Catheterrelated bloodstream infections Wanida Paoin Thammasat University Catheterrelated

Catheter-related bloodstream infections Wanida Paoin Thammasat University

Catheter-related bloodstream infections n n Incidence in PICU: 7. 3 -13. 8 central line-associated bloodstream infections/1000 cath days Risk factors q q q premature < 1000 g low immunity multiple CVC long term CVC insertion reduced ICU Nurse: Patient Ratio using Non-ICU-Trained Nurses in ICUs
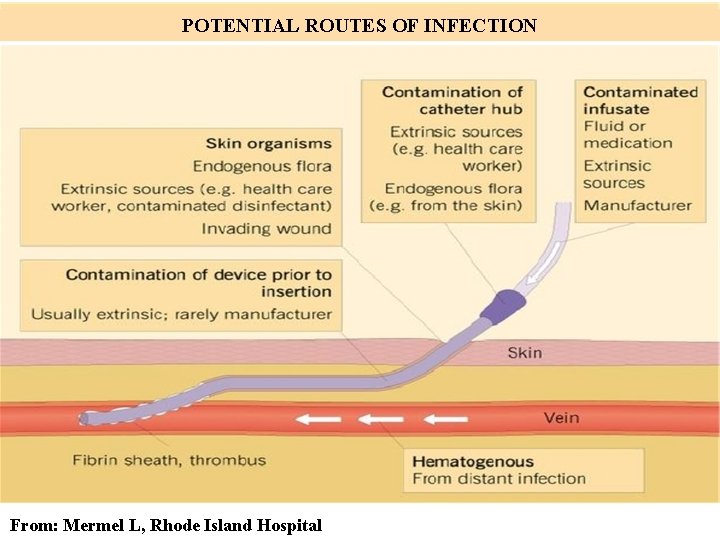
POTENTIAL ROUTES OF INFECTION From: Mermel L, Rhode Island Hospital

Strategies for Prevention of Catheter. Related Infections n n Quality Assurance and Continuing Education Surveillance q q n Monitor the catheter sites visually or by palpation through the intact dressing on a regular basis, Manifestations suggesting local or BSI, the dressing should be removed to allow thorough examination of the site Hand Hygiene and Aseptic Technique q q q Maximal sterile barrier precautions (e. g. , cap, mask, sterile gown, sterile gloves, and large sterile drape) during the insertion of CVCs Hand hygiene before and after inserting, replacing, accessing, or dressing an intravascular catheter. Washing hands with conventional antiseptic-containing soap and water or with waterless alcohol-based gels or foams.

Catheter Insertion Efficacy of Barrier Precautions During CVC Insertion Cath colonization Cath sepsis *p<0. 05 Raadet al, ICHE 1994 Barrier precautions Minimal Maximal 7. 2% 2. 3%* 3. 6% 0. 6%*

Strategies for Prevention of CR Infections n Site of Catheter Insertion q q Femoral catheters: relatively high colonization rates, higher risk for deep venous thrombosis Internal jugular cath: higher risk for infection than those inserted into a subclavian or femoral vein

Catheter Placement Prospective, Randomized, Multicenter Study of Femoral vs Subclavian CVC Insertion Femoral cath Cath colonization Cath sepsis Cath thrombosis Merrer et al JAMA 2001 14. 2% 4. 4% 6% Subclavian cath 2. 2% 1. 5% 0%

Strategies for Prevention of CR Infections n Skin Antisepsis q 2% aqueous chlorhexidine gluconate lowered BSI rates compared with site preparation with 10% povidone-iodine or 70% alcohol

Cutaneous Antisepsis CR Infection Prevention w/ Chlorhexidine Cath colonization CRBSI CHX Control 2. 3% 7%*† 0. 5% 2. 6% (Maki `91( 2% 7%* 0. 6% (Sheehan`93( 4. 7% 9. 3%* 0 0. 5% (Garland `95( 312/10 ) 30. 9/10 30. 1/10 *331/10 Minoz`96( 34% 27%* 3. 5/103 4. 1/103 (Humar`97( red values = p<0. 05 =*povidone iodine † = alcohol

Strategies for Prevention of CR Infections n Catheter Site Dressing Regimens q Transparent, semipermeable polyurethane dressings: n n n Reliably secure the device, Permit continuous visual inspection of the catheter site, Permit patients to bathe Require less frequent changes than do standard gauze A meta-analysis: The risk for CRBSIs did not differ between the groups using transparent dressings versus groups using gauze dressing. If blood is oozing from the catheter insertion site, gauze dressing might be preferred.

Strategies for Prevention of CR Infections n Catheter Site Dressing Regimens q Chlorhexidine-impregnated sponge (Biopatch™) placed over the site n n A multi-center study: reduced the risk for catheter colonization and CRBSI. No adverse systemic effects resulted from use of this device.
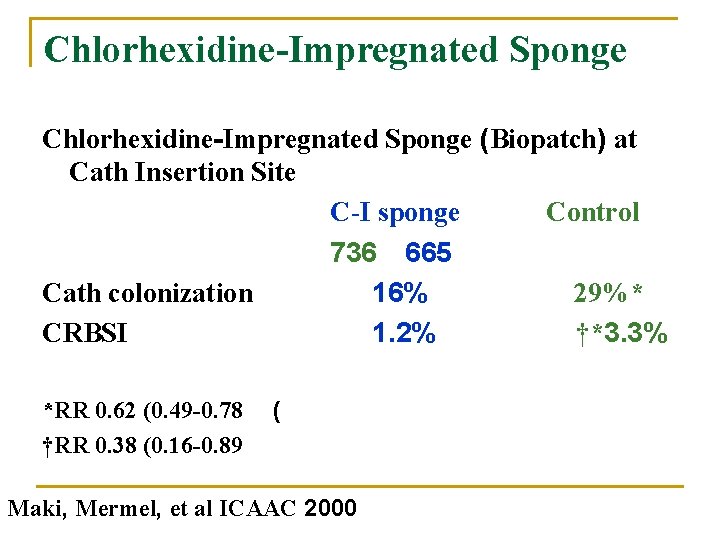
Chlorhexidine-Impregnated Sponge (Biopatch) at Cath Insertion Site C-I sponge Control 736 665 Cath colonization 16% 29%* CRBSI 1. 2% †*3. 3% *RR 0. 62 (0. 49 -0. 78 †RR 0. 38 (0. 16 -0. 89 ( Maki, Mermel, et al ICAAC 2000

