Casebased Review of Endovascular Renal Interventions Primer for

Case-based Review of Endovascular Renal Interventions: Primer for Radiology Residents and Fellows Jay Patel, MD, Sean Calhoun, DO, Thaddeus Yablonsky, MD, Michael Miller, Jr, MD, Juan C. Camacho, MD, Courtney C. Moreno, MD, Frank H. Miller, MD, Pardeep K. Mittal, MD Department of Radiology and Imaging Sciences, Emory University (J. P. , M. M. , C. C. M. , P. K. M. ); Department of Radiology, Morristown Medical Center, Morristown, NJ (S. C. , T. Y. ); Department of Radiology, Medical University of South Carolina, Charleston, SC (J. C. C. ); and Department of Radiology, Northwestern University Feinberg School of Medicine, Chicago, Ill (F. H. M. ). Presented as an education exhibit at the 2016 RSNA Annual Meeting (VI 129 -ED-X). All authors have disclosed no relevant relationships. Address correspondence to P. K. M. , Department of Radiology and Imaging, Medical College of Georgia, 1120 15 th St, Augusta, GA 30912 (e-mail: pmittal@augusta. edu).

Learning Objectives • Identify indications, patient preparation, and equipment for various renal vascular interventions • Review various embolic options, including mechanical devices, particulate agents, and liquid agents • Discuss renal interventional treatment options, technical considerations, and common complications of these interventions

Background • Renal interventions are commonly performed procedures at most institutions and can be broadly divided into two categories: endovascular and percutaneous. • The purpose of this presentation is to give trainees a better understanding of commonly performed endovascular renal interventions, including technical aspects and associated imaging findings of each procedure. • Endovascular interventions can be used to treat a variety of renal pathologic conditions, including vascular abnormalities and benign and malignant tumors, and can be used preoperatively as an adjunct to surgery and ablation. • Interventional and diagnostic radiologists and trainees will often encounter a variety of renal pathologic conditions for which intervention may be performed.

Topics Covered • • • Basic patient preparation Overview of embolic agents Endovascular interventions • • Malformation • Arteriovenous malformation (AVM) Traumatic or iatrogenic • Arteriovenous fistula (AVF) • Pseudoaneurysm and active arterial extravasation Neoplasms • Renal cell carcinoma (RCC) • Angiomyolipoma (AML) Renal artery stenosis (RAS) • Atherosclerotic disease • Fibromuscular dysplasia (FMD)

Basic Patient Preparation Preprocedural Laboratory Testing and Management • Per Society of Interventional Radiology (SIR) consensus guidelines, procedures can be categorized into low, moderate, and high risk of bleeding. Each category has specific recommendations regarding laboratory work and patient medications. • Common laboratory work includes international normalized ratio/prothrombin time, activated partial thromboplastin time (a. PTT), platelet count, and hematocrit level. • Common medications that may need to be held: warfarin, clopidogrel, aspirin, unfractionated heparin, low molecular weight heparin, and others including factor Xa inhibitors. • Endovascular procedures such as embolization, stent placement, and angioplasty have a moderate risk of bleeding. Check and correct international normalized ratio greater than 1. 5 times the normal value and platelet count less than 50 x 109/L. Also check a. PTT levels, but only 73% consensus to correct a. PTT greater than 1. 5 times the normal value. Some medications need to be held for varying lengths of time. • Varying data on anticoagulation requirements for endovascular procedures performed by using radial access • Patients with concomitant cardiac disease that precludes discontinuation of antiplatelet and anticoagulation medications are often switched to a reversible agent such as heparin.

Basic Patient Preparation Informed consent Purpose, nature, method, risks, benefits, and alternative of procedure Fasting status • If patient is a candidate for moderate sedation—No solid food and no full liquids for 6– 8 hours and no clear liquids for 2– 3 hours prior to planned procedure Preprocedural antibiotics • As per SIR consensus guidelines, procedure can be divided into four classes: clean, clean-contaminated, and dirty. • Endovascular interventions are categorized as clean procedures and only require antibiotic prophylaxis for skin flora. • Antibiotics should be administered within 1 hour of procedure.
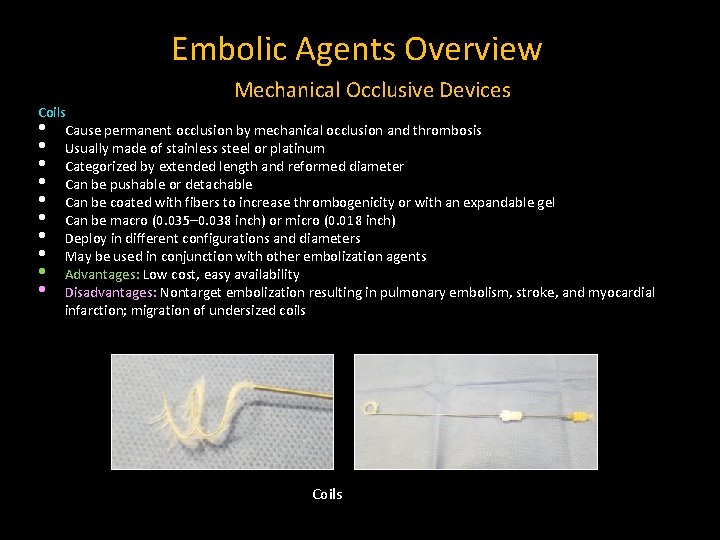
Embolic Agents Overview Mechanical Occlusive Devices Coils • Cause permanent occlusion by mechanical occlusion and thrombosis • Usually made of stainless steel or platinum • Categorized by extended length and reformed diameter • Can be pushable or detachable • Can be coated with fibers to increase thrombogenicity or with an expandable gel • Can be macro (0. 035– 0. 038 inch) or micro (0. 018 inch) • Deploy in different configurations and diameters • May be used in conjunction with other embolization agents • Advantages: Low cost, easy availability • Disadvantages: Nontarget embolization resulting in pulmonary embolism, stroke, and myocardial infarction; migration of undersized coils Coils

Embolic Agents Overview Mechanical Occlusive Devices Plugs • Self-expanding and detachable nitinol wire mesh • Secured on both ends by platinum marker bands • Usually mounted to a long pusher wire by screw thread • Available in various sizes and configurations • Allows for precise deployment and modification of deployment • Advantages: Good control over position deployment. Can be recaptured and repositioned • Disadvantages: Expensive, device migration if undersized; may be difficult to advance into distal or tortuous vessels because of stiff delivery system Vascular plug

Embolic Agents Overview Particulate Agents Permanent • Tris-acryl gelatin microspheres (TAGMs) • Made from a hydrophilic acrylic polymer and precisely calibrated by size • Available in multiple sizes, 40– 120 µm to 900– 1200 µm • Are positively charged and repel each other resulting in less clumping and increased attraction to the negatively charged blood vessel wall • Advantages: Predictable level of embolization, permanent embolization, effectively embolize large tumors by occluding at the arteriole level • Disadvantages: Particle reflux and nontarget embolization Microspheres

