Catabolic and Anabolic Reactions Learning Objectives 5 1

Catabolic and Anabolic Reactions Learning Objectives 5 -1 Define metabolism, and describe the fundamental differences between anabolism and catabolism. 5 -2 Identify the role of ATP as an intermediate between catabolism and anabolism.

Catabolic and Anabolic Reactions § Metabolism: the sum of the chemical reactions in an organism

Catabolic and Anabolic Reactions § Catabolism: provides energy and building blocks for anabolism § Anabolism: uses energy and building blocks to build large molecules
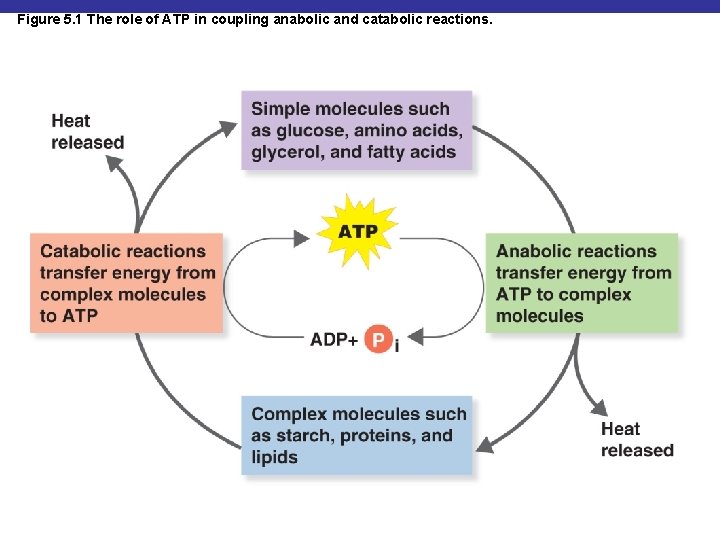
Figure 5. 1 The role of ATP in coupling anabolic and catabolic reactions.

Catabolic and Anabolic Reactions § A metabolic pathway is a sequence of enzymatically catalyzed chemical reactions in a cell § Metabolic pathways are determined by enzymes § Enzymes are encoded by genes

Collision Theory § The collision theory states that chemical reactions can occur when atoms, ions, and molecules collide § Activation energy is needed to disrupt electronic configurations § Reaction rate is the frequency of collisions with enough energy to bring about a reaction § Reaction rate can be increased by enzymes or by increasing temperature or pressure
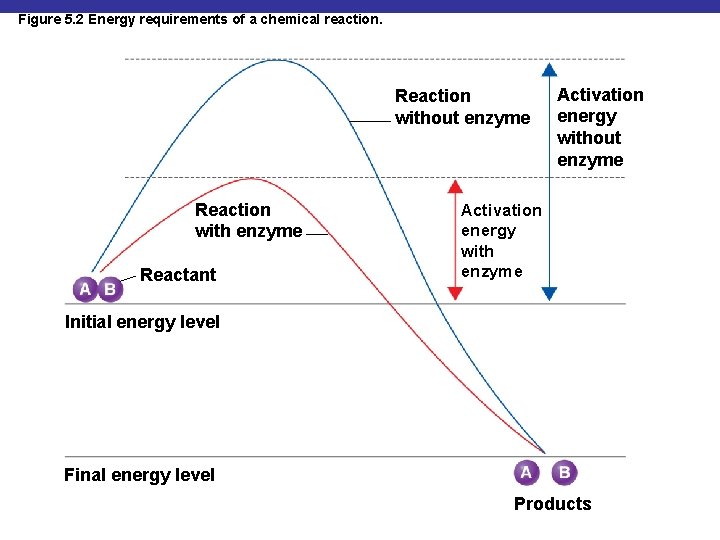
Figure 5. 2 Energy requirements of a chemical reaction. Reaction without enzyme Reaction with enzyme Reactant Activation energy without enzyme Activation energy with enzyme Initial energy level Final energy level Products

Enzyme Components § Biological catalysts § Specific for a chemical reaction; not used up in that reaction § Apoenzyme: protein § Cofactor: nonprotein component § Coenzyme: organic cofactor § Holoenzyme: apoenzyme plus cofactor
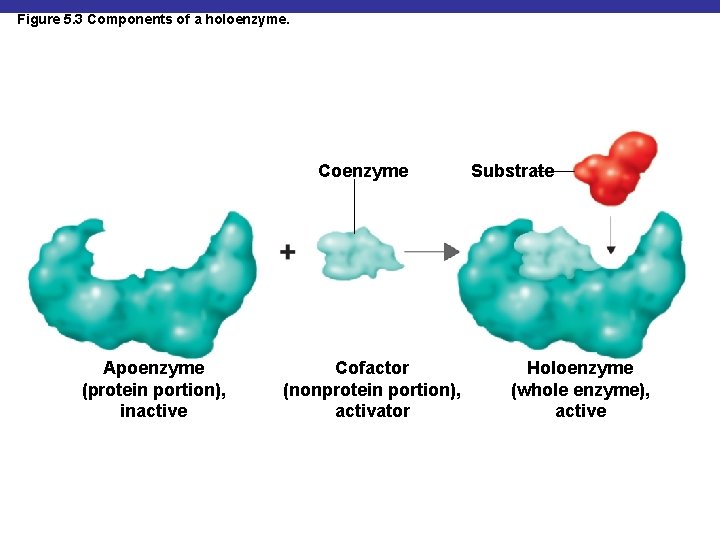
Figure 5. 3 Components of a holoenzyme. Coenzyme Apoenzyme (protein portion), inactive Cofactor (nonprotein portion), activator Substrate Holoenzyme (whole enzyme), active

Important Coenzymes § § NAD+ NADP+ FAD Coenzyme A

Enzyme Specificity and Efficiency § The turnover number is generally 1 to 10, 000 molecules per second
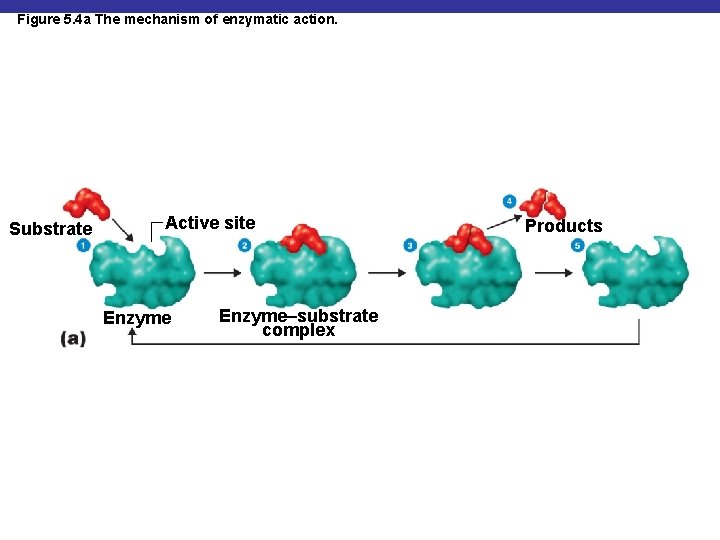
Figure 5. 4 a The mechanism of enzymatic action. Substrate Active site Enzyme–substrate complex Products
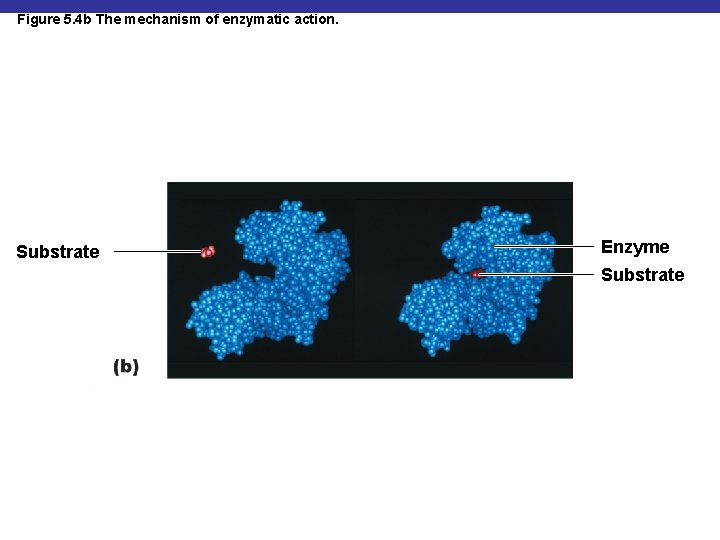
Figure 5. 4 b The mechanism of enzymatic action. Substrate Enzyme Substrate

