Catalyst 5 min 92611 Electron Orbitals EQ How

Catalyst (5 min) 9/26/11 Electron Orbitals EQ: How are electrons organized in an atom? How were the models of JJ Thomson, Niels Bohr, and Erwin Schrodinger different from each other with respect to their ideas about electrons in an atom? Draw pictures if it will help you explain yourself. (Hint: Use your foldable. )
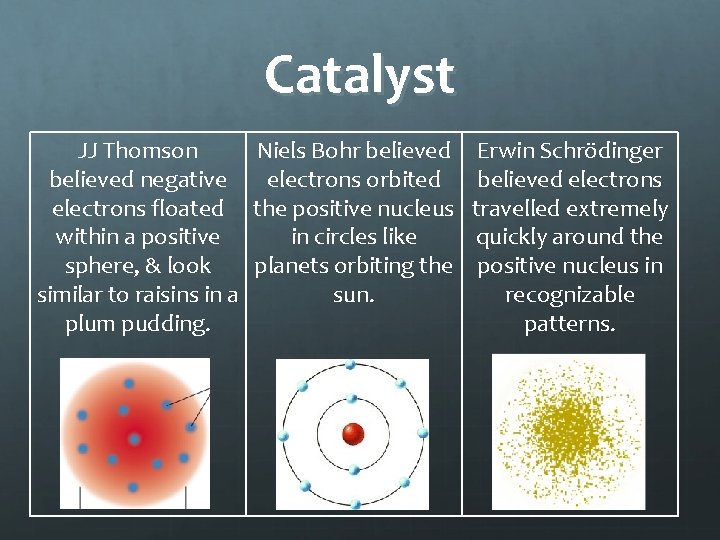
Catalyst JJ Thomson Niels Bohr believed negative electrons orbited electrons floated the positive nucleus within a positive in circles like sphere, & look planets orbiting the similar to raisins in a sun. plum pudding. Erwin Schrödinger believed electrons travelled extremely quickly around the positive nucleus in recognizable patterns.

Modern Orbital Theory There are four kinds of electron orbitals: s, p, d, & f Lower energy orbitals are closer to the nucleus and higher energy orbitals are farther from the nucleus. Each orbital can hold a maximum of 2 electrons
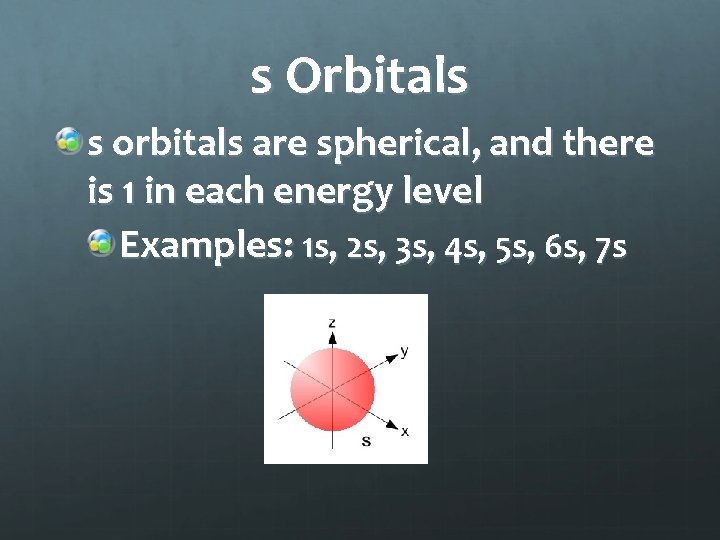
s Orbitals s orbitals are spherical, and there is 1 in each energy level Examples: 1 s, 2 s, 3 s, 4 s, 5 s, 6 s, 7 s
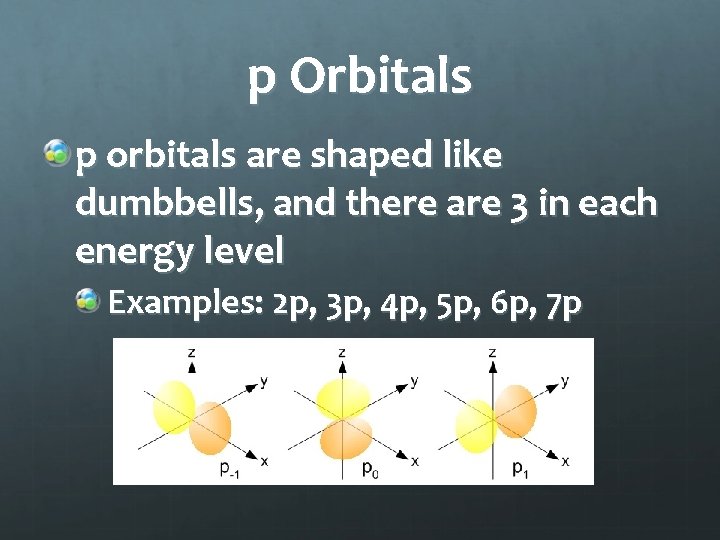
p Orbitals p orbitals are shaped like dumbbells, and there are 3 in each energy level Examples: 2 p, 3 p, 4 p, 5 p, 6 p, 7 p
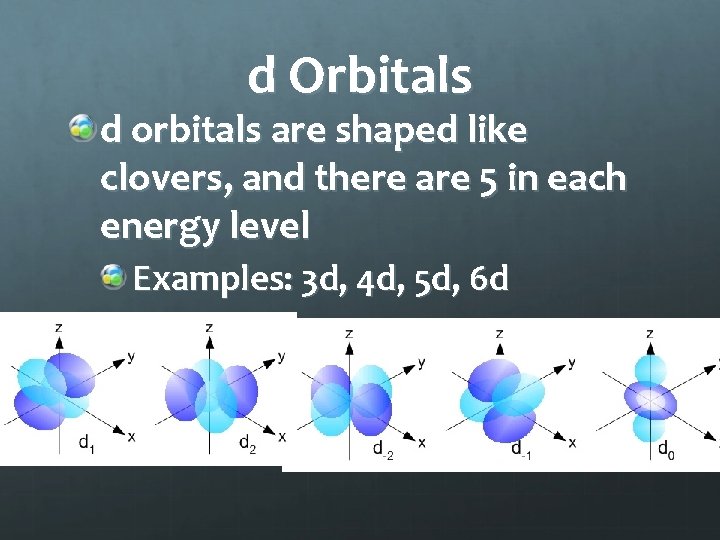
d Orbitals d orbitals are shaped like clovers, and there are 5 in each energy level Examples: 3 d, 4 d, 5 d, 6 d
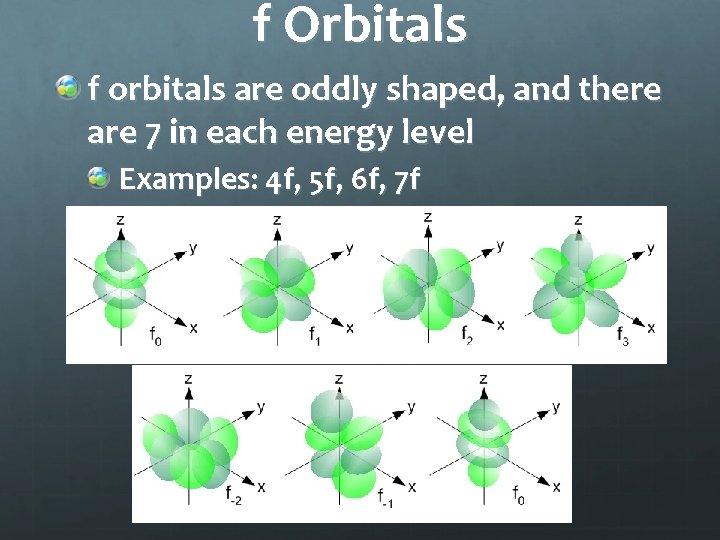
f Orbitals f orbitals are oddly shaped, and there are 7 in each energy level Examples: 4 f, 5 f, 6 f, 7 f