Embolic Agents Overview Particulate Agents Permanent • Polyvinyl alcohol (PVA) • Synthetic polymer available in a range of sizes of approximately 100– 1100 µm • Advantages: Particles clog distal vessel causing thrombosis (devascularization of hypervascular tumors). • Disadvantages: PVA preparations have a tendency to form clumps of particles in the catheter or vessels of varying size; potential of nontarget embolization; difficult to predict level of embolization PVA particles

Embolic Agents Overview Particulate Agents Temporary • Gelatin sponge • Dissolvable gelatin sponge that usually comes as a sheet • Usually cut into small pledgets and soaked in contrast material prior to injection • Gelatin foam causes mechanical obstruction, slows blood flow, and aids thrombus formation. • Advantages: Temporary agent with potential of vessel recanalization in a few days to weeks • Disadvantages: Infection due to trapped air bubbles that support aerobic organism growth, delayed bleeding due to recanalization Gelatin sponge

Embolic Agents Overview Liquid Agents • Absolute alcohol • Causes cell death by dehydration resulting in denudation of endothelial cell from vascular wall and subsequent thrombosis • Usually mixed with ethiodized oil for visualization • Advantages: Excellent means of achieving tumor necrosis • Disadvantages: Skin and tissue necrosis, nerve damage, hemoglobinuria, deep vein thrombosis, pulmonary embolism • Maximum dose of 30 m. L to minimize risk of acute pulmonary artery hypertension and cardiac collapse (increased pulmonary artery pressure occurs because of ethanol-induced vasospasms at the precapillary level, pulmonary artery hypertension may lead to right ventricular failure and subsequent left ventricular failure, ultimately cardiopulmonary collapse) • n-Butyl cyanoacrylate (n-BCA) • Synthetic glue • Usually mixed with ethiodized oil and/or tantalum powder to make the adhesive radiopaque • Mixing ratio determined by flow rate through the target with more concentrated glue used for lesions with high flow rate • Advantages: Instant, completely occludes the vessel and is permanent • Disadvantages: Catheter can be entrapped in the occluded vessel • Ethylene vinyl alcohol copolymer (EVOH) • Synthetic copolymer mixed with tantalum powder for opacity • Dimethyl sulfoxide (DMSO)–compatible catheter must be used • Advantages: Nonadhesive agent provides controlled delivery • Disadvantages: DMSO-compatible catheter must be used; longer embolization times

Choosing Embolic Agent Endovascular Renal Interventions Indications and Contraindications • • • The embolic agent is chosen on the basis of anatomic and flow characteristics, as well as determining if endothelial or tumor necrosis is the intended outcome. Indications: Based on presence of symptoms, which are grouped as: • Occlusion: Aneurysms, pseudoaneurysms, AVMs, and AVFs • Hemorrhage: Posttraumatic, iatrogenic (renal biopsy), hemorrhagic neoplasms • Devascularization: Malignant and benign tumors: RCC, AML • Vascular: RAS, FMD Contraindications • Relative: Coagulopathy, sepsis, renal insufficiency • Absolute: Communication between target organ and an adjacent vital structure, arteriovenous shunting to lungs, right-to-left shunt Angle et al. Quality improvement guidelines for percutaneous transcatheter embolization: Society of Interventional Radiology Standards of Practice Committee. J Vasc Interv Radiol 2010; 21(10): 1479– 1486.
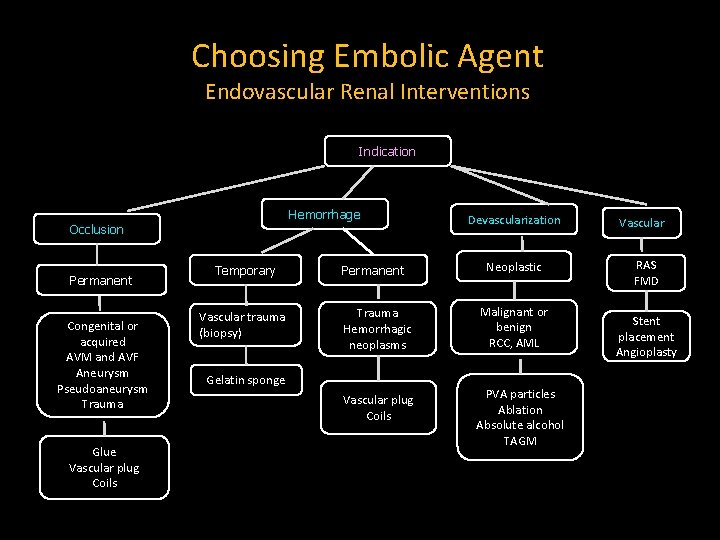
Choosing Embolic Agent Endovascular Renal Interventions Indication Hemorrhage Occlusion Permanent Congenital or acquired AVM and AVF Aneurysm Pseudoaneurysm Trauma Glue Vascular plug Coils Temporary Vascular trauma (biopsy) Permanent Devascularization Neoplastic Trauma Hemorrhagic neoplasms Malignant or benign RCC, AML Vascular plug Coils PVA particles Ablation Absolute alcohol TAGM Gelatin sponge Vascular RAS FMD Stent placement Angioplasty

