CAUSALITY ASSESSMENT OF REPORTED ADVERSE DRUG REACTIONS Ana

















![REFERENCES [1] MACEDO, A. F. [et al. ] - Causality assessment of adverse drug REFERENCES [1] MACEDO, A. F. [et al. ] - Causality assessment of adverse drug](https://slidetodoc.com/presentation_image_h/2cb7aae65925da20e6e3b8d59e1b12e9/image-24.jpg)
- Slides: 24

CAUSALITY ASSESSMENT OF REPORTED ADVERSE DRUG REACTIONS Ana Filipa Macedo Faculty of Health Sciences Portugal Ana Filipa Macedo 2004

No drug which is pharmacologically effective is entirely without hazard Efficacy Safety Quality Safety is a relative concept Ana Filipa Macedo 2004

HISTORICAL PERSPECTIVE 2000 b. C. – HAMMURABI CODE: “The Doctor who causes death should loose his hands” 500 b. C. – HIPÓCRATES: “Primum non nocere” 200 – GALENO: “Drugs have a main action and a secondary action ” Ana Filipa Macedo 2004

PERSPECTIVA HISTÓRICA … 1937 – Sulfanilamide in Dietilenoglicol / Renal Impairment 1961 – Thalidomide / Phocomelia - 10 000 Victims 1971 – Dietilstilbestrol / Vaginal Carcinoma in the daughters, 10 to 20 years after exposition Ana Filipa Macedo 2004

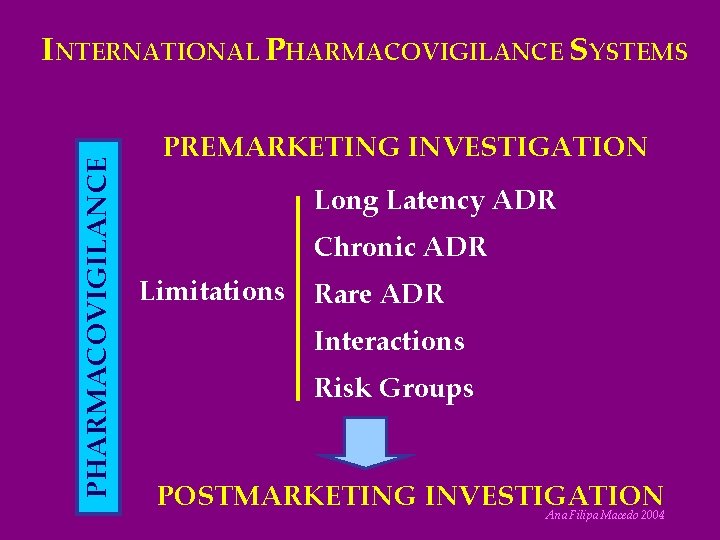
PHARMACOVIGILANCE INTERNATIONAL PHARMACOVIGILANCE SYSTEMS PREMARKETING INVESTIGATION Long Latency ADR Chronic ADR Limitations Rare ADR Interactions Risk Groups POSTMARKETING INVESTIGATION Ana Filipa Macedo 2004

ADVERSE DRUG REACTION “a response to a drug which is noxious and unintended, and which occurs at dose normally used in humans for the prophylaxis, diagnosis or therapy of disease, or for a modification of physiological function. ” W. H. O 1972 Ana Filipa Macedo 2004

EPIDEMIOLOGY OF ADVERSE DRUG REACTIONS Adverse Effects Medication Error (Preventable) Preventable ADRs not Preventable Quality Problems • 4 th to 6 th cause of deat in U. S. A. , 1994 • 5% of Hospital Admissions • 11% of Hospital Patients • Doubles costs, lengt of stay and mortality risk • 50% are potentially preventable • Less than 1% are reported Ana Filipa Macedo 2004

