Caspofungin Breakthrough Treatment in the Management of Patients

Caspofungin Breakthrough Treatment in the Management of Patients with Invasive Candidiasis

Overview: Growing Threat • Serious fungal infections are on the rise • Invasive Candida infections – 4 th most common nosocomial bloodstream infection in the United States* Pathogen No. of Isolates Incidence (%) 3908 1928 1354 934 31. 9 15. 7 11. 1 7. 6 Coagulase-negative staphylococci Staphylococcus aureus Enterococci Candida species * In a 3 -year (1995 -1998) surveillance study of 49 hospitals in the United States. Adapted from Edmond MB et al Clin Infect Dis 1999; 29: 239 -244. Andriole VT J Antimicrob Chemother 1999; 44: 151 -162; Uzun O, Anaissie EJ Ann Oncol 2000; 11: 1517 -1521; Edmond MB, Wallace SE, Mc. Clish DK, Pfaller MA, Jones RN, Wenzel RP Clin Infect Dis 1999; 29: 239 -244.
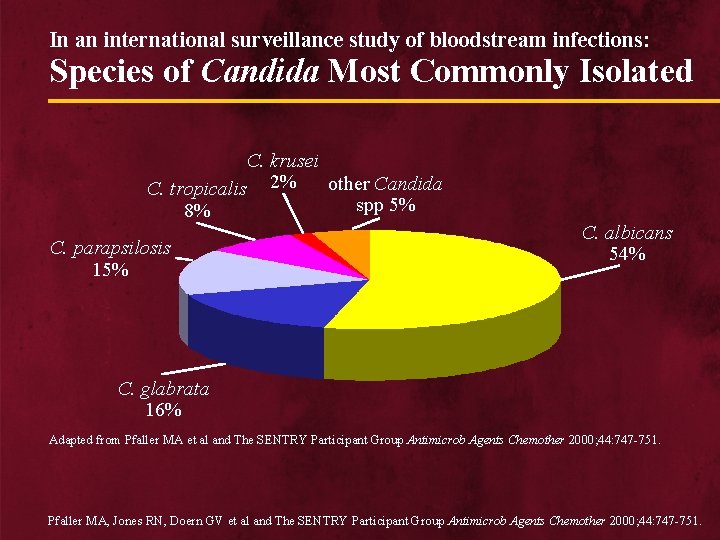
In an international surveillance study of bloodstream infections: Species of Candida Most Commonly Isolated C. krusei other Candida C. tropicalis 2% spp 5% 8% C. parapsilosis 15% C. albicans 54% C. glabrata 16% Adapted from Pfaller MA et al and The SENTRY Participant Group Antimicrob Agents Chemother 2000; 44: 747 -751. Pfaller MA, Jones RN, Doern GV et al and The SENTRY Participant Group Antimicrob Agents Chemother 2000; 44: 747 -751.
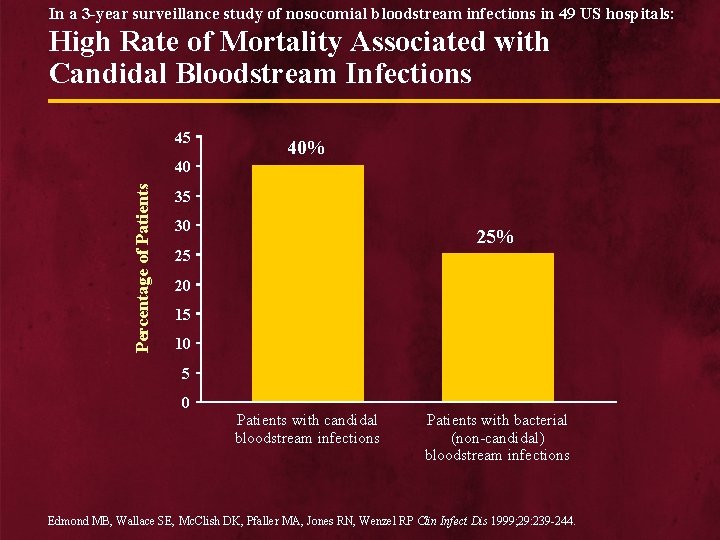
In a 3 -year surveillance study of nosocomial bloodstream infections in 49 US hospitals: High Rate of Mortality Associated with Candidal Bloodstream Infections 45 Percentage of Patients 40 40% 35 30 25% 25 20 15 10 5 0 Patients with candidal bloodstream infections Patients with bacterial (non-candidal) bloodstream infections Edmond MB, Wallace SE, Mc. Clish DK, Pfaller MA, Jones RN, Wenzel RP Clin Infect Dis 1999; 29: 239 -244.