Renal Arteriovenous Malformation • Abnormal communication between a renal artery and vein through a network of abnormal vessels known as the nidus • Classified as cirsoid (having multiple arteries feeding into the nidus) or cavernous (having a single feeding artery) • Can present as flank pain, gross hematuria, and hypertension; blood clots can also be seen in the collecting system. • Most common indication for intervention is symptomatic patient with gross hematuria. • Goal of treatment is to ablate the nidus of the AVMs are usually high-flow lesions; therefore, viscous agents such as glue are preferred over alcohol. Surgery is usually reserved for complicated cases not amendable to renal artery embolization (RAE). • Incomplete embolization of the nidus leads to recurrence. • Postembolization syndrome—combination of flulike symptoms, including fever, malaise, pain at the site of ablation, nausea, and/or vomiting. Symptoms are usually mild and self-limiting and tend to resolve spontaneously by 2 weeks.
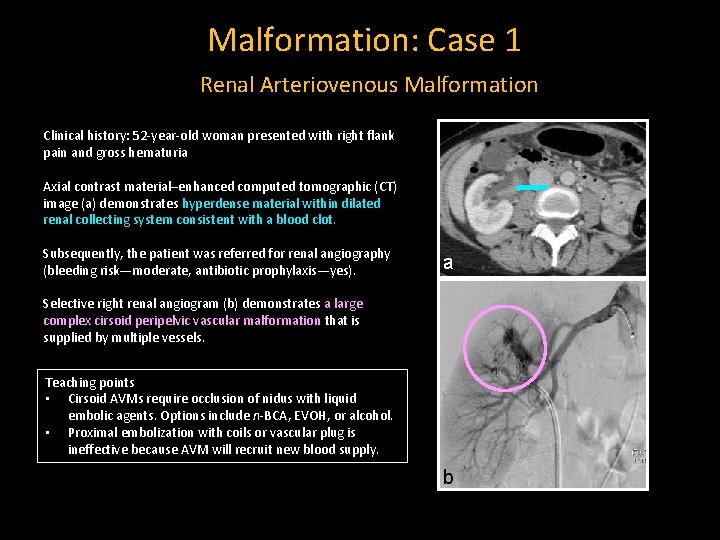
Malformation: Case 1 Renal Arteriovenous Malformation Clinical history: 52 -year-old woman presented with right flank pain and gross hematuria Axial contrast material–enhanced computed tomographic (CT) image (a) demonstrates hyperdense material within dilated renal collecting system consistent with a blood clot. Subsequently, the patient was referred for renal angiography (bleeding risk—moderate, antibiotic prophylaxis—yes). a Selective right renal angiogram (b) demonstrates a large complex cirsoid peripelvic vascular malformation that is supplied by multiple vessels. Teaching points • Cirsoid AVMs require occlusion of nidus with liquid embolic agents. Options include n-BCA, EVOH, or alcohol. • Proximal embolization with coils or vascular plug is ineffective because AVM will recruit new blood supply. b
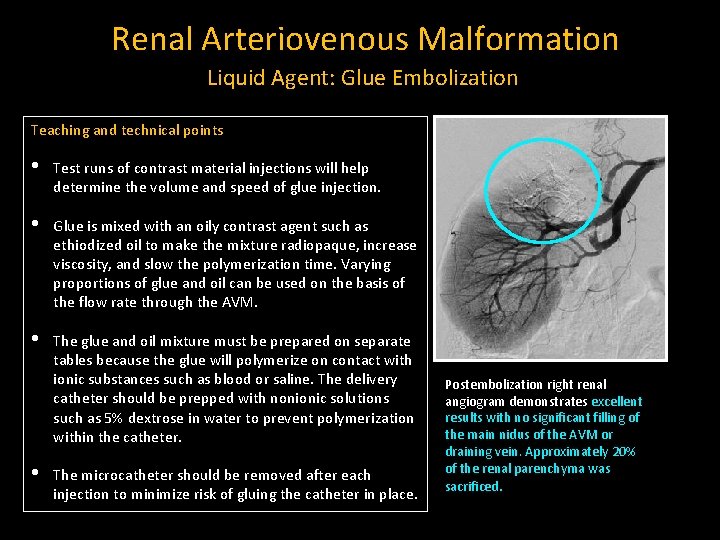
Renal Arteriovenous Malformation Liquid Agent: Glue Embolization Teaching and technical points • Test runs of contrast material injections will help determine the volume and speed of glue injection. • Glue is mixed with an oily contrast agent such as ethiodized oil to make the mixture radiopaque, increase viscosity, and slow the polymerization time. Varying proportions of glue and oil can be used on the basis of the flow rate through the AVM. • The glue and oil mixture must be prepared on separate tables because the glue will polymerize on contact with ionic substances such as blood or saline. The delivery catheter should be prepped with nonionic solutions such as 5% dextrose in water to prevent polymerization within the catheter. • The microcatheter should be removed after each injection to minimize risk of gluing the catheter in place. Postembolization right renal angiogram demonstrates excellent results with no significant filling of the main nidus of the AVM or draining vein. Approximately 20% of the renal parenchyma was sacrificed.
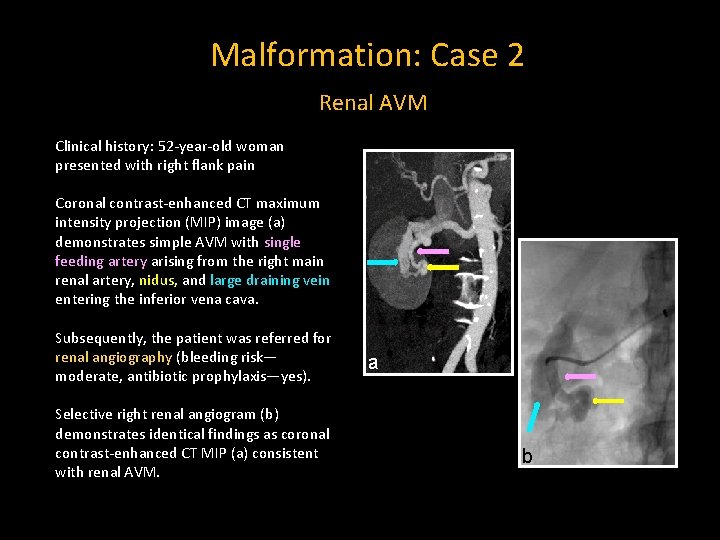
Malformation: Case 2 Renal AVM Clinical history: 52 -year-old woman presented with right flank pain Coronal contrast-enhanced CT maximum intensity projection (MIP) image (a) demonstrates simple AVM with single feeding artery arising from the right main renal artery, nidus, and large draining vein entering the inferior vena cava. Subsequently, the patient was referred for renal angiography (bleeding risk— moderate, antibiotic prophylaxis—yes). Selective right renal angiogram (b) demonstrates identical findings as coronal contrast-enhanced CT MIP (a) consistent with renal AVM. a b
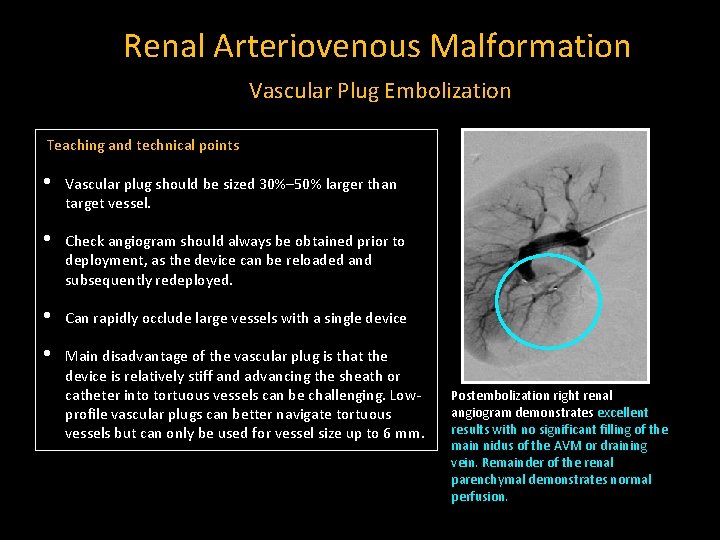
Renal Arteriovenous Malformation Vascular Plug Embolization Teaching and technical points • Vascular plug should be sized 30%– 50% larger than target vessel. • Check angiogram should always be obtained prior to deployment, as the device can be reloaded and subsequently redeployed. • Can rapidly occlude large vessels with a single device • Main disadvantage of the vascular plug is that the device is relatively stiff and advancing the sheath or catheter into tortuous vessels can be challenging. Lowprofile vascular plugs can better navigate tortuous vessels but can only be used for vessel size up to 6 mm. Postembolization right renal angiogram demonstrates excellent results with no significant filling of the main nidus of the AVM or draining vein. Remainder of the renal parenchymal demonstrates normal perfusion.

