CatheterBased Closure of Adult ASD and PFO Richard

Catheter-Based Closure of Adult ASD and PFO Richard A. Krasuski, M. D. Director of the Adult Congenital Heart Disease Center Duke University Medical Center

Disclosures Richard A. Krasuski, MD Consultant Actelion Non-paid scientific advisory board member Ventripoint Institutional Primary Investigator Abbott: ASO 522 Actelion: OVATION (Current) Edward Life Sciences: COMPASSION 3 (Current) NMT Medical: CLOSURE-1 Gore: PFO-REDUCE

Overview • Demographics of atrial septal abnormalities • Guideline based management of ASD • Device closure of ASD and PFO – Patient evaluation and preparation – Procedural details – Follow-up and complications
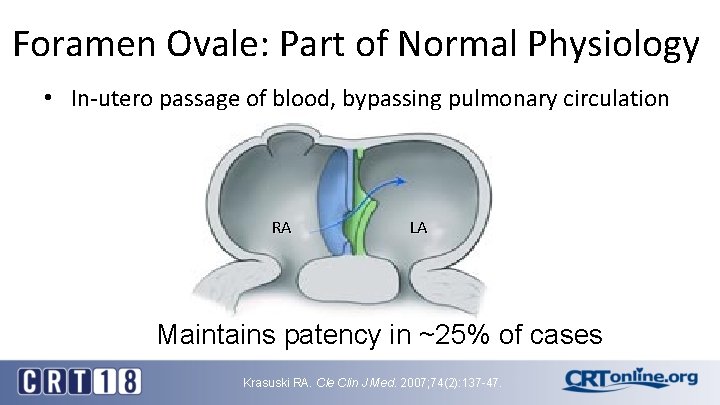
Foramen Ovale: Part of Normal Physiology • In-utero passage of blood, bypassing pulmonary circulation RA LA Maintains patency in ~25% of cases Krasuski RA. Cle Clin J Med. 2007; 74(2): 137 -47.
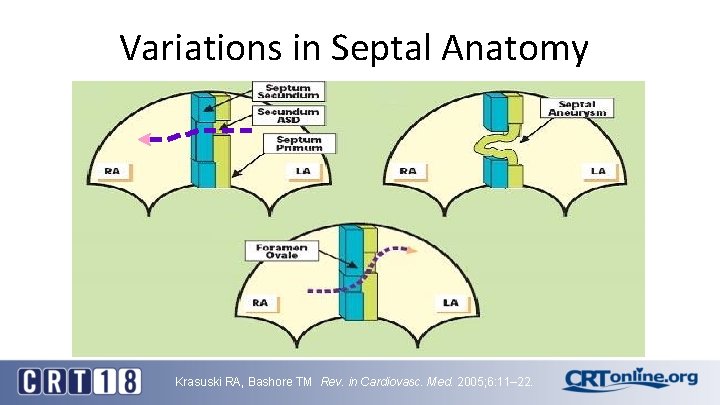
Variations in Septal Anatomy Krasuski RA, Bashore TM Rev. in Cardiovasc. Med. 2005; 6: 11– 22.
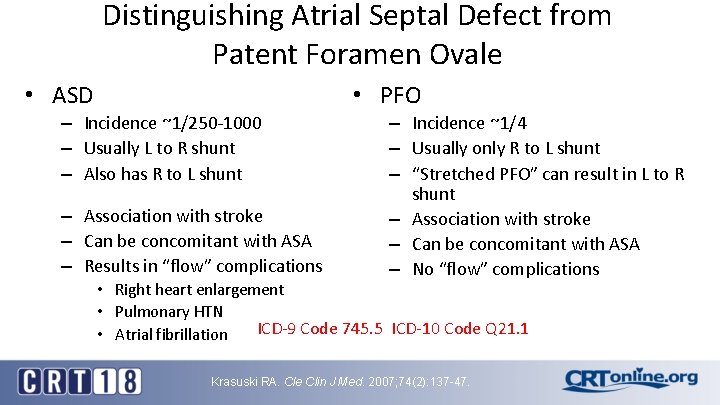
Distinguishing Atrial Septal Defect from Patent Foramen Ovale • PFO • ASD – Incidence ~1/250 -1000 – Usually L to R shunt – Also has R to L shunt – Association with stroke – Can be concomitant with ASA – Results in “flow” complications – Incidence ~1/4 – Usually only R to L shunt – “Stretched PFO” can result in L to R shunt – Association with stroke – Can be concomitant with ASA – No “flow” complications • Right heart enlargement • Pulmonary HTN ICD-9 Code 745. 5 ICD-10 Code Q 21. 1 • Atrial fibrillation Krasuski RA. Cle Clin J Med. 2007; 74(2): 137 -47.
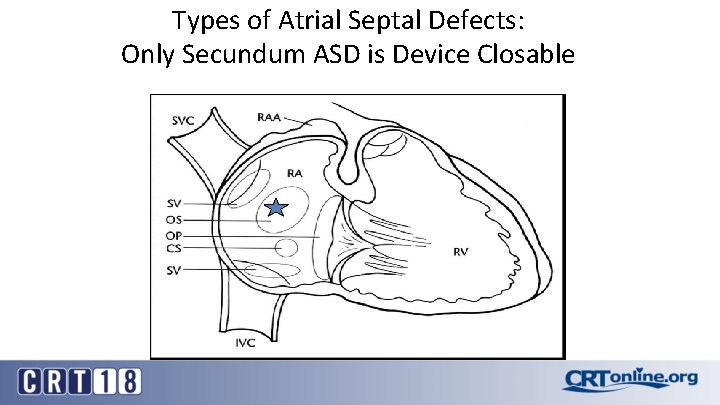
Types of Atrial Septal Defects: Only Secundum ASD is Device Closable

