Catalyst What happens when you change the number

Catalyst • What happens when you change the number of electrons in an atom? • What happens when you change the number of protons in an atom? • What happens when you change the number of neutrons in an atom? End


LESSON 2. 5 – NUCLEAR CHEMISTRY: FISSION, FUSION, and RADIOACTIVITY

Today’s Learning Targets • LT 2. 11 – I can explain the forces that exist within the nucleus of an atom. I can compare the strength of the nuclear force to other (electromagnetic, gravity, etc) forces that exist. • LT 2. 12 – I can explain why energy is released in a nuclear fusion/fission reaction and I can calculate the amount of energy released.

Today’s Focus Question • What is nuclear energy?

What are forces?

I. Forces • A force is a push or pull • 4 fundamental forces in the universe 1. Gravity 2. Coulombic Force 3. Strong Nuclear Force 4. Weak Nuclear Force

What are the fundamental forces?

I. We Have a Problem • The nucleus of an atom contains many positive changes (protons) • If the nucleus contains a bunch of positive charges, why doesn’t the nucleus of the atom split apart?

II. Strong Nuclear Force • The strong nuclear force is the force that exists between particles in the nucleus of an atom • Much stronger then coulombic forces, so the attraction between particles in nucleus is much stronger then the proton repulsion

III. Weak Nuclear Forces • The weak nuclear force is the force that allows certain elements to become radioactive.

Strength of the Forces Strong Nuclear Force Strongest Force Electromagnetic Force Weak Nuclear Force Gravitational Force Weakest Force

SUMMARIZE Use the following terms in your summary: • Force • Gravitational • Electromagnetic • Strong • Weak

What are nuclear fusion and fission?

I. Nuclear Fusion • Fusion – When two things combine into one • Nuclear fusion is when two atomic nuclei fuse into one new, HEAVIER, atom
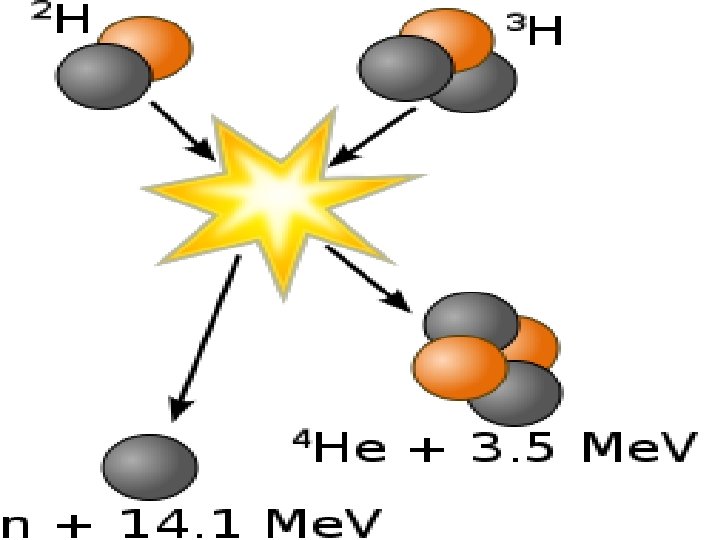

II. Nuclear Fission • Fission – Splitting of an item into two • Nuclear fission is when a very heavy nucleus splits into two smaller, more stable nucleuses
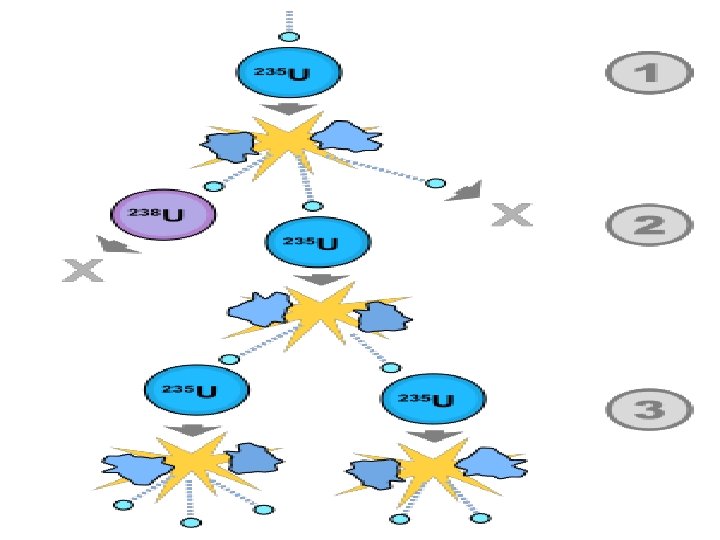