Enzyme Classification § § § Oxidoreductase: oxidation-reduction reactions Transferase: transfer functional groups Hydrolase: hydrolysis Lyase: removal of atoms without hydrolysis Isomerase: rearrangement of atoms Ligase: joining of molecules; uses ATP

Factors Influencing Enzyme Activity § § Temperature p. H Substrate concentration Inhibitors

Factors Influencing Enzyme Activity § Temperature and p. H denature proteins

Figure 5. 6 Denaturation of a protein. Active (functional) protein Denatured protein
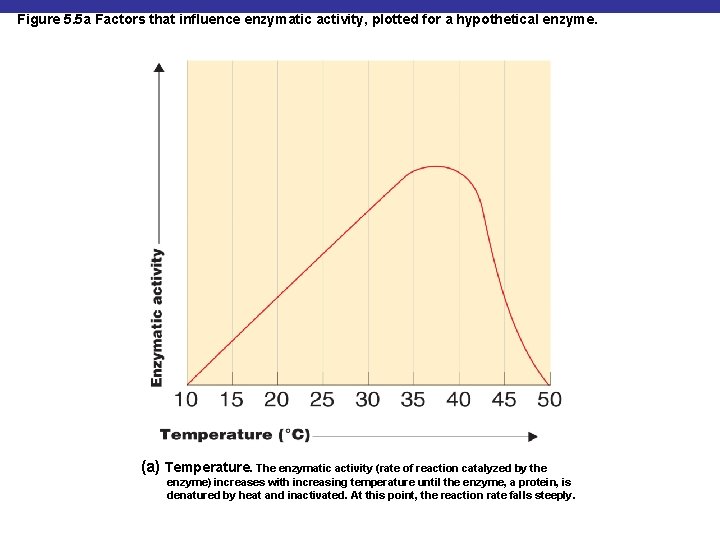
Figure 5. 5 a Factors that influence enzymatic activity, plotted for a hypothetical enzyme. (a) Temperature. The enzymatic activity (rate of reaction catalyzed by the enzyme) increases with increasing temperature until the enzyme, a protein, is denatured by heat and inactivated. At this point, the reaction rate falls steeply.
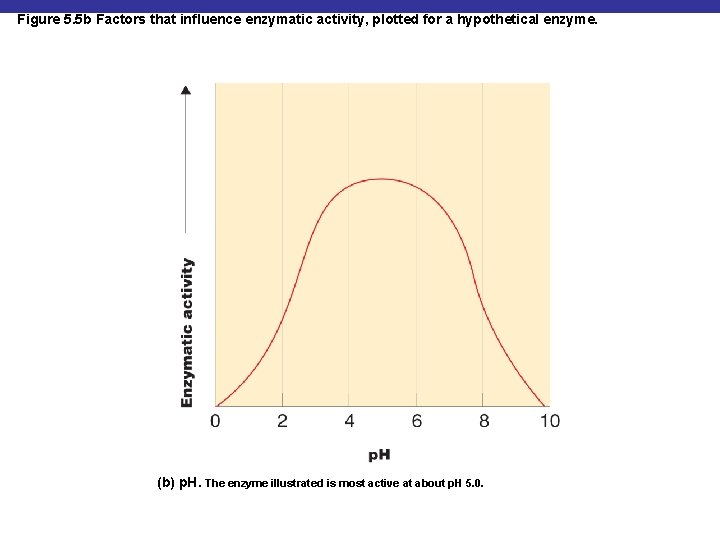
Figure 5. 5 b Factors that influence enzymatic activity, plotted for a hypothetical enzyme. (b) p. H. The enzyme illustrated is most active at about p. H 5. 0.
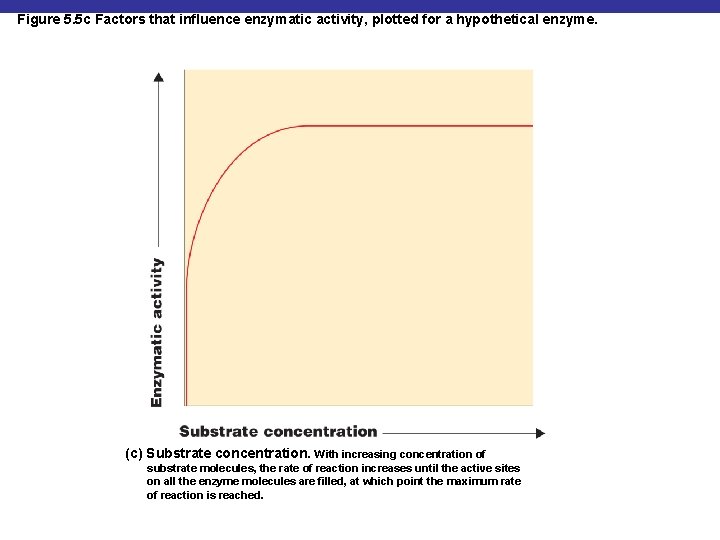
Figure 5. 5 c Factors that influence enzymatic activity, plotted for a hypothetical enzyme. (c) Substrate concentration. With increasing concentration of substrate molecules, the rate of reaction increases until the active sites on all the enzyme molecules are filled, at which point the maximum rate of reaction is reached.
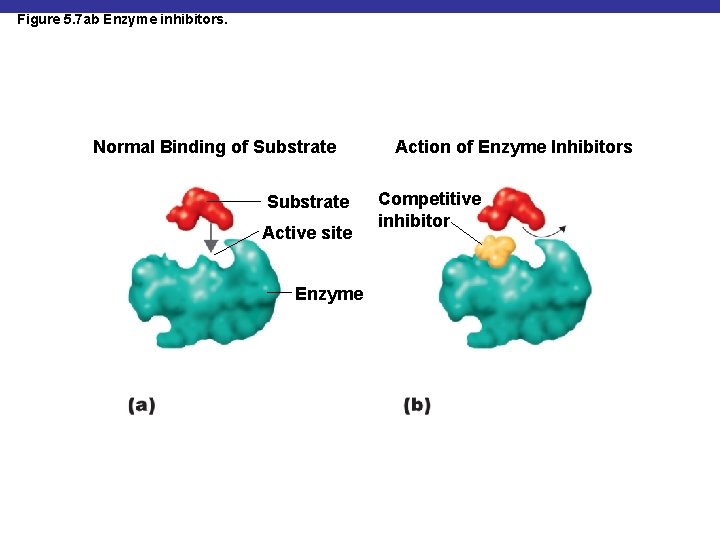
Figure 5. 7 ab Enzyme inhibitors. Normal Binding of Substrate Active site Enzyme Action of Enzyme Inhibitors Competitive inhibitor
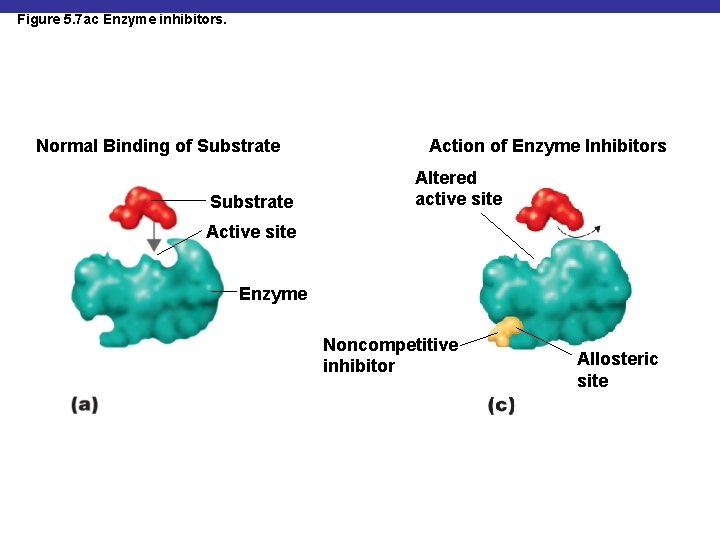
Figure 5. 7 ac Enzyme inhibitors. Normal Binding of Substrate Action of Enzyme Inhibitors Altered active site Active site Enzyme Noncompetitive inhibitor Allosteric site