Reviewing orbitals 2 electrons. Each orbital can hold ____ 1 kind of s orbital. There is ____ 2 electrons. Therefore, s can hold ____ 3 kinds of p orbitals. There are ___ 6 electrons. Therefore, p can hold ___ 5 There are ___ kinds of d orbitals. 10 electrons. Therefore, d can hold ___ 7 kinds of f orbitals. There are ___ 14 electrons. Therefore, f can hold ___

Orbitals Orbital names have 2 parts The energy level is the first part. For example: n=1, n=2, n=3, etc. The shape of the orbital is the second part. For example: s, p, d, or f Examples of orbital names include 1 s, 2 p, 3 d, 3 f, 4 d, 5 s, 5 d, etc.
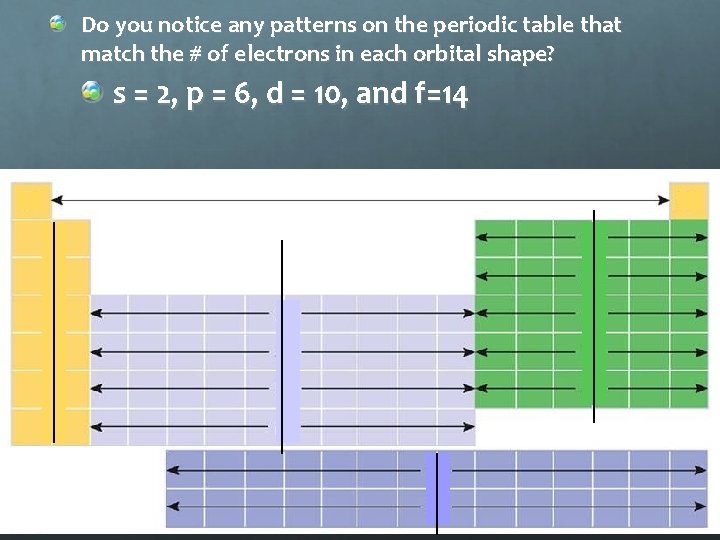
Do you notice any patterns on the periodic table that match the # of electrons in each orbital shape? s = 2, p = 6, d = 10, and f=14

Notes: Atomic Orbitals Summary of Principal Energy Levels, Sublevels and Orbitals: Principal Energy Level Type of Sublevel n=1 1 s n=2 2 s, 2 p n=3 3 s, 3 p, 3 d n=4 4 s, 4 p, 4 d, 4 f Maximum Number of Electrons 2 8 (2+6) 18 (2+6+10) 32 (2+6+10+14)

Notes: Orbital Diagrams • Fold your paper so that you have two columns – the left side column should be larger than the right Orbital Diagrams Electron Configuration

Notes: Orbital Diagrams Energy Hydrogen 3 s 2 p 2 s 1 s 2 p 2 p The Aufbau Principle states that an electron will occupy the lowest energy level orbital available

Notes: Orbital Diagrams Energy Helium 3 s 2 p 2 s 1 s 2 p 2 p The Pauli Exclusion Principle states that two electrons in the same orbital will have opposite spins

