Catalyst Characterization by Temperature Programmed Methods temperature programmed

Catalyst Characterization by Temperature Programmed Methods temperature programmed Thermoanalytical techniques measurement of a physical or chemical property of a solid as the temperature of the solid is varied in a predetermined manner
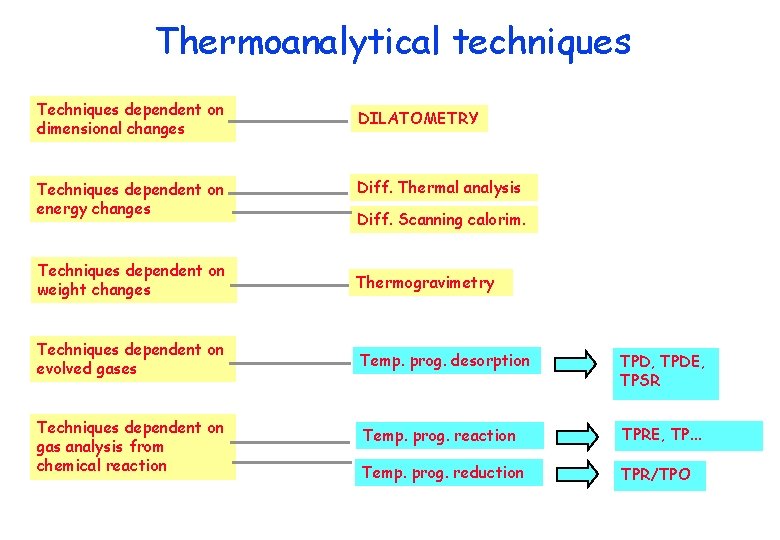
Thermoanalytical techniques Techniques dependent on dimensional changes Techniques dependent on energy changes DILATOMETRY Diff. Thermal analysis Diff. Scanning calorim. Techniques dependent on weight changes Thermogravimetry Techniques dependent on evolved gases Temp. prog. desorption TPD, TPDE, TPSR Temp. prog. reaction TPRE, TP. . . Temp. prog. reduction TPR/TPO Techniques dependent on gas analysis from chemical reaction
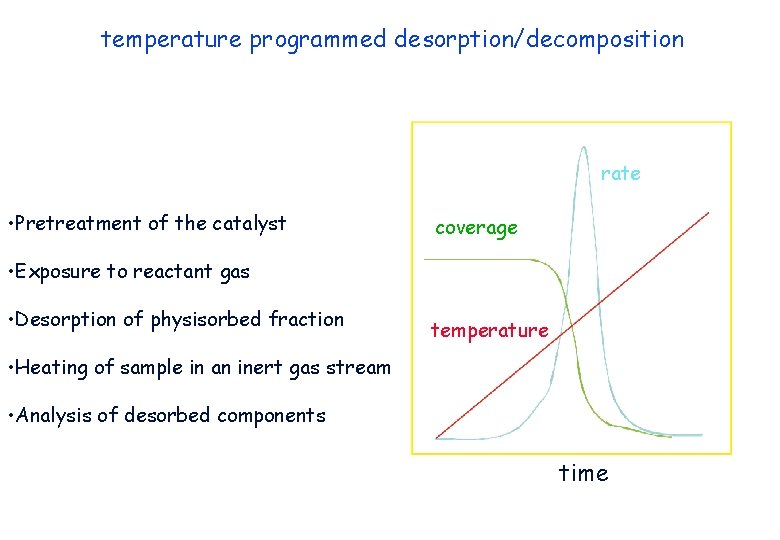
temperature programmed desorption/decomposition rate • Pretreatment of the catalyst coverage • Exposure to reactant gas • Desorption of physisorbed fraction temperature • Heating of sample in an inert gas stream • Analysis of desorbed components time
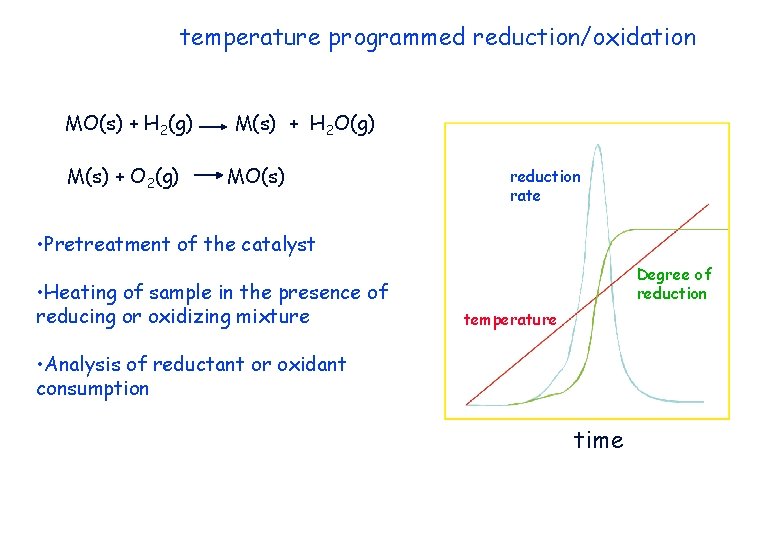
temperature programmed reduction/oxidation MO(s) + H 2(g) M(s) + O 2(g) M(s) + H 2 O(g) MO(s) reduction rate • Pretreatment of the catalyst • Heating of sample in the presence of reducing or oxidizing mixture Degree of reduction temperature • Analysis of reductant or oxidant consumption time

temperature programmed reaction Coadsorption of two gases and heating in inert carrier Adsorption of one component and heating in reactive carrier gas Heating in reactive atmosphere containing reagents Temperature programmed methanation, hydrogenation, sulphidation, combustion…….