Oxidation-Reduction Reactions § Oxidation: removal of electrons § Reduction: gain of electrons § Redox reaction: an oxidation reaction paired with a reduction reaction
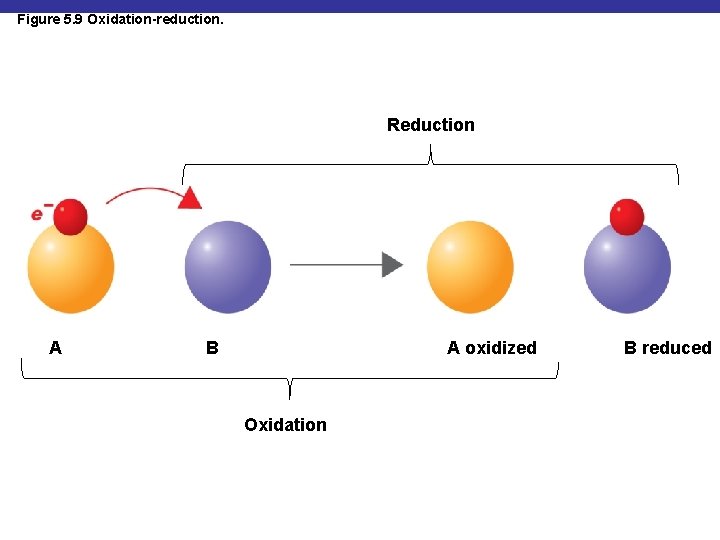
Figure 5. 9 Oxidation-reduction. Reduction A A oxidized B Oxidation B reduced

Oxidation-Reduction Reactions § In biological systems, the electrons are often associated with hydrogen atoms § Biological oxidations are often dehydrogenations
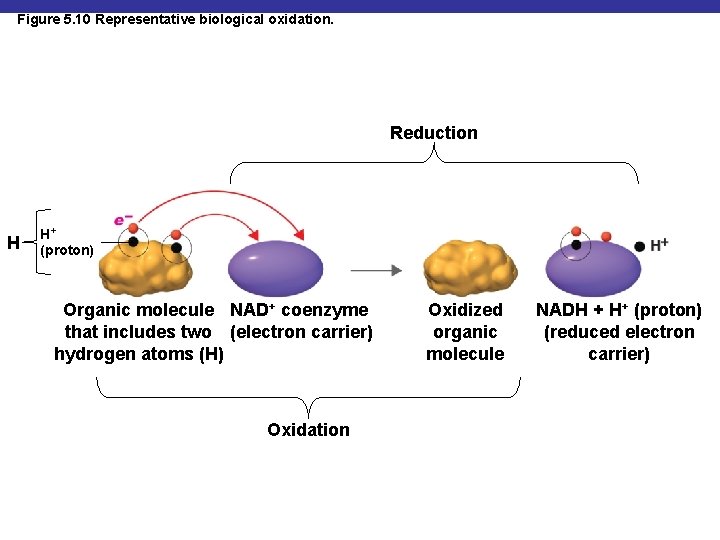
Figure 5. 10 Representative biological oxidation. Reduction H H+ (proton) Organic molecule NAD+ coenzyme that includes two (electron carrier) hydrogen atoms (H) Oxidation Oxidized organic molecule NADH + H+ (proton) (reduced electron carrier)

The Generation of ATP § ATP is generated by the phosphorylation of ADP
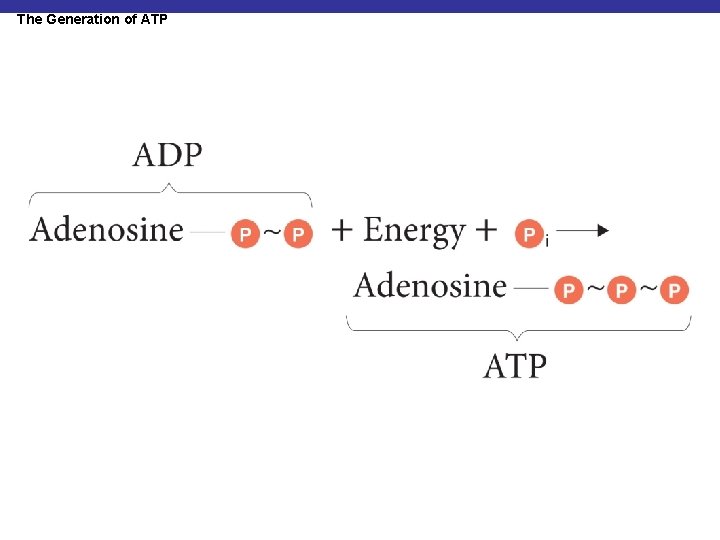
The Generation of ATP

Substrate-Level Phosphorylation § Energy from the transfer of a high-energy PO 4– to ADP generates ATP

Substrate-Level Phosphorylation

Oxidative Phosphorylation § Energy released from transfer of electrons (oxidation) of one compound to another (reduction) is used to generate ATP in the electron transport chain
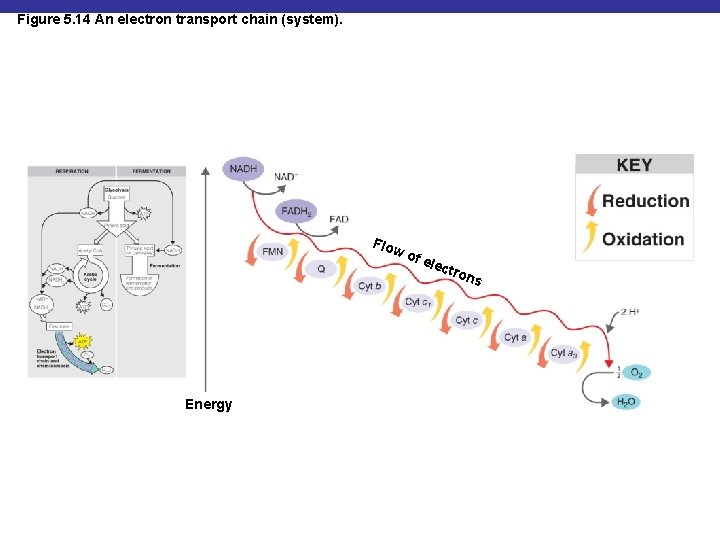
Figure 5. 14 An electron transport chain (system). Flo w of e le ctro Energy ns

Photophosphorylation § Light causes chlorophyll to give up electrons § Energy released from transfer of electrons (oxidation) of chlorophyll through a system of carrier molecules is used to generate ATP
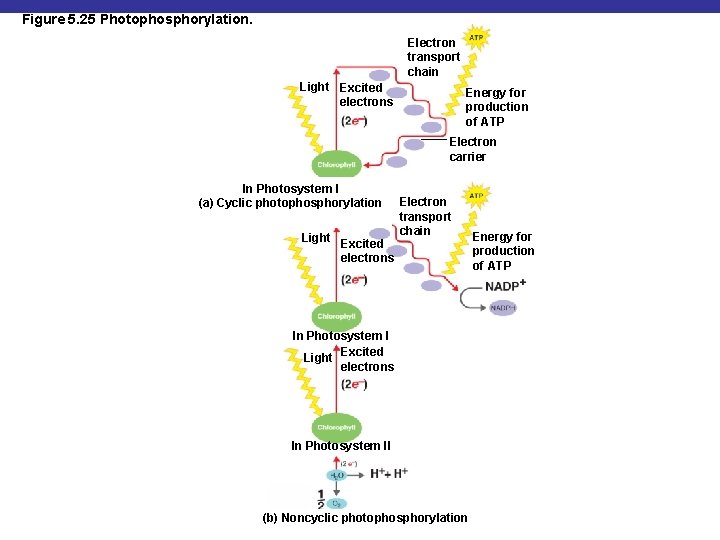
Figure 5. 25 Photophosphorylation. Electron transport chain Light Excited electrons Energy for production of ATP Electron carrier In Photosystem I (a) Cyclic photophosphorylation Light Excited electrons Electron transport chain In Photosystem I Light Excited electrons In Photosystem II (b) Noncyclic photophosphorylation Energy for production of ATP

Carbohydrate Catabolism § The breakdown of carbohydrates to release energy § Glycolysis § Krebs cycle § Electron transport chain