Notes: Orbital Diagrams Energy Lithium 3 s 2 p 2 s 1 s 2 p 2 p

Notes: Orbital Diagrams Energy Beryllium 3 s 2 p 2 s 1 s 2 p 2 p
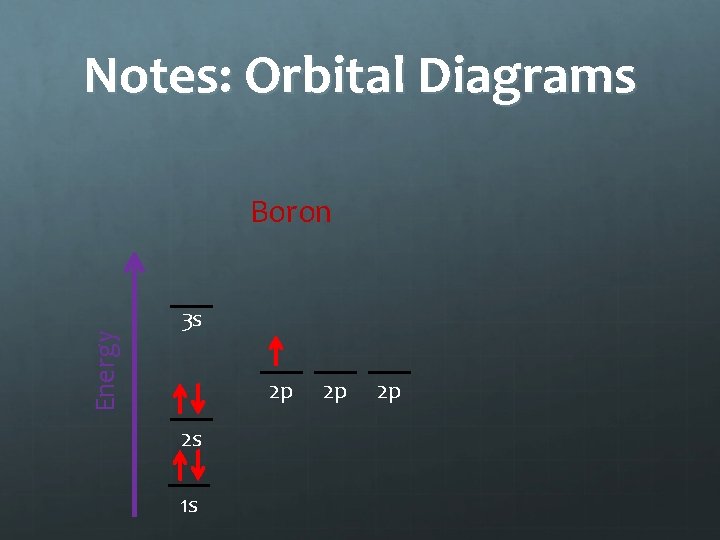
Notes: Orbital Diagrams Energy Boron 3 s 2 p 2 s 1 s 2 p 2 p

Notes: Orbital Diagrams Energy Carbon 3 s 2 p 2 s 1 s 2 p 2 p Hund’s Rule states that electrons in an equal energy level orbital will go in singly before doubling up

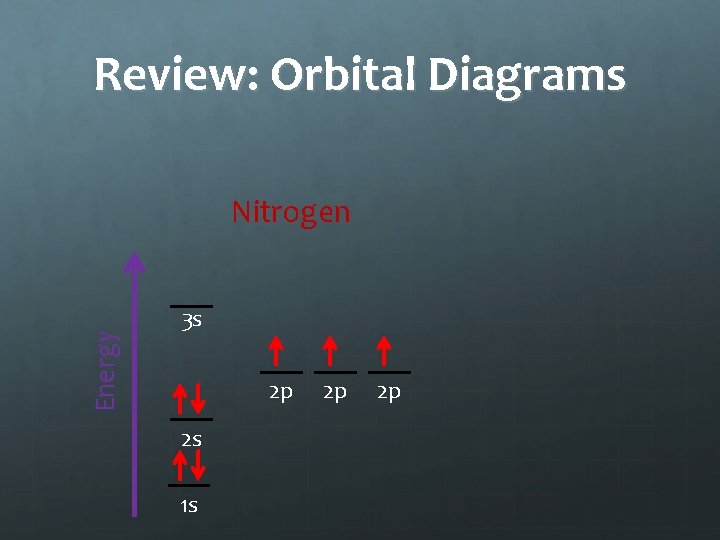
Notes: Orbital Diagrams Energy Nitrogen 3 s 2 p 2 s 1 s 2 p 2 p Hund’s Rule states that electrons in an equal energy level orbital will go in singly before doubling up
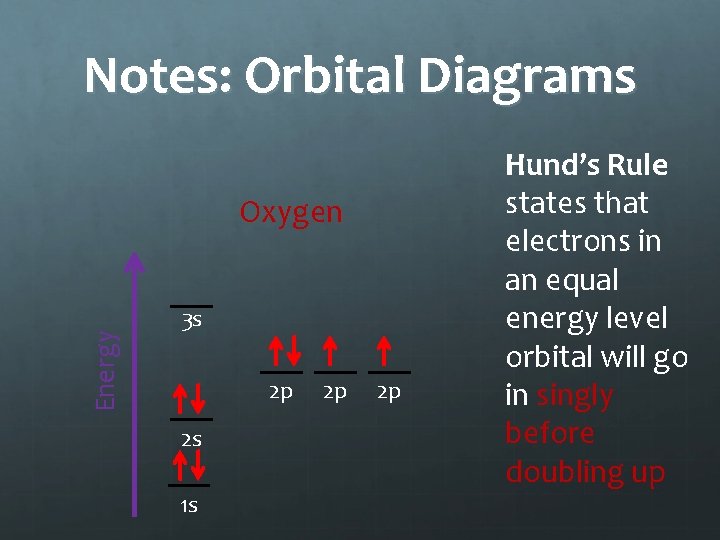
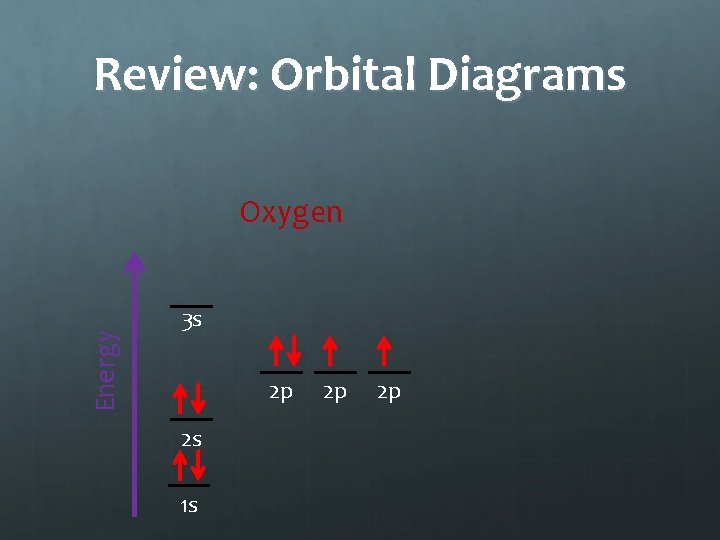
Notes: Orbital Diagrams Energy Oxygen 3 s 2 p 2 s 1 s 2 p 2 p Hund’s Rule states that electrons in an equal energy level orbital will go in singly before doubling up
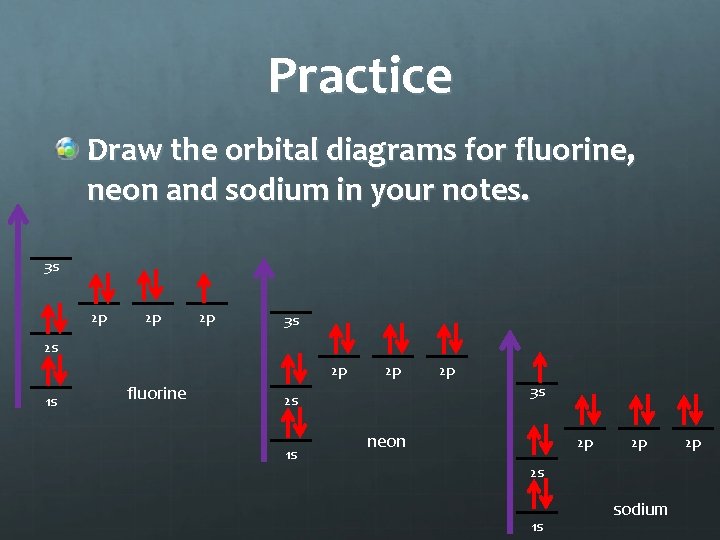
Practice Draw the orbital diagrams for fluorine, neon and sodium in your notes. 3 s 2 p 2 p 2 p 3 s 2 s 2 p 1 s fluorine 2 p 3 s 2 s 1 s 2 p neon 2 p 2 p 2 s 1 s sodium 2 p

