CaseControl Studies Afshin Ostovar Bushehr University of Medical

Case-Control Studies Afshin Ostovar Bushehr University of Medical Sciences Bushehr, 2011 6/11/2021 1
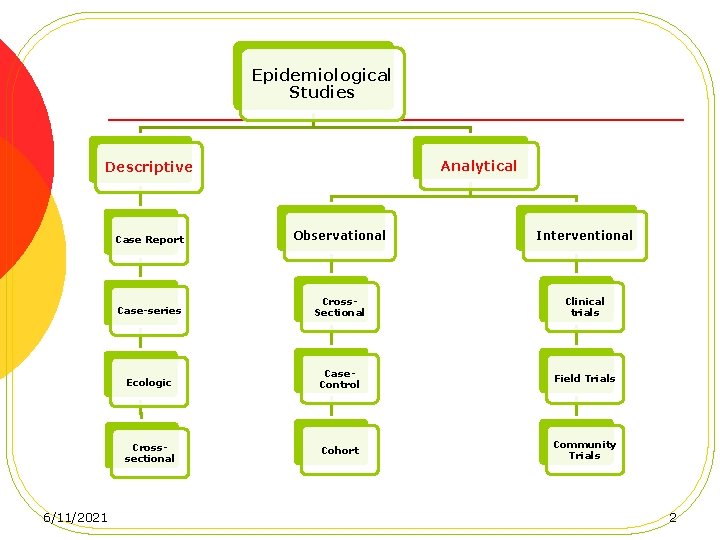
Epidemiological Studies Analytical Descriptive 6/11/2021 Case Report Observational Interventional Case-series Cross. Sectional Clinical trials Ecologic Case. Control Field Trials Crosssectional Cohort Community Trials 2

l The sophisticated use and understanding of case -control studies is the most outstanding methodologic development of modern epidemiology. (Rothman 1986, p. 62) l A primary goal is to reach the same conclusions in a retrospective study as would have been obtained from a forward study, if one had been done. (Mantel and Haenszel 1959, p. 722) 6/11/2021 3

Key advantages 6/11/2021 l Statistically efficient for rare conditions l Logistically efficient for prolonged induction or latency diseases l Can examine many exposures in one study l Ethical - cannot affect onset of disease 4

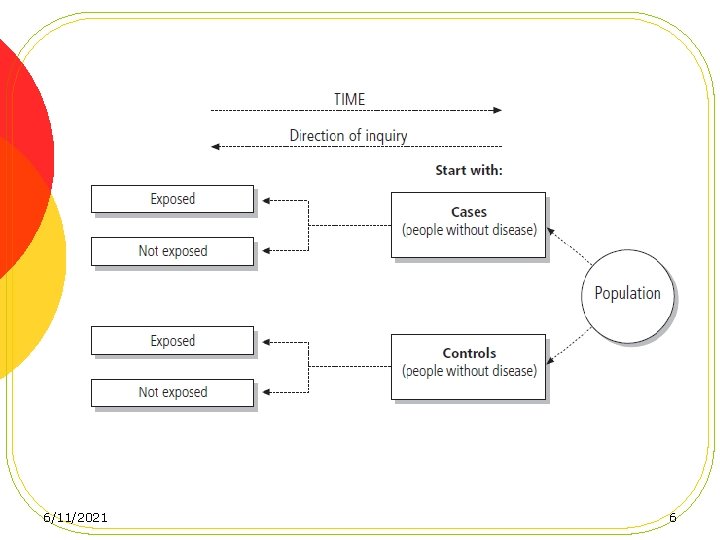
Case-Control Studies l Examines the association between disease and potential risk factors by taking separate samples of diseased cases and of controls at risk of developing disease. l Information may be collected for both cases and controls on genetic, social, behavioral, environmental or other determinants of disease risk. 6/11/2021 5
6/11/2021 6

