CATHODIC PROTECTION OF REINFORCED CONCRETE STRUCTURES Center for















































- Slides: 51

CATHODIC PROTECTION OF REINFORCED CONCRETE STRUCTURES Center for Engineering Research Institute, KFUPM

REINFORCEMENT CORROSION § Passivity • High p. H leading to formation of passive layer • Chemical binding of chlorides • Dense and relatively impermeable structure of concrete § Depassivation • Chloride ingress • Carbonation

Chloride-induced Reinforcement Corrosion • Due to external chlorides • Due to chloride contamination from the mix constituents

Chloride Limits § ACI 318 (0. 1 – 0. 15%; water soluble) § ACI 224 (0. 2%; acid soluble) § BS 8110 (0. 4%; total)

CARBONATION • Ca(OH)2 + CO 2 Ca. CO 3 + H 2 O • Reduction in p. H (up to 8. 5)

FACTORS AFFECTING CARBONATION • • • Concrete quality Concrete cover Moisture condition Temperature Chloride-sulfate contamination

PREVENTION THROUGH DESIGN § § Cover reinforcing steel Water-cement ratio Cement content Cement type

PROTECTION OF STEEL • • Metallic coatings Non-Metallic coatings Inhibitors Electrochemical protection • Cathodic protection • Chloride removal • Realkalization

CATHODIC PROTECTION § Sacrificial anode § Impressed current

Corrosion cell in concrete

Corrosion cell in a repaired concrete

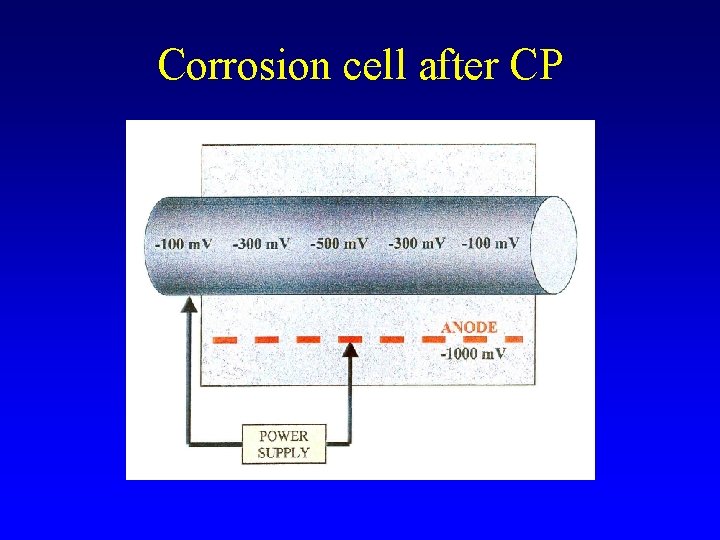
Corrosion cell after CP

Comparison of CP systems (Merits) Impressed current system Sacrificial anode system Longer anode life Inherently simple Current can be controlled No monitoring and maintenance No requirement for electrical isolation Risk of hydrogen embrittlement on high strength steel is minimal Saw cutting and concrete encapsulation for anodes is not required

Comparison of CP systems (Demerits) Impressed current system Sacrificial anode system Requires monitoring and maintenance Short anode life Electrical isolation required between anode and steel Anode current delivery is dependent on anode chemistry and surrounding environment Conduit and wiring required Current cannot be adjusted or controlled

Sacrifical Anode System § Anode material • Alloys of Mg, Al, Zn § Anode shapes • Zinc foil (Zinc hydrogel) • Metallized zinc (thermal spray) • Jackets • Zinc capsules or discs encased in mortar

Corroded pile in sea

Pier with SACP

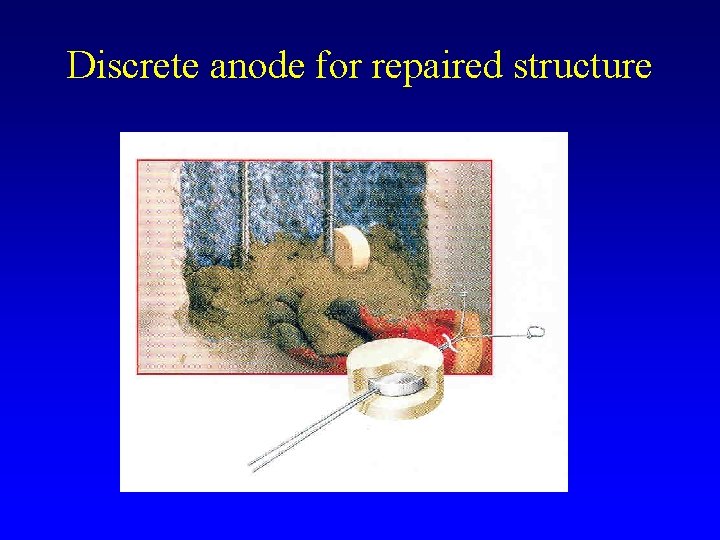
Discrete anode for repaired structure

Discrete anode

Placement of discrete anodes

Mesh anode

Placement of anodes

Specimens with discrete anodes

Specimens with jacket anodes

Specimens with mesh anodes

Specimens and measurement boxes

Sacrificial Anode

Impressed Current CP

ICCP Anodes § Conductive coating • Sparyed, rolled or brushed over a nibium wire (300 um thickness) § Arc sprayed zinc • Spraying metal on concrete with compressed air (300 -400 um) § Titanium anode mesh jackets § Titanium ribbon § Discrete anodes § Thermally sprayed titanium