Notes: Orbital Diagrams Energy 4 s 3 p 3 p 3 p 2 p 2 p 2 p 3 s 2 s 1 s

Homework Draw orbital diagrams for: Aluminum Phosphorous Chlorine

Catalyst (5 min) 9/28/11 Electron Configurations EQ: How do we represent the organization of electrons in an atom? Draw an electron orbital diagram for sulfur.
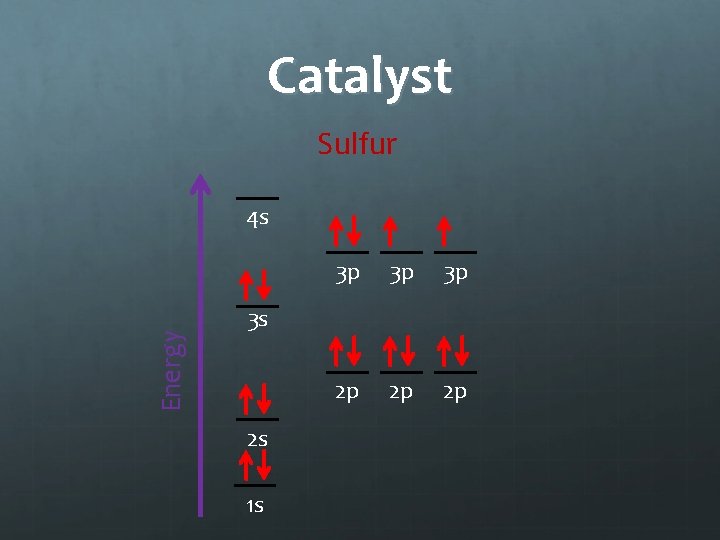
Catalyst Sulfur Energy 4 s 3 p 3 p 3 p 2 p 2 p 2 p 3 s 2 s 1 s

