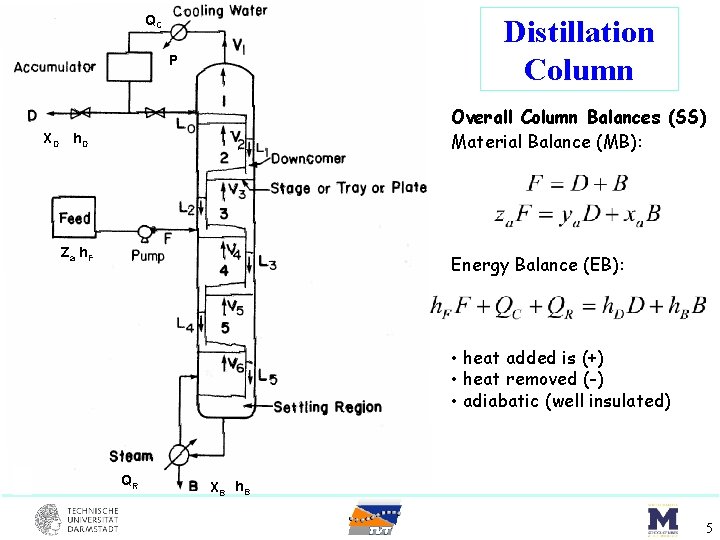
QC Distillation Column P Overall Column Balances (SS) Material Balance (MB): XD h. D Za h. F Energy Balance (EB): • heat added is (+) • heat removed (-) • adiabatic (well insulated) QR XB h. B 5

Mc. Cabe-Thiele Graphical Method (binary) Used to simplify analysis of binary distillation (ease of understanding) Assumptions: • Pure components a, b have equal latent heats of vaporization / mole ( ) and they stay constant. • are much larger than • Sensible heat changes • Heats of mixing • Column is adiabatic (well – insulated) • Constant pressure (P) throughout the column (i. e. no P in the column) Called Constant Molal Overflow (CMO) • Assumes for every 1 mole of light material vaporized that 1 mole of heavy material condenses from the vapor phase • Net result: • Total molar flowrates (i. e. L and V) remain constant within that column section (rectifying or stripping, or other) • Do not need a stage by stage energy balance Mc. Cabe-Thiele is done with MB and thermodynamic information. 6