SUMMARIZE Use the following terms in your summary: • Fission • Fusion • Strong Nuclear Force

LESSON 2. 5 – ISOTOPES AND RADIOACTIVE DECAY

Today’s Learning Targets • LT 2. 13 – I can compare and contrast normal isotopes and radioactive isotopes. I can discuss the three forms of decay that radioactive isotopes can undergo and how the nucleus is altered in each type of decay. • LT 2. 14 – I can list the three different type of radiation and the impact that each one of these types of decay can have on myself and other living matter

What are isotopes?

I. Isotopes • Isotope – Atoms of the same element have different numbers of neutrons and thus have different atomic masses.




What is radioactive decay?

I. Radioactive Decay • Atoms can be unstable and breakdown • Radioactive Decay – The spontaneous breakdown of the nucleus into a smaller nucleus, accompanied by the release of particles, radiation, or both.

What are the 3 types of radioactive decay?

I. Alpha (α) Decay • Alpha Decay – When an atom splits nucleus into: 1. A smaller nucleus and 2. A Helium atom (2 protons, 2 neutrons). • Observed often in very heavy atoms • Alpha particles on travel a few cm in air • Paper blocks alpha particles
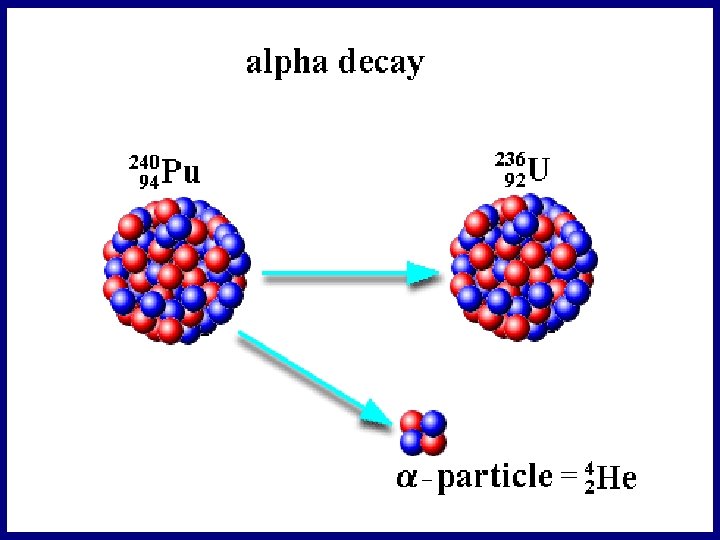
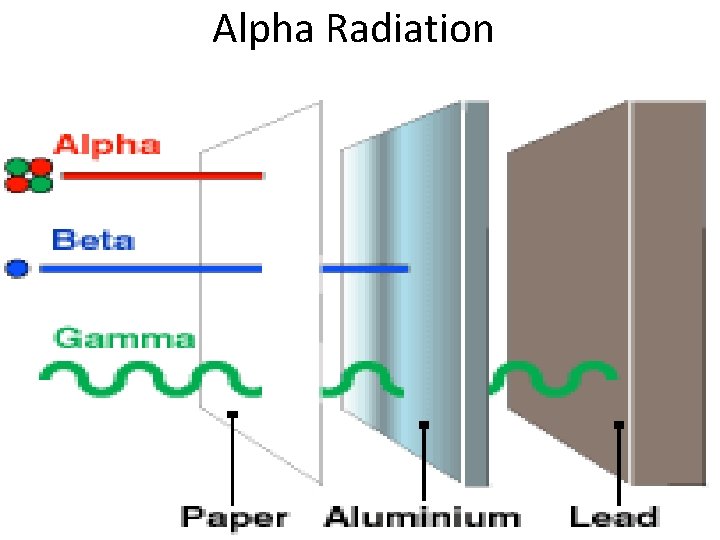
Alpha Radiation

