Catalysis Starter What do you know about catalysts
Catalysis

Starter: What do you know about catalysts? • • • Substance that changes the speed/rate of a chemical reaction, but is not used up in the reaction. Speed up reactions by providing an alternative route with a lower activation energy (Do not alter ∆G, ∆H or ∆S for a reaction, only the activation energy). Speed the rate at which an equilibrium is reached by speeding up the forward and reverse reaction They do not alter the position of the equilibrium (or the value of Kc –the equilibrium constant). Catalysts that slow down reactions are called inhibitors (or negative catalysts).

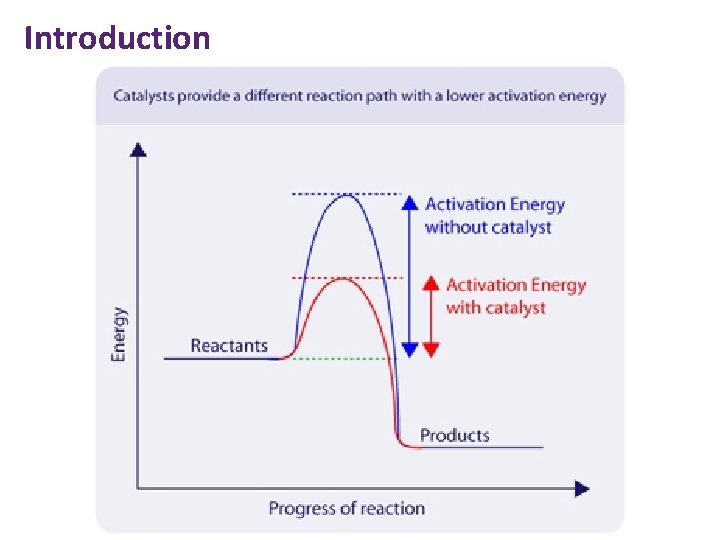
Introduction • Catalyst = substance that speeds up reaction without being used up. • They provide an alternative mechanism with lower activation energy. • For an equilibrium, they speed up both reactions equally and so do not effect equilibrium position. • Heterogeneous catalyst is in different phase to the reactants. • Homogeneous catalyst is in the same phase as the reactants.
Introduction

Common Features of Catalysts • They can be very specific and only catalyse a certain reaction or class of similar reactions • Size and surface area: Often only needed in very small amounts (e. g. , 2 g of Pt can catalyse the decomposition of 1000 m 3 of H 2 O 2). • Solid catalysts more effective when they are a powder or as a thin layer – their surface area to volume ratio is very large.

Heterogeneous Catalysts • Heterogeneous catalyst is in different phase to the reactants. • Usually a solid catalyst and gaseous reactants. • Most industrial catalysts are like this • (e. g. Haber Process – Fe; Contact Process – V 2 O 5).
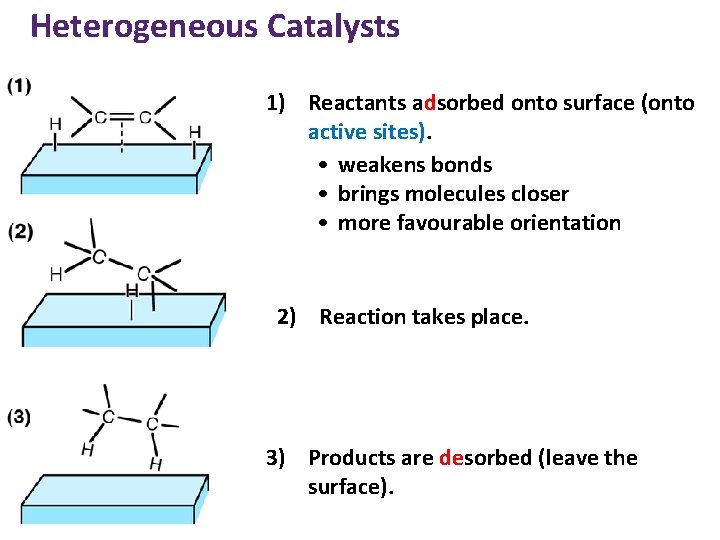
Heterogeneous Catalysts 1) Reactants adsorbed onto surface (onto active sites). • weakens bonds • brings molecules closer • more favourable orientation 2) Reaction takes place. 3) Products are desorbed (leave the surface).
Heterogeneous Catalysts