Odds Ratio (cross-product ratio) 6/11/2021 7

l The initial interpretation of the case-control study was the comparison of exposure histories for a group of diseased cases with those for non-diseased controls. l The critics argued that such comparisons provided no information about the quantities of true epidemiologic interest, namely the disease rates. l Cornfield (1951) corrected this misconception by demonstrating that the exposure odds ratio for cases vs. controls was equal to the disease odds ratio for exposed vs. non-exposed. 6/11/2021 8

l Selection of cases and controls l Matching l Case-control studies based on a defined cohort 6/11/2021 9

Selection of cases 6/11/2021 l Sources for cases l Problems in case selection l Incident or Prevalent Cases 10

Sources for cases l Hospitals l Physicians— practices, or clinics l Registries 6/11/2021 11

Problems in case selection l If cases are selected from a single hospital, any risk factors that are identified may be unique to that hospital as a result of referral patterns or other factors, and the results may not be generalizable to all patients with the disease. l Furthermore, if the hospital from which the cases are drawn is a tertiary care facility, which selectively admits severely ill patients, any risk factors identified in the study may be risk factors only in persons with severe forms of the disease. 6/11/2021 12

Incident or Prevalent Cases 6/11/2021 l Number of cases available l Survival bias 13

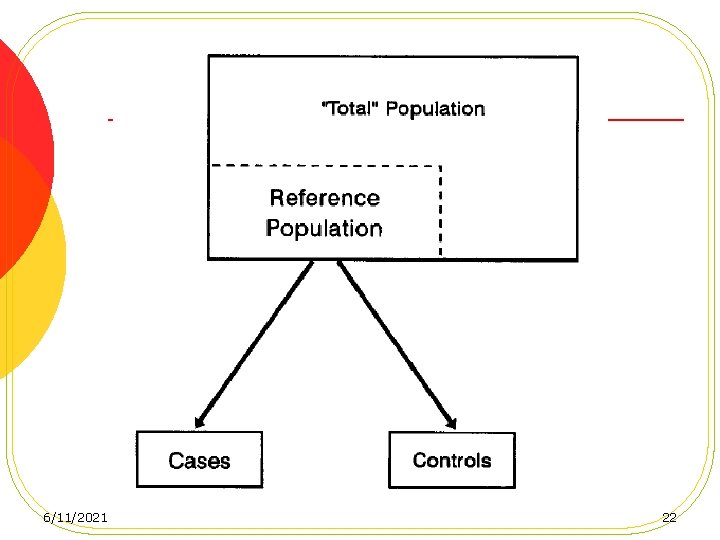
Principles of Control Selection l The “study-base” Principle l The Deconfounding Principle l The Comparable Accuracy Principle 6/11/2021 14

The “study-base” Principle l Controls be randomly selected from disease-free members of the underlying cohort, also known as the source population or study-base at the times that cases are being ascertained l When controls are in fact selected later, it sometimes mandates the random selection of a reference date for each control so that the distributions of the case diagnosis dates and control reference dates are comparable. 6/11/2021 15

The “study-base” Principle. Cont. 6/11/2021 l Only exposures occurring prior to the reference|diagnosis date would be taken into account. l This principle also implies that whatever exclusion criteria have been applied to the cases must also be applied equally to the controls. 16

The Deconfounding Principle l 6/11/2021 This principle underlies the stratified sampling of controls to render possible, or improve the efficiency of, an adjusted analysis designed to control confounding 17

The Comparable Accuracy Principle l 6/11/2021 Controls be selected so that the errors of measurement of their exposures and covariates are comparable to the measurement errors of the cases. 18

Selection of Controls l Sources of Controls l Non-hospitalized l Hospitalized 6/11/2021 Persons Patients 19

Non-hospitalized Persons l Probability sample of the total population l School rosters l Selective service lists l Insurance company lists l Neighborhood controls l Random-digit dialing l Best friend control l Spouse or sibling 6/11/2021 20

Hospitalized Patients as Controls l “captive population” = more economical l Ill-defined reference population l “All other patients admitted to the hospital” Vs “a specific “other diagnosis” 6/11/2021 21
6/11/2021 22