Patients at High Risk Potential risk factors include: Non-Neutropenic • Acute renal failure • Cancer • Parenteral nutrition • Transplantation • Anti-anaerobic agents • Broad spectrum anti-anaerobic antibiotic use • Prior vancomycin use • Intralipid agents • Immunocompromised state • Prior surgery • Surgery • Indwelling triple-lumen catheters • Indwelling catheters Blumberg HM, Jarvis WR, Soucie JM et al and the NEMIS Study Group Clin Infect Dis 2001; 33: 177 -186; Garber G Drugs 2001; 61(suppl 1): 1 -12. National Epidemiology of Mycosis Survey (NEMIS) was a prospective, multicenter study conducted at 6 US sites from 1993 -1995 to examine rates of risk factors for the development of candidal bloodstream infections (CBSIs) among patients in surgical and neonatal ICUs >48 h. Among 4276 patients, 42 CBSIs occurred.
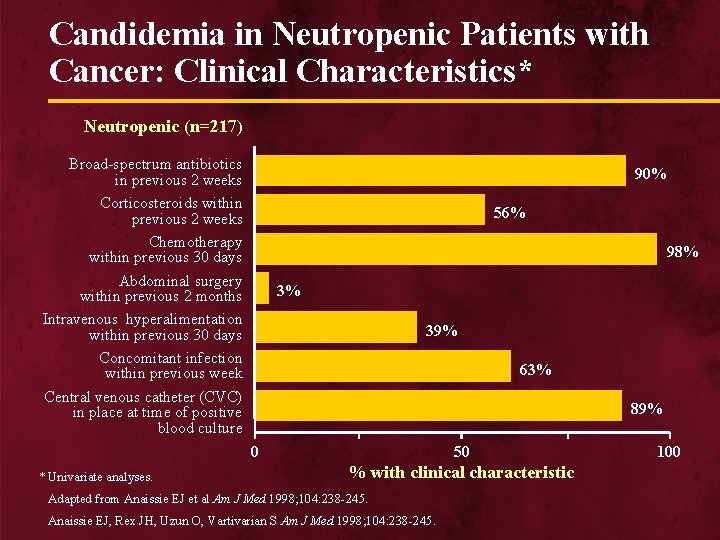
Candidemia in Neutropenic Patients with Cancer: Clinical Characteristics* Neutropenic (n=217) Broad-spectrum antibiotics in previous 2 weeks Corticosteroids within previous 2 weeks Chemotherapy within previous 30 days Abdominal surgery within previous 2 months Intravenous hyperalimentation within previous 30 days Concomitant infection within previous week Central venous catheter (CVC) in place at time of positive blood culture 90% 56% 98% 3% 39% 63% 89% 0 * Univariate analyses. 50 % with clinical characteristic Adapted from Anaissie EJ et al Am J Med 1998; 104: 238 -245. Anaissie EJ, Rex JH, Uzun O, Vartivarian S Am J Med 1998; 104: 238 -245. 100
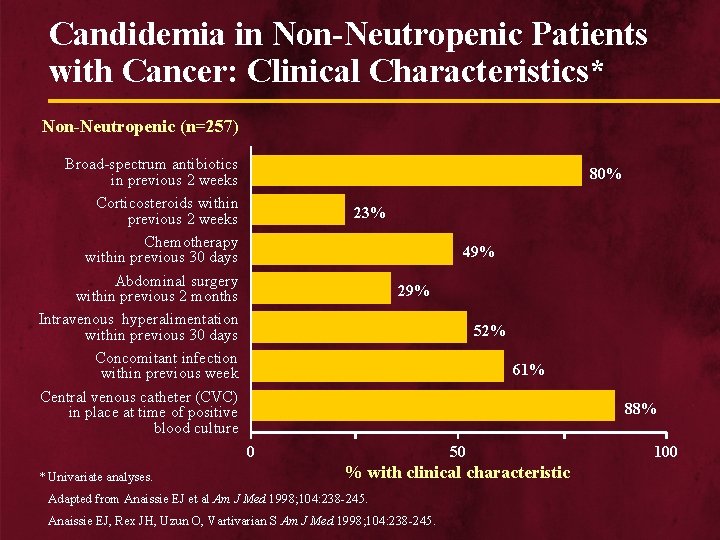
Candidemia in Non-Neutropenic Patients with Cancer: Clinical Characteristics* Non-Neutropenic (n=257) Broad-spectrum antibiotics in previous 2 weeks Corticosteroids within previous 2 weeks Chemotherapy within previous 30 days Abdominal surgery within previous 2 months Intravenous hyperalimentation within previous 30 days Concomitant infection within previous week Central venous catheter (CVC) in place at time of positive blood culture 80% 23% 49% 29% 52% 61% 88% 0 * Univariate analyses. 50 % with clinical characteristic Adapted from Anaissie EJ et al Am J Med 1998; 104: 238 -245. Anaissie EJ, Rex JH, Uzun O, Vartivarian S Am J Med 1998; 104: 238 -245. 100
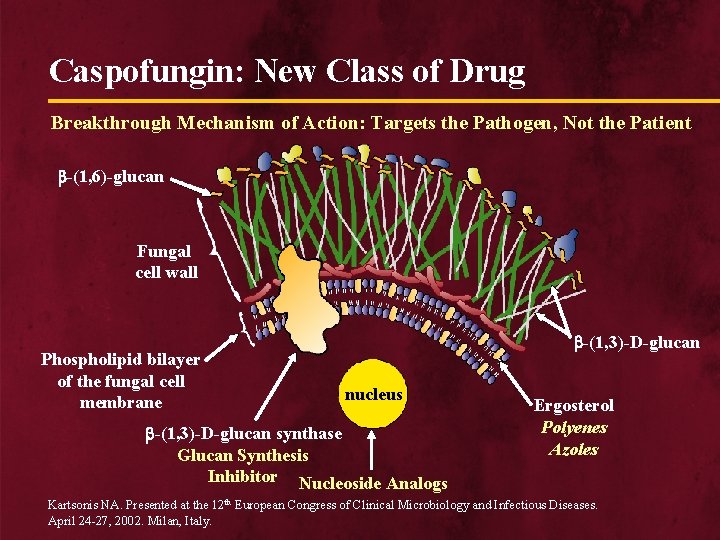
Caspofungin: New Class of Drug Breakthrough Mechanism of Action: Targets the Pathogen, Not the Patient -(1, 6)-glucan Fungal cell wall Phospholipid bilayer of the fungal cell membrane -(1, 3)-D-glucan nucleus -(1, 3)-D-glucan synthase Glucan Synthesis Inhibitor Nucleoside Analogs Ergosterol Polyenes Azoles Kartsonis NA. Presented at the 12 th European Congress of Clinical Microbiology and Infectious Diseases. April 24 -27, 2002. Milan, Italy.