Glycolysis § The oxidation of glucose to pyruvic acid produces ATP and NADH
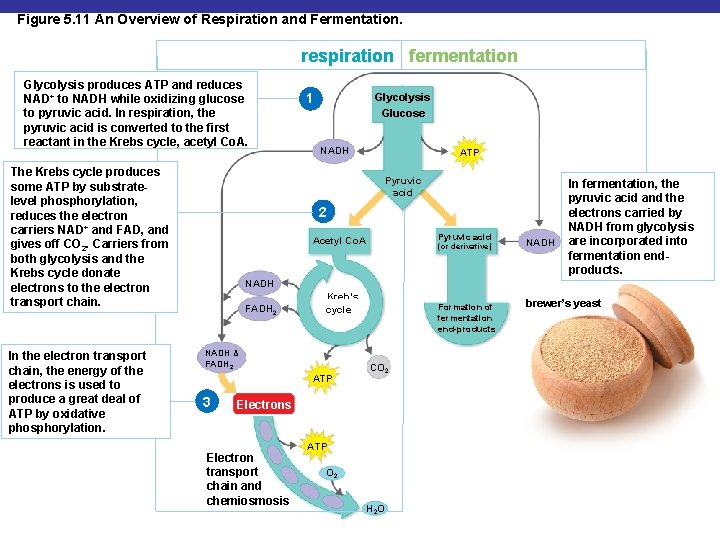
Figure 5. 11 An Overview of Respiration and Fermentation. respiration fermentation Glycolysis produces ATP and reduces NAD+ to NADH while oxidizing glucose to pyruvic acid. In respiration, the pyruvic acid is converted to the first reactant in the Krebs cycle, acetyl Co. A. The Krebs cycle produces some ATP by substratelevel phosphorylation, reduces the electron carriers NAD+ and FAD, and gives off CO 2. Carriers from both glycolysis and the Krebs cycle donate electrons to the electron transport chain. In the electron transport chain, the energy of the electrons is used to produce a great deal of ATP by oxidative phosphorylation. 1 Glycolysis Glucose NADH ATP Pyruvic acid 2 Pyruvic acid Acetyl Co. A (or derivative) NADH In fermentation, the pyruvic acid and the electrons carried by NADH from glycolysis are incorporated into fermentation endproducts. NADH FADH 2 Kreb’s cycle NADH & FADH 2 ATP 3 Formation of fermentation end-products CO 2 Electrons Electron transport chain and chemiosmosis ATP O 2 H 2 O brewer’s yeast

Preparatory Stage of Glycolysis § 2 ATP are used § Glucose is split to form 2 glucose-3 -phosphate
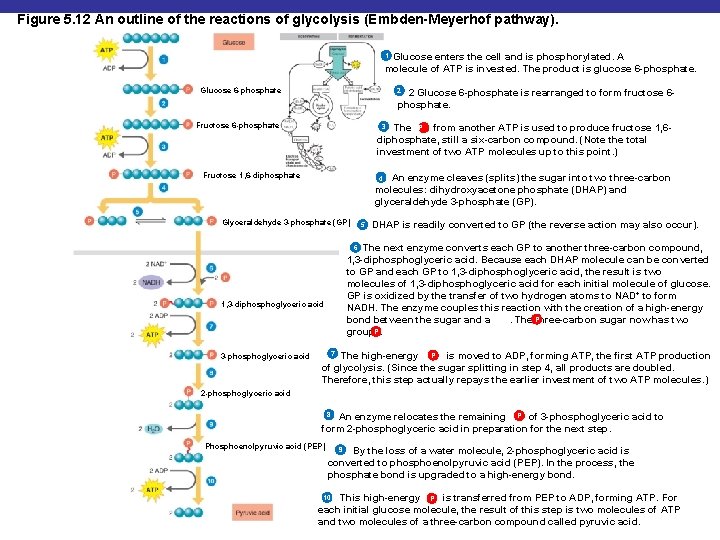
Figure 5. 12 An outline of the reactions of glycolysis (Embden-Meyerhof pathway). 1 Glucose enters the cell and is phosphorylated. A molecule of ATP is invested. The product is glucose 6 -phosphate. Glucose 6 -phosphate 2 2 Glucose 6 -phosphate is rearranged to form fructose 6 phosphate. Fructose 6 -phosphate 3 The P from another ATP is used to produce fructose 1, 6 diphosphate, still a six-carbon compound. (Note the total investment of two ATP molecules up to this point. ) Fructose 1, 6 -diphosphate 4 An enzyme cleaves (splits) the sugar into two three-carbon molecules: dihydroxyacetone phosphate (DHAP) and glyceraldehyde 3 -phosphate (GP). Glyceraldehyde 3 -phosphate (GP) DHAP is readily converted to GP (the reverse action may also occur). 6 The next enzyme converts each GP to another three-carbon compound, 1, 3 -diphosphoglyceric acid. Because each DHAP molecule can be converted to GP and each GP to 1, 3 -diphosphoglyceric acid, the result is two molecules of 1, 3 -diphosphoglyceric acid for each initial molecule of glucose. GP is oxidized by the transfer of two hydrogen atoms to NAD+ to form NADH. The enzyme couples this reaction with the creation of a high-energy bond between the sugar and a. The Pthree-carbon sugar now has two P groups. 1, 3 -diphosphoglyceric acid 3 -phosphoglyceric acid 5 7 The high-energy P is moved to ADP, forming ATP, the first ATP production of glycolysis. (Since the sugar splitting in step 4, all products are doubled. Therefore, this step actually repays the earlier investment of two ATP molecules. ) 2 -phosphoglyceric acid 8 An enzyme relocates the remaining P of 3 -phosphoglyceric acid to form 2 -phosphoglyceric acid in preparation for the next step. Phosphoenolpyruvic acid (PEP) 9 By the loss of a water molecule, 2 -phosphoglyceric acid is converted to phosphoenolpyruvic acid (PEP). In the process, the phosphate bond is upgraded to a high-energy bond. This high-energy P is transferred from PEP to ADP, forming ATP. For each initial glucose molecule, the result of this step is two molecules of ATP and two molecules of a three-carbon compound called pyruvic acid. 10

Energy-Conserving Stage of Glycolysis § 2 glucose-3 -phosphate are oxidized to 2 pyruvic acid § 4 ATP are produced § 2 NADH are produced

Glycolysis § Glucose + 2 ATP + 2 ADP + 2 PO 4– + 2 NAD+ 2 pyruvic acid + 4 ATP + 2 NADH + 2 H+

Cellular Respiration § Oxidation of molecules liberates electrons for an electron transport chain § ATP is generated by oxidative phosphorylation

Intermediate Step § Pyruvic acid (from glycolysis) is oxidized and decarboxylated

The Krebs Cycle § Oxidation of acetyl Co. A produces NADH and FADH 2
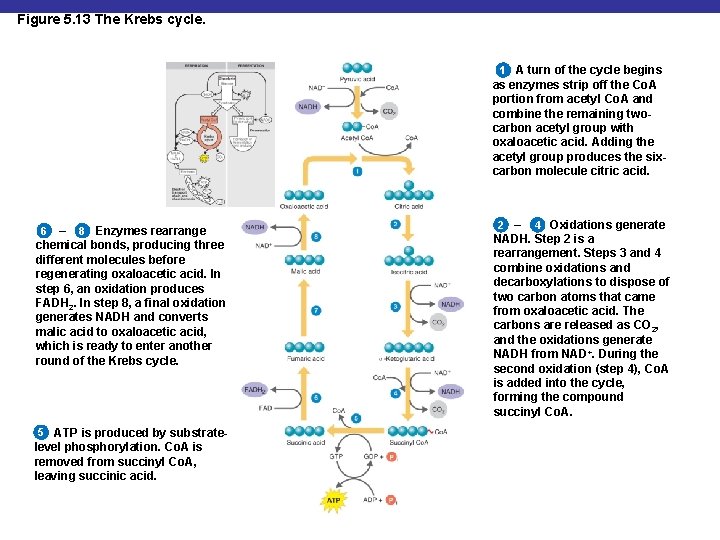
Figure 5. 13 The Krebs cycle. 1 A turn of the cycle begins as enzymes strip off the Co. A portion from acetyl Co. A and combine the remaining twocarbon acetyl group with oxaloacetic acid. Adding the acetyl group produces the sixcarbon molecule citric acid. – 8 Enzymes rearrange chemical bonds, producing three different molecules before regenerating oxaloacetic acid. In step 6, an oxidation produces FADH 2. In step 8, a final oxidation generates NADH and converts malic acid to oxaloacetic acid, which is ready to enter another round of the Krebs cycle. 6 5 ATP is produced by substratelevel phosphorylation. Co. A is removed from succinyl Co. A, leaving succinic acid. 2 – 4 Oxidations generate NADH. Step 2 is a rearrangement. Steps 3 and 4 combine oxidations and decarboxylations to dispose of two carbon atoms that came from oxaloacetic acid. The carbons are released as CO 2, and the oxidations generate NADH from NAD+. During the second oxidation (step 4), Co. A is added into the cycle, forming the compound succinyl Co. A.