How Many Controls per Case? l For a fixed number of study subjects, statistical power for testing the null hypothesis is optimized by having equal numbers of cases and controls. l When the disease is extremely rare or acquisition of cases particularly expensive, however, it may be important and costeffective to increase the numbers of controls. 6/11/2021 23

How Many Controls per Case? l In order to have the same statistical power (to reject the null hypothesis of no exposure effect against local alternatives) as a design with equal numbers of cases and controls, a design with M controls per case would need only (M+1)|2 M as many cases. l For a fixed number of cases, the relative efficiency of a design with M controls per case relative to one that uses an unlimited number of controls is therefore only M|(M + 1). 24 6/11/2021

How Many Control Groups? l multiple control groups were recommended by Dorn (1959) to improve the case-control study so that it would “provide a more valid basis for generalization”. If a whole series of control groups, e. g. , of patients with different diseases, gives much the same answer and only the one affected group differs, the evidence is clearly much stronger than if the affected group differs from merely one other group. ” 6/11/2021 l 25

How Many Control Groups? l Failure to detect a difference among control groups may give a false sense of security unless they were deliberately selected to differ with respect to unmeasured potential confounders. l Recent reviews of case-control methods have tended to shy away from the use of multiple control groups. They argue that there is usually a single “best” control group, and that since the discovery of an adjusted exposure difference with other control groups will force these to be discarded, the effort involved will have been wasted. 6/11/2021 26

Case-Control Types l Primary-based Vs Secondary-based l Population-based Vs Hospital-based 6/11/2021 27

Matching l Group Matching (frequency matching) l Individual Matching (matched pairs) 6/11/2021 28