Caspofungin: Broad Spectrum of Activity CANDIDA ALBICANS C. g C. labr pa ata C. rap tr s C. opic ilosi a s k C. ruse lis gu i C. illi lip erm C. oly on d d ti C. ublin ca ii k i C. efyr ensis lu C. sita r ni C. ugos ae ps a eu do tro A. pic fla ali v s u A. fum s A. iga ter tu A. reu s nig s A. er nid ula ns C. alb i ca ns Expanded Spectrum of In Vitro Activity CANDIDA NON-ALBICANS ASPERGILLUS Data on file, MSD; Bartizal K, Gill CJ, Abruzzo GK et al Antimicrob Agents Chemother 1997; 41: 2326 -2332.

Unique Mechanism of Action (MOA) Offers Favorable Resistance Profile • Active in vitro against fluconazole-, amphotericin B-, or flucytosine-resistant Candida • Not cross-resistant with azoles or polyenes • Not intrinsically resistant to Candida isolates Data on file, MSD; Graybill JR Int J Clin Pract 2001; 55(9): 633 -638; Pfaller MA, Jones RN, Doern GV et al Diagn Microbiol Infect Dis 1999; 35: 19 -25.

Caspofungin: Indication • NEW: Invasive candidiasis including candidemia in neutropenic and non-neutropenic patients In addition to: • Invasive aspergillosis in patients who are refractory to or intolerant of standard therapies • Esophageal candidiasis • Oropharyngeal candidiasis Data on file, MSD.

Caspofungin: Proven Antifungal Efficacy against Invasive Candidiasis Clinical Trial: Protocol 014 Caspofungin vs. Amphotericin B Deoxycholate in the Treatment of Invasive Candidiasis in Neutropenic and Non-Neutropenic Patients Data on file, MSD.

Protocol 014: Objective To compare the proportion of caspofungin acetate patients with both a favorable clinical response and a favorable microbiological assessment at the time of discontinuing IV antifungal therapy with that of amphotericin B patients Data on file, MSD.

Protocol 014: Design • Multicenter, randomized, double-blind, comparative study • To compare the proportion of caspofungin patients with a favorable clinical response and a favorable microbiological assessment at the time of discontinuing IV antifungal therapy with that of amphotericin B patients • Patients ( 18 years old) stratified by neutropenic status Caspofungin: 114 pts (92 with candidemia) Amphotericin B: 125 pts (92 with candidemia) — 50 mg/day — 0. 7– 1. 0 mg/kg/day (70 mg loading dose on day 1) neutropenic patients — 0. 6– 0. 7 mg/kg/day non-neutropenic patients Data on file, MSD.
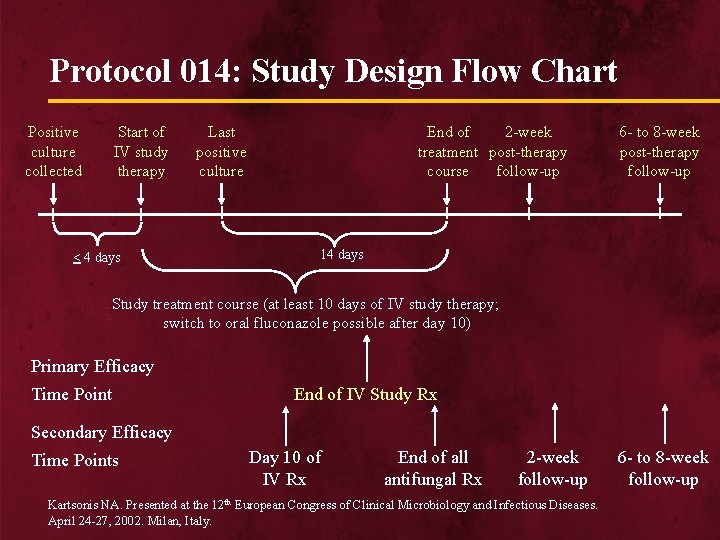
Protocol 014: Study Design Flow Chart Positive culture collected Start of IV study therapy < 4 days Last positive culture End of 2 -week treatment post-therapy course follow-up 6 - to 8 -week post-therapy follow-up 14 days Study treatment course (at least 10 days of IV study therapy; switch to oral fluconazole possible after day 10) Primary Efficacy Time Point End of IV Study Rx Secondary Efficacy Time Points Day 10 of IV Rx End of all antifungal Rx 2 -week follow-up Kartsonis NA. Presented at the 12 th European Congress of Clinical Microbiology and Infectious Diseases. April 24 -27, 2002. Milan, Italy. 6 - to 8 -week follow-up

Protocol 014: Efficacy Evaluation— Diagnostic Criteria • Favorable clinical response – Complete resolution of signs/symptoms of Candida • Favorable microbiological response or presumptive eradication – Candida eradication from follow-up cultures • Definition of comparability – 95. 6% confidence interval (CI) difference between groups Data on file, MSD.