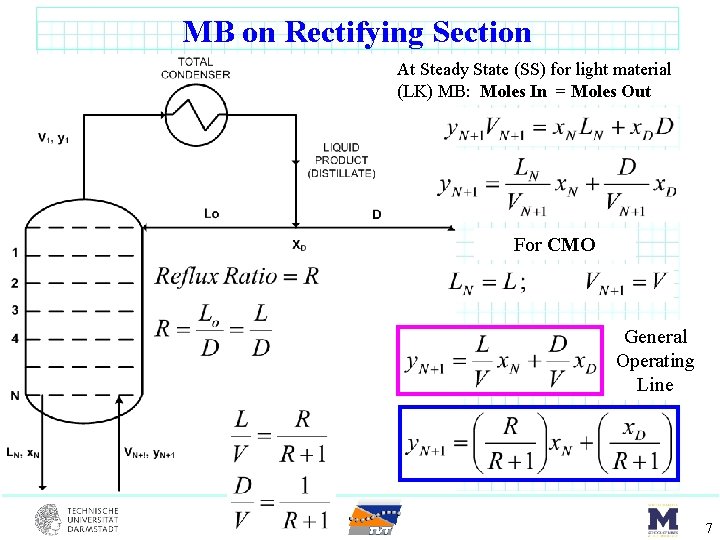
MB on Rectifying Section At Steady State (SS) for light material (LK) MB: Moles In = Moles Out For CMO General Operating Line 7
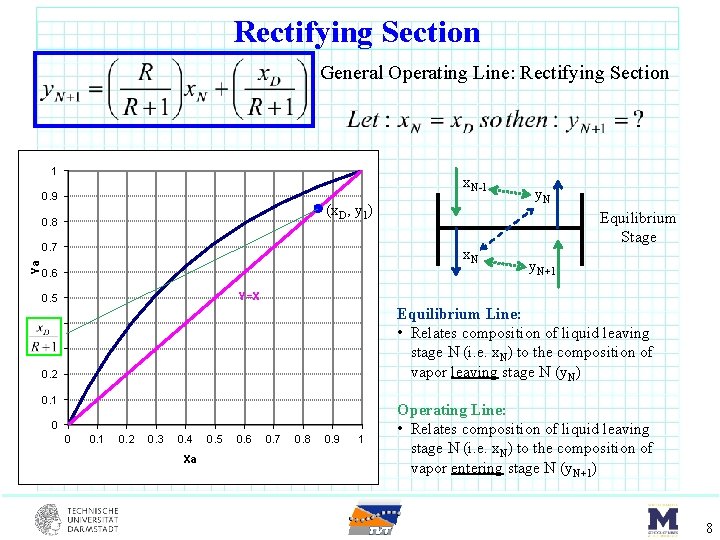
Rectifying Section General Operating Line: Rectifying Section 1 x. N-1 0. 9 (x. D, y 1) 0. 8 Ya 0. 7 x. N 0. 6 y. N Equilibrium Stage y. N+1 Y=X 0. 5 Equilibrium Line: • Relates composition of liquid leaving stage N (i. e. x. N) to the composition of vapor leaving stage N (y. N) 0. 4 0. 3 0. 2 0. 1 0 0 0. 1 0. 2 0. 3 0. 4 Xa 0. 5 0. 6 0. 7 0. 8 0. 9 1 Operating Line: • Relates composition of liquid leaving stage N (i. e. x. N) to the composition of vapor entering stage N (y. N+1) 8
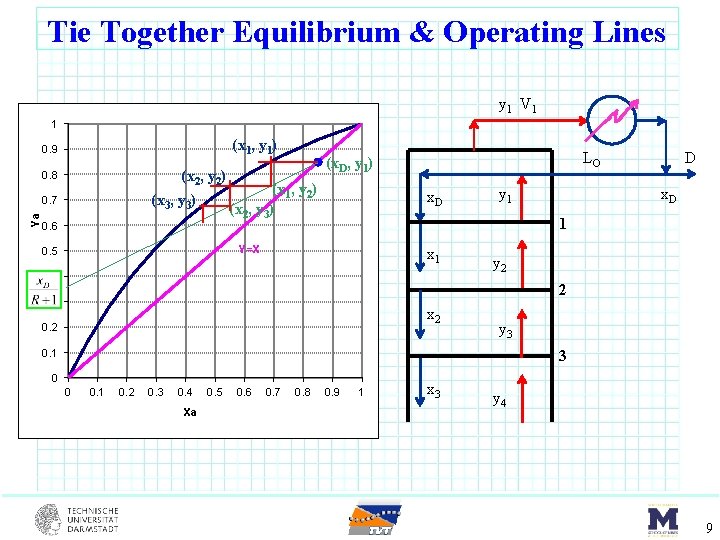