Principles of Orbital Filling The Aufbau Principle states that an electron will occupy the lowest energy level orbital available The Pauli Exclusion Principle states that two electrons in the same orbital will have opposite spins Hund’s Rule states that electrons in an equal energy level orbital will go in singly before doubling up
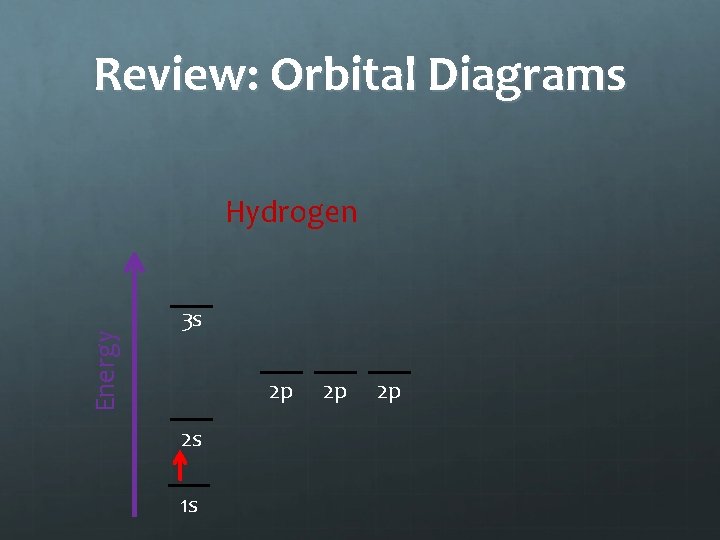
Review: Orbital Diagrams Energy Hydrogen 3 s 2 p 2 s 1 s 2 p 2 p
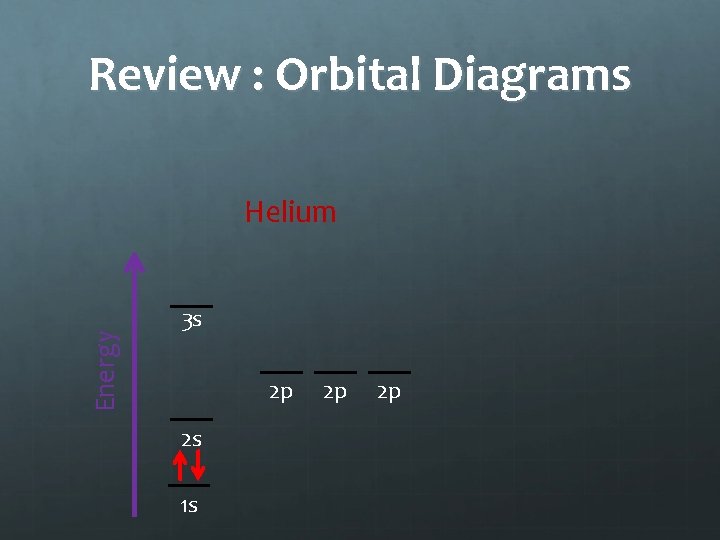
Review : Orbital Diagrams Energy Helium 3 s 2 p 2 s 1 s 2 p 2 p
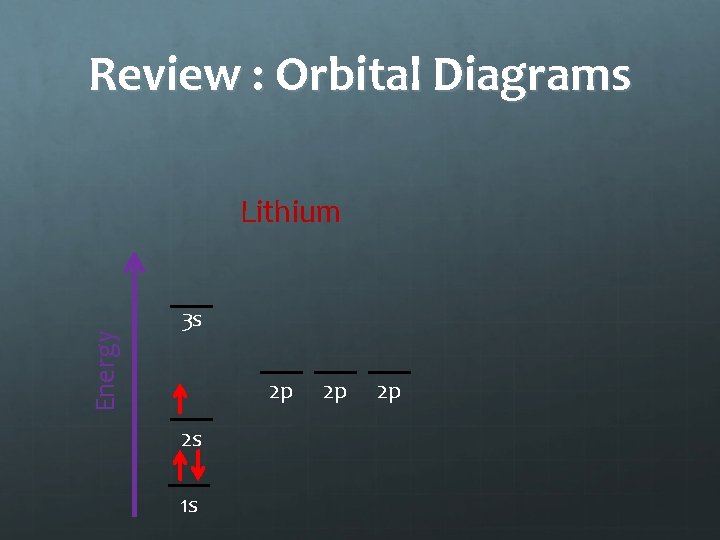
Review : Orbital Diagrams Energy Lithium 3 s 2 p 2 s 1 s 2 p 2 p
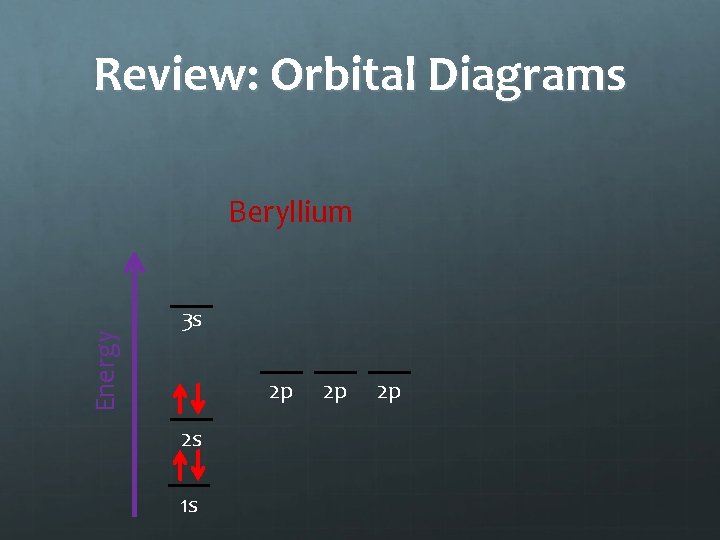
Review: Orbital Diagrams Energy Beryllium 3 s 2 p 2 s 1 s 2 p 2 p
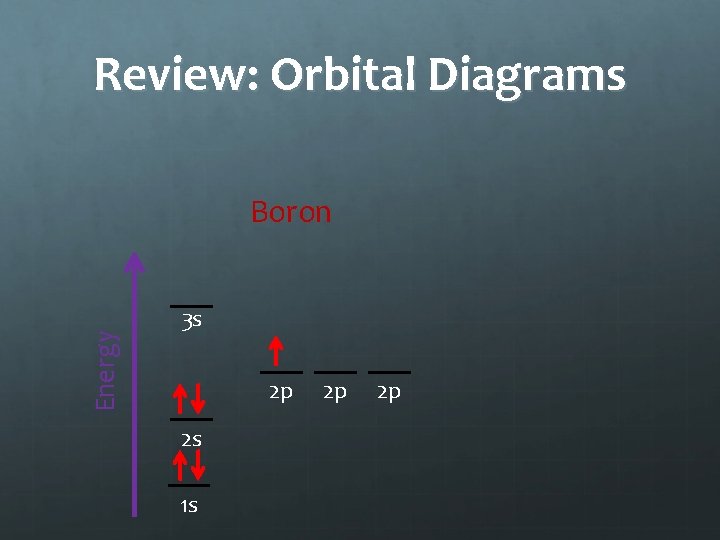
Review: Orbital Diagrams Energy Boron 3 s 2 p 2 s 1 s 2 p 2 p
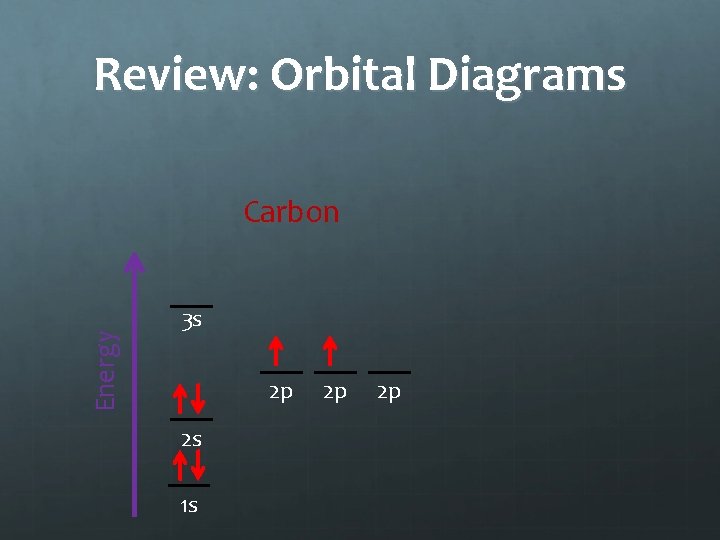
Review: Orbital Diagrams Energy Carbon 3 s 2 p 2 s 1 s 2 p 2 p