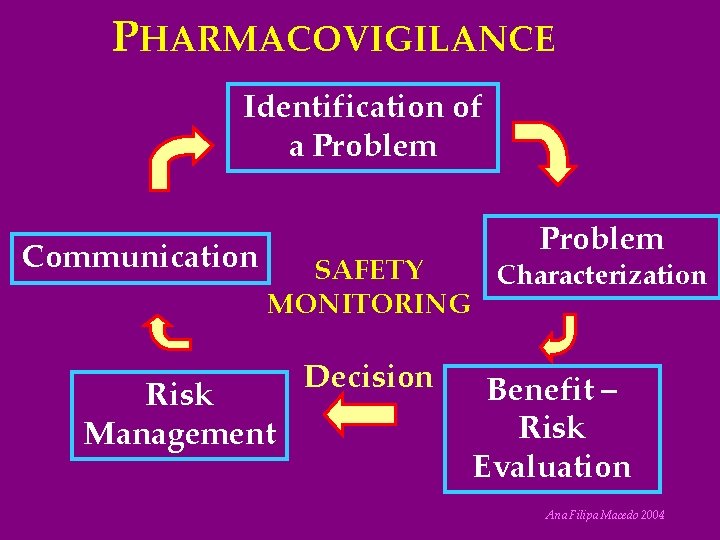
PHARMACOVIGILANCE Identification of a Problem Communication Problem SAFETY Characterization MONITORING Risk Management Decision Benefit – Risk Evaluation Ana Filipa Macedo 2004

“Drug Safety is a field where can be smoke without fire” Waller, P. Drug Exposition Reported Adverse Effect IMPUTATION Dynamic Process Ana Filipa Macedo 2004

DIFFICULT. . . • Complex Nature of Adverse Events • Individual Clinical Variability • Retrospective Spontaneous Report Ana Filipa Macedo 2004

GLOBAL INTROSPECTION “ Clinical Judgement of Experts” • Subjective • Problems of reproducibility • Not calibrated Ana Filipa Macedo 2004

DECISIONAL ALGORITHMS “Systematic Strategies of Decision in Uncertainty Conditions” • Explicit • Reproducible • Simple • Possible Automation • Improve Reporting Ana Filipa Macedo 2004

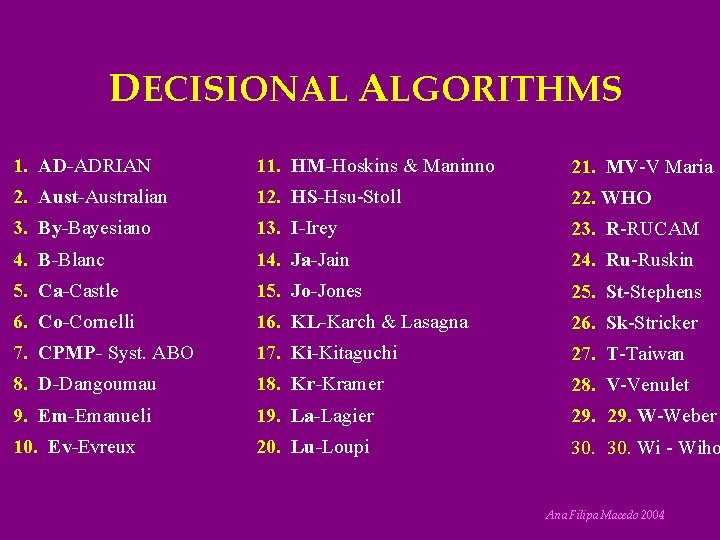
DECISIONAL ALGORITHMS 1. AD-ADRIAN 11. HM-Hoskins & Maninno 21. MV-V Maria 2. Aust-Australian 12. HS-Hsu-Stoll 22. WHO 3. By-Bayesiano 13. I-Irey 23. R-RUCAM 4. B-Blanc 14. Ja-Jain 24. Ru-Ruskin 5. Ca-Castle 15. Jo-Jones 25. St-Stephens 6. Co-Cornelli 16. KL-Karch & Lasagna 26. Sk-Stricker 7. CPMP- Syst. ABO 17. Ki-Kitaguchi 27. T-Taiwan 8. D-Dangoumau 18. Kr-Kramer 28. V-Venulet 9. Em-Emanueli 19. La-Lagier 29. W-Weber 10. Ev-Evreux 20. Lu-Loupi 30. Wi - Wiho Ana Filipa Macedo 2004

