Catalytic Segregation of Graphene Using Silver Nanoparticles Brandon

Catalytic Segregation of Graphene Using Silver Nanoparticles Brandon R. Reed CHEM 318

Introduction • A fast developing field • Desirable to cut correct shapes • Few techniques currently available • This research illustrates reproducible results. 2

Catalytic Environment • Catalysis occurs at the “step edges” of graphene. • At high temperatures, silver nanoparticles located at these edges are able to channel trenches through the graphene. nanoparticle 3

Catalytic Environment Mechanism of channeling • Particles cling to surface defects • Energetically favorable • Reaction involves oxygen chemisorbed by particle (Silver oxide). • Oxidize atoms of graphene into CO 2. • Follows the receding edge 4

resonate 3 nm graphene fold 5

Ag Nanoparticles-Multiple Methods • Na. BH 4 and Ag. NO 3 on ice • Laser reflection colloidal solution. • No aggregation, citrate buffer: double layer of charge, stabilizes particles and prevents aggregation 6
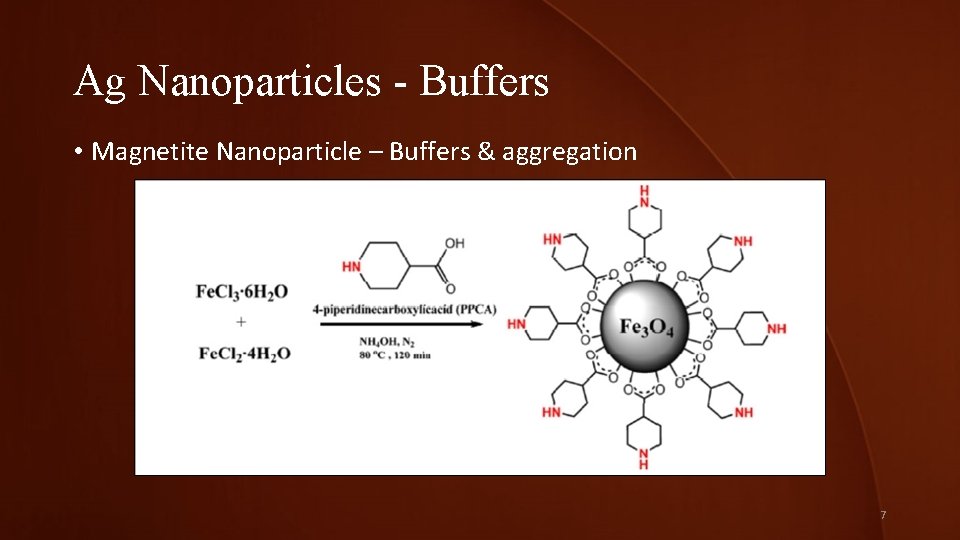
Ag Nanoparticles - Buffers • Magnetite Nanoparticle – Buffers & aggregation 7

Materials & Methods • “highly oriented pyrolytic graphene (HOPG)” • Aqueous Ag. NO 2 vs Ag. NO 3 • High temp (650 °C ). • In heated quartz tube (D = 3 cm). • Imaging Ag particles : Scanning Force Microscopy (SFM) & Scanning Tunneling Microscope (STM). 8
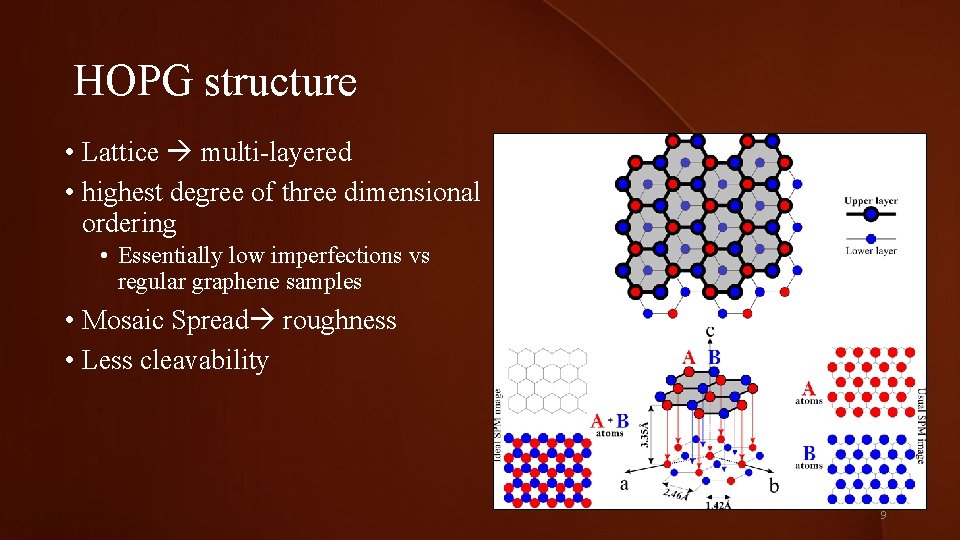
HOPG structure • Lattice multi-layered • highest degree of three dimensional ordering • Essentially low imperfections vs regular graphene samples • Mosaic Spread roughness • Less cleavability 9