Review: Orbital Diagrams Energy Nitrogen 3 s 2 p 2 s 1 s 2 p 2 p
Review: Orbital Diagrams Energy Oxygen 3 s 2 p 2 s 1 s 2 p 2 p
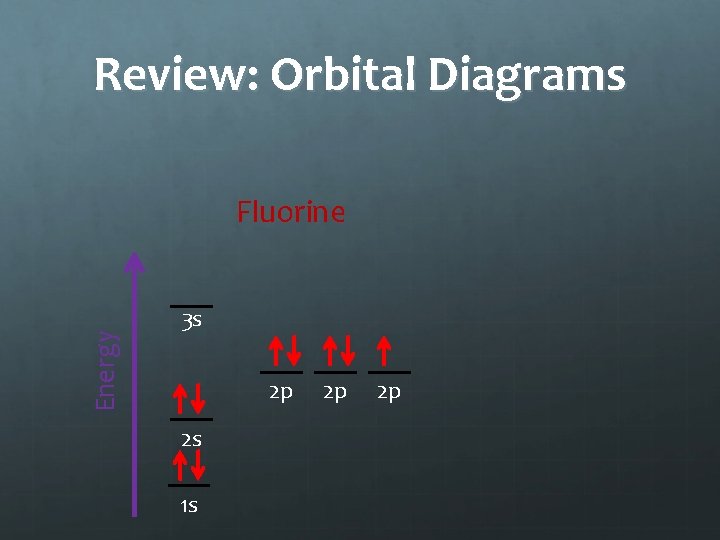
Review: Orbital Diagrams Energy Fluorine 3 s 2 p 2 s 1 s 2 p 2 p
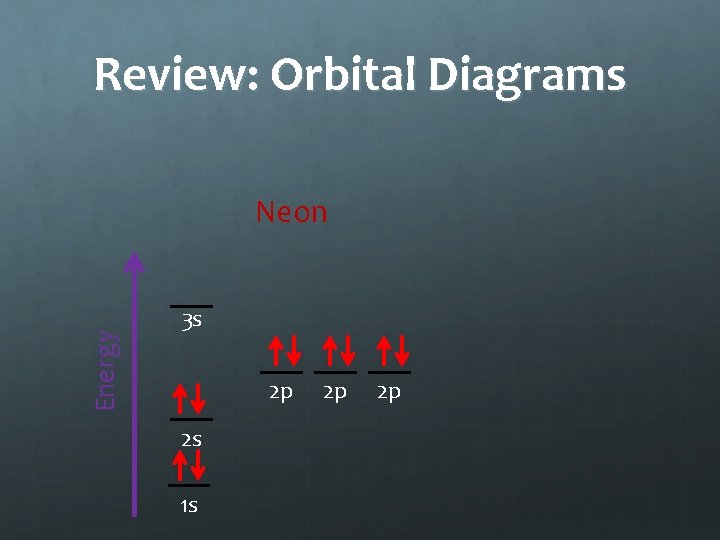
Review: Orbital Diagrams Energy Neon 3 s 2 p 2 s 1 s 2 p 2 p
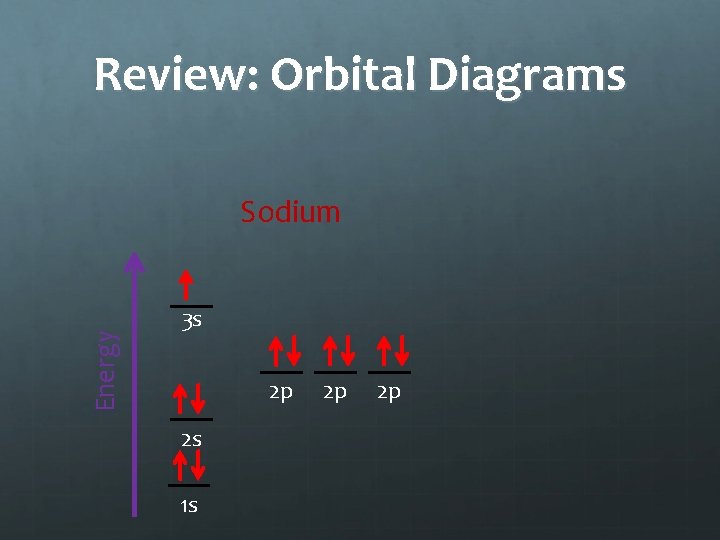
Review: Orbital Diagrams Energy Sodium 3 s 2 p 2 s 1 s 2 p 2 p
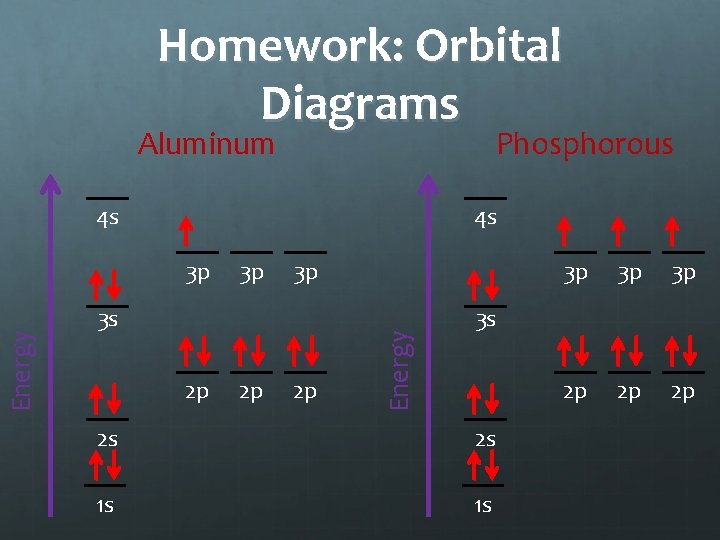
Homework: Orbital Diagrams Aluminum Phosphorous 4 s 4 s 3 p 3 p 3 s 2 p 2 p 2 p Energy 3 p 3 p 2 p 2 p 2 p 3 s 2 s 2 s 1 s 1 s
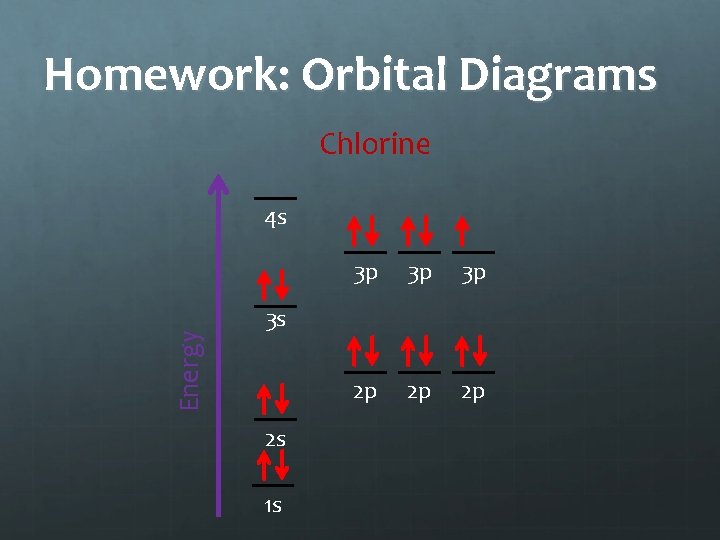
Homework: Orbital Diagrams Chlorine Energy 4 s 3 p 3 p 3 p 2 p 2 p 2 p 3 s 2 s 1 s
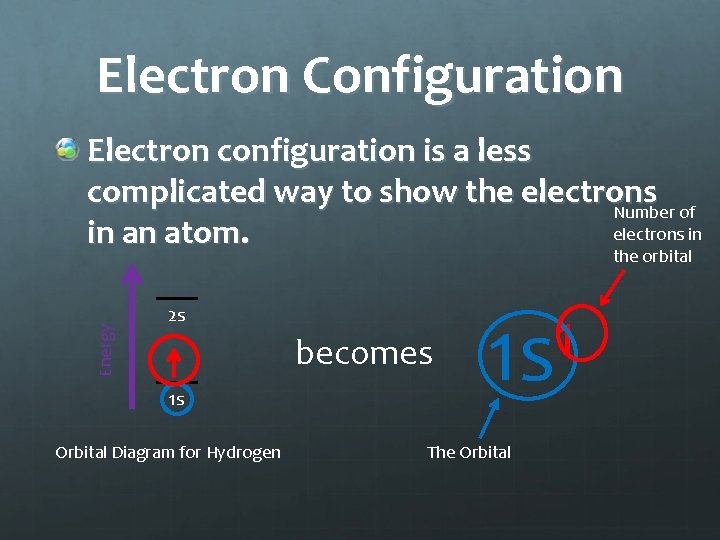
Electron Configuration Electron configuration is a less complicated way to show the electrons Number of electrons in in an atom. Energy the orbital 2 s becomes 1 s Orbital Diagram for Hydrogen 1 1 s The Orbital