Tie Together Equilibrium & Operating Lines y 1 V 1 1 (x 1, y 1) 0. 9 (x 2, y 2) (x 3, y 3) 0. 8 Ya 0. 7 0. 6 (x 1, y 2) (x 2, y 3) x. D y 1 D x. D 1 Y=X 0. 5 LO (x. D, y 1) x 1 0. 4 y 2 2 0. 3 x 2 0. 2 y 3 0. 1 3 0 0 0. 1 0. 2 0. 3 0. 4 Xa 0. 5 0. 6 0. 7 0. 8 0. 9 1 x 3 y 4 9
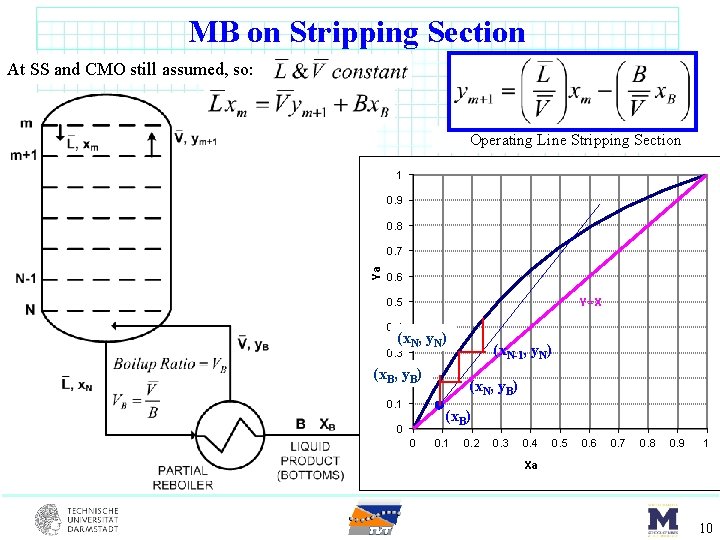
MB on Stripping Section At SS and CMO still assumed, so: Operating Line Stripping Section 1 0. 9 0. 8 Ya 0. 7 0. 6 Y=X 0. 5 0. 4 (x. N, y. N) (x. N-1, y. N) 0. 3 (x. B 0. 2 , y. B) 0. 1 (x. N, y. B) (x. B) 0 0 0. 1 0. 2 0. 3 0. 4 0. 5 0. 6 0. 7 0. 8 0. 9 1 Xa 10
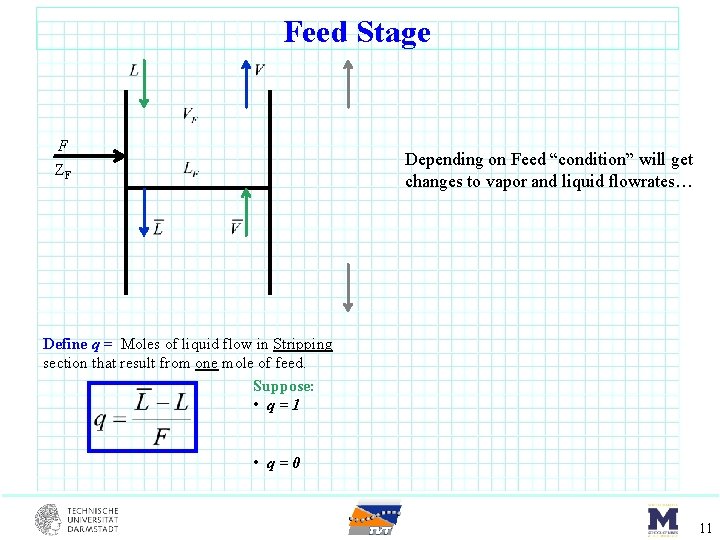
Feed Stage F ZF Depending on Feed “condition” will get changes to vapor and liquid flowrates… Define q = Moles of liquid flow in Stripping section that result from one mole of feed. Suppose: • q=1 • q=0 11
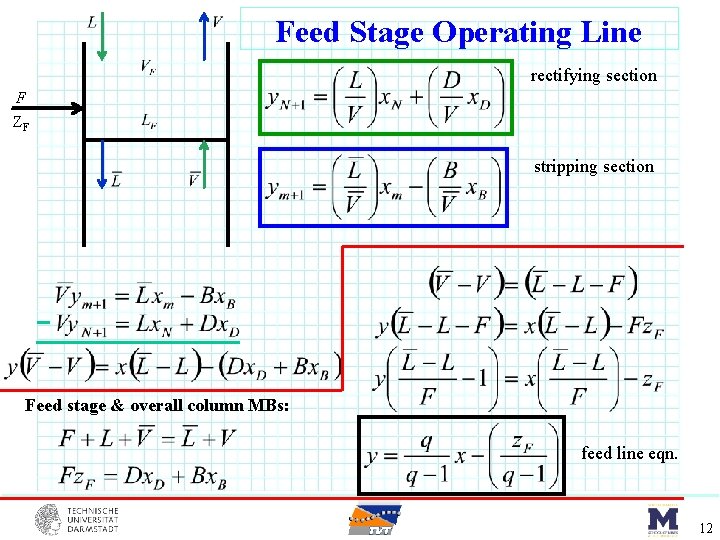
Feed Stage Operating Line rectifying section F ZF stripping section Feed stage & overall column MBs: feed line eqn. 12
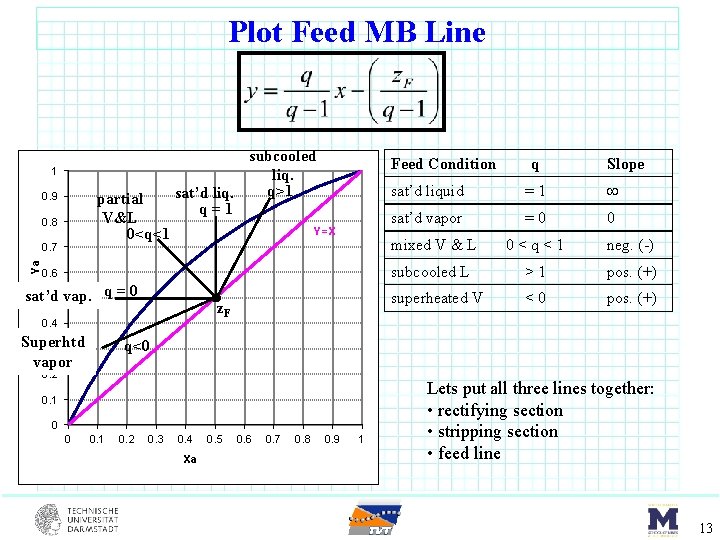