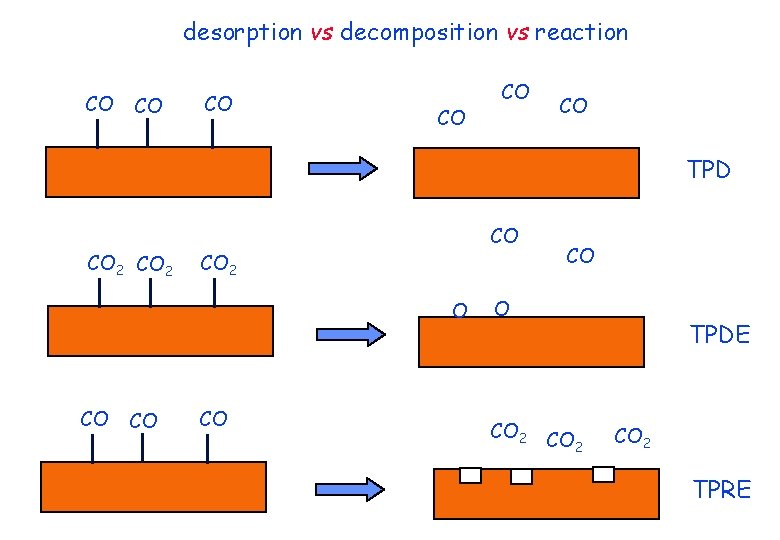
desorption vs decomposition vs reaction CO CO CO TPD CO 2 CO CO 2 O CO CO 2 TPDE CO 2 TPRE

information that can be obtained… • Characterization of reducibility of catalysts • Determination of binding energy of adsorbed molecules • Acidity • Kinetic of catalytic reaction (combustion, oxidation, methanation…. ) • Characterization of surface carbon deposits • Physical parameters (surface area, dispersion…)
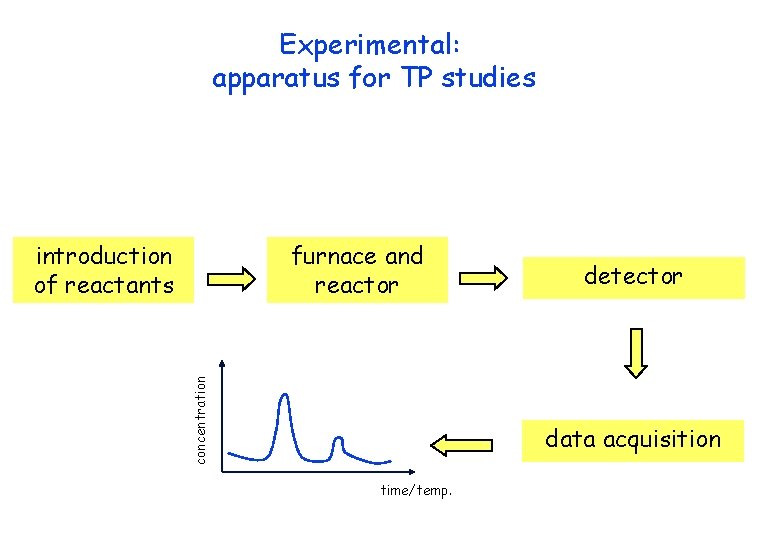
Experimental: apparatus for TP studies furnace and reactor concentration introduction of reactants detector data acquisition time/temp.
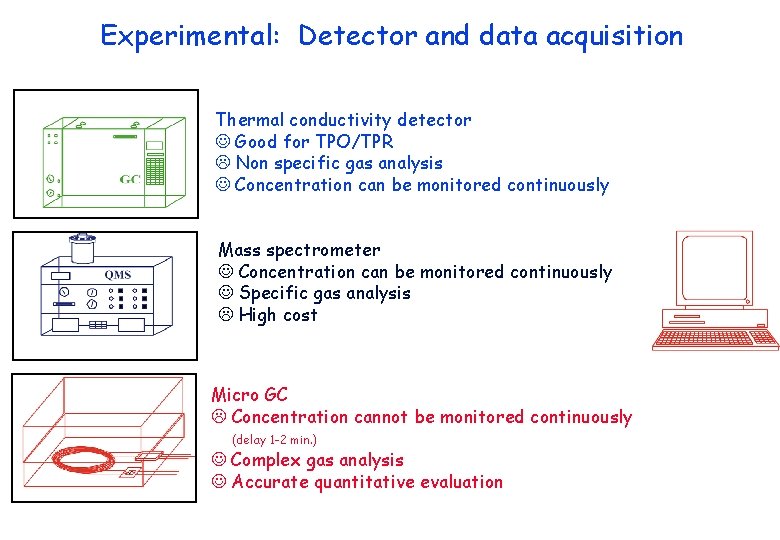
Experimental: Detector and data acquisition Thermal conductivity detector Good for TPO/TPR Non specific gas analysis Concentration can be monitored continuously Mass spectrometer Concentration can be monitored continuously Specific gas analysis High cost Micro GC Concentration cannot be monitored continuously (delay 1 -2 min. ) Complex gas analysis Accurate quantitative evaluation

Experimental: 5. Practical consideration Gas flow rate Sample mass Sample particle size Geometry of the bed Heating rate Leaks and carrier-gas impurities smaller particles decrease the High enaugh torates avoid time delay Because of large carrier gas flow Too thin layer results in an irregular Mass of sample should be kept to a High heating result in better possibility of diffusional between desorption/ reaction and rate relative to intraparticle the quantity bed minimum to avoid backpressure defined peaks, less time per of run and limitations and allow in better detection adsorbed gas, extreme care must problems and temperature less time for changes flowgradients rate and thermocouple contact Too deep results in back pressure and baseline be taken in gas purification within the bed flow changes Low flow rate might cause pressure small particles can create Programmer must be able to maintain a diffusion problems Probably best arrangement is to have drop orprofile, even fall the rates reactor linear toothrough high heating roughly equal depth and width support can result in diffusional limitations

TPD-title Temperature Programmed Desorption Determining the strength of an adsorbate bond to the surface.