Strategies for Prevention of CR Infections n Antimicrobial/Antiseptic Impregnated Catheters and Cuffs q q Chlorhexidine/Silver sulfadiazine. Minocycline/Rifampin. Platinum/Silver cuffs

Chlorhexidine-Impregnated Sponge Chlorhexidine-Silver Sulfadiazine-Impregnated 2 nd. Generation Catheters #CVCs Cath duration Cath coloniz †p=0. 006 Rupp et al, ICAAC 2001 CHSS 368 7 d 6. 4% (9/103 CD) Control 374 7 d 12. 8% (19/103 CD†(
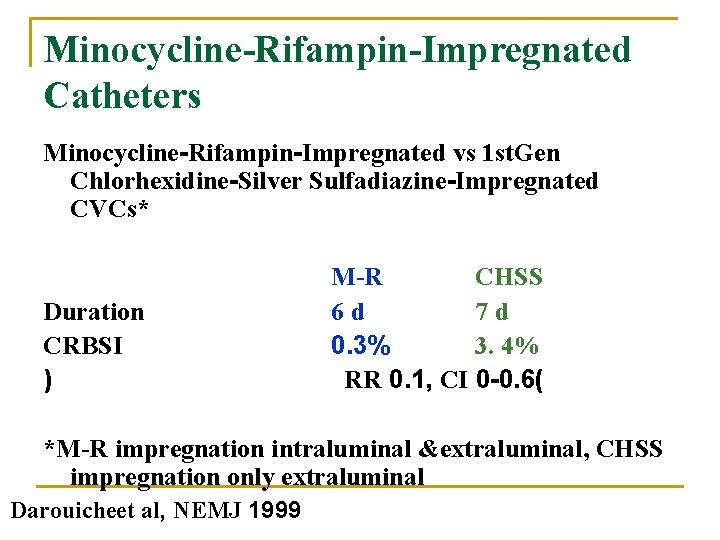
Minocycline-Rifampin-Impregnated Catheters Minocycline-Rifampin-Impregnated vs 1 st. Gen Chlorhexidine-Silver Sulfadiazine-Impregnated CVCs* Duration CRBSI ) M-R CHSS 6 d 7 d 0. 3% 3. 4% RR 0. 1, CI 0 -0. 6( *M-R impregnation intraluminal &extraluminal, CHSS impregnation only extraluminal Darouicheet al, NEMJ 1999

Silver Iontophoretic Catheter CRBSI Silver-Iontophoretic 3. 1% 1% Combined OR 0. 23 (0. 07 -0. 66( *Bong et al, ICAAC 2001 †Ibanez-Nolla et al, ICAAC 2001 Control 8%* 3. 9%†

Silver Iontophoretic Catheter type growth Control Chlorhexidine/silver sulfadiazine Silver iontophoretic Caths w/ significant 100% 67% 20% insertion site of lab animals inoculated w/ S. aureus, caths quantitatively cultured at 7 d Raadet al, JID 1996

Strategies for Prevention of CR Infections n Antibiotic/Antiseptic Ointments q q n Povidone-iodine ointment Mupirocin ointment Antibiotic Lock Prophylaxis q q Flushing and filling the lumen of the catheter with an antibiotic solution and leaving the solution to dwell in the lumen of the catheter. Heparin plus 25 micrograms/ml of vancomycin Vancomycin/ciprofloxacin/heparin combination Minocycline and ethylenediaminetetraraacetic acid (EDTA)

Strategies for Prevention of CR Infections n Anticoagulants q q Prevent catheter thrombosis Heparin flush Heparin-bonded coating Warfarin

Strategies for Prevention of CR Infections n Replacement of intravascular catheters q q q Replace CVCs if purulence is observed at the insertion site Replace CVCs if the patient is hemodynamically unstable and CRBSI is suspected Do not use guidewire techniques to replace catheters in patients suspected of having catheter-related infection

Strategies for Prevention of CR Infections n Replacement of administration sets q q q Replace administration sets, and add-on devices, no more frequently than at 72 -hour intervals, unless catheter-related infection is suspected Replace tubing used to administer blood, blood products, or lipid emulsions within 24 hours If the solution contains only dextrose and amino acids, the administration set does not need to be replaced more frequently than every 72 hours

Strategies for Prevention of CR Quality Assurance and Continuing Education Infections A subclavian site instead of a jugular or femoral site Hands hygiene, maximal barrier precautions for CVC insertion Changing administration sets at appropriate intervals Remove caths after intended use Chlorhexidine-containing cutaneous antiseptics Transparent, semipermeable polyurethane dressings Antimicrobial/Antiseptic Impregnated Catheters and Cuffs Anticoagulants Antibiotic/Antiseptic Ointments +++ +++ +++ + + +, + + -
- Slides: 22