Traumatic or Iatrogenic • Most common indications for intervention are active extravasation, pseudoaneurysm, or AVF. • Major bleeding complication related to biopsy can be minimized by correcting coagulation parameters prior to the procedure, using good technique, and controlling hypertension in the periprocedural period with a goal of less than 140/90 mm Hg. • Postbiopsy tract embolization with a gelatin-based agent may also decrease the risk of major bleeding complication. • The kidney is the third most common organ injured in abdominal trauma after the spleen and liver. • Presence of hematuria after abdominal injury is a good predictor of renal injury. • A variety of embolic agents can be used. Vascular plugs and macrocoils for larger vessels, microcoils and gelatin sponge for smaller vessels • Use of liquid embolic agents is limited in trauma because of increased risk of tissue necrosis.

Renal Arteriovenous Fistula • Abnormal direct communication between a renal artery and vein • Often acquired secondary to iatrogenic causes, such as renal biopsy, percutaneous nephrostomy, or penetrating injuries • A feature of AVF is early opacification of vein during the arterial phase of contrastenhanced CT or digital subtraction angiography. • Color Doppler ultrasonographic (US) evaluation may show direct communication between artery and vein, and the draining vein will have an arterialized flow pattern. • Usually asymptomatic, but may have hematuria or hypertension; if large, can present with high-output heart failure • For small AVFs, conservative management in conjunction with US surveillance is recommended, as up to 70% resolve spontaneously (wait and watch). Symptomatic or persistent AVFs can be effectively treated with transcatheter embolization.
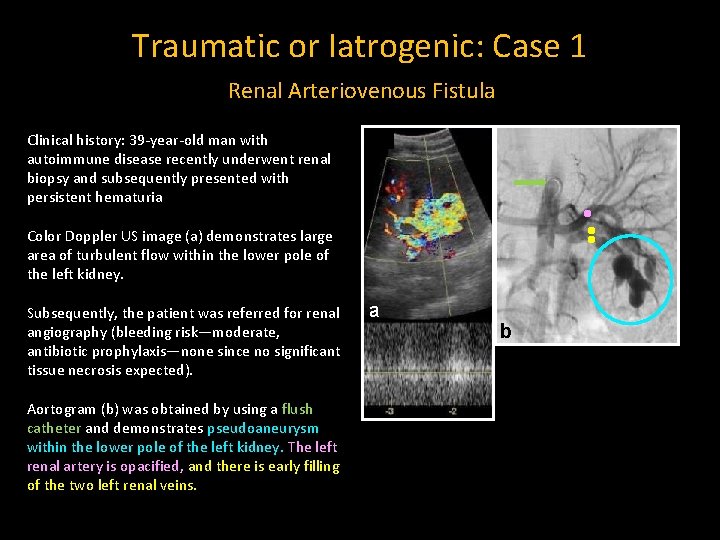
Traumatic or Iatrogenic: Case 1 Renal Arteriovenous Fistula Clinical history: 39 -year-old man with autoimmune disease recently underwent renal biopsy and subsequently presented with persistent hematuria Color Doppler US image (a) demonstrates large area of turbulent flow within the lower pole of the left kidney. Subsequently, the patient was referred for renal angiography (bleeding risk—moderate, antibiotic prophylaxis—none since no significant tissue necrosis expected). Aortogram (b) was obtained by using a flush catheter and demonstrates pseudoaneurysm within the lower pole of the left kidney. The left renal artery is opacified, and there is early filling of the two left renal veins. a b
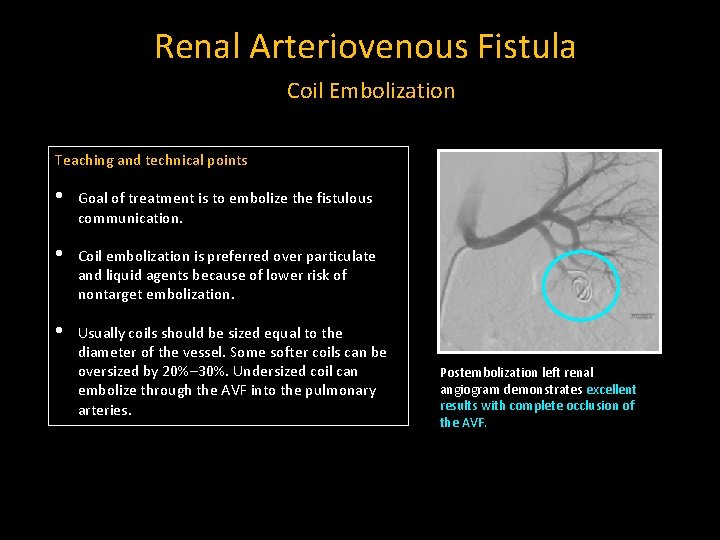
Renal Arteriovenous Fistula Coil Embolization Teaching and technical points • Goal of treatment is to embolize the fistulous communication. • Coil embolization is preferred over particulate and liquid agents because of lower risk of nontarget embolization. • Usually coils should be sized equal to the diameter of the vessel. Some softer coils can be oversized by 20%– 30%. Undersized coil can embolize through the AVF into the pulmonary arteries. Postembolization left renal angiogram demonstrates excellent results with complete occlusion of the AVF.
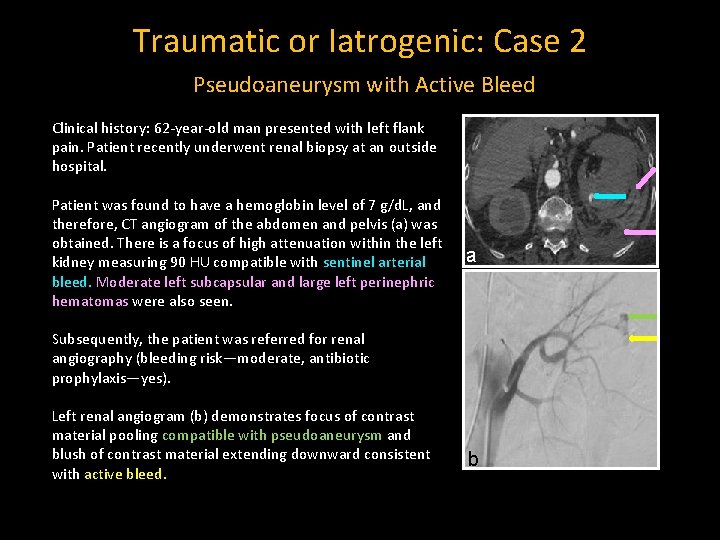