Data & Results • SFM surface morphology • SFM images graphene 1 min anneal = no etching. • Cutting speed/trench length correlated to size of the nanoparticles. Properties Findings Trench Length 9. 0 u. M Trench Width 6 -100 nm Max Speed 250 nm s-1 10
11

Trenching Types • Silver nanoparticles stay along the defects or step edges of the graphene. • 3 types of trenches: • Straight segmented trenches • Spiral tranches • Zig zag trenches • Alternate trench formation causes • Pollution of sample Sulfur Dioxide 12
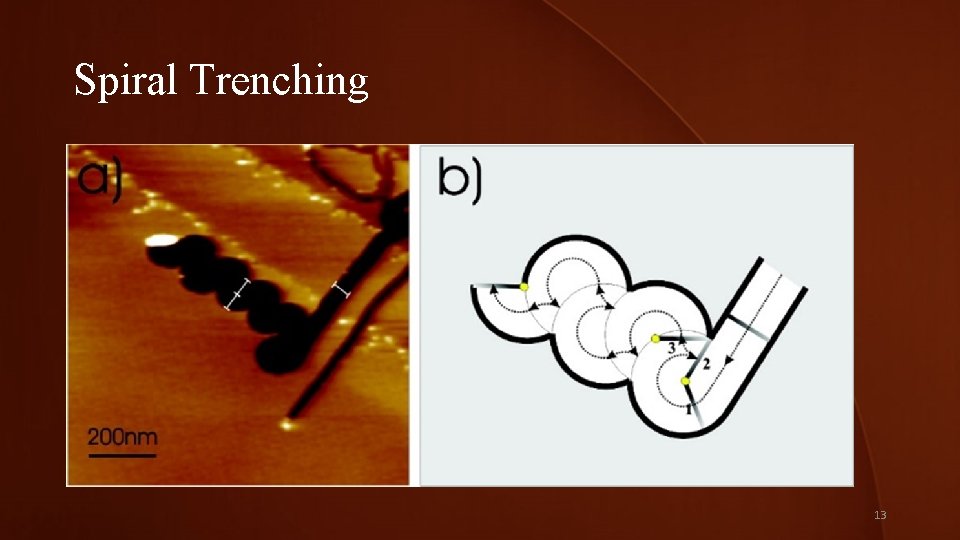
Spiral Trenching 13

Pollution Insight • HOPG covered with silver nitrite in the presence of a small amount of sulfur. 1 -min run. • Annealing of a new sample brief ventilation • Spiral trenching. • Insight poisoning of the catalyst responsible formation of spiral trenches • Further studies needed. 14

Conclusions • Fast channeling of large nanoparticles vs small • Rate-limiting step is the adsorption of molecular oxygen • Larger particles assemble at higher step edges • Max channeling speed 250 nm/s is of importance • Implications: any size particle capable given enough oxygen 15

Future Possibilities? YES! • New applications • High precision lithography on graphenes. • Catalysis applications probe tip. 16