Problems with matching l Practical Problems l Conceptual Problems 6/11/2021 29

Problem of Recall l Limitations in Recall l Recall Bias 6/11/2021 30

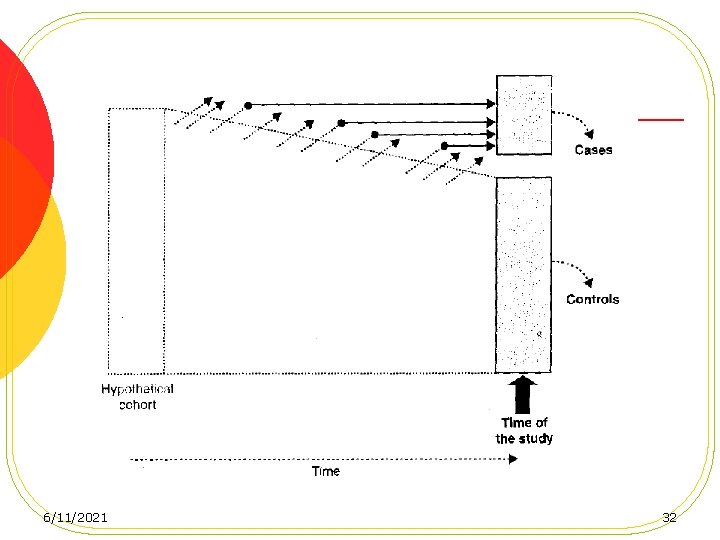
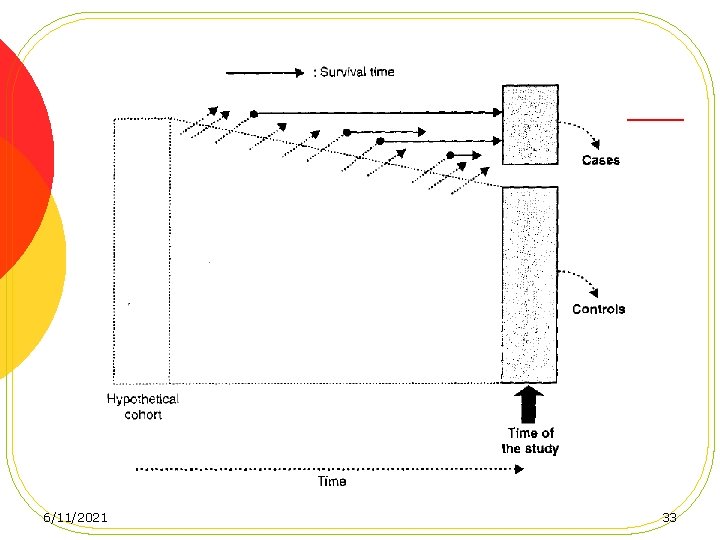
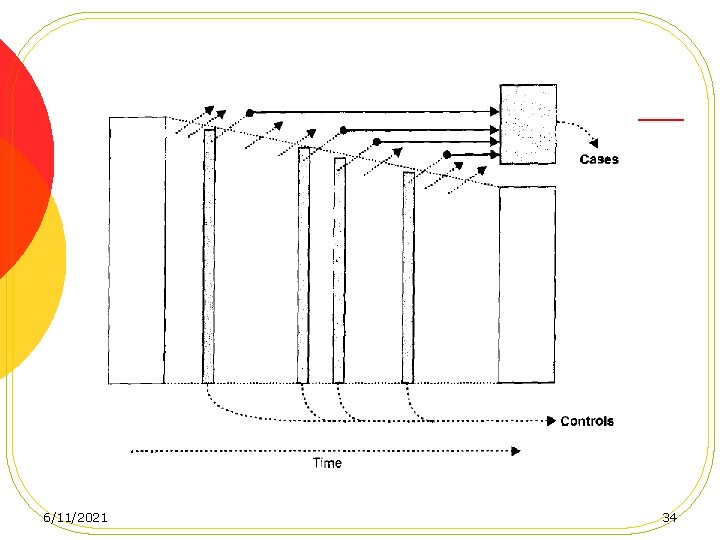
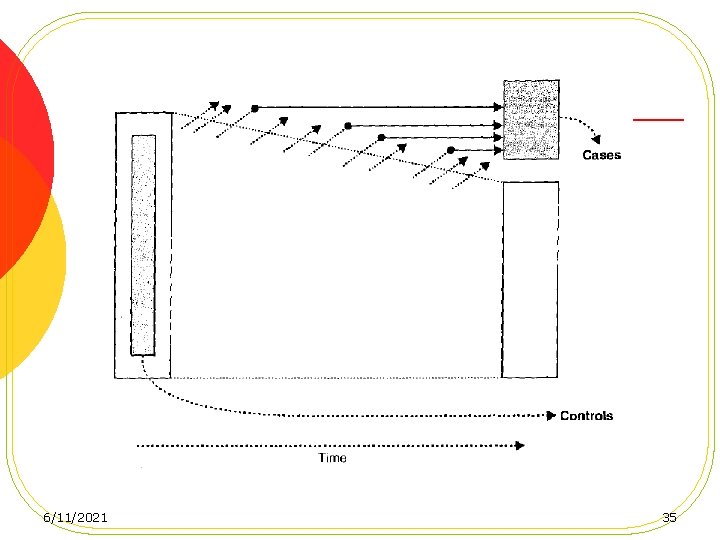
Case-control studies based on a defined cohort l Nested Case-Control Studies l Case-Cohort Studies 6/11/2021 31
6/11/2021 32
6/11/2021 33
6/11/2021 34
6/11/2021 35

advantages of conducting a casecontrol study in a defined cohort 1. No recall 2. Temporality is established 3. Efficiency 4. Compariblity 6/11/2021 36

Case-Crossover Design l Primarily used for studying the etiology of acute outcomes such as myocardial infarctions or deaths from acute events in situations where the suspected exposure is transient and its effect occurs over a short time. l The question being asked is: Was there any difference in exposure between the time period immediately preceding the outcome and a time period in the more remote past which was not immediately followed by any adverse health effect? 6/11/2021 37
- Slides: 37