The Electron Transport Chain § A series of carrier molecules that are, in turn, oxidized and reduced as electrons are passed down the chain § Energy released can be used to produce ATP by chemiosmosis
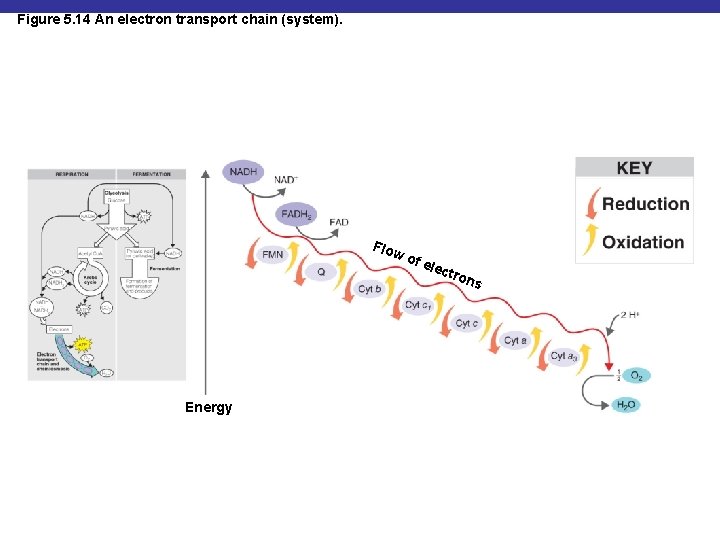
Figure 5. 14 An electron transport chain (system). Flo w of e le ctro Energy ns
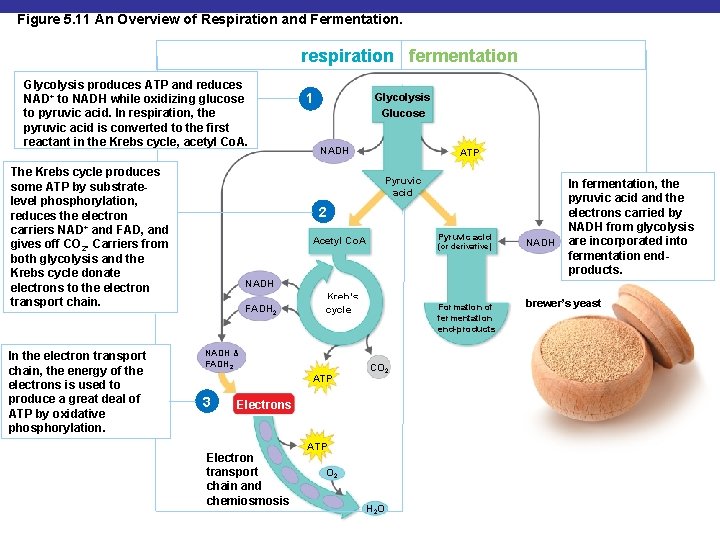
Figure 5. 11 An Overview of Respiration and Fermentation. respiration fermentation Glycolysis produces ATP and reduces NAD+ to NADH while oxidizing glucose to pyruvic acid. In respiration, the pyruvic acid is converted to the first reactant in the Krebs cycle, acetyl Co. A. The Krebs cycle produces some ATP by substratelevel phosphorylation, reduces the electron carriers NAD+ and FAD, and gives off CO 2. Carriers from both glycolysis and the Krebs cycle donate electrons to the electron transport chain. In the electron transport chain, the energy of the electrons is used to produce a great deal of ATP by oxidative phosphorylation. 1 Glycolysis Glucose NADH ATP Pyruvic acid 2 Pyruvic acid Acetyl Co. A (or derivative) NADH In fermentation, the pyruvic acid and the electrons carried by NADH from glycolysis are incorporated into fermentation endproducts. NADH FADH 2 Kreb’s cycle NADH & FADH 2 ATP 3 Formation of fermentation end-products CO 2 Electrons Electron transport chain and chemiosmosis ATP O 2 H 2 O brewer’s yeast
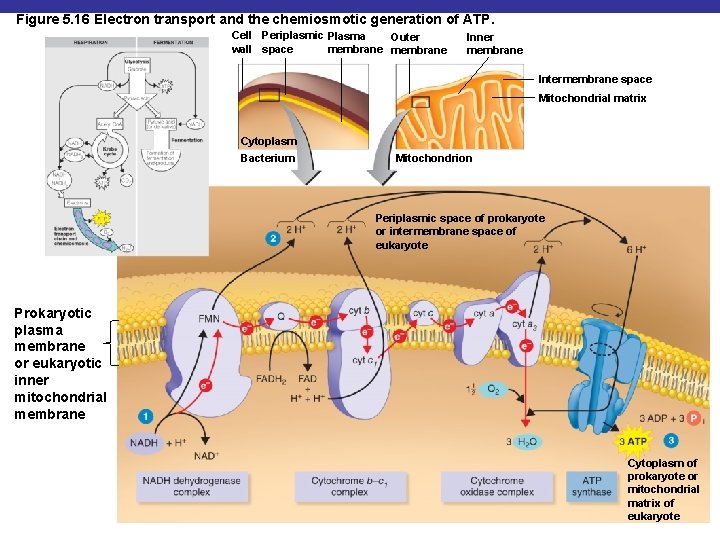
Figure 5. 16 Electron transport and the chemiosmotic generation of ATP. Cell Periplasmic Plasma Outer wall space membrane Inner membrane Intermembrane space Mitochondrial matrix Cytoplasm Bacterium Mitochondrion Periplasmic space of prokaryote or intermembrane space of eukaryote Prokaryotic plasma membrane or eukaryotic inner mitochondrial membrane Cytoplasm of prokaryote or mitochondrial matrix of eukaryote
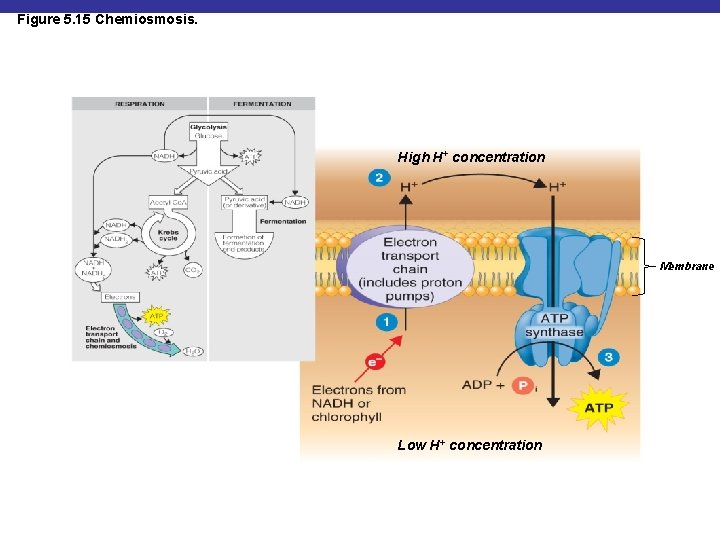
Figure 5. 15 Chemiosmosis. High H+ concentration Membrane Low H+ concentration

A Summary of Respiration § Aerobic respiration: the final electron acceptor in the electron transport chain is molecular oxygen (O 2) § Anaerobic respiration: the final electron acceptor in the electron transport chain is NOT O 2 § Yields less energy than aerobic respiration because only part of the Krebs cycle operates under anaerobic conditions

Carbohydrate Catabolism Pathway Eukaryote Prokaryote Glycolysis Cytoplasm Intermediate step Cytoplasm Krebs cycle Mitochondrial matrix Cytoplasm ETC Mitochondrial inner membrane Plasma membrane
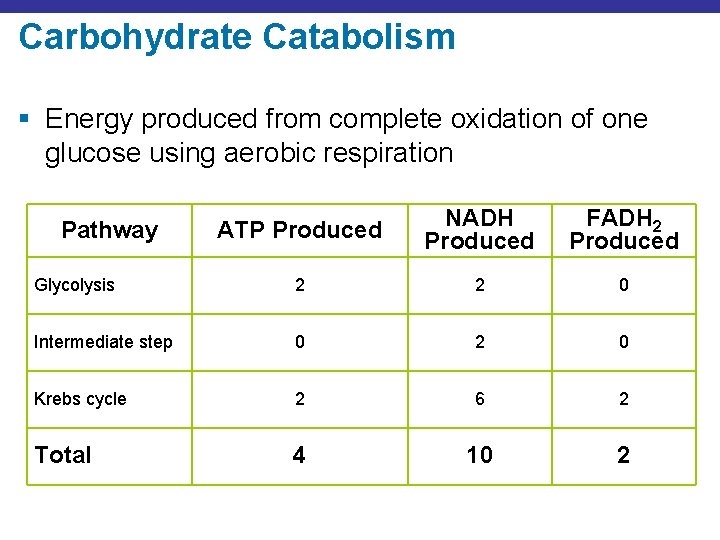
Carbohydrate Catabolism § Energy produced from complete oxidation of one glucose using aerobic respiration ATP Produced NADH Produced FADH 2 Produced Glycolysis 2 2 0 Intermediate step 0 2 0 Krebs cycle 2 6 2 Total 4 10 2 Pathway