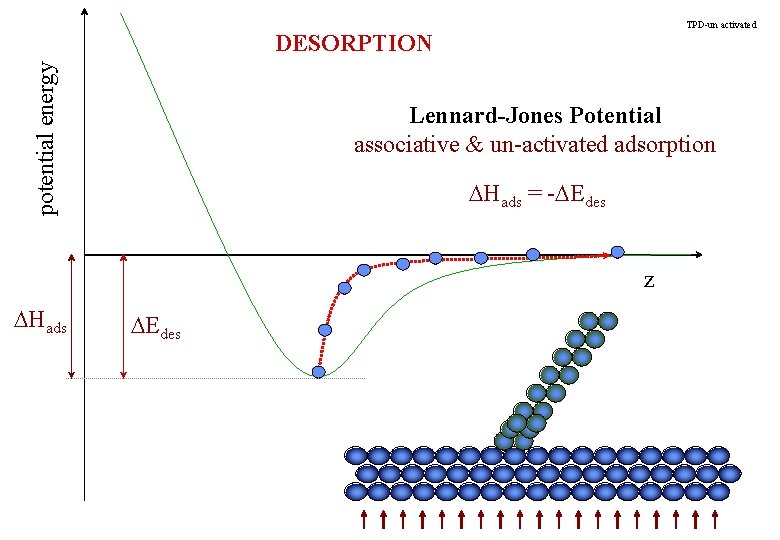
TPD-un activated potential energy DESORPTION Lennard-Jones Potential associative & un-activated adsorption DHads = -DEdes z DHads DEdes
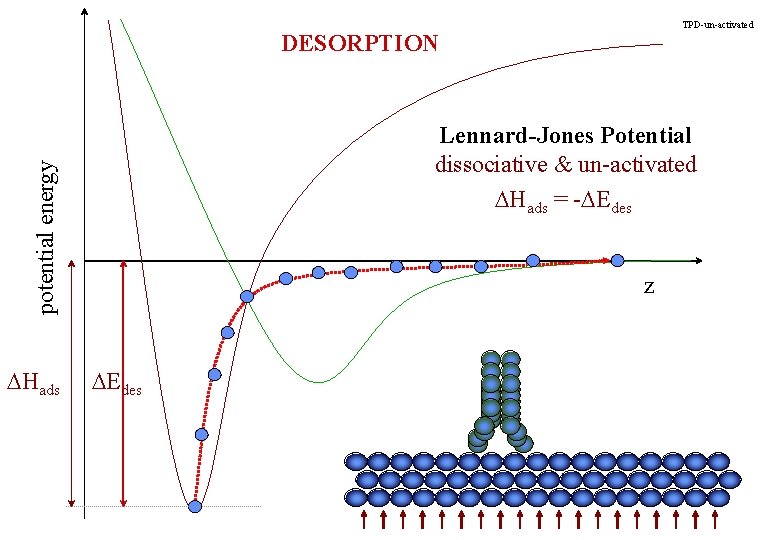
TPD-un-activated DESORPTION potential energy Lennard-Jones Potential dissociative & un-activated DHads = -DEdes DHads z DEdes
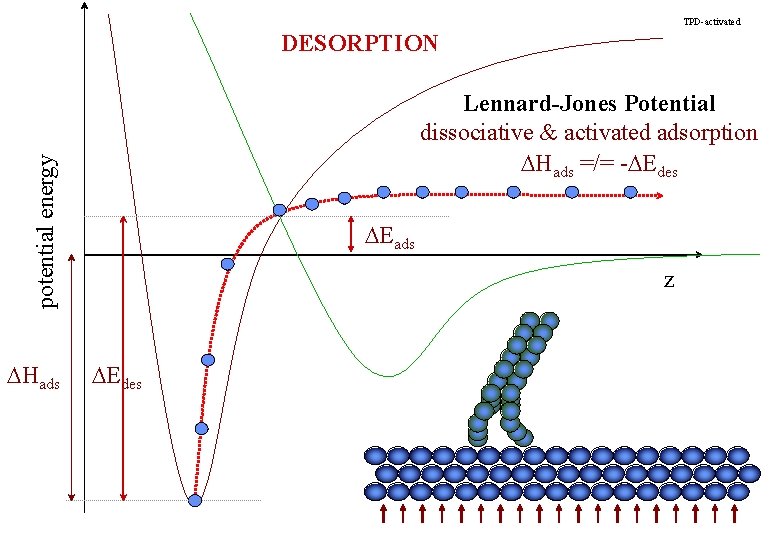
TPD-activated DESORPTION potential energy Lennard-Jones Potential dissociative & activated adsorption DHads =/= -DEdes DHads DEads z DEdes
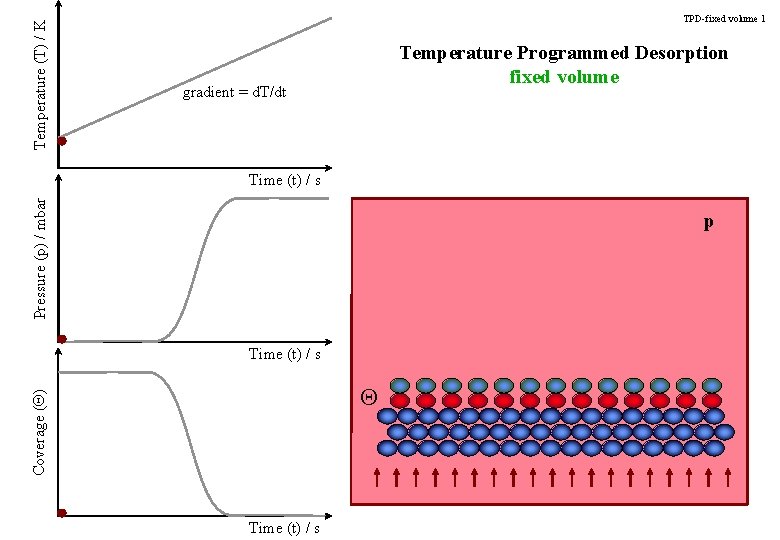
Temperature (T) / K TPD-fixed volume 1 Temperature Programmed Desorption fixed volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Time (t) / s Coverage (Q) Q Time (t) / s
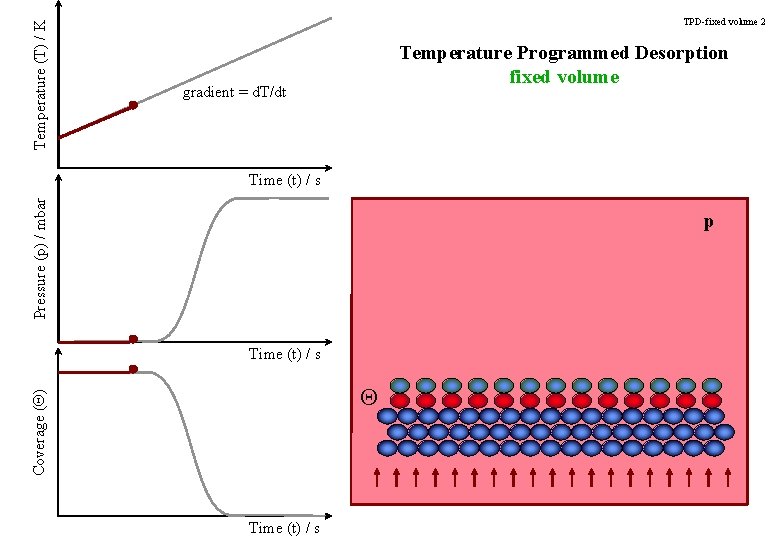
Temperature (T) / K TPD-fixed volume 2 Temperature Programmed Desorption fixed volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Time (t) / s Coverage (Q) Q Time (t) / s
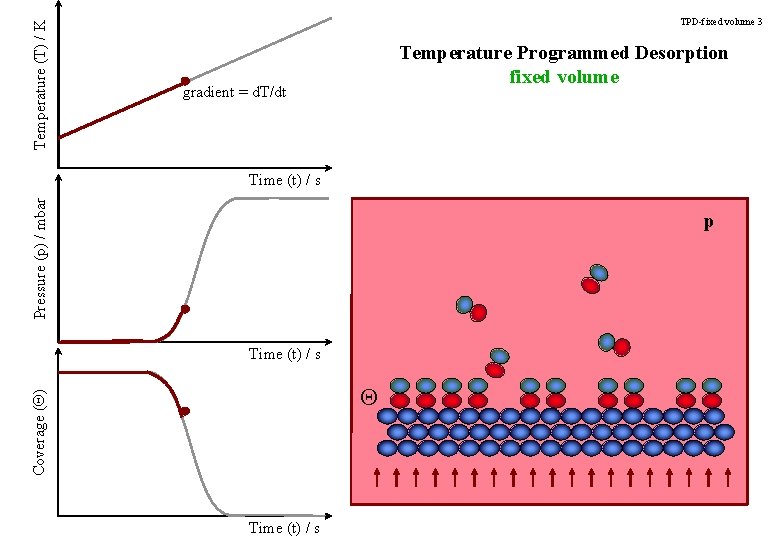
Temperature (T) / K TPD-fixed volume 3 Temperature Programmed Desorption fixed volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Time (t) / s Coverage (Q) Q Time (t) / s
Temperature (T) / K TPD-fixed volume 4 Temperature Programmed Desorption fixed volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Time (t) / s Coverage (Q) Q Time (t) / s