Current requirements § Atmospheric zones • 5 to 15 m. A/m 2 § Under water • 0. 2 to 2 m. A/m 2 § Decreases with time

ICCP – Power System § § Transformers/Rectifiers Constant voltage/current system Stable over long period of time Solar- or wind-powered units are also used

ICCP - Connections § § § Durable and corrosion resistant Low-voltage wires Diameter for minimum voltage drop Color coded Multiple connections and cables recommended

ICCP Monitoring System § Potential measurements § Embeddable reference electrodes • Ag/Ag. Cl • Mn/Mn. O 2

Anode mesh fixed on concrete

Conductor bar welded to anode mesh

Negative connection to the steel bar

Connection of embedded items

Reference electrode

Installing overlay

Finishing operation

Anode and steel cage

Details of anode

Connection of anode ribbon to the titanium conductor bar

Steel connection

Placement of electrodes and connections to the steel bar

Pouring of concrete

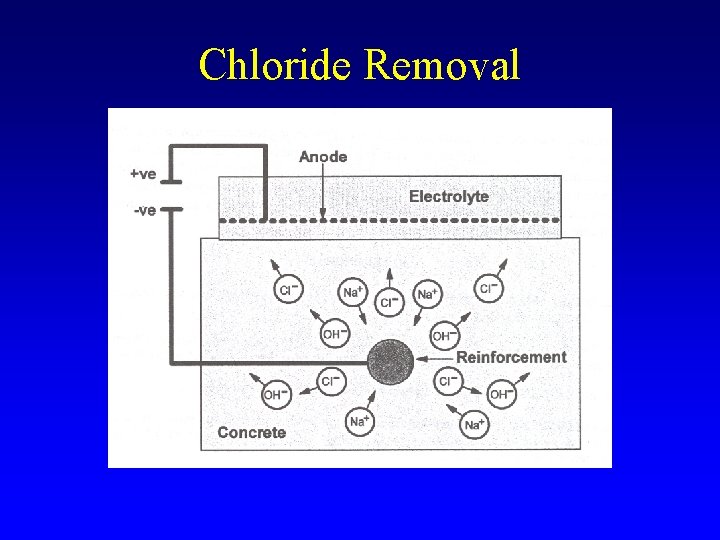
Chloride Removal

CE - Details § Anode • Titanium or steel mesh § Electrolyte • Water or saturated calcium hydroxide solution § Current • 1 to 2 A/m 2 § Time • 6 to 10 weeks § Not advisable for prestressed concrete § Impressed/sacrificial anode cathodic protection systems may be used § Side effects – Alkali-silica reaction

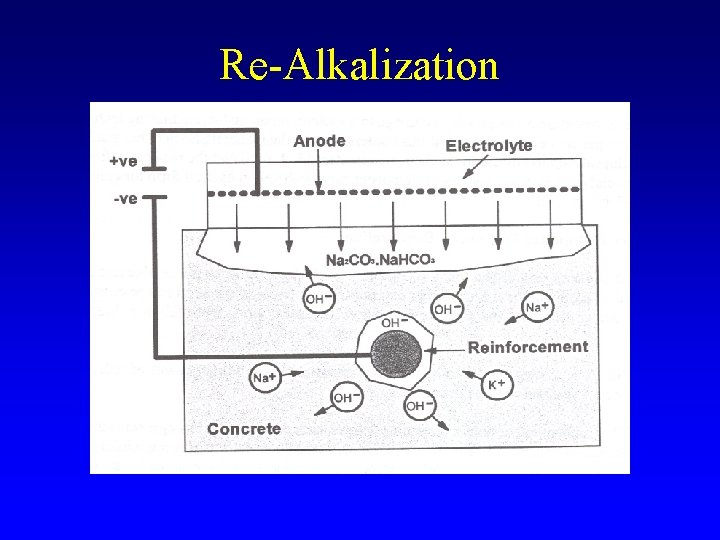
Re-Alkalization

Realkalization - details § Anode • Steel or titanium mesh § Electrolyte • Sodium carbonate solution (1 mole/L) § Current • 1 to 2 A/m 2 § Time • Days to weeks § Not advisable for prestressed concrete § Impressed/sacrificial anode cathodic protection systems may be used § Side effects – Alkali-silica reaction

THANK YOU