Carbohydrate Catabolism § ATP produced from complete oxidation of one glucose using aerobic respiration Pathway By Substrate-Level Phosphorylation By Oxidative Phosphorylation From NADH From FADH Glycolysis 2 6 0 Intermediate step 0 6 0 Krebs cycle 2 18 4 Total 4 30 4

Carbohydrate Catabolism § 36 ATPs are produced in eukaryotes Pathway By Substrate-Level Phosphorylation By Oxidative Phosphorylation From NADH From FADH Glycolysis 2 6 0 Intermediate step 0 6 0 Krebs cycle 2 18 4 Total 4 30 4

Fermentation § Any spoilage of food by microorganisms (general use) § Any process that produces alcoholic beverages or acidic dairy products (general use) § Any large-scale microbial process occurring with or without air (common definition used in industry)

Fermentation § Scientific definition: § § Releases energy from oxidation of organic molecules Does not require oxygen Does not use the Krebs cycle or ETC Uses an organic molecule as the final electron acceptor
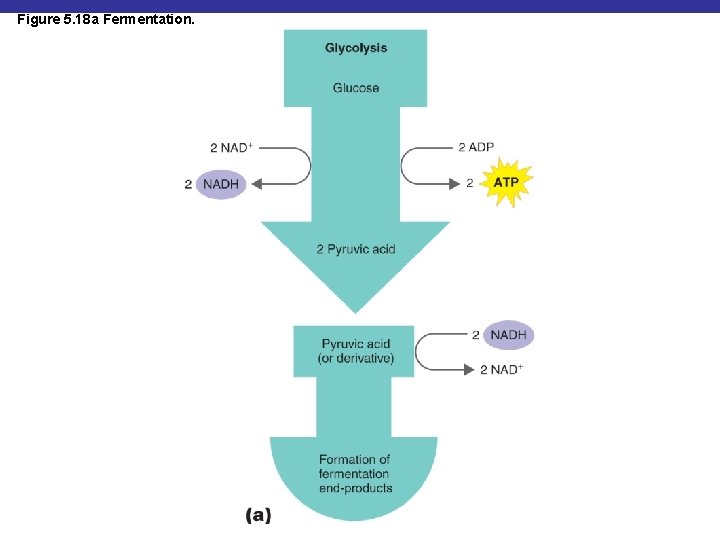
Figure 5. 18 a Fermentation.
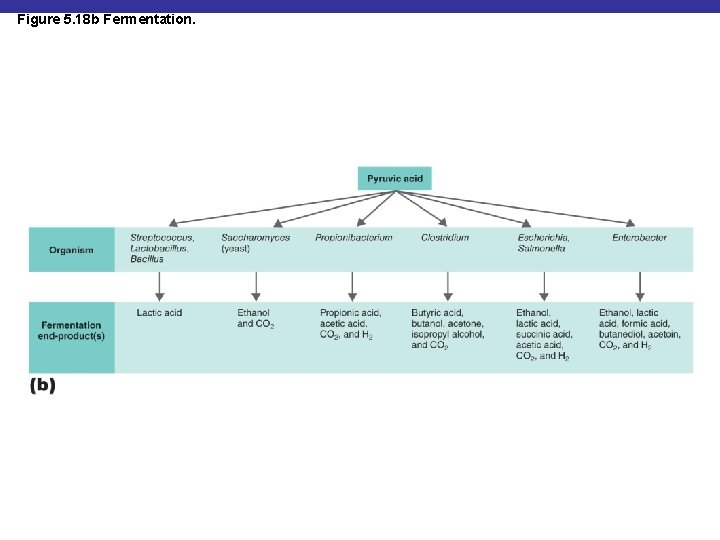
Figure 5. 18 b Fermentation.

Fermentation § Alcohol fermentation: produces ethanol + CO 2 § Lactic acid fermentation: produces lactic acid § Homolactic fermentation: produces lactic acid only § Heterolactic fermentation: produces lactic acid and other compounds
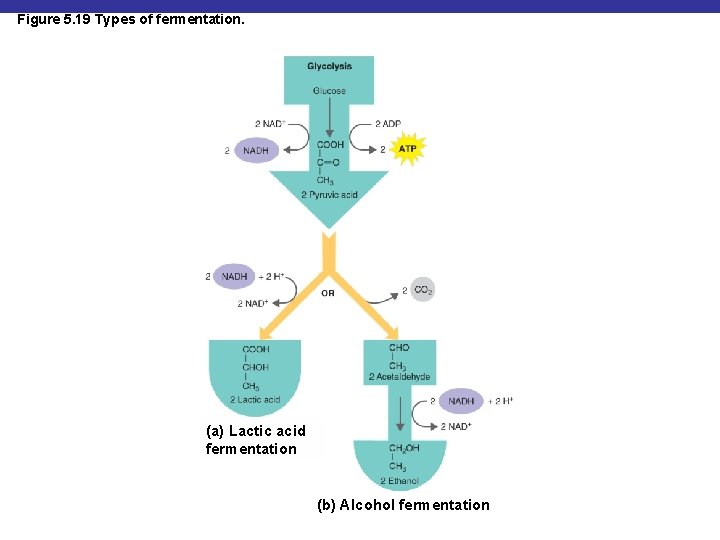
Figure 5. 19 Types of fermentation. (a) Lactic acid fermentation (b) Alcohol fermentation