II. Beta (β) Decay • Some elements are unstable because their proton to neutron ratio is too large. • Beta decay converts neutrons to protons and releases an electron known as a beta particle neutron proton + β • Travel a few meters in the air Electron • Metals provide a shield against beta radiation.
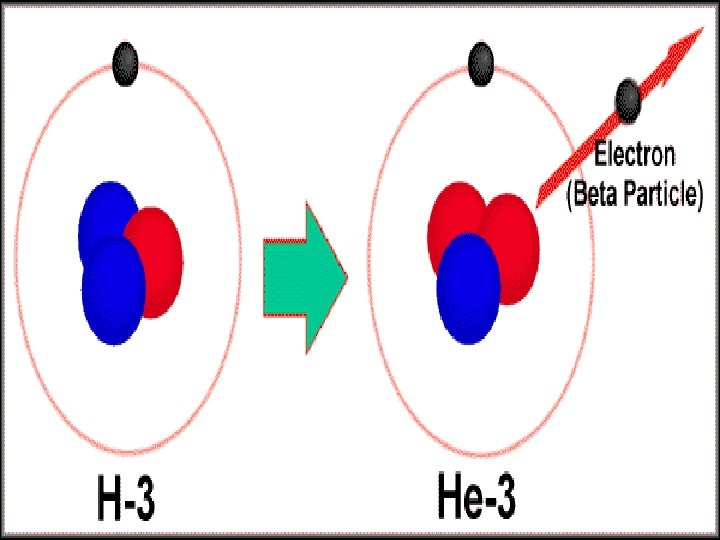
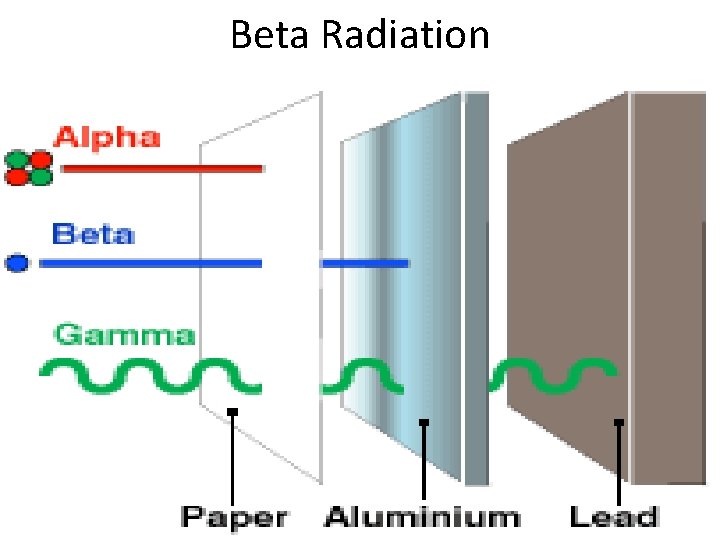
Beta Radiation

III. Gamma (γ) Decay • Gamma rays are high energy waves that are released when nucleus goes from high energy state to low energy. • Gamma radiation is the most dangerous. It can lead to DNA mutations and cancers. • Gamma radiation requires thick pieces of lead/concrete in order to stop.