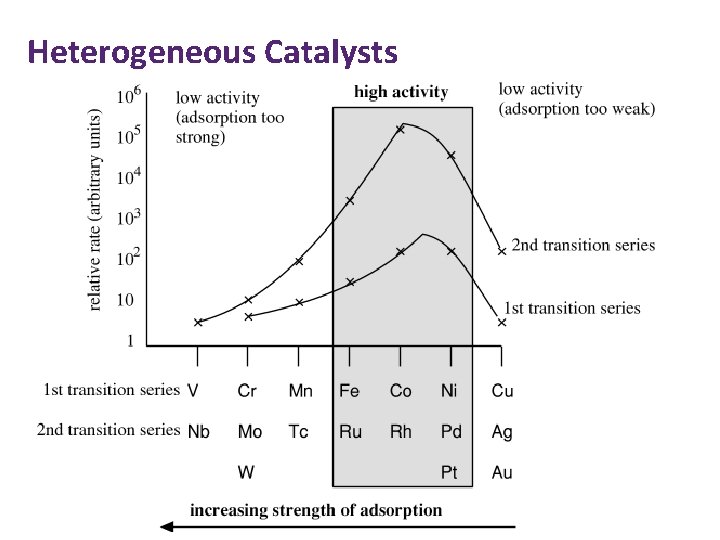
Heterogeneous Catalysts Strength of adsorption: Too strong (e. g. W) • Reactants cannot move round surface. • Products cannot desorb. Too weak (e. g. Ag) • Reactants not adsorbed. Ideal (e. g. Ni, Pt)

WHAT IS THE CONTACT PROCESS?

Importance of Variable Oxidation States • Variable oxidation states very important in heterogeneous catalysis • E. g. , use of V 2 O 5 in the Contact Process • In the second stage of H 2 SO 4 manufacture, sulfur(IV) oxide is oxidised to sulfur(VI) oxide (2 -step mechanism): 1. The vanadium(V) oxide catalyst oxidises sulfur(IV) oxide to sulfur(VI) oxide. SO 2(g) + V 2 O 5(s) → SO 3(g) + V 2 O 4(s) 2. The vanadium(IV) oxide reacts with oxygen and the Vanadium(V) oxide is regenerated V 2 O 4(s) + 1/2 O 2(g) → V 2 O 5(s) Overall: V 2 O 5 catalyst SO 2(g) + 1/2 O 2(g) ↔ SO 3(g)

HOW DOES A CATALYTIC CONVERTOR WORK?

Heterogeneous Catalysts Nature of catalyst • Large surface area. • Spread thinly over ceramic honeycomb.
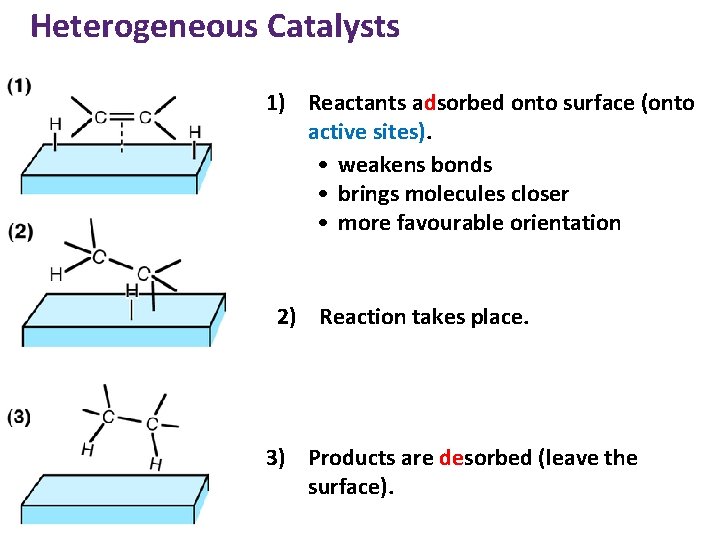
Heterogeneous Catalysts 1) Reactants adsorbed onto surface (onto active sites). • weakens bonds • brings molecules closer • more favourable orientation 2) Reaction takes place. 3) Products are desorbed (leave the surface).

Homogeneous Catalysts • Homogeneous catalyst is in the same phase as the reactants. • Most examples involve reactions in solution with catalyst in solution. • Some gas phase examples.