Figure 5. 23 A fermentation test.
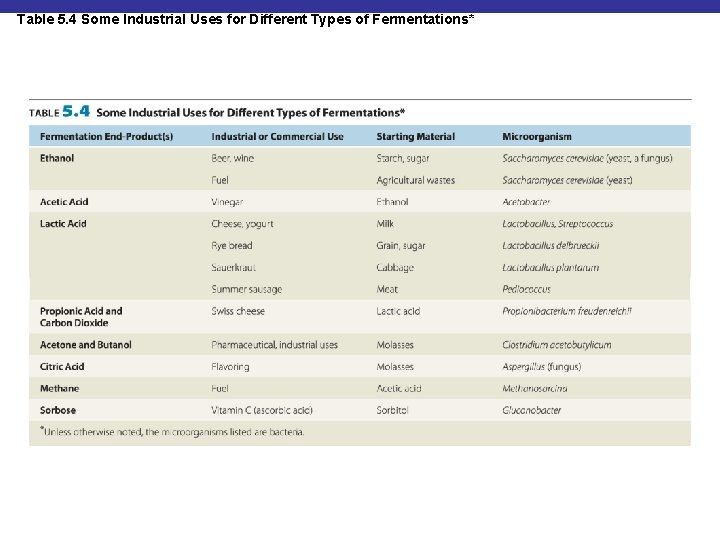
Table 5. 4 Some Industrial Uses for Different Types of Fermentations*
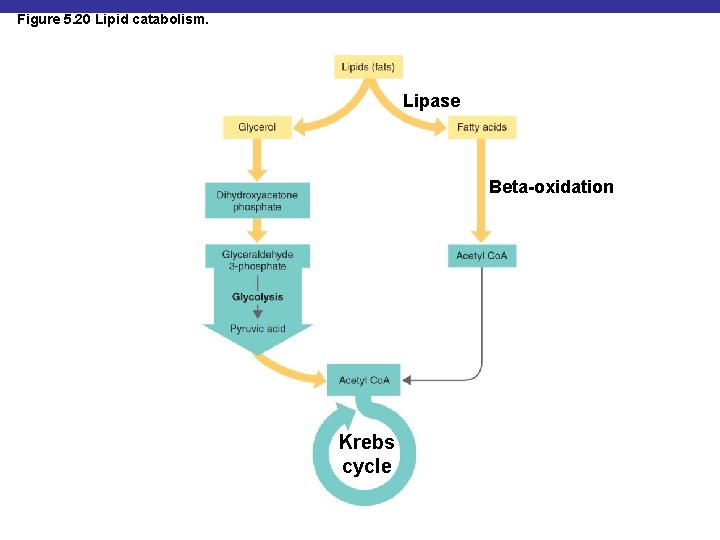
Figure 5. 20 Lipid catabolism. Lipase Beta-oxidation Krebs cycle
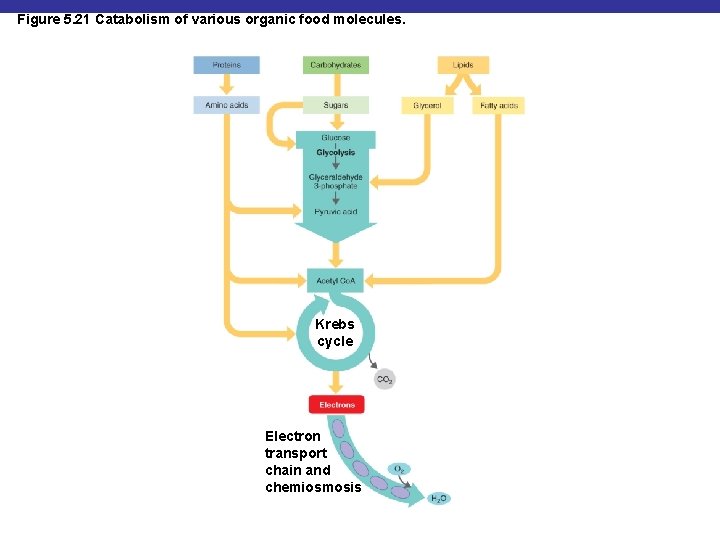
Figure 5. 21 Catabolism of various organic food molecules. Krebs cycle Electron transport chain and chemiosmosis
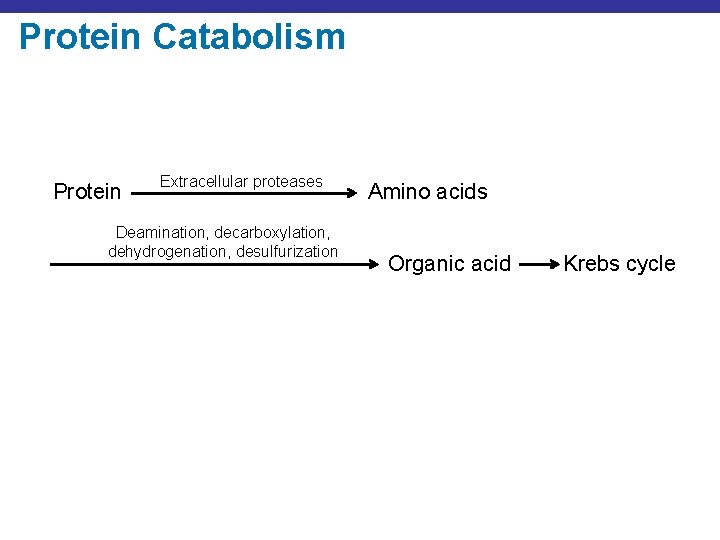
Protein Catabolism Protein Extracellular proteases Deamination, decarboxylation, dehydrogenation, desulfurization Amino acids Organic acid Krebs cycle
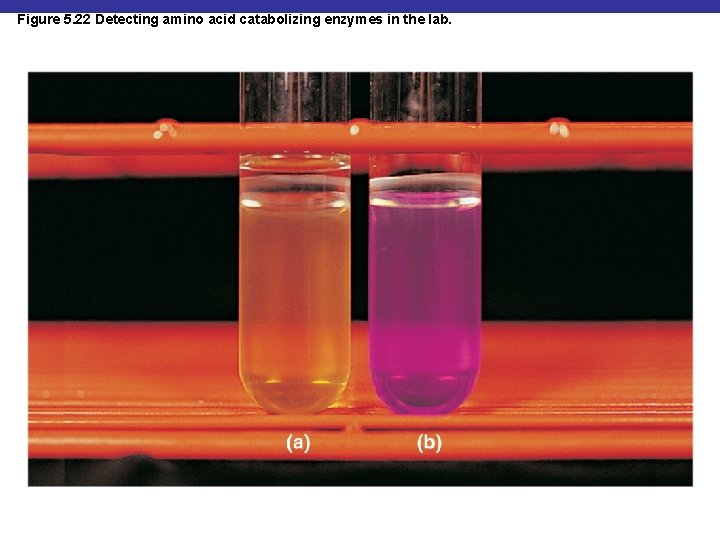
Figure 5. 22 Detecting amino acid catabolizing enzymes in the lab.
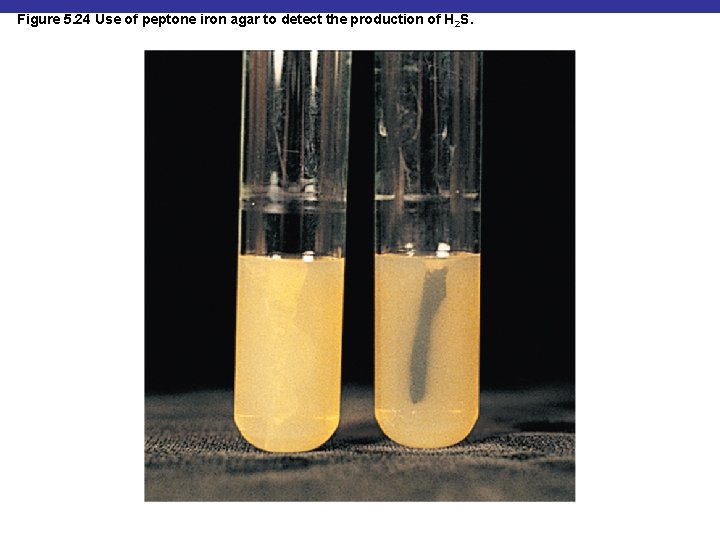
Figure 5. 24 Use of peptone iron agar to detect the production of H 2 S.
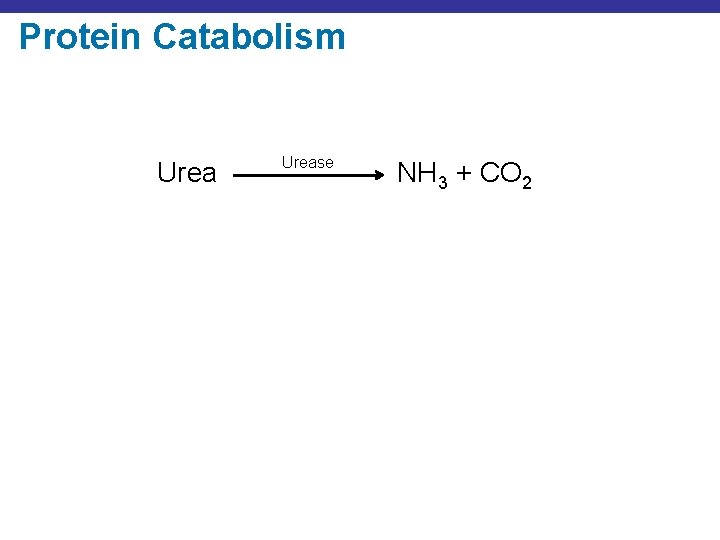
Protein Catabolism Urease NH 3 + CO 2

Clinical Focus: Human Tuberculosis – Dallas, Texas Figure B The urease test.

Biochemical Tests § Used to identify bacteria
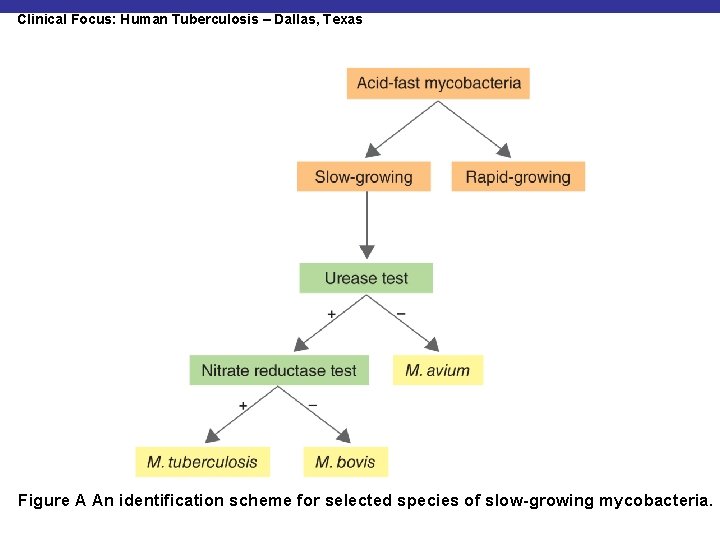
Clinical Focus: Human Tuberculosis – Dallas, Texas Figure A An identification scheme for selected species of slow-growing mycobacteria.