Protocol 014: Primary Efficacy Endpoint • Proportion of patients with favorable overall response (favorable clinical and microbiological response) at end of IV therapy – Modified Intent-To-Treat (MITT): primary assessment criteria • Patients received 1 day IV study therapy – Evaluable Patients (EP): secondary assessment analysis • Patients met entry criteria, received IV study therapy 5 days, and had full efficacy evaluation at the end of IV study therapy Data on file, MSD.
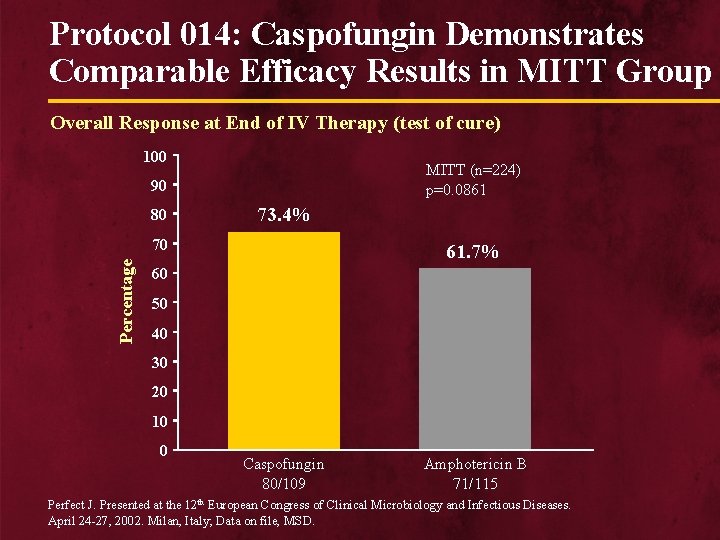
Protocol 014: Caspofungin Demonstrates Comparable Efficacy Results in MITT Group Overall Response at End of IV Therapy (test of cure) 100 MITT (n=224) p=0. 0861 90 80 73. 4% Percentage 70 61. 7% 60 50 40 30 20 10 0 Caspofungin 80/109 Amphotericin B 71/115 Perfect J. Presented at the 12 th European Congress of Clinical Microbiology and Infectious Diseases. April 24 -27, 2002. Milan, Italy; Data on file, MSD.
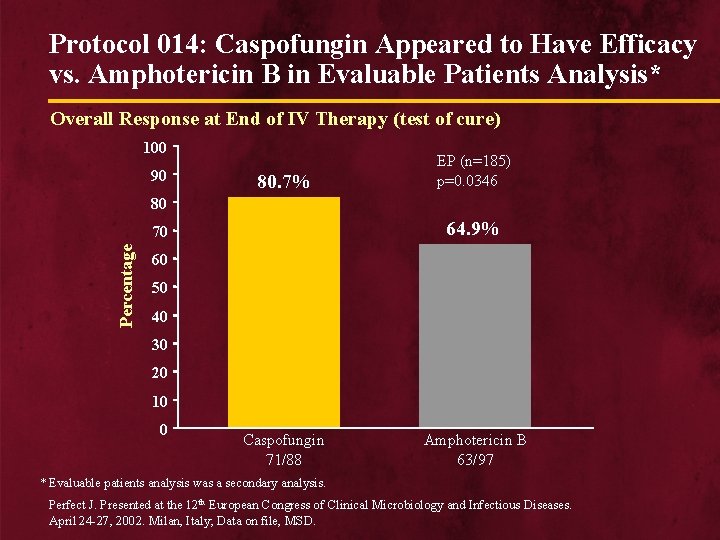
Protocol 014: Caspofungin Appeared to Have Efficacy vs. Amphotericin B in Evaluable Patients Analysis* Overall Response at End of IV Therapy (test of cure) 100 90 80. 7% EP (n=185) p=0. 0346 80 64. 