Temperature (T) / K TPD-fixed volume 5 Temperature Programmed Desorption fixed volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Time (t) / s Coverage (Q) Q Time (t) / s
Temperature (T) / K TPD-pumped volume 1 Temperature Programmed Desorption pumped volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Coverage (Q) Q Time (t) / s Q -d. Q/dt Time (t) / s
Temperature (T) / K TPD-pumped volume 2 Temperature Programmed Desorption pumped volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Coverage (Q) Q Time (t) / s Q -d. Q/dt Time (t) / s
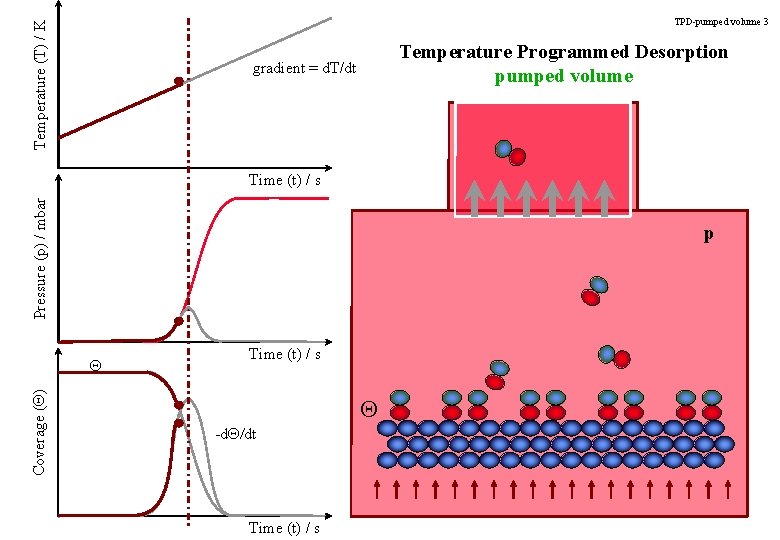
Temperature (T) / K TPD-pumped volume 3 Temperature Programmed Desorption pumped volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Coverage (Q) Q Time (t) / s Q -d. Q/dt Time (t) / s
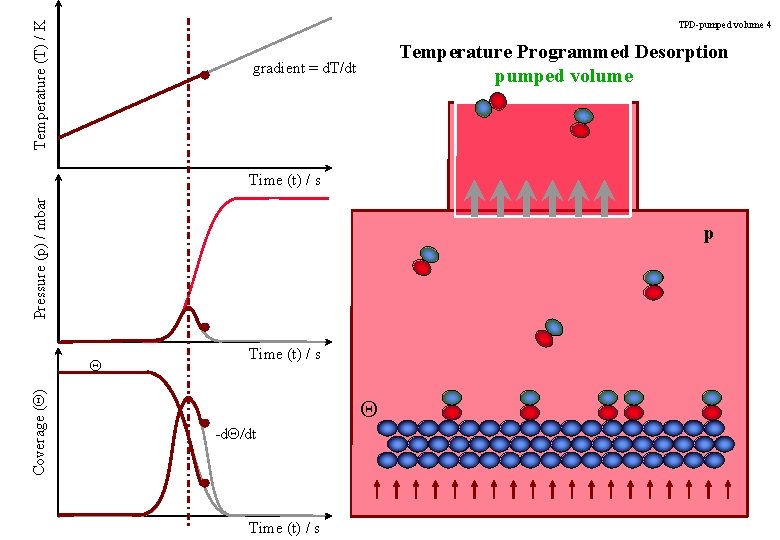
Temperature (T) / K TPD-pumped volume 4 Temperature Programmed Desorption pumped volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Coverage (Q) Q Time (t) / s Q -d. Q/dt Time (t) / s
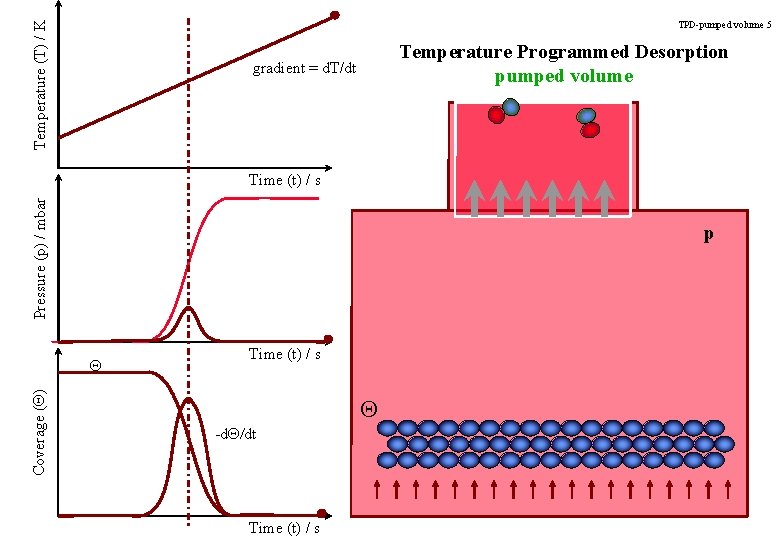
Temperature (T) / K TPD-pumped volume 5 Temperature Programmed Desorption pumped volume gradient = d. T/dt Pressure (p) / mbar Time (t) / s p Coverage (Q) Q Time (t) / s Q -d. Q/dt Time (t) / s
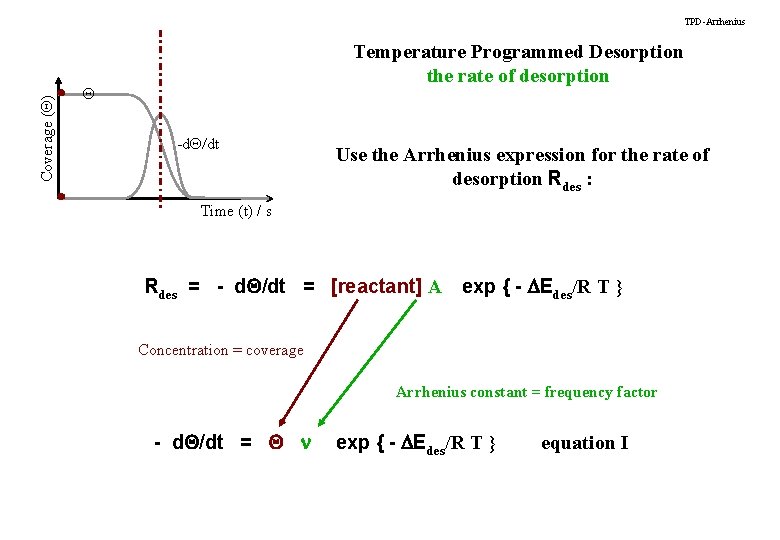