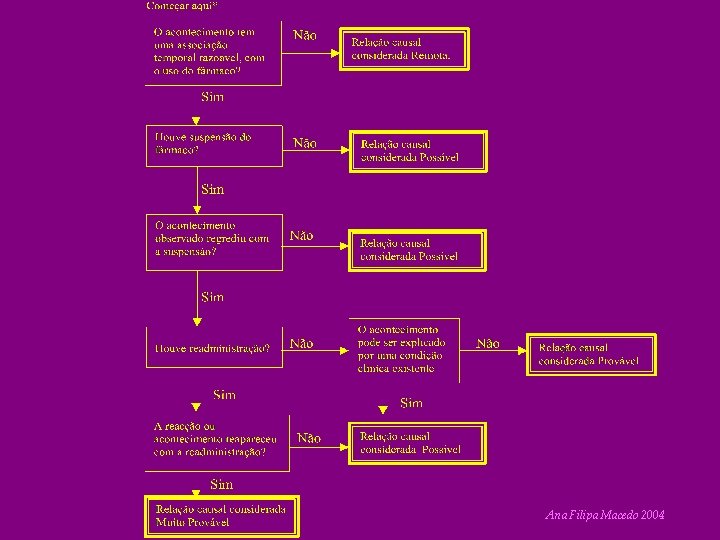
CRITERIA 1. Challenge 2. Dechallenge 3. Rechallenge 4. Bibliographic Description • Etiologic Alternatives Ana Filipa Macedo 2004

Ana Filipa Macedo 2004

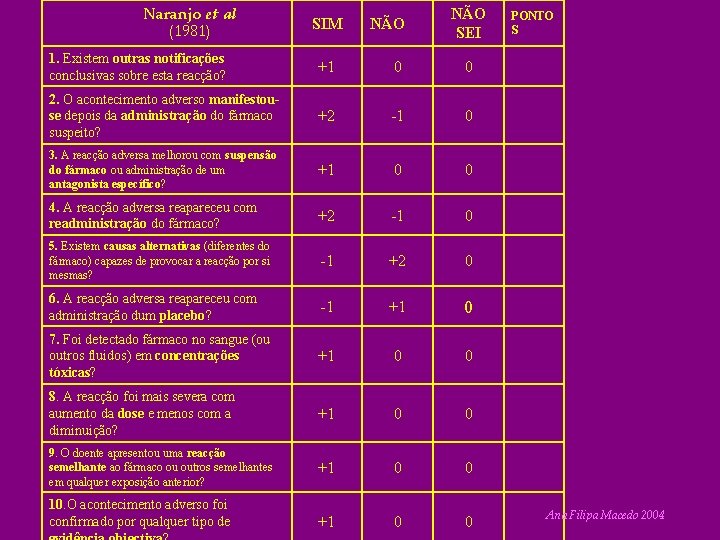
Naranjo et al (1981) SIM NÃO SEI 1. Existem outras notificações conclusivas sobre esta reacção? +1 0 0 2. O acontecimento adverso manifestouse depois da administração do fármaco suspeito? +2 -1 0 3. A reacção adversa melhorou com suspensão do fármaco ou administração de um antagonista específico? +1 0 0 4. A reacção adversa reapareceu com readministração do fármaco? +2 -1 0 5. Existem causas alternativas (diferentes do fármaco) capazes de provocar a reacção por si mesmas? -1 +2 0 6. A reacção adversa reapareceu com administração dum placebo? -1 +1 0 7. Foi detectado fármaco no sangue (ou outros fluidos) em concentrações tóxicas? +1 0 0 8. A reacção foi mais severa com aumento da dose e menos com a diminuição? +1 0 0 9. O doente apresentou uma reacção semelhante ao fármaco ou outros semelhantes em qualquer exposição anterior? +1 0 0 10. O acontecimento adverso foi confirmado por qualquer tipo de PONTO S Ana Filipa Macedo 2004