Homogeneous Catalysts 1) Catalyst reacts with a reactant to form intermediate. 2) Intermediate reacts to form product faster than original reactant (and regenerates catalyst). e. g. acid catalyst X + Y products 1) X + H+ → HX+ 2) HX+ + Y → products + H+
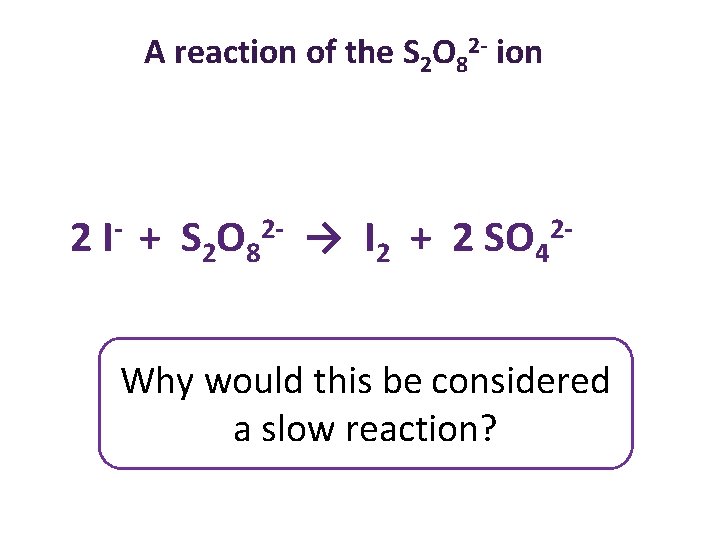
A reaction of the S 2 O 82 - ion 2 I- + S 2 O 82 - → I 2 + 2 SO 42 Why would this be considered a slow reaction?

A reaction of the S 2 O 82 - ion Fe 2+ 2 I- + S 2 O 82 - → I 2 + 2 SO 42 -

Homogeneous Catalysts 1) 2 Fe 2+ + S 2 O 82 - → 2 Fe 3+ + 2 SO 422) 2 Fe 3+ + 2 I- → 2 Fe 2+ + I 2 Fe 2+ is regenerated, so the two steps can repeat continuously.

Can also happen the other way around… 1) 2 Fe 3+ + 2 I- → 2 Fe 2+ + I 2 1) 2 Fe 2+ + S 2 O 82 - → 2 Fe 3+ + 2 SO 42 Fe 3+ is regenerated, so the two steps can repeat continuously.

Redox Titrations • May want to measure the conc of an oxidising or reducing agent • Do a redox titration • Similar to acid-base titration (find out how much acid is needed to react with a certain volume of base [or vice versa]) • C 2 O 42 - react with Mn. O 4 - acidified with excess dilute sulfuric acid (H+)

Homogeneous Catalysts: Autocatalysis • The reaction is catalysed by one of the products (Mn 2+) – this is called autocatalysis. • Such a reaction starts slowly – uncatalysed rate • As conc of the products (also the catalyst) builds up, the reaction speeds up to catalysed rate. • Then behaves as normal reaction. . . Slows down as reactants are used up e. g. transition metal catalyst: works by metal varying oxidation state Oxidation of ethanedioic acid by manganate(VII) ions Mn 2+ 2 Mn. O 4 - (aq)+16 H+ (aq) + 5 C 2 O 42 - (aq) → 2 Mn 2+ (aq) + 8 H 2 O (aq) +10 CO 2 (aq)
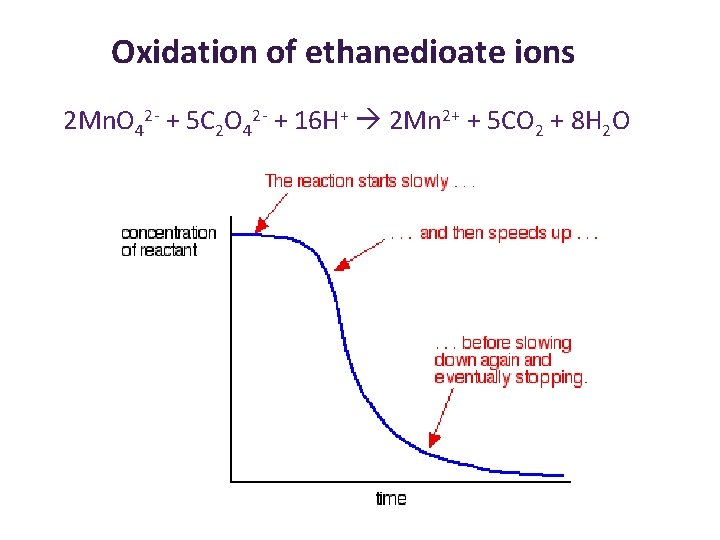
Oxidation of ethanedioate ions 2 Mn. O 42 - + 5 C 2 O 42 - + 16 H+ 2 Mn 2+ + 5 CO 2 + 8 H 2 O

EXAM QUESTIONS
- Slides: 24