Coverage (Q) TPD-Arrhenius Temperature Programmed Desorption the rate of desorption Q -d. Q/dt Use the Arrhenius expression for the rate of desorption Rdes : Time (t) / s Rdes = - d. Q/dt = [reactant] A exp { - DEdes/R T } Concentration = coverage Arrhenius constant = frequency factor - d. Q/dt = Q n exp { - DEdes/R T } equation I
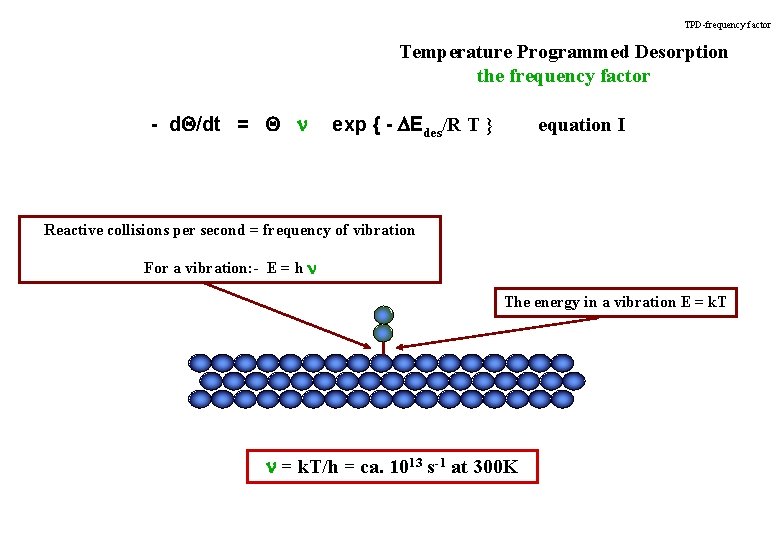
TPD-frequency factor Temperature Programmed Desorption the frequency factor - d. Q/dt = Q n exp { - DEdes/R T } equation I Reactive collisions per second = frequency of vibration For a vibration: - E = h n The energy in a vibration E = k. T n = k. T/h = ca. 1013 s-1 at 300 K
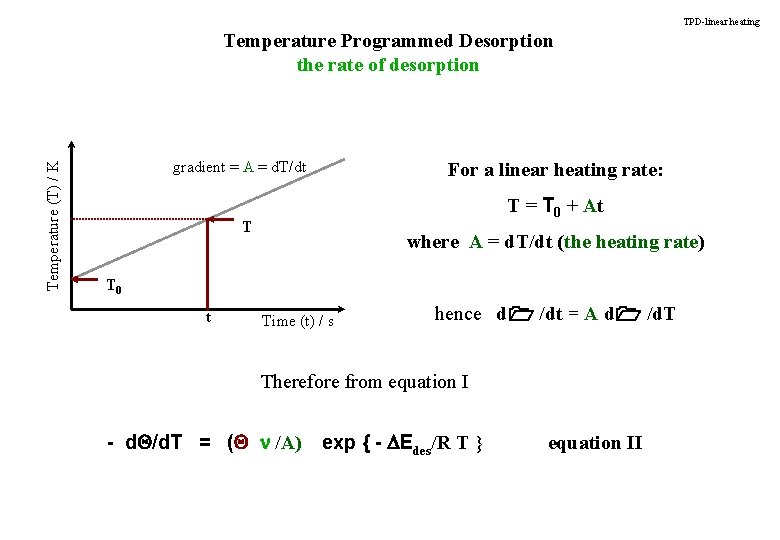
TPD-linear heating Temperature (T) / K Temperature Programmed Desorption the rate of desorption gradient = A = d. T/dt For a linear heating rate: T = T 0 + At T where A = d. T/dt (the heating rate) T 0 t Time (t) / s hence d 1 /dt = A d 1 /d. T Therefore from equation I - d. Q/d. T = (Q n /A) exp { - DEdes/R T } equation II
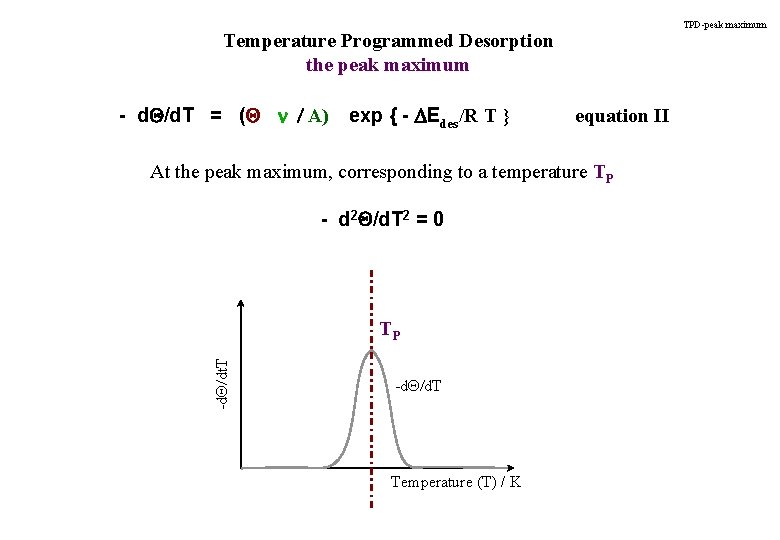
TPD-peak maximum Temperature Programmed Desorption the peak maximum - d. Q/d. T = (Q n / A) exp { - DEdes/R T } equation II At the peak maximum, corresponding to a temperature TP - d 2 Q/d. T 2 = 0 -d. Q/dt. T TP -d. Q/d. T Temperature (T) / K