Traumatic or Iatrogenic: Case 2 Pseudoaneurysm with Active Bleed Clinical history: 62 -year-old man presented with left flank pain. Patient recently underwent renal biopsy at an outside hospital. Patient was found to have a hemoglobin level of 7 g/d. L, and therefore, CT angiogram of the abdomen and pelvis (a) was obtained. There is a focus of high attenuation within the left kidney measuring 90 HU compatible with sentinel arterial bleed. Moderate left subcapsular and large left perinephric hematomas were also seen. a Subsequently, the patient was referred for renal angiography (bleeding risk—moderate, antibiotic prophylaxis—yes). Left renal angiogram (b) demonstrates focus of contrast material pooling compatible with pseudoaneurysm and blush of contrast material extending downward consistent with active bleed. b
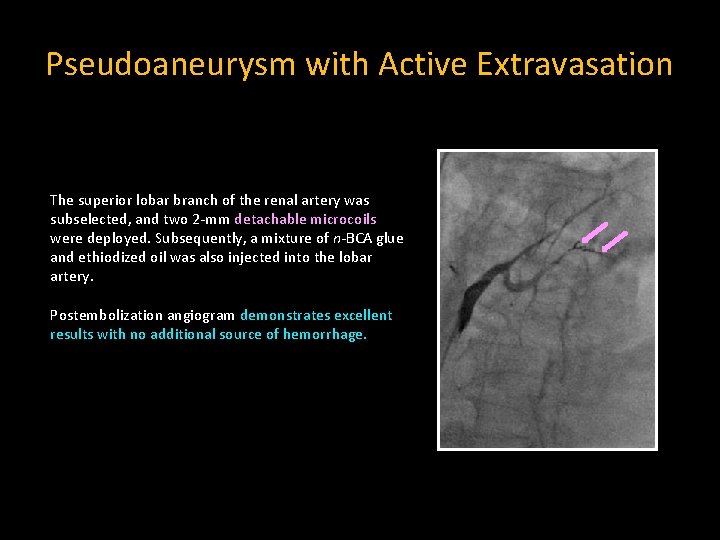
Pseudoaneurysm with Active Extravasation The superior lobar branch of the renal artery was subselected, and two 2 -mm detachable microcoils were deployed. Subsequently, a mixture of n-BCA glue and ethiodized oil was also injected into the lobar artery. Postembolization angiogram demonstrates excellent results with no additional source of hemorrhage.
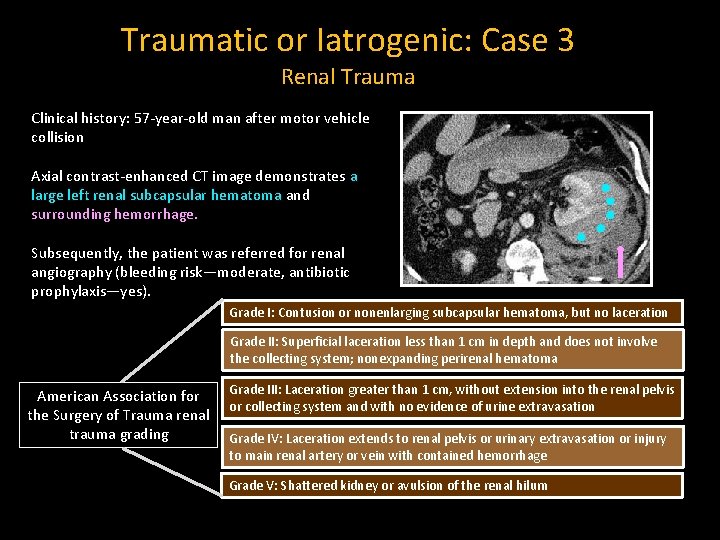
Traumatic or Iatrogenic: Case 3 Renal Trauma Clinical history: 57 -year-old man after motor vehicle collision Axial contrast-enhanced CT image demonstrates a large left renal subcapsular hematoma and surrounding hemorrhage. Subsequently, the patient was referred for renal angiography (bleeding risk—moderate, antibiotic prophylaxis—yes). Grade I: Contusion or nonenlarging subcapsular hematoma, but no laceration Grade II: Superficial laceration less than 1 cm in depth and does not involve the collecting system; nonexpanding perirenal hematoma American Association for the Surgery of Trauma renal trauma grading Grade III: Laceration greater than 1 cm, without extension into the renal pelvis or collecting system and with no evidence of urine extravasation Grade IV: Laceration extends to renal pelvis or urinary extravasation or injury to main renal artery or vein with contained hemorrhage Grade V: Shattered kidney or avulsion of the renal hilum
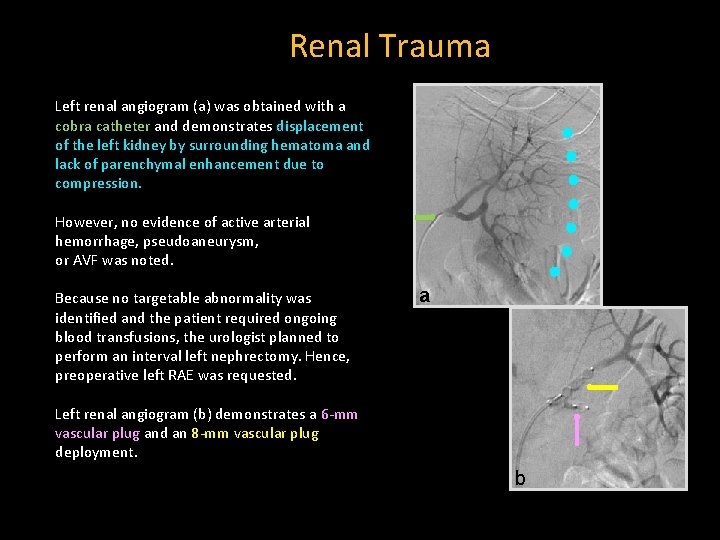
Renal Trauma Left renal angiogram (a) was obtained with a cobra catheter and demonstrates displacement of the left kidney by surrounding hematoma and lack of parenchymal enhancement due to compression. However, no evidence of active arterial hemorrhage, pseudoaneurysm, or AVF was noted. Because no targetable abnormality was identified and the patient required ongoing blood transfusions, the urologist planned to perform an interval left nephrectomy. Hence, preoperative left RAE was requested. a Left renal angiogram (b) demonstrates a 6 -mm vascular plug and an 8 -mm vascular plug deployment. b
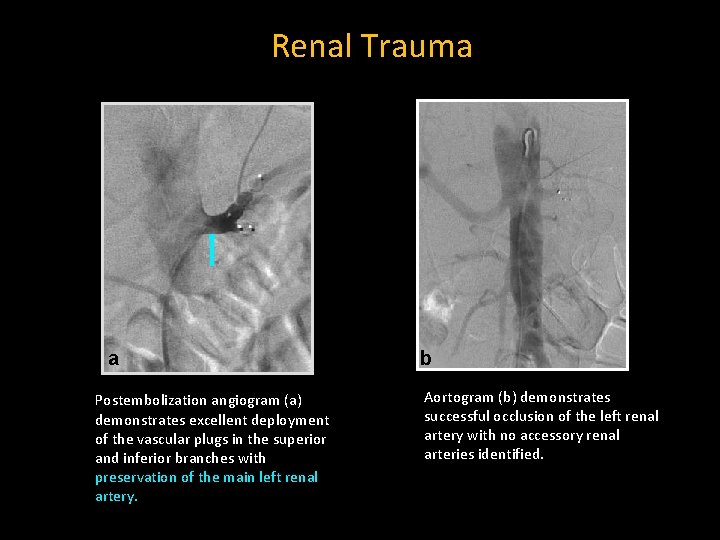
Renal Trauma a Postembolization angiogram (a) demonstrates excellent deployment of the vascular plugs in the superior and inferior branches with preservation of the main left renal artery. b Aortogram (b) demonstrates successful occlusion of the left renal artery with no accessory renal arteries identified. b