9% Percentage 70 60 50 40 30 20 10 0 Caspofungin 71/88 Amphotericin B 63/97 * Evaluable patients analysis was a secondary analysis. Perfect J. Presented at the 12 th European Congress of Clinical Microbiology and Infectious Diseases. April 24 -27, 2002. Milan, Italy; Data on file, MSD.
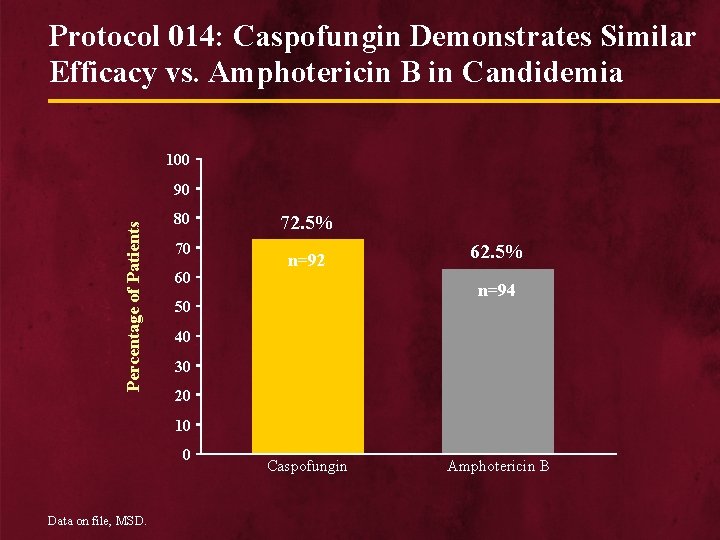
Protocol 014: Caspofungin Demonstrates Similar Efficacy vs. Amphotericin B in Candidemia 100 Percentage of Patients 90 80 70 60 72. 5% n=92 62. 5% n=94 50 40 30 20 10 0 Data on file, MSD. Caspofungin Amphotericin B
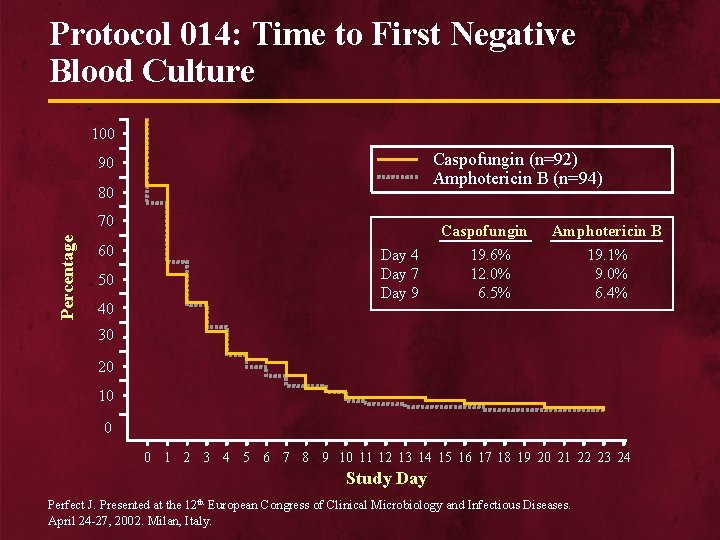
Protocol 014: Time to First Negative Blood Culture 100 Caspofungin (n=92) Amphotericin B (n=94) 90 80 Percentage 70 60 50 40 Caspofungin Day 4 Day 7 Day 9 Amphotericin B 19. 6% 12. 0% 6. 5% 19. 1% 9. 0% 6. 4% 30 20 10 0 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 Study Day Perfect J. Presented at the 12 th European Congress of Clinical Microbiology and Infectious Diseases. April 24 -27, 2002. Milan, Italy.
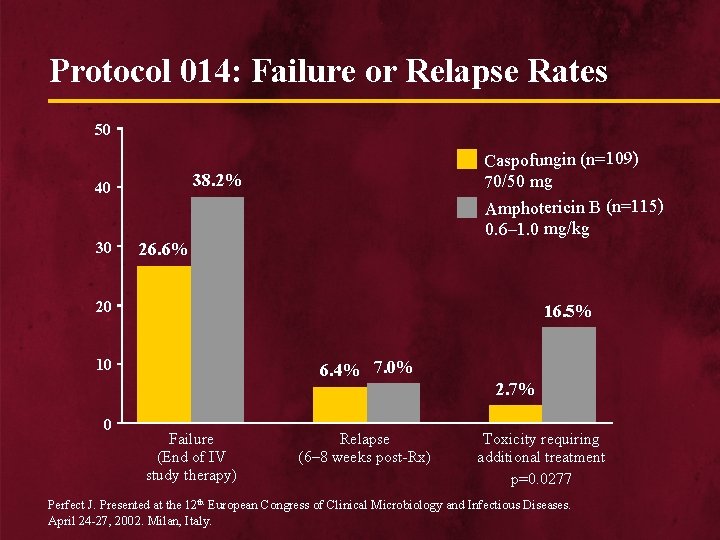