TPD-differentiation Temperature Programmed Desorption differentiation Differentiate - d. Q/d. T with respect to T to obtain - d 2 Q/d. T 2 Using d(U*V)/d. T = U d. V/d. T + V d. U/d. T - d. Q/d. T = (n /A) constant { Q exp { - DEdes/R T } } - d 2 Q/d. T 2 = 0 = { n /A (Q DEdes/R T 2 ) exp { - DEdes/R T } + d. Q /d. T exp { - DEdes/R T } Therefore since at T=TP, - d 2 Q/d. T 2 = 0 : d. Q /d. T = Q DEdes/R TP 2 equation III }
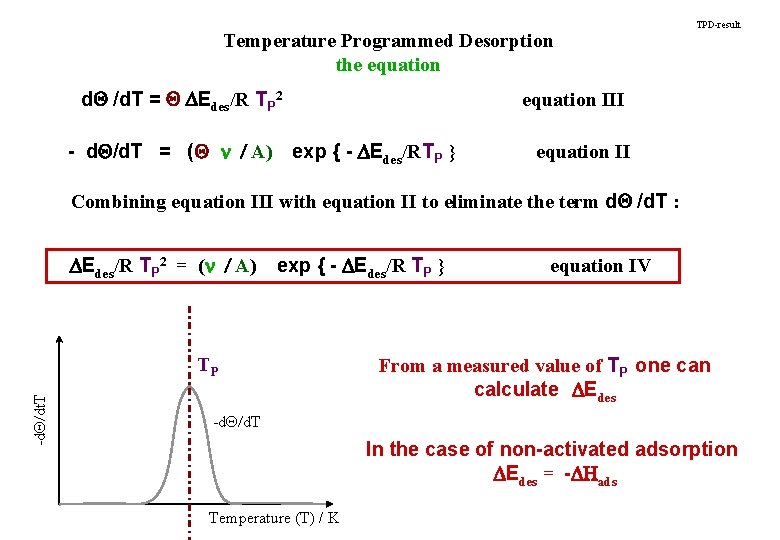
Temperature Programmed Desorption the equation d. Q /d. T = Q DEdes/R TP 2 - d. Q/d. T = (Q n / A) TPD-result equation III exp { - DEdes/RTP } equation II Combining equation III with equation II to eliminate the term d. Q /d. T : DEdes/R TP 2 = (n / A) exp { - DEdes/R TP } -d. Q/dt. T TP equation IV From a measured value of TP one can calculate DEdes -d. Q/d. T In the case of non-activated adsorption DEdes = -DHads Temperature (T) / K

TPD-calculation Temperature Programmed Desorption the calculation DEdes/R TP 2 = (n / A) exp { - DEdes/R TP } equation IV The easiest way to obtain – DEdes from TP is to use an iterative method on a re-arranged form of equation IV: DEdes/R TP 2 = (n / A) exp { - DEdes/R TP } equation IV DEdes = R TP ln {n R TP 2 / A DEdes ie. guess a sensible value for DEdes (eg 100 k. J mol-1 for chem. ); substitute in RHS, calculate )Edes; Substitute new value in RHS, re-calculate, etc The solution quickly converges!