Photosynthesis § Photo: conversion of light energy into chemical energy (ATP) § Light-dependent (light) reactions § Synthesis: § Carbon fixation: fixing carbon into organic molecules § Light-independent (dark) reaction: Calvin-Benson cycle
Figure 4. 15 Chromatophores © 2013 Pearson Education, Inc.
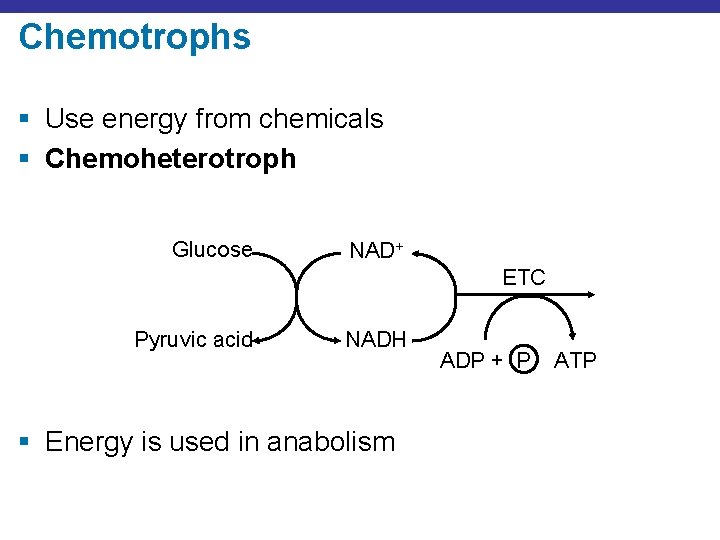
Chemotrophs § Use energy from chemicals § Chemoheterotroph Glucose NAD+ ETC Pyruvic acid NADH § Energy is used in anabolism ADP + P ATP
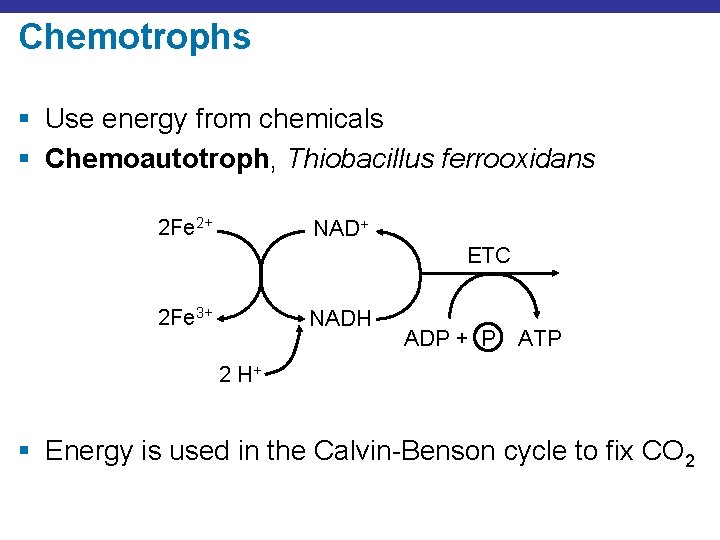
Chemotrophs § Use energy from chemicals § Chemoautotroph, Thiobacillus ferrooxidans 2 Fe 2+ NAD+ ETC 2 Fe 3+ NADH ADP + P ATP 2 H+ § Energy is used in the Calvin-Benson cycle to fix CO 2

Phototrophs § Use light energy Chlorophyll ETC Chlorophyll oxidized ADP + P ATP § Photoautotrophs use energy in the Calvin-Benson cycle to fix CO 2 § Photoheterotrophs use energy
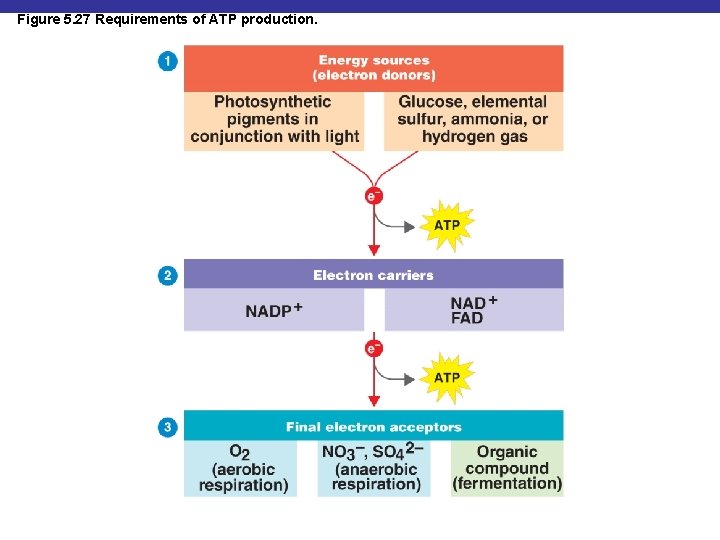
Figure 5. 27 Requirements of ATP production.
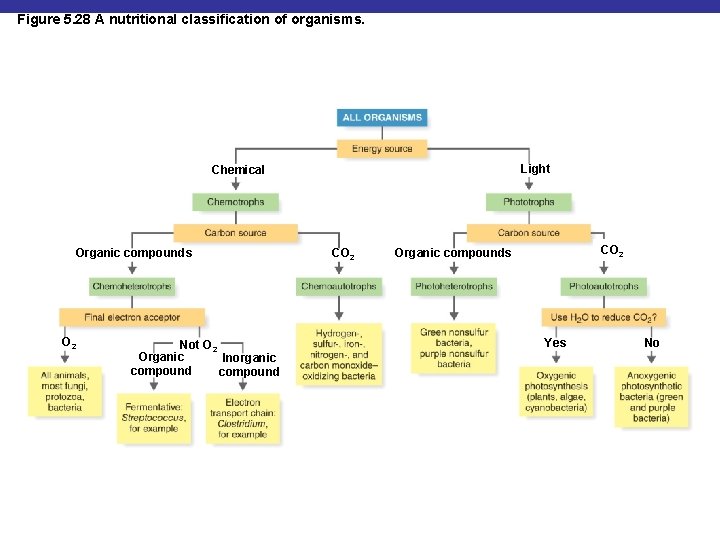
Figure 5. 28 A nutritional classification of organisms. Light Chemical Organic compounds O 2 Not O 2 Organic Inorganic compound CO 2 Organic compounds Yes No
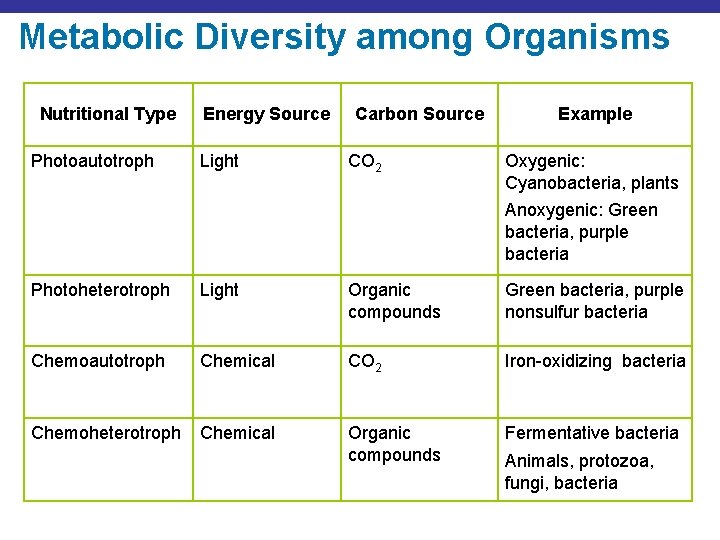
Metabolic Diversity among Organisms Nutritional Type Photoautotroph Energy Source Light Carbon Source CO 2 Example Oxygenic: Cyanobacteria, plants Anoxygenic: Green bacteria, purple bacteria Photoheterotroph Light Organic compounds Green bacteria, purple nonsulfur bacteria Chemoautotroph Chemical CO 2 Iron-oxidizing bacteria Chemoheterotroph Chemical Organic compounds Fermentative bacteria Animals, protozoa, fungi, bacteria
- Slides: 80