Protocol 014: Failure or Relapse Rates 50 38. 2% 40 30 Caspofungin (n=109) 70/50 mg Amphotericin B (n=115) 0. 6– 1. 0 mg/kg 26. 6% 20 16. 5% 10 0 6. 4% 7. 0% Failure (End of IV study therapy) Relapse (6– 8 weeks post-Rx) 2. 7% Toxicity requiring additional treatment p=0. 0277 Perfect J. Presented at the 12 th European Congress of Clinical Microbiology and Infectious Diseases. April 24 -27, 2002. Milan, Italy.
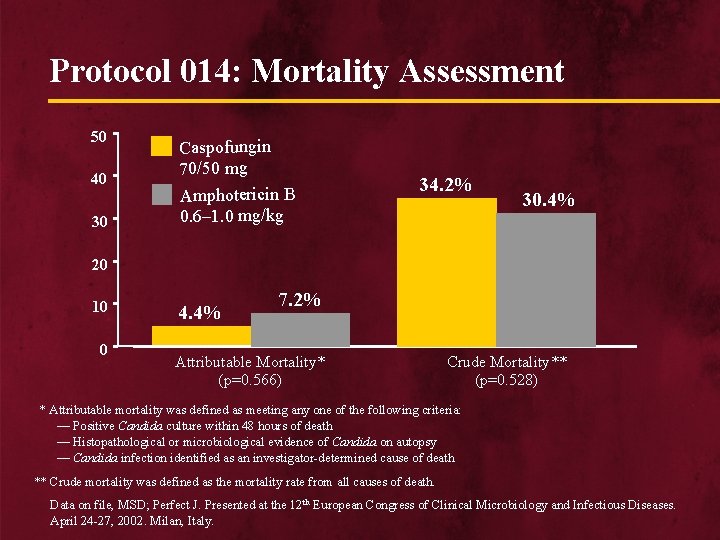
Protocol 014: Mortality Assessment 50 40 30 Caspofungin 70/50 mg Amphotericin B 0. 6– 1. 0 mg/kg 34. 2% 30. 4% 20 10 0 4. 4% 7. 2% Attributable Mortality* (p=0. 566) Crude Mortality** (p=0. 528) * Attributable mortality was defined as meeting any one of the following criteria: — Positive Candida culture within 48 hours of death — Histopathological or microbiological evidence of Candida on autopsy — Candida infection identified as an investigator-determined cause of death ** Crude mortality was defined as the mortality rate from all causes of death. Data on file, MSD; Perfect J. Presented at the 12 th European Congress of Clinical Microbiology and Infectious Diseases. April 24 -27, 2002. Milan, Italy.
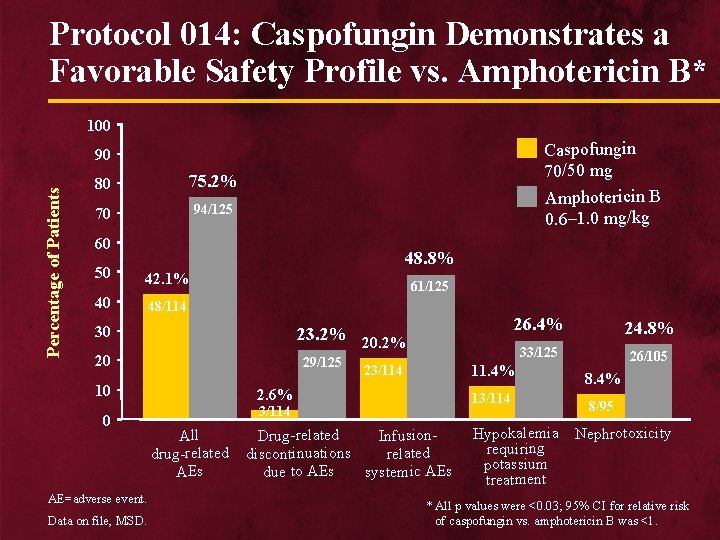