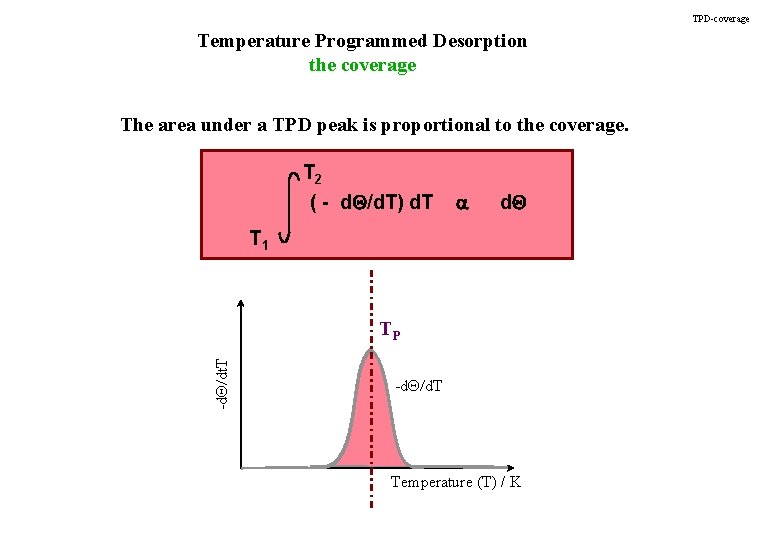
TPD-coverage Temperature Programmed Desorption the coverage The area under a TPD peak is proportional to the coverage. T 2 ( - d. Q/d. T) d. T a d. Q T 1 -d. Q/dt. T TP -d. Q/d. T Temperature (T) / K
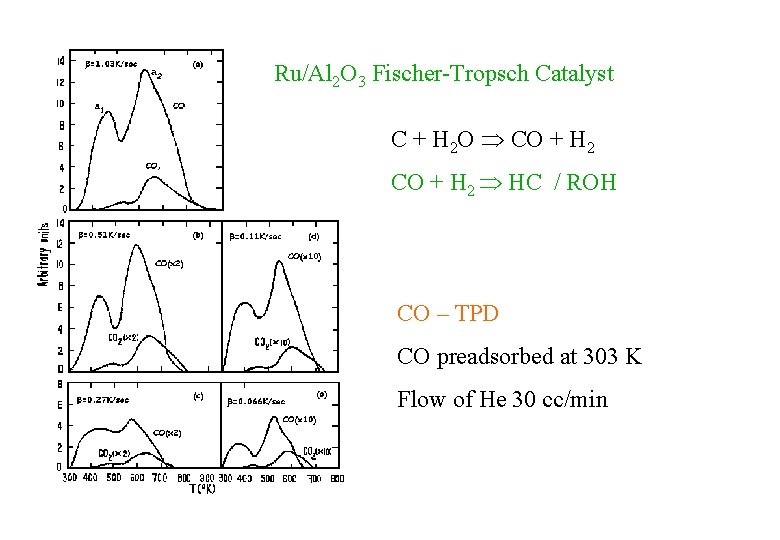
Ru/Al 2 O 3 Fischer-Tropsch Catalyst C + H 2 O CO + H 2 HC / ROH CO – TPD CO preadsorbed at 303 K Flow of He 30 cc/min
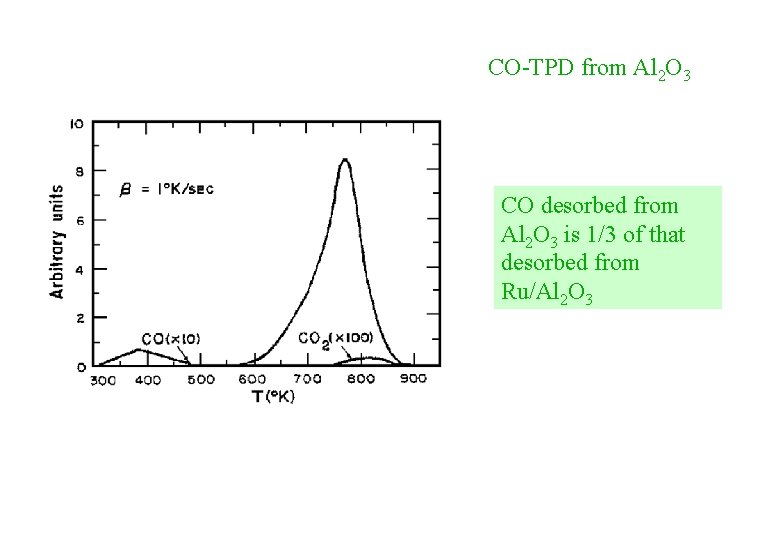
CO-TPD from Al 2 O 3 CO desorbed from Al 2 O 3 is 1/3 of that desorbed from Ru/Al 2 O 3
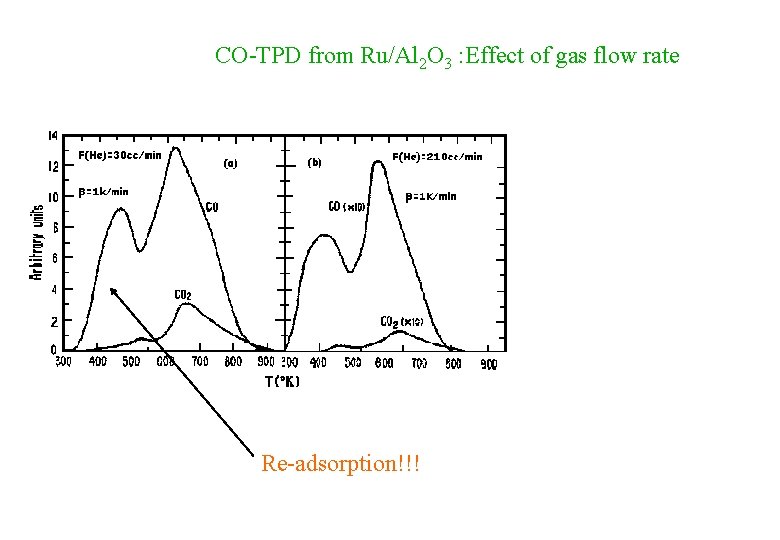
CO-TPD from Ru/Al 2 O 3 : Effect of gas flow rate Re-adsorption!!!