Renal Cell Carcinoma • Most common malignant neoplasm of the kidney. 70% of RCCs are clear cell. RCCs are often encapsulated and highly vascular lesions with propensity to invade the renal pelvis and renal vein. • Hereditary renal cell cancers account for 3%– 5% of all renal cancers, and genetic syndromes associated with increased risk of renal cancer include von Hippel–Lindau syndrome, hereditary papillary RCC, Birt-Hogg-Dubé syndrome, tuberous sclerosis, etc. • Radical nephrectomy is standard treatment. Partial nephrectomy or ablation can be performed if the tumor is peripheral, exophytic, and less than 5 cm in size. • Indications for treatment of RCC with RAE include preoperative devascularization to improve resection plane and decrease blood loss, palliative treatment in nonsurgical candidates, and treatment of hemorrhagic complications. • Combined therapy with RAE prior to ablation is thought to have a synergistic effect with decreased blood flow from embolization resulting in decreased heat sink drain during radiofrequency ablation. • Proximal main renal artery should be preserved for cross-clamping during nephrectomy.
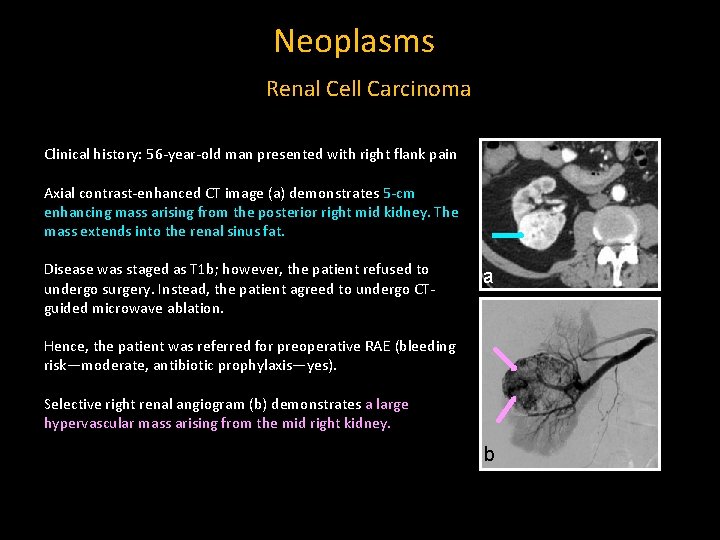
Neoplasms Renal Cell Carcinoma Clinical history: 56 -year-old man presented with right flank pain Axial contrast-enhanced CT image (a) demonstrates 5 -cm enhancing mass arising from the posterior right mid kidney. The mass extends into the renal sinus fat. Disease was staged as T 1 b; however, the patient refused to undergo surgery. Instead, the patient agreed to undergo CTguided microwave ablation. a Hence, the patient was referred for preoperative RAE (bleeding risk—moderate, antibiotic prophylaxis—yes). Selective right renal angiogram (b) demonstrates a large hypervascular mass arising from the mid right kidney. b
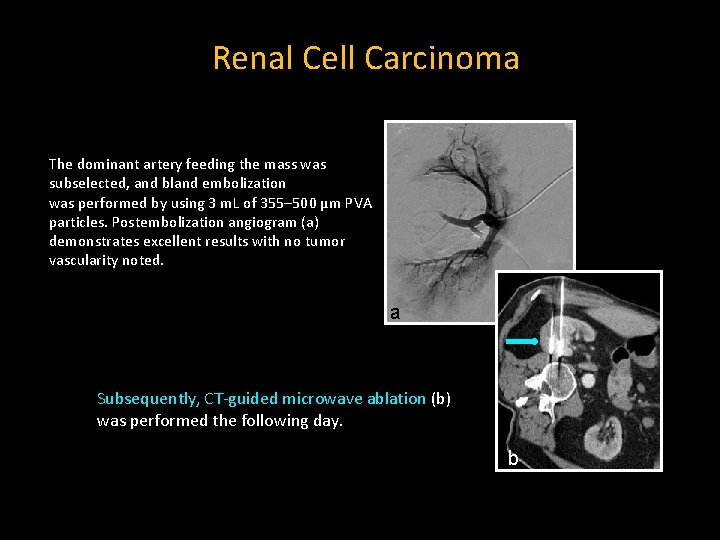
Renal Cell Carcinoma The dominant artery feeding the mass was subselected, and bland embolization was performed by using 3 m. L of 355– 500 µm PVA particles. Postembolization angiogram (a) demonstrates excellent results with no tumor vascularity noted. a Subsequently, CT-guided microwave ablation (b) was performed the following day. b