Surgical Closure of Secundum ASD • First attempted in 1939 at Columbia Presbyterian Hospital • Variety of techniques used from 1939 -1953 • May 6, 1953 - first successful open-heart operation performed using heart-lung machine by Dr. John H. Gibbon at Jefferson University Medical Center • Suture approximation (small) or patch repair (large) now standard • Minimally invasive techniques • Operative mortality 1 -3% Moore J et al. J Am Coll Cardiol Intv. 2013; 6(5): 433 -42.
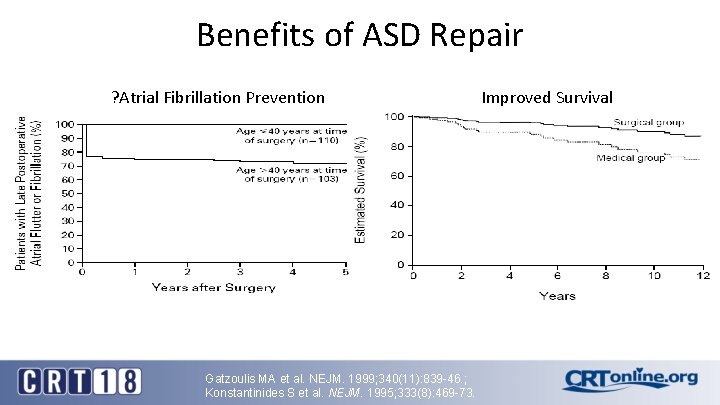
Benefits of ASD Repair ? Atrial Fibrillation Prevention Gatzoulis MA et al. NEJM. 1999; 340(11): 839 -46. ; Konstantinides S et al. NEJM. 1995; 333(8): 469 -73. Improved Survival

Evolution of Devices • King and Mills - 2 umbrella system – 80% success – 23 F system • Rashkind Device – Irretrievable • Constantly evolving series of devices – Amplatzer Septal Occluder™/Amplatzer PFO Occluder™/ Amplatzer Cribriform Device™ (Abbott Medical) – Gore Cardioform Device™ (Gore Medical) Krasuski RA. Cle Clin J Med. 2007; 74(2): 137 -47.

Evolution of ASD Occluders Amplatzer Angel Wing ASDOS Sideris Button Cardio. Seal Helex Star. Flex Guardian Angel Cardioform
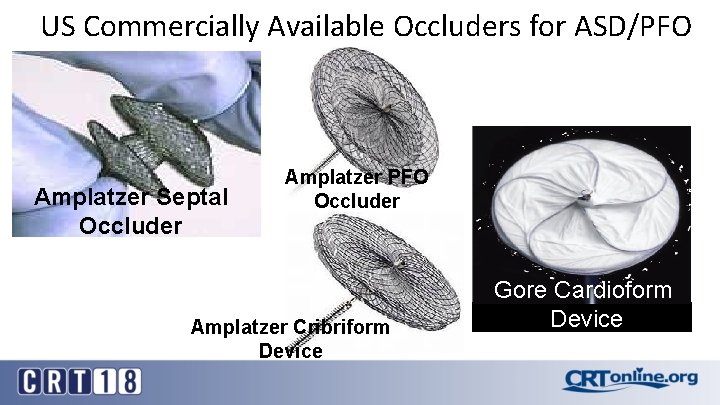
US Commercially Available Occluders for ASD/PFO Amplatzer Septal Occluder Amplatzer PFO Occluder Amplatzer Cribriform Device Gore Cardioform Device

When Should Repair of an ASD be Considered? – ACHD Guidelines Closure of ASD either percutaneously/surgically indicated for: Right atrial and RV enlargement with or without symptoms After paradoxical embolism With documented platypnea-orthodeoxia Warnes CA et al. J Am Coll Cardiol. 2008; 52(23): e 1 -121.