Electron Configuration Energy He 2 s becomes 2 1 1 s 2 s 1 s Energy Li 2 s 1 s
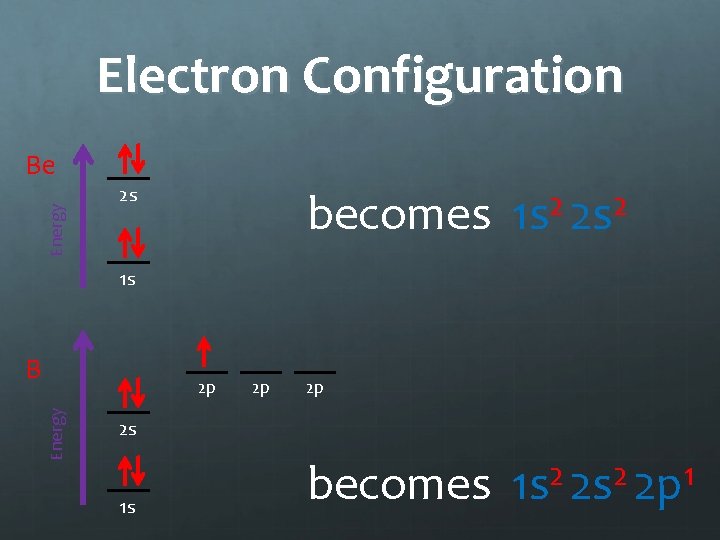
Electron Configuration Energy Be 2 s becomes 2 2 1 s 2 s 1 s B Energy 2 p 2 p 2 p 2 s 1 s becomes 1 s 2 2 p 1