Plot Feed MB Line 1 partial V&L 0<q<1 0. 9 0. 8 Ya 0. 7 subcooled liq. q>1 sat’d liq. q=1 Feed Condition Y=X 0. 6 sat’d 0. 5 vap. q = 0 z. F 0. 4 Superhtd 0. 3 vapor Slope sat’d liquid =1 sat’d vapor =0 0 mixed V & L 0<q<1 neg. (-) subcooled L >1 pos. (+) superheated V <0 pos. (+) q<0 0. 2 0. 1 0 0 q 0. 1 0. 2 0. 3 0. 4 Xa 0. 5 0. 6 0. 7 0. 8 0. 9 1 Lets put all three lines together: • rectifying section • stripping section • feed line 13
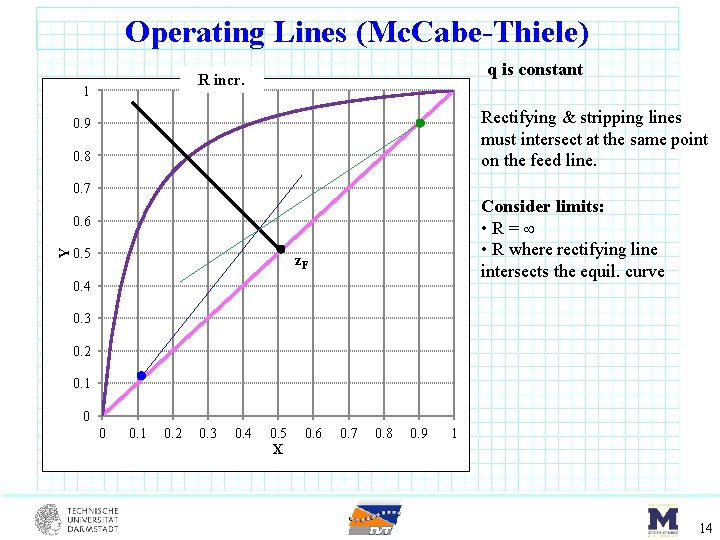
Operating Lines (Mc. Cabe-Thiele) q is constant R incr. 1 Rectifying & stripping lines must intersect at the same point on the feed line. 0. 9 0. 8 0. 7 Consider limits: • R= • R where rectifying line intersects the equil. curve Y 0. 6 0. 5 z. F 0. 4 0. 3 0. 2 0. 1 0 0 0. 1 0. 2 0. 3 0. 4 0. 5 X 0. 6 0. 7 0. 8 0. 9 1 14
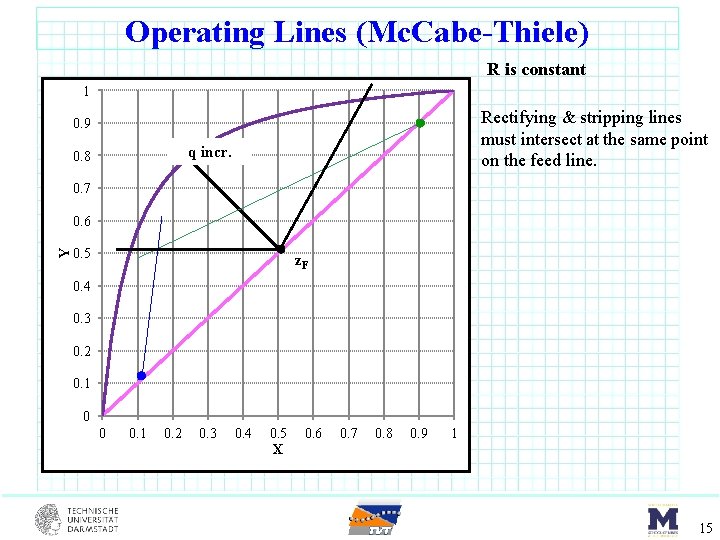