Renal AML • Most common benign tumor of kidney; composed of blood vessels, smooth muscle, and fat • Incidence of renal AML can be sporadic, which presents as a single lesion most commonly, or associated with tuberous sclerosis, which tends to be multiple. 80% of patients with tuberous sclerosis have renal AMLs. • Indications for intervention include prophylaxis for tumors larger than 4 cm in diameter (greater than 50% risk of rupture and hemorrhage) and treatment of acute hemorrhage due to spontaneous rupture of AML. • Absence of arteriovenous shunting and the presence of microaneurysms help distinguish AML from RCC on angiogram. • Usually treated with transarterial embolization with a variety of embolic agents. Since AMLs are highly vascular lesions, treatment with liquid or particulate embolic agents is recommended over mechanical embolization. The reduction of volume is more noticeable in the vascular component compared with the fat component of AML. • Less risk of nontarget embolization compared with AVM or AVF treatment • Continued surveillance required because of risk of recurrence (higher in patients with tuberous sclerosis) • 30% risk of postembolization syndrome
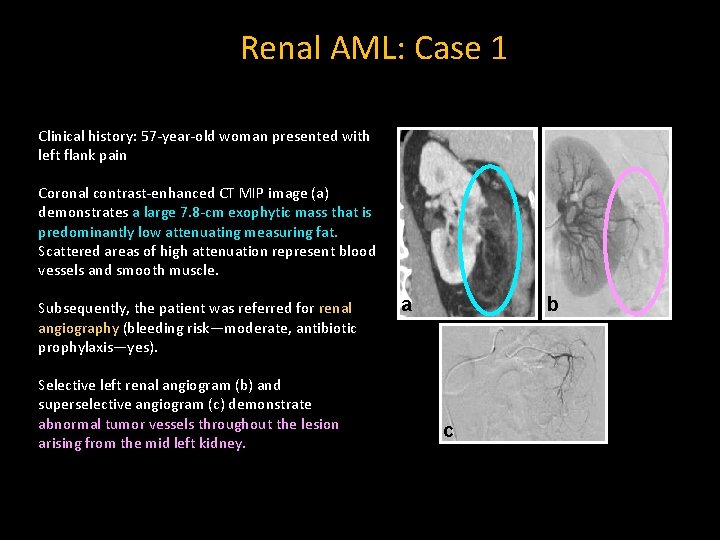
Renal AML: Case 1 Clinical history: 57 -year-old woman presented with left flank pain Coronal contrast-enhanced CT MIP image (a) demonstrates a large 7. 8 -cm exophytic mass that is predominantly low attenuating measuring fat. Scattered areas of high attenuation represent blood vessels and smooth muscle. Subsequently, the patient was referred for renal angiography (bleeding risk—moderate, antibiotic prophylaxis—yes). Selective left renal angiogram (b) and superselective angiogram (c) demonstrate abnormal tumor vessels throughout the lesion arising from the mid left kidney. a b c
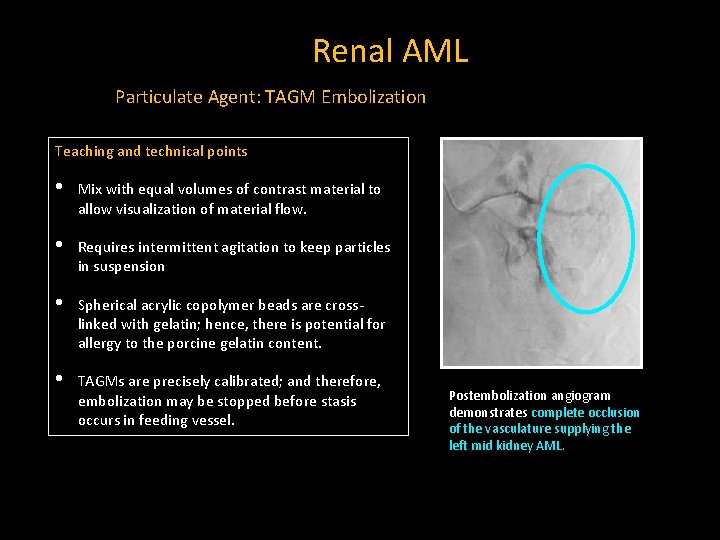
Renal AML Particulate Agent: TAGM Embolization Teaching and technical points • Mix with equal volumes of contrast material to allow visualization of material flow. • Requires intermittent agitation to keep particles in suspension • Spherical acrylic copolymer beads are crosslinked with gelatin; hence, there is potential for allergy to the porcine gelatin content. • TAGMs are precisely calibrated; and therefore, embolization may be stopped before stasis occurs in feeding vessel. Postembolization angiogram demonstrates complete occlusion of the vasculature supplying the left mid kidney AML.
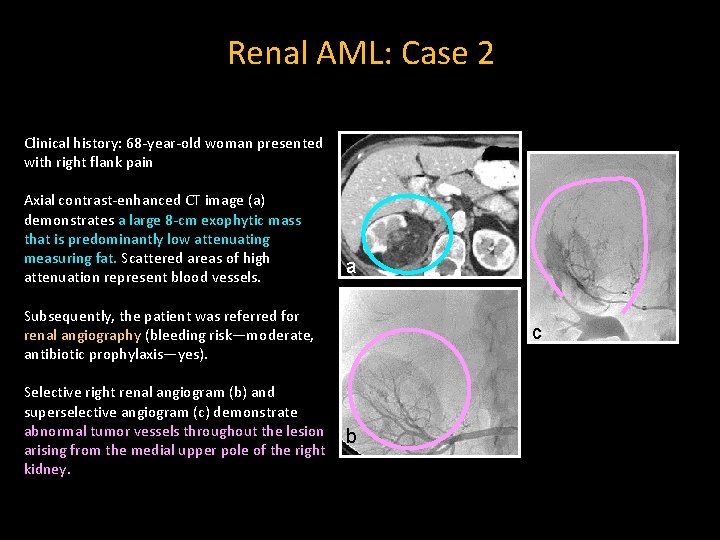
Renal AML: Case 2 Clinical history: 68 -year-old woman presented with right flank pain Axial contrast-enhanced CT image (a) demonstrates a large 8 -cm exophytic mass that is predominantly low attenuating measuring fat. Scattered areas of high attenuation represent blood vessels. a Subsequently, the patient was referred for renal angiography (bleeding risk—moderate, antibiotic prophylaxis—yes). Selective right renal angiogram (b) and superselective angiogram (c) demonstrate abnormal tumor vessels throughout the lesion arising from the medial upper pole of the right kidney. c b
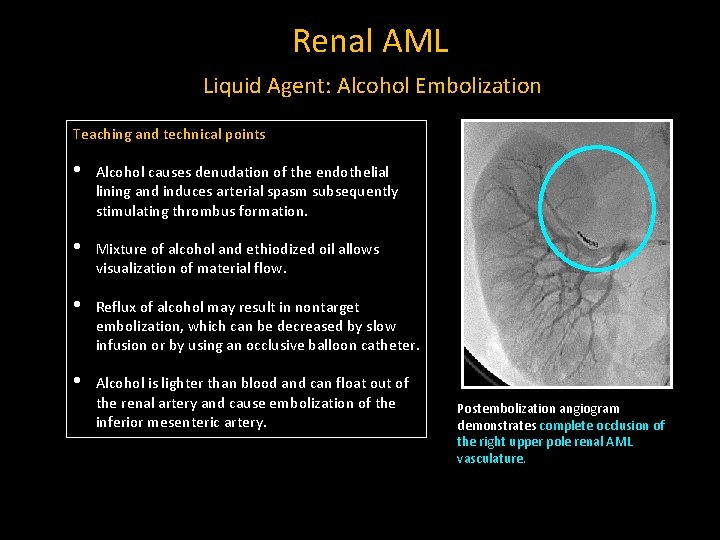
Renal AML Liquid Agent: Alcohol Embolization Teaching and technical points • Alcohol causes denudation of the endothelial lining and induces arterial spasm subsequently stimulating thrombus formation. • Mixture of alcohol and ethiodized oil allows visualization of material flow. • Reflux of alcohol may result in nontarget embolization, which can be decreased by slow infusion or by using an occlusive balloon catheter. • Alcohol is lighter than blood and can float out of the renal artery and cause embolization of the inferior mesenteric artery. Postembolization angiogram demonstrates complete occlusion of the right upper pole renal AML vasculature.