Practice Write the electron configuration for carbon through neon. Carbon = 1 s 22 p 2 Nitrogen = 1 s 22 p 3 Oxygen = 1 s 22 p 4 Fluorine = 1 s 22 p 5 Neon = 1 s 22 p 6

Classwork/Homework On a separate sheet of paper, make 2 columns just like you did on Thursday. Keep going! I need to see orbital diagrams & electron configurations for elements #11 -22 (sodium through titanium). Orbital Diagrams Electron Configuration

Classwork/Homework 4 p 4 p 4 p 3 d Energy 4 s 3 p 3 p 3 p 2 p 2 p 2 p 3 s 2 s 1 s 3 d 3 d

Homework Write electron configurations for Al, P, Cl, & S (aluminum, phosphorous, chlorine, and sulfur). Hint: You should have the electron orbital diagrams for all of these from last night’s homework and today’s warm-up.

Catalyst (5 min) 9/28/11 Electron Configurations EQ: How do we represent the organization of electrons in an atom? 1. How many types of s, p, d, & f orbitals are there? 2. How many electrons will fit in one orbital? 3. If phosphorous has 15 electrons, where is each one located?

Catalyst Review 1 & 2: s = 1 type, 2 electrons p = 3 types, 6 electrons d = 5 types, 10 electrons f = 7 types, 14 electrons 3: Phosphorous has 15 electrons. 1 s 2 2 s 22 p 6 3 s 23 p 3 1 st energy level has 2 electrons in the 1 s orbital 2 nd energy level has 8 electrons in the 2 s & 2 p orbitals 2 electrons in 2 s, 6 electrons in 2 p 3 rd energy level has 5 electrons in the 3 s & 3 p orbitals 2 electrons in 3 s, 3 electrons in 2 p
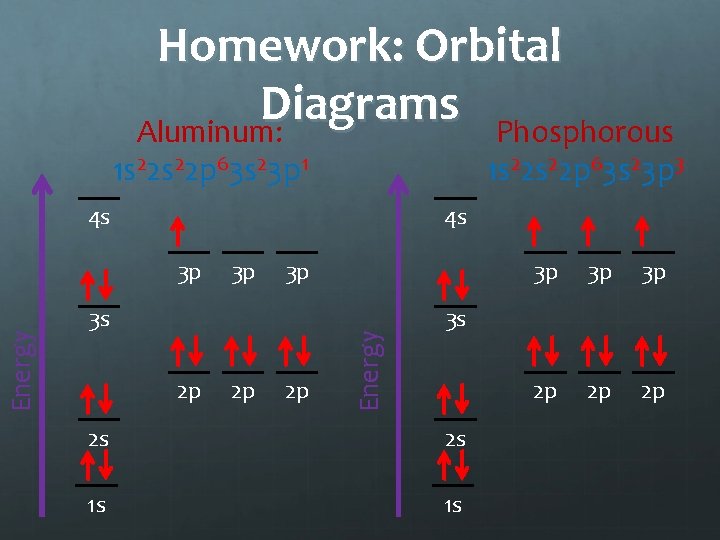
Homework: Orbital Diagrams Aluminum: Phosphorous 1 s 22 p 63 s 23 p 1 1 s 22 p 63 s 23 p 3 4 s 4 s 3 p 3 p 3 s 2 p 2 p 2 p Energy 3 p 3 p 2 p 2 p 2 p 3 s 2 s 2 s 1 s 1 s

Homework: Orbital Diagrams Chlorine Sulfur 1 s 22 p 63 s 23 p 5 1 s 22 p 63 s 23 p 4 4 s 4 s 3 p 3 p 3 s 2 p 2 p 2 p Energy 3 p 3 p 2 p 2 p 2 p 3 s 2 s 2 s 1 s 1 s
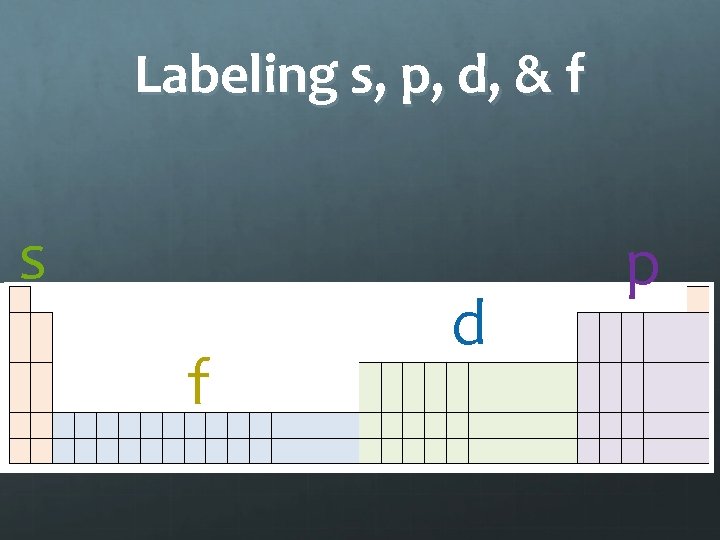
Labeling s, p, d, & f s f d p

Writing Electron Configurations Carbon s 2 1 s f 2 2 2 s 2 p d p C

Writing Electron Configurations Magnesium s Mg 2 1 s f 2 6 2 2 s 2 p 3 s d p
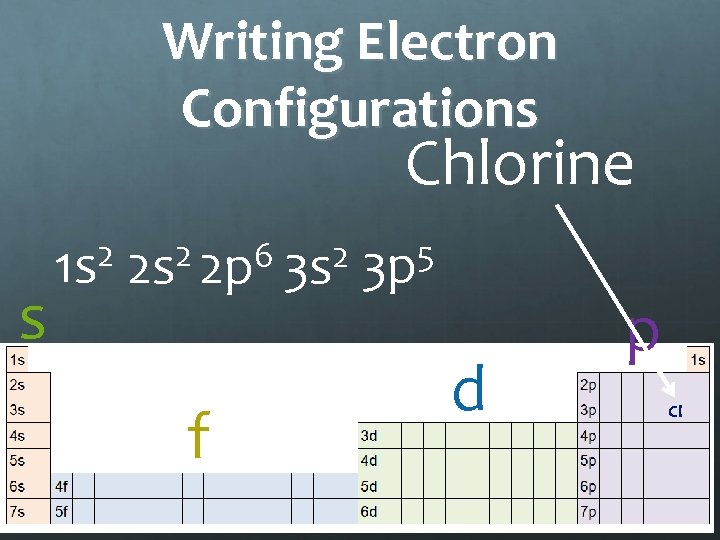
Writing Electron Configurations Chlorine s 2 1 s 2 6 2 2 s 2 p 3 s f 5 3 p d p Cl

Writing Electron Configurations Arsenic 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 3 s f d p As

Identifying Elements 1 s 22 p 3 Nitrogen 1 s 22 p 63 s 23 p 64 s 23 d 104 p 1 Gallium

Identifying Elements 1 s 22 p 63 s 23 p 3 Phosphorous 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 1 Rubidium

Catalyst (5 min) 9/29/11 Valence Electrons EQ: What are valence electrons? Write the electron configuration for neon. Write the electron configuration for aluminum. Write the electron configuration for argon. Write the electron configuration for calcium.

Catalyst Review Neon 1 s 22 p 6 Aluminum 1 s 22 p 63 s 23 p 1 Argon Calcium 1 s 22 p 63 s 23 p 6 2 2 6 2 1 s 2 s 2 p 3 s 3 p 4 s

- Slides: 60