Protocol 014: Caspofungin Demonstrates a Favorable Safety Profile vs. Amphotericin B* 100 Caspofungin 70/50 mg Amphotericin B 0. 6– 1. 0 mg/kg Percentage of Patients 90 80 75. 2% 70 94/125 60 48. 8% 50 42. 1% 40 48/114 61/125 30 23. 2% 20 10 0 AE=adverse event. Data on file, MSD. 29/125 26. 4% 24. 8% 33/125 26/105 11. 4% 23/114 2. 6% 13/114 All Infusion. Drug-related drug-related discontinuations AEs due to AEs systemic AEs Hypokalemia requiring potassium treatment 8. 4% 8/95 Nephrotoxicity * All p values were <0. 03; 95% CI for relative risk of caspofungin vs. amphotericin B was <1.
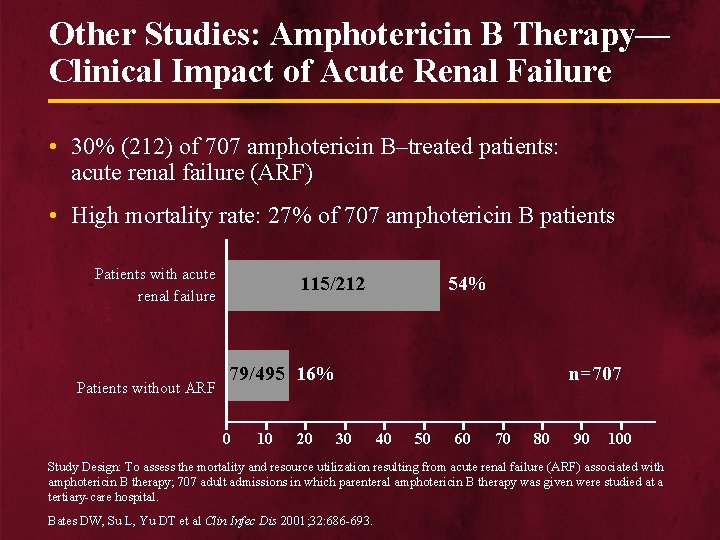
Other Studies: Amphotericin B Therapy— Clinical Impact of Acute Renal Failure • 30% (212) of 707 amphotericin B–treated patients: acute renal failure (ARF) • High mortality rate: 27% of 707 amphotericin B patients Patients with acute renal failure Patients without ARF 115/212 54% 79/495 16% 0 10 20 n=707 30 40 50 60 70 80 90 100 Study Design: To assess the mortality and resource utilization resulting from acute renal failure (ARF) associated with amphotericin B therapy; 707 adult admissions in which parenteral amphotericin B therapy was given were studied at a tertiary-care hospital. Bates DW, Su L, Yu DT et al Clin Infec Dis 2001; 32: 686 -693.