Current Research Developments - kirigami 17

Current Research Developments - kirigami 18
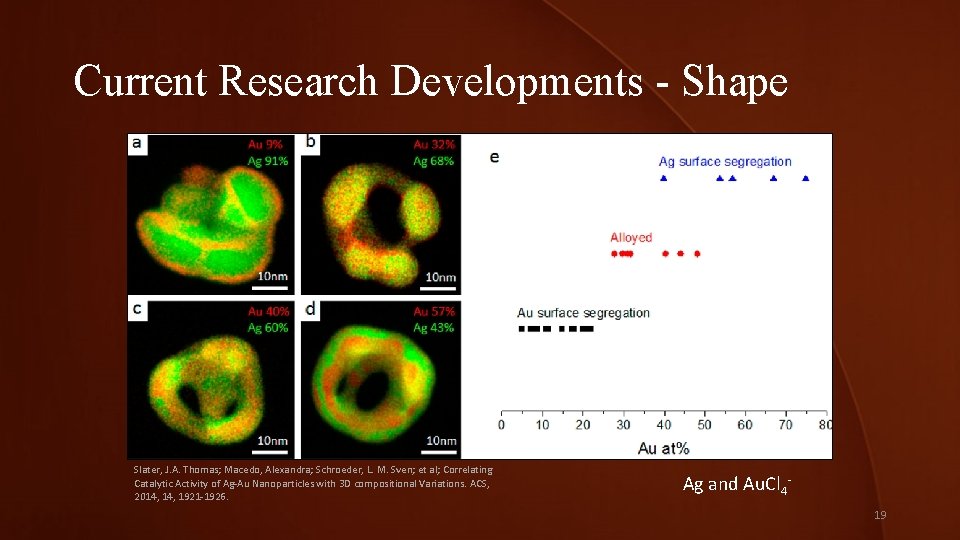
Current Research Developments - Shape Slater, J. A. Thomas; Macedo, Alexandra; Schroeder, L. M. Sven; et al; Correlating Catalytic Activity of Ag-Au Nanoparticles with 3 D compositional Variations. ACS, 2014, 1921 -1926. Ag and Au. Cl 419
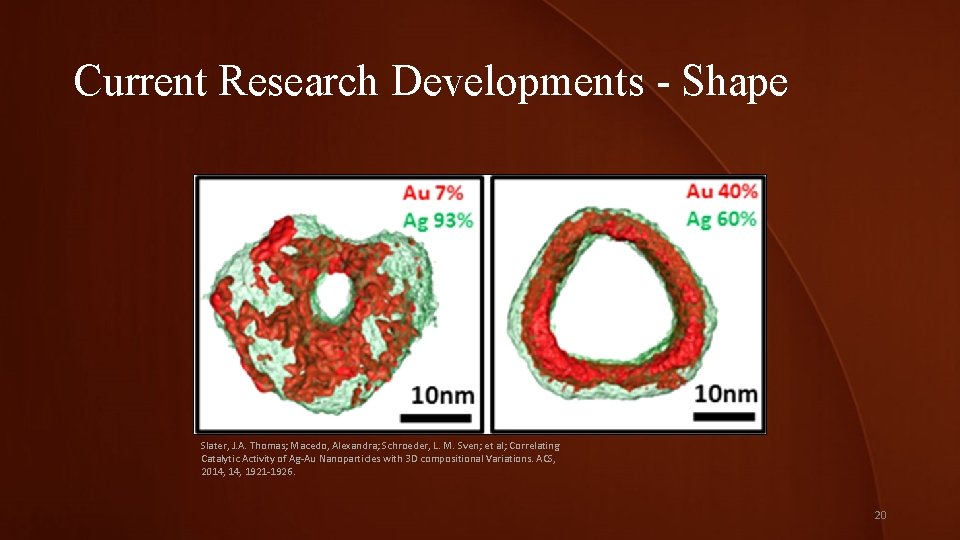
Current Research Developments - Shape Slater, J. A. Thomas; Macedo, Alexandra; Schroeder, L. M. Sven; et al; Correlating Catalytic Activity of Ag-Au Nanoparticles with 3 D compositional Variations. ACS, 2014, 1921 -1926. 20

