Catalytic Converter NO NO OO N O NO


- Slides: 8

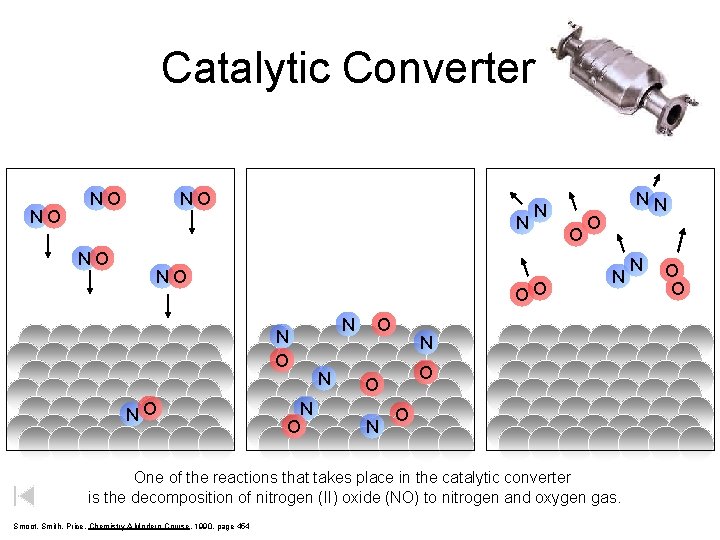
Catalytic Converter NO NO OO N O NO N N O O NN O One of the reactions that takes place in the catalytic converter is the decomposition of nitrogen (II) oxide (NO) to nitrogen and oxygen gas. Smoot, Smith, Price, Chemistry A Modern Course, 1990, page 454 N O O

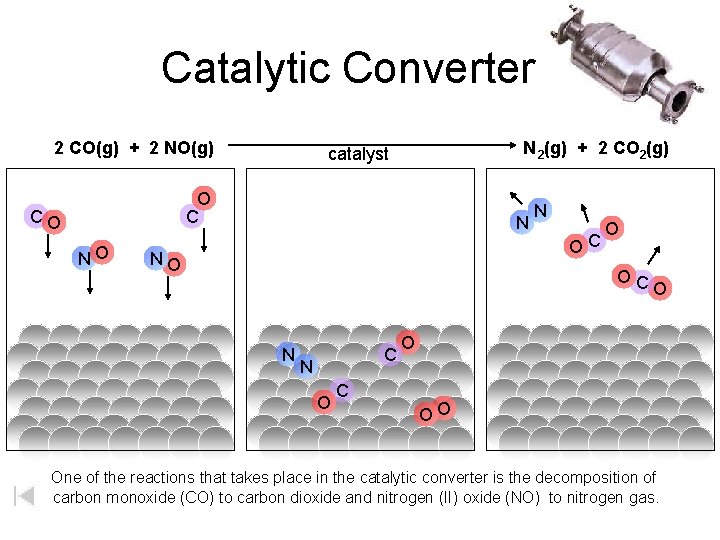
Catalytic Converter 2 CO(g) + 2 NO(g) N 2(g) + 2 CO 2(g) catalyst O C CO NO N OC O OC N O C O O OO One of the reactions that takes place in the catalytic converter is the decomposition of carbon monoxide (CO) to carbon dioxide and nitrogen (II) oxide (NO) to nitrogen gas.

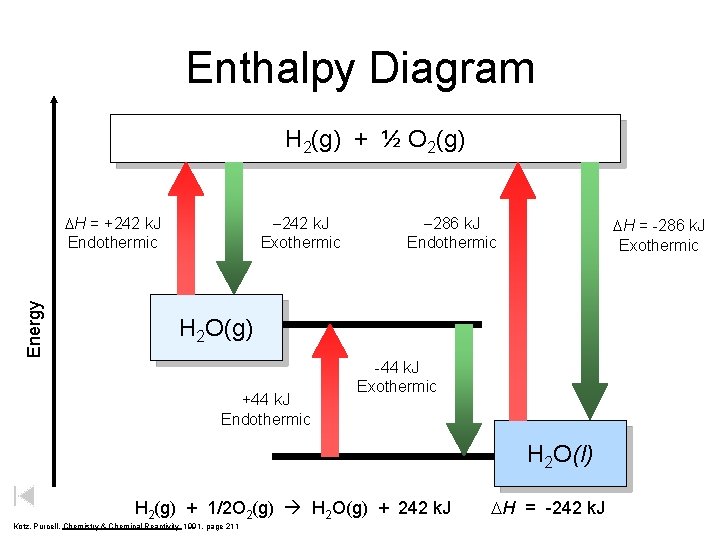
Enthalpy Diagram H 2(g) + ½ O 2(g) Energy DH = +242 k. J Endothermic -242 k. J Exothermic -286 k. J Endothermic DH = -286 k. J Exothermic H 2 O(g) +44 k. J Endothermic -44 k. J Exothermic H 2 O(l) H 2(g) + 1/2 O 2(g) H 2 O(g) + 242 k. J Kotz, Purcell, Chemistry & Chemical Reactivity 1991, page 211 DH = -242 k. J

Hess’s Law Calculate the enthalpy of formation of carbon dioxide from its elements. C(g) + 2 O(g) CO 2(g) Use the following data: 2 O(g) O 22(g) C(g) C(s)2(g) + O 2(g) CO CO C(s) + O 2(g) DH DH DH C(g) + 2 O(g) CO 2(g) DH = -1360 k. J Smith, Smoot, Himes, pg 141 = = = - 250 k. J - 720 k. J +720 - 390 k. J +390

In football, as in Hess's law, only the initial and final conditions matter. A team that gains 10 yards on a pass play but has a five-yard penalty, has the same net gain as the team that gained only 5 yards. 10 yard pass 5 yard net gain 5 yard penalty initial position of ball final position of ball

Conversion Factors Conversions http: //www. unit 5. org/christjs/temp T 27 d. Fields-Jeff/Matter 1. htm

Atoms, Mass, and the Mole key http: //www. unit 5. org/christjs/temp T 27 d. Fields-Jeff/Matter 1. htm

Mole Pattern http: //www. unit 5. org/christjs/temp T 27 d. Fields-Jeff/Matter 1. htm