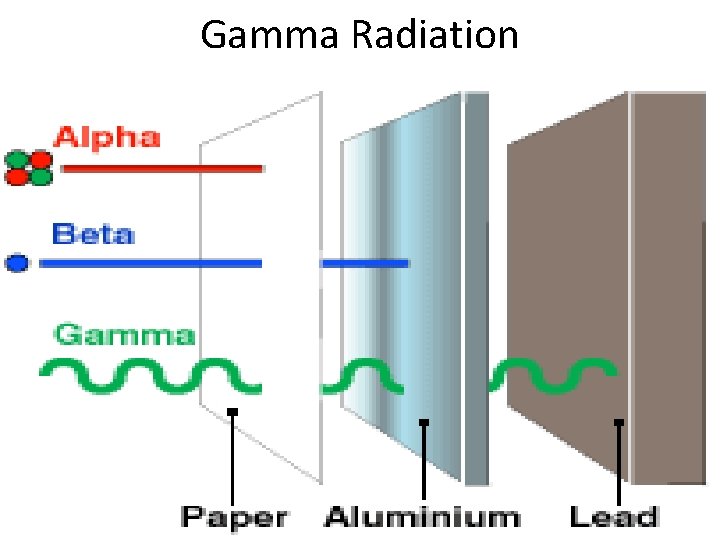
Gamma Radiation
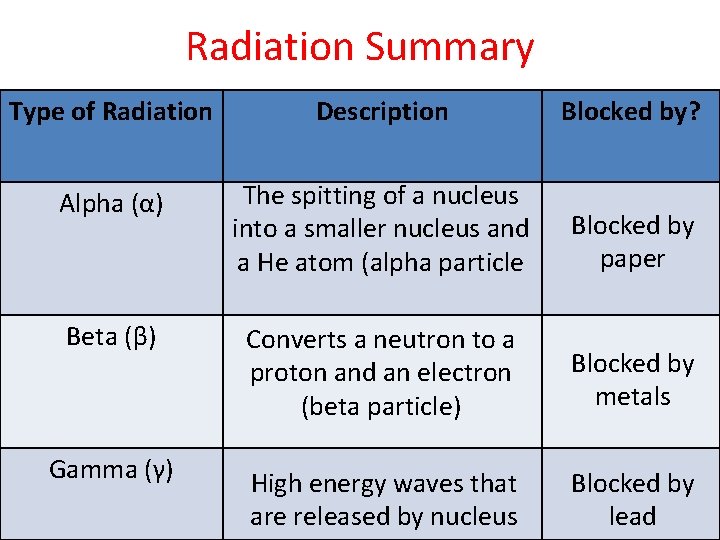
Radiation Summary Type of Radiation Description Blocked by? Alpha (α) The spitting of a nucleus into a smaller nucleus and a He atom (alpha particle Blocked by paper Beta (β) Converts a neutron to a proton and an electron (beta particle) Blocked by metals Gamma (γ) High energy waves that are released by nucleus Blocked by lead

Class Example • Label the following decay as alpha, beta or gamma decay 43 K 43 Ca + electron 19 20
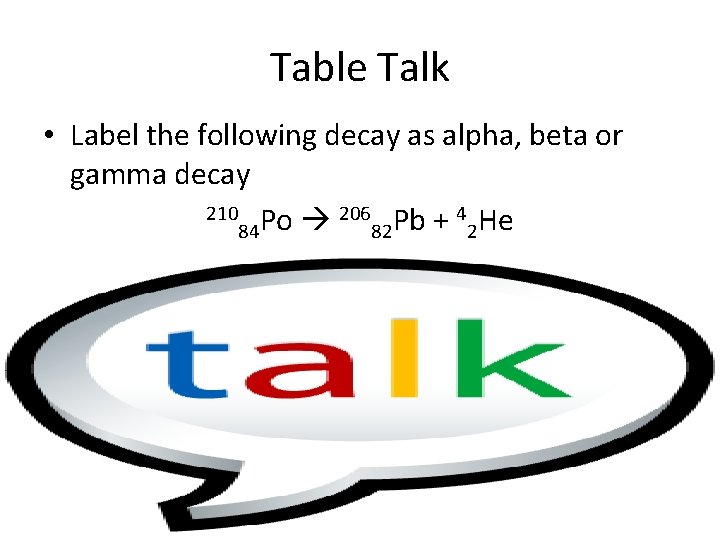
Table Talk • Label the following decay as alpha, beta or gamma decay 210 Po 206 Pb + 4 He 84 82 2

Table Talk • Complete the following reaction if it undergoes beta decay: 14 N ______ + _______ 7

SUMMARIZE Use the following terms in your summary: • Alpha • Beta • Gamma • Decay • Radioactive

Work Time • Work on Practice for the next 10 minutes.
Online Simulator • Please open the two simulators on the computer and complete the simulator handout with a partner.
Learning Target Log Assessment • Rate your current level of understanding of LT 9. 3 and 9. 4. Rate yourself 1 – 4.

Exit Slip 1. What is fusion? 2. What is gamma decay? What will stop gamma radiation? 3. What type of decay is shown below: 231 Pa 227 Ac + 4 He 91 89 2

Exit Ticket 1. What is a force? 2. How does the strong nuclear force allow for the nucleus of an atom to remain together? 3. Rank the 4 fundamental forces from weakest to strongest.
Learning Target Log Assessment • Rate your current level of understanding of LT 9. 3 and 9. 4. Rate yourself 1 – 4.



Closing Time • Homework
- Slides: 53