Renal Artery Stenosis • Most common cause of secondary hypertension • Most common cause of RAS is atherosclerotic disease (90%) followed by FMD (10%). • Significant atherosclerotic RAS is defined as a decrease of at least 60% in luminal diameter. • Data on progression of atherosclerotic RAS are inconsistent, with progressive stenosis reported in 51% 5 years after diagnosis. • According to 2015 SIR guidelines, minimum indication for stent placement is 50% diameter narrowing and 10% gradient. • Intervention may be warranted for renal insufficiency and preservation of renal function. However, intervention is not recommended if renal function has remained stable during the past 6– 12 months. • Treatment with aspirin before the procedure, heparin during the procedure, and dual antiplatelet therapy with aspirin and clopidogrel after the procedure for 6 months is recommended. • Complications of renal artery stent placement include vasospasm, dissection, rupture, and distal embolization.
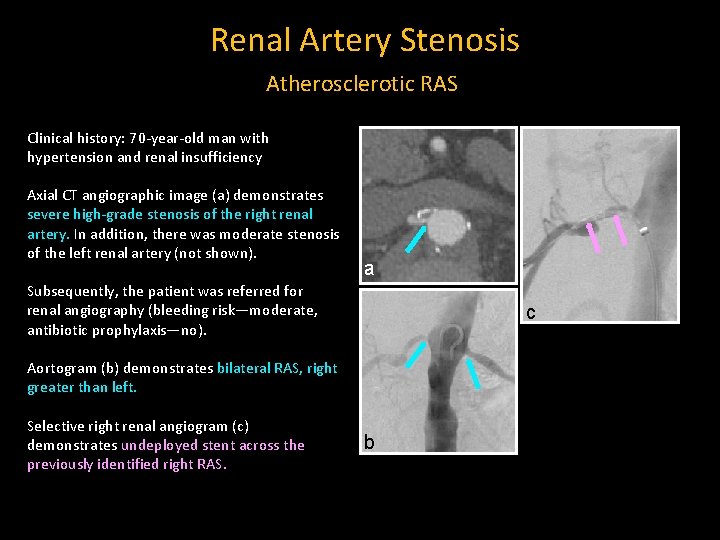
Renal Artery Stenosis Atherosclerotic RAS Clinical history: 70 -year-old man with hypertension and renal insufficiency Axial CT angiographic image (a) demonstrates severe high-grade stenosis of the right renal artery. In addition, there was moderate stenosis of the left renal artery (not shown). a Subsequently, the patient was referred for renal angiography (bleeding risk—moderate, antibiotic prophylaxis—no). c Aortogram (b) demonstrates bilateral RAS, right greater than left. Selective right renal angiogram (c) demonstrates undeployed stent across the previously identified right RAS. b
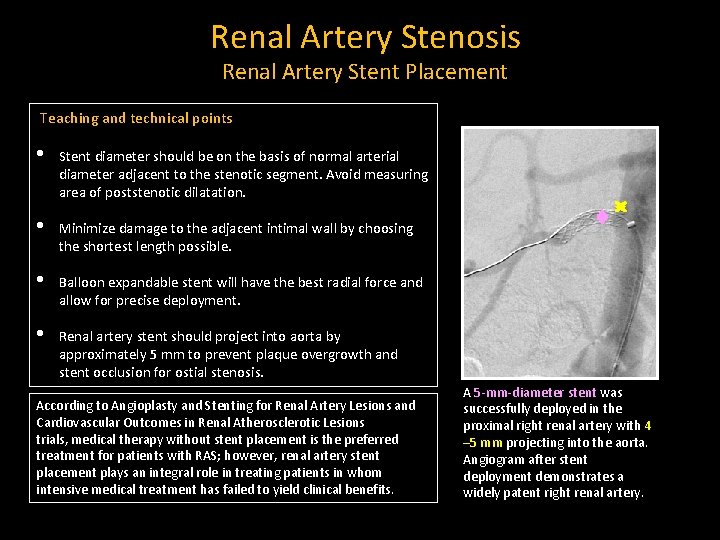
Renal Artery Stenosis Renal Artery Stent Placement Teaching and technical points • Stent diameter should be on the basis of normal arterial diameter adjacent to the stenotic segment. Avoid measuring area of poststenotic dilatation. • Minimize damage to the adjacent intimal wall by choosing the shortest length possible. • Balloon expandable stent will have the best radial force and allow for precise deployment. • Renal artery stent should project into aorta by approximately 5 mm to prevent plaque overgrowth and stent occlusion for ostial stenosis. According to Angioplasty and Stenting for Renal Artery Lesions and Cardiovascular Outcomes in Renal Atherosclerotic Lesions trials, medical therapy without stent placement is the preferred treatment for patients with RAS; however, renal artery stent placement plays an integral role in treating patients in whom intensive medical treatment has failed to yield clinical benefits. A 5 -mm-diameter stent was successfully deployed in the proximal right renal artery with 4 – 5 mm projecting into the aorta. Angiogram after stent deployment demonstrates a widely patent right renal artery.

Fibromuscular Dysplasia • Common presentation is new onset of refractory hypertension in female patients aged 15– 50 years. • Noninflammatory, nonatherosclerotic disorder caused by segmental areas of collagen deposition and smooth muscle overgrowth • Classified based on involved layer or arterial wall: intimal (<10%), medial (90%), and adventitial (<1%) • Most commonly involves mid to distal renal arteries (60%– 75%), with 50% chance of bilateral occurrence • Percutaneous angioplasty cures or improves hypertension in more than 95% of cases, with excellent long-term prognosis after treatment. • Complications of renal artery angioplasty include transient renal insufficiency, vasospasm, dissection, or rupture.