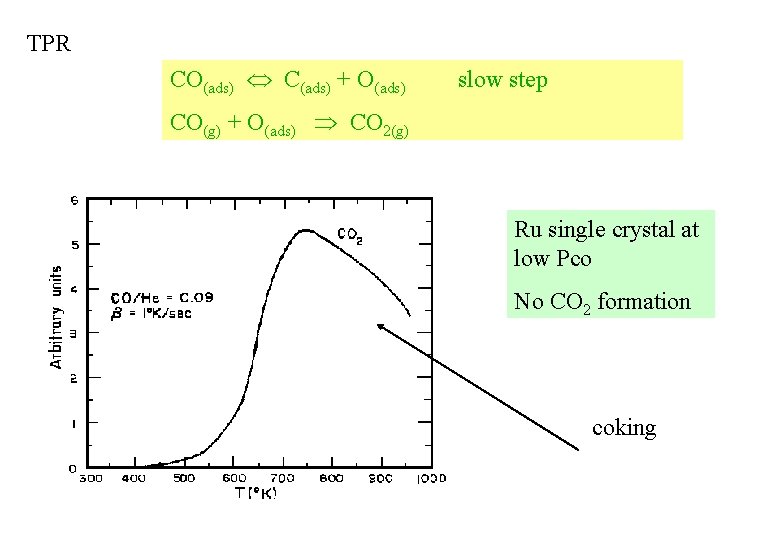
TPR CO(ads) C(ads) + O(ads) slow step CO(g) + O(ads) CO 2(g) Ru single crystal at low Pco No CO 2 formation coking
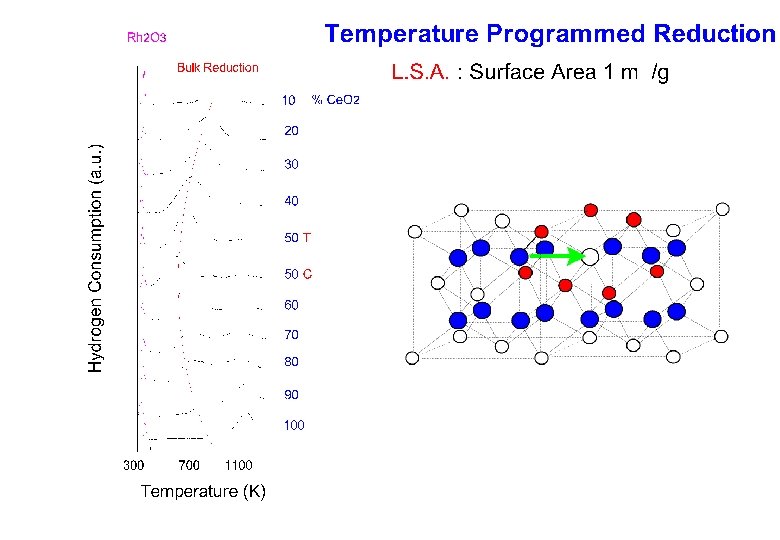
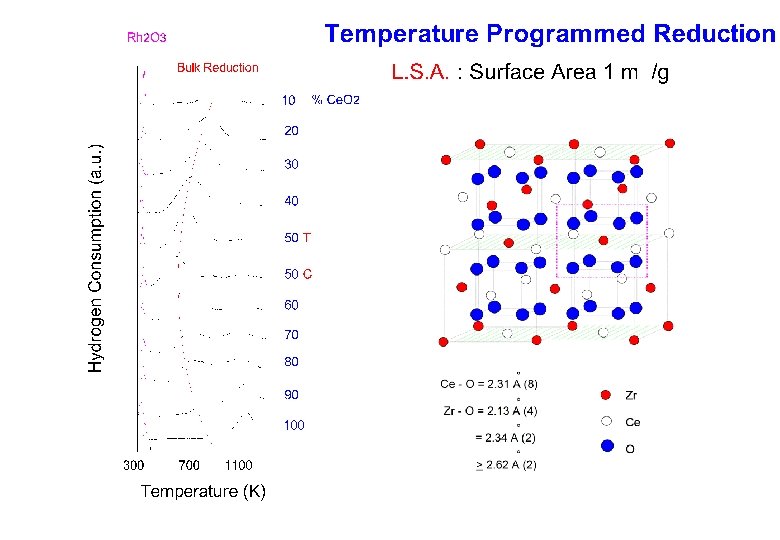
- Slides: 38