Ana Filipa Macedo 2004

Ana Filipa Macedo 2004

LIMITATIONS • Fixed Scoring • Arbitrary Scoring • Disagreement between algorithms None universally accepted as gold standard Ana Filipa Macedo 2004

CAUSALITY ASSESSMENT GLOBAL INTROSPECTION SELECTED ALGORITHMS 1. Aust-Australian 6. HS-Hsu-Stoll 2. B-Blanc 7. I-Irey 3. Co-Cornelli 8. Jo-Jones 4. D-Dangoumau 9. KL-Karch & Lasagna 5. Em-Emanueli 10. Ki-Kitaguchi 11. Kr-Kramer 12. N -Naranjo 13. St-Stephens 14. V-Venulet 15. W-Weber Ana Filipa Macedo 2004

RESULTS v. Agreement between algorithms and GI was 43% in average v 100% agreement was not found for any algorithm v None of the adverse events was equally imputed by all the algorithms v Sensitivity 93% and Specificity 7% in average Ana Filipa Macedo 2004

CONCLUSIONS A reference method was not identified Decisional algorithms are not definite alternatives in the individual causality assessment of adverse drug reactions. Ana Filipa Macedo 2004

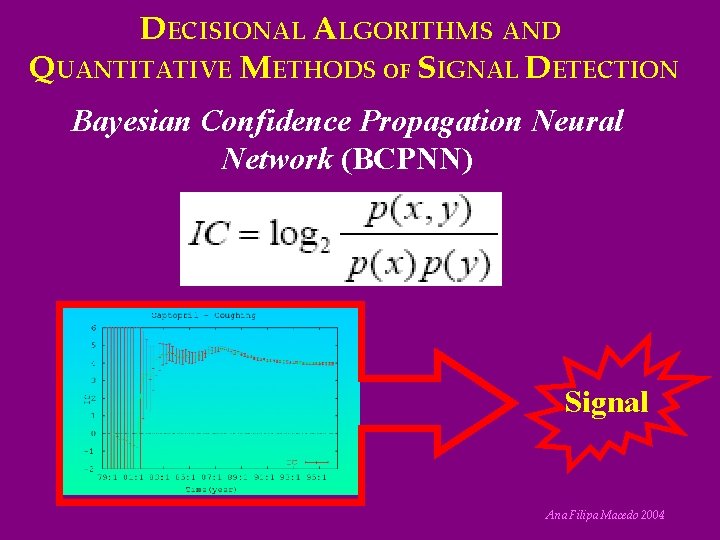
DECISIONAL ALGORITHMS AND QUANTITATIVE METHODS OF SIGNAL DETECTION Bayesian Confidence Propagation Neural Network (BCPNN) Signal Ana Filipa Macedo 2004
![REFERENCES 1 MACEDO A F et al Causality assessment of adverse drug REFERENCES [1] MACEDO, A. F. [et al. ] - Causality assessment of adverse drug](https://slidetodoc.com/presentation_image_h/2cb7aae65925da20e6e3b8d59e1b12e9/image-24.jpg)
REFERENCES [1] MACEDO, A. F. [et al. ] - Causality assessment of adverse drug reactions: comparison of the results obtained from published decisional algorithms and from the evaluations of an expert panel, according to different levels of imputability. J Clin Pharm Ther. 2003. 28: 137 -143. [2] STAHL, M. [et al. ] – Introducing triage logic as a new strategy for the detection of signals in the WHO Drug Monitoring Database. Pharmacoepidemiol Drug Safe. 2004; 13: 355 -363. Ana Filipa Macedo 2004