Operating Lines (Mc. Cabe-Thiele) R is constant 1 Rectifying & stripping lines must intersect at the same point on the feed line. 0. 9 q incr. 0. 8 0. 7 Y 0. 6 0. 5 z. F 0. 4 0. 3 0. 2 0. 1 0 0 0. 1 0. 2 0. 3 0. 4 0. 5 X 0. 6 0. 7 0. 8 0. 9 1 15
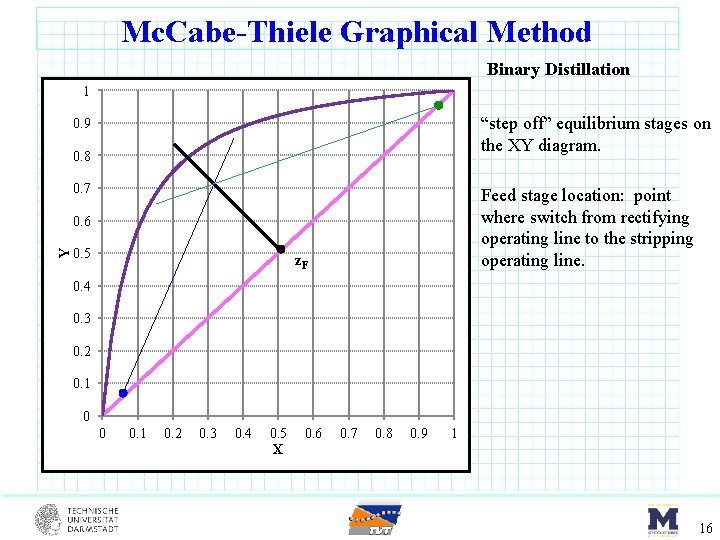
Mc. Cabe-Thiele Graphical Method Binary Distillation 1 “step off” equilibrium stages on the XY diagram. 0. 9 0. 8 0. 7 Feed stage location: point where switch from rectifying operating line to the stripping operating line. Y 0. 6 0. 5 z. F 0. 4 0. 3 0. 2 0. 1 0 0 0. 1 0. 2 0. 3 0. 4 0. 5 X 0. 6 0. 7 0. 8 0. 9 1 16
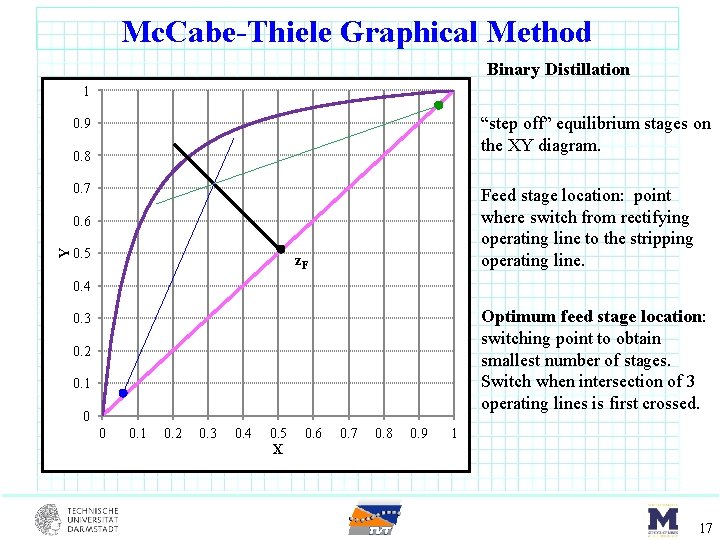
Mc. Cabe-Thiele Graphical Method Binary Distillation 1 “step off” equilibrium stages on the XY diagram. 0. 9 0. 8 0. 7 Feed stage location: point where switch from rectifying operating line to the stripping operating line. Y 0. 6 0. 5 z. F 0. 4 Optimum feed stage location: switching point to obtain smallest number of stages. Switch when intersection of 3 operating lines is first crossed. 0. 3 0. 2 0. 1 0 0 0. 1 0. 2 0. 3 0. 4 0. 5 X 0. 6 0. 7 0. 8 0. 9 1 17