Deciding Between Surgical and Percutaneous Treatment in ASD Surgical closure of secundum ASD is reasonable when concomitant surgical repair/replacement of a tricuspid valve is considered or when the anatomy of the defect precludes the use of a percutaneous device Concomitant Maze procedure may be considered for intermittent or chronic atrial tachyarrhythmias in adults with ASDs Warnes CA et al. J Am Coll Cardiol. 2008; 52(23): e 1 -121. 14

PFO Closure Prior to 2017: Confusion http: //www. fda. gov/Medical. Devices/Productsand. Medical. Procedures accessed 2/20/2017; Messe SR et al. Neurology 2016; 87(8): 815 -21.

Latest Systematic Meta-Analysis of Trials • 5 RCTs of 3630 patients of patients following first cryptogenic stroke • Device closure reduced subsequent stroke risk (HR=0. 3, 95% CI=0. 020. 57) p=0. 034) by ITT analysis • No difference in bleeding or serious adverse events • Significantly increased risk for atrial fibrillation (HR= 4. 8, 95% CI= 2. 210. 7) p<0. 001 Riaz H and Krasuski RA. Am Heart J 2018; 199: 44 -50.

Key for PFO Closure: Ensure Appropriate Patient Selection • Thorough medical work-up is essential – Neurological imaging – Transesophageal echocardiography – Hypercoagability testing • Adequate monitoring to exclude atrial fibrillation – Study examining implantable loop vs. conventional f/u in 441 pts >40 yo with no AF after 24 hours monitoring – Primary endpoint - AF lasting >30 sec – 8. 9/12. 4% of implants vs. 1. 4/2. 0% controls at 6/12 months Sanna T et al. N Engl J Med 2014; 370: 2478 -86.

Selecting Appropriate ASD Patients for Device Closure: Obtaining Critical Medical History • • Palpitations and documented arrhythmias Unexplained neurological symptoms Exertional limitations +/- observed cyanosis History of coagulation abnormalities Tolerance of antiplatelet therapy Allergy history – nickel Exclusion of recent or current infection

Selecting Appropriate ASD Patients for Device Closure: Assessing Anatomy and Physiology • Transthoracic echocardiography to assess concomitant lesions – MVP and mitral regurgitation – Tricuspid regurgitation • Severity • Assessment of right ventricular systolic pressure • Holter or event monitor to assess for arrhythmia • Transesophageal echo or cardiac MRI – Septal Rims – Pulmonary venous return
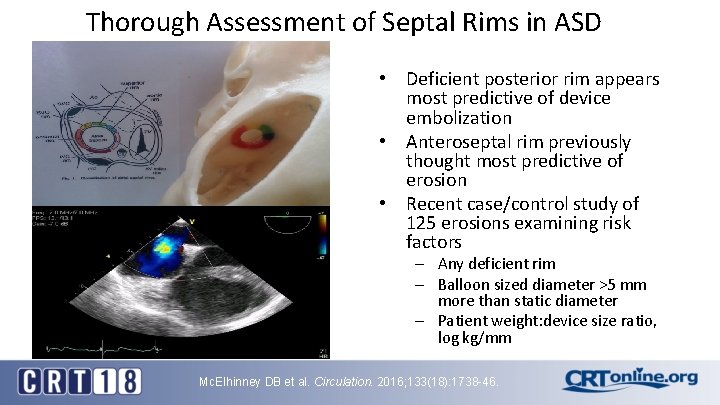
Thorough Assessment of Septal Rims in ASD • Deficient posterior rim appears most predictive of device embolization • Anteroseptal rim previously thought most predictive of erosion • Recent case/control study of 125 erosions examining risk factors – Any deficient rim – Balloon sized diameter >5 mm more than static diameter – Patient weight: device size ratio, log kg/mm Mc. Elhinney DB et al. Circulation. 2016; 133(18): 1738 -46.
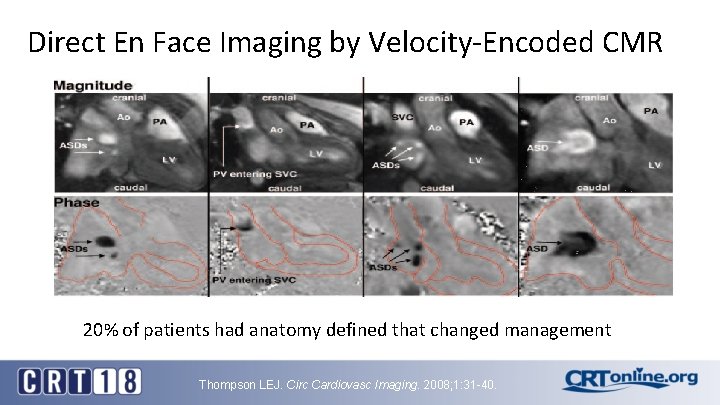
Direct En Face Imaging by Velocity-Encoded CMR 20% of patients had anatomy defined that changed management Thompson LEJ. Circ Cardiovasc Imaging. 2008; 1: 31 -40.

Overview of Closure Procedure • Preparing the Patient – – • • • Proper patient selection + informed consent Decide what to do about anticoagulation Start aspirin +/- clopidogrel Antibiotic on-call to the cath lab Right heart pressure and saturation assessment +/- coronary angio Access the LA via the defect Transesophageal or intracardiac echo assessment of septum +/- Angiography of the defect Compliant balloon to assess defect size and morphology Proper implant and size selection

Choosing Between Available Devices Gore Cardioform Device • • • Nitinol wire frame and PTFE covering with hydrophilic coating Available in 15, 20, 25, 30 mm sizes ~1. 7 x balloon size Fully retrievable even after release Earlier design (Helex) associated with higher risk of residual shunt More flexible and conforming - no documented erosions Amplatzer Septal Occluder • • Nitinol mesh and Dacron fabric 2 discs and central waist • LA disc 12 -16 mm > waist • RA disc 8 -10 mm > waist • • Available 4 -38 mm waists (2 mm increments) - ≤ 2 mm > balloon size Easily retrievable/redeployable until released Highest successful closure rate of all occluder devices Erosions reported in 0. 043 -0. 3%
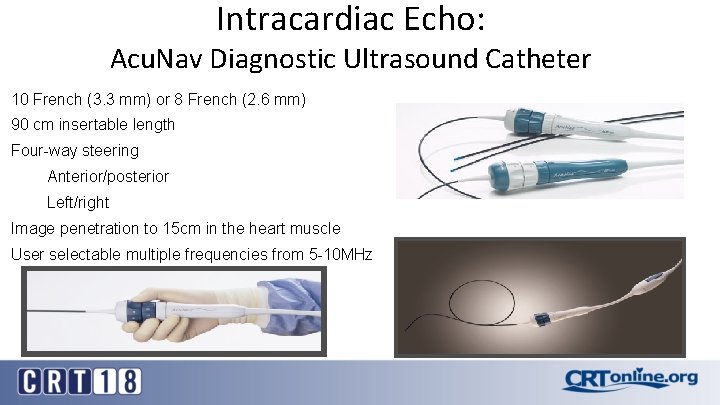
Intracardiac Echo: Acu. Nav Diagnostic Ultrasound Catheter 10 French (3. 3 mm) or 8 French (2. 6 mm) 90 cm insertable length Four-way steering Anterior/posterior Left/right Image penetration to 15 cm in the heart muscle User selectable multiple frequencies from 5 -10 MHz

Overview of Closure Procedure • • • Delivery sheath in LA/upper pulmonary vein Left atrial side opened Device Pulled back to contact left atrial septum Right atrial side opened Device release only after echo and fluoroscopic confirmation of position

Sheath Modification for Larger Defects Vasquez AF and Lasala J. Cardiol Clin 2013; 31: 385– 400.

Postprocedural Follow-up • Admit patient overnight – 1 or 2 more doses of antibiotics – Echocardiogram and chest x-ray prior to d/c • Follow-up office visit and imaging at 1 month, 6 months and 1 year • Follow-up and imaging every 3 -5 years afterwards

Long-term Complications of Closure • Meta-analysis of 28, 142 patients from 203 studies • Included PFO and ASD patients – Complication rates slightly higher in ASD • Major complications – 1. 6% (embolization/malposition surgery, erosion, heart block) • Minor complications – 1. 4% (effusion) • Neurological events (1. 3%) and device thrombosis 1. 2%) most common events on follow-up • Device-related mortality 0. 1% at 2 years – Related to erosions or stroke Abaci A et al. Cath and Cardiovasc Interv. 2013; 82: 1123– 38.

THANK YOU FOR YOUR ATTENTION
- Slides: 29