References 1. 2. 3. 4. 5. 6. 7. Severin, N. ; Kirstein, I. M. Sokolov; Rabe, J. P. ; Rapid Trench Channeling of Graphenes with Catalytic Silver Nanoparticles. 2009, 9, 457 -461. Slater, J. A. ; Macedo, A. ; Schroeder, L. M. Sven; et al; Correlating Catalytic Activity of Ag-Au Nanoparticles with 3 D compositional Variations. ACS, 2014, 1921 -1926. Barberio, M. ; Barone, P. ; Imbrogno, A. ; Xu, Fang. ; CO 2 adsorption on silver nanoparticle/carbon nanotube nanocomposites: A study of adsorption characteristics, J. physica status solidi, 2015, Vol. 252, 9, 1955– 1959. Bahadory, M. S. ; Synthesis of Silver Nanoparticles, Journal of Chemical Education, 2007, 84, 322 -325. Oldenburg, J. S. ; Silver Nanoparticles: Uses and Applications, Sigma Aldrich, Nano. Composix Inc. 2015. HOPG Detailed Description, http: //nanoprobes. aist-nt. com/apps/HOPG%20 info. htm, 2014, accessed: 8 October 2015. Blees, M. ; Nature-Video Research-Graphene Kirigami. http: //blogs. nature. com/aviewfromthebridge/2015/07/29/graphene-structures-at-the-cuttingedge/, 2015, accessed: 14 September 2015. 21

Questions • Additional Notes/ Questions 22

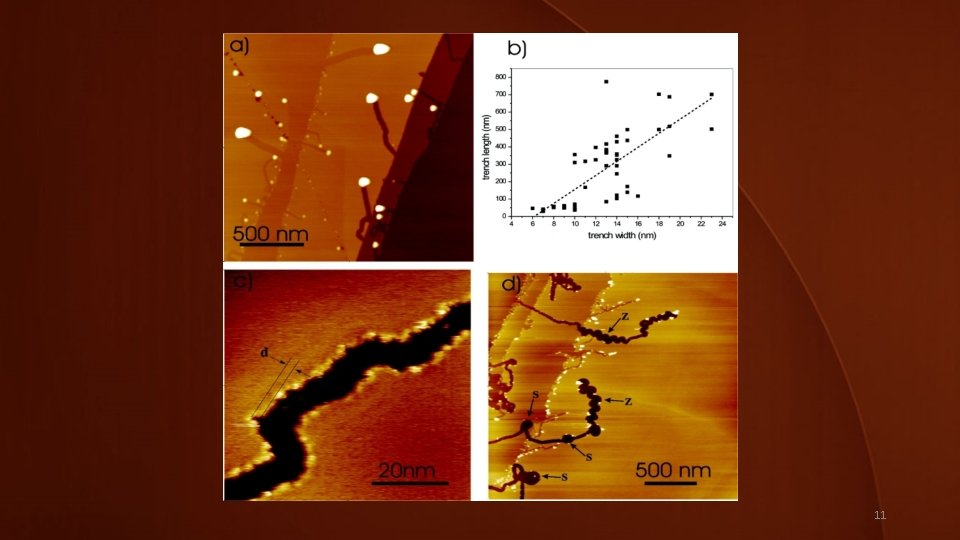
Quad image-notes • (a) SFM height image of a sample annealed for 45 s. The area is chosen to demonstrate that trenches channeled through graphene layers of different thickness. • (b) Dependence of trench length on trench width for trenches formed in graphene bilayer on a sample annealed for 1 min. The dashed line is the linear fit. • (c) STM image of a monolayer trench on a sample annealed for 1 min reveals smooth edges with peak-to-peak roughness below 2 nm. The width of the imaged trench is about 9 nm. • (d) SFM height image of a sample annealed for 1 min, a few examples of spiral (S) and zigzag (Z) trenches are outlined. Notice that particles can switch back and forth between spiral or zigzag and straight channeling. 23

Sulfur Dioxide Pollution-Notes To propve pollution of catalyst, team annealed HOPG covered with silver nitrite in the presence of a small amount of sulfur. SFM imaging of results showed inhibition of channeling: the trenches were no longer than 50 nm (not shown). Annealing of a new sample after a brief ventilation produced mostly spiral type of trenches, which proves the poisoning of the catalyst to be responsible for the formation of spiral trenches. Thus these experiments give a nanoscopic insight into the poisoning mechanism of catalysts. Quantitative characterization of catalyst poisoning will require further investigations. NOT DETERMINITIVE 24

25
- Slides: 25