Other Studies: Caspofungin Maintains Favorable Safety/Tolerability Profile • Overall safety assessed in 876 patients • 394 patients enrolled in Phase I studies • Most patients with Candida infections had serious underlying medical conditions, including hematologic or other malignancy, recent major surgery, or HIV • In a clinical study among patients with oropharyngeal or esophageal candidiasis, caspofungin (n=83) demonstrated a comparable tolerability profile vs. fluconazole (n=94) • In a clinical study among patients with invasive candidiasis, caspofungin (n=114) demonstrated a superior tolerability profile to amphotericin B (n=125) • In an open-label, noncomparative aspergillosis (n=69) study, caspofungin maintained its favorable profile Data on file, MSD.

Caspofungin: Minimal Drug Interactions • Not a P 450 (CYP) inhibitor • No antagonistic interaction with amphotericin B or itraconazole • Has been used with antirejection drugs tacrolimus and/or mycophenolate Data on file, MSD; Bartizal K et al Antimicrob Agents Chemother 1997; 41: 2326 -2332.

Caspofungin: Dosing/Administration • Once-daily dosing with 50 mg standard dose • 70 mg loading dose on day 1 for aspergillosis and invasive candidiasis • No premedication necessary • Recommended infusion time: 1 hour • No dosage adjustment in many cases* * For patients with moderate hepatic insufficiency (Child-Pugh score 7 -9), a dose adjustment to 35 mg daily is recommended. There is no clinical experience in patients with severe hepatic insufficiency (Child-Pugh score >9). Data on file, MSD.

Conclusions: Caspofungin— The New Gold Standard • Invasive candidiasis: – Caspofungin is comparable to amphotericin B (MITT analysis) – Caspofungin appears to be superior to amphotericin B (EP analysis) – Candidemia: Caspofungin is comparable to amphotericin B • Overall safety/tolerability profile: – Caspofungin has a favorable tolerability profile Data on file, MSD.

References

References
- Slides: 31