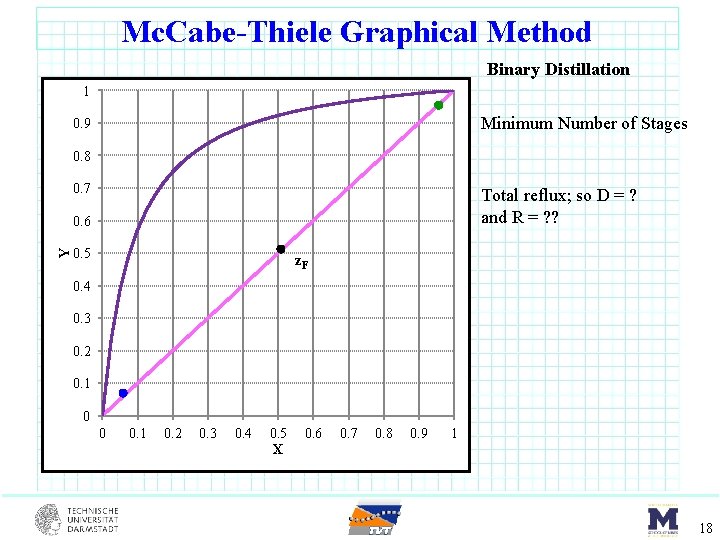
Mc. Cabe-Thiele Graphical Method Binary Distillation 1 Minimum Number of Stages 0. 9 0. 8 0. 7 Total reflux; so D = ? and R = ? ? Y 0. 6 0. 5 z. F 0. 4 0. 3 0. 2 0. 1 0 0 0. 1 0. 2 0. 3 0. 4 0. 5 X 0. 6 0. 7 0. 8 0. 9 1 18
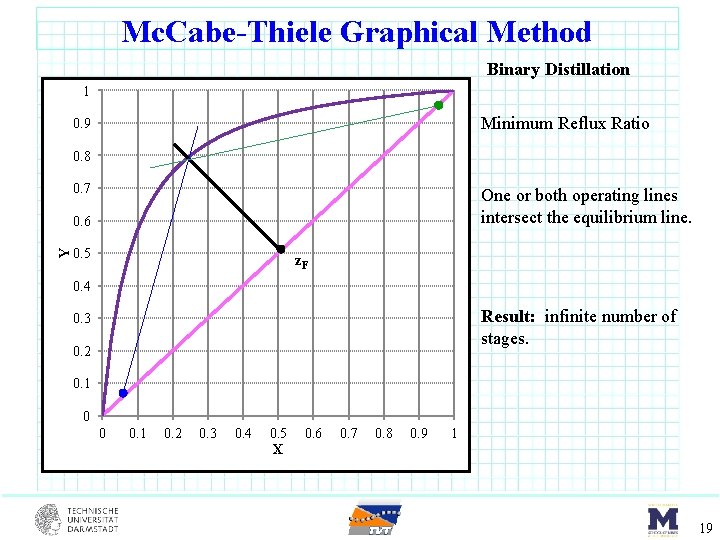
Mc. Cabe-Thiele Graphical Method Binary Distillation 1 Minimum Reflux Ratio 0. 9 0. 8 0. 7 One or both operating lines intersect the equilibrium line. Y 0. 6 0. 5 z. F 0. 4 Result: infinite number of stages. 0. 3 0. 2 0. 1 0 0 0. 1 0. 2 0. 3 0. 4 0. 5 X 0. 6 0. 7 0. 8 0. 9 1 19
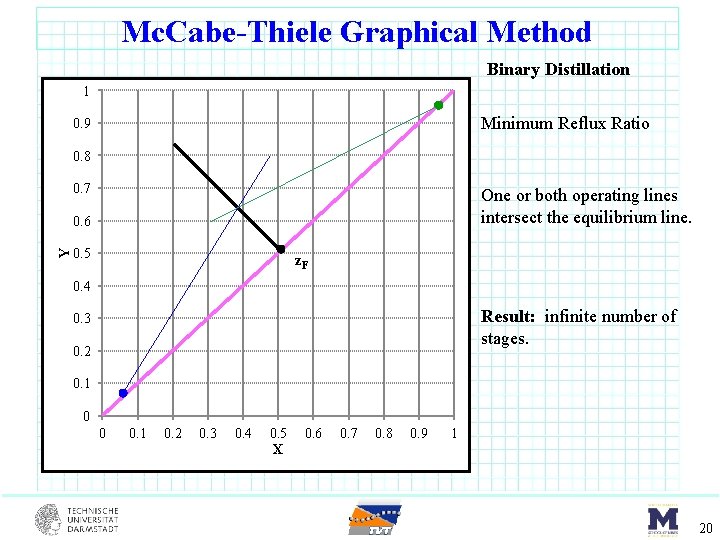
Mc. Cabe-Thiele Graphical Method Binary Distillation 1 Minimum Reflux Ratio 0. 9 0. 8 0. 7 One or both operating lines intersect the equilibrium line. Y 0. 6 0. 5 z. F 0. 4 Result: infinite number of stages. 0. 3 0. 2 0. 1 0 0 0. 1 0. 2 0. 3 0. 4 0. 5 X 0. 6 0. 7 0. 8 0. 9 1 20

Mc. Cabe-Thiele Graphical Method (binary) Used to simplify analysis of binary distillation (ease of understanding) Assumptions: • Pure components a, b have equal latent heats of vaporization / mole ( ) and they stay constant. • are much larger than • Sensible heat changes • Heats of mixing • Column is adiabatic (well – insulated) • Constant pressure (P) throughout the column (i. e. no P in the column) Called Constant Molal Overflow (CMO) • Assumes for every 1 mole of light material vaporized that 1 mole of heavy material condenses from the vapor phase • Net result: • Total molar flowrates (i. e. L and V) remain constant within that column section (rectifying or stripping, or other) • Do not need a stage by stage energy balance Mc. Cabe-Thiele is done with MB and thermodynamic information. 21
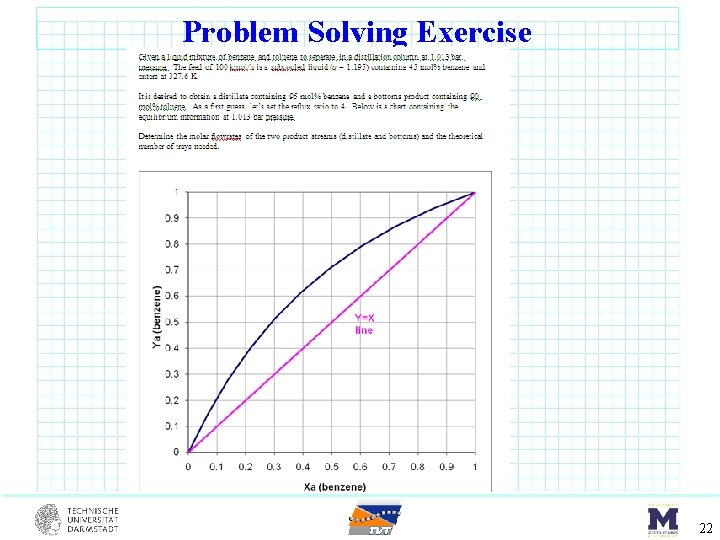
Problem Solving Exercise 22

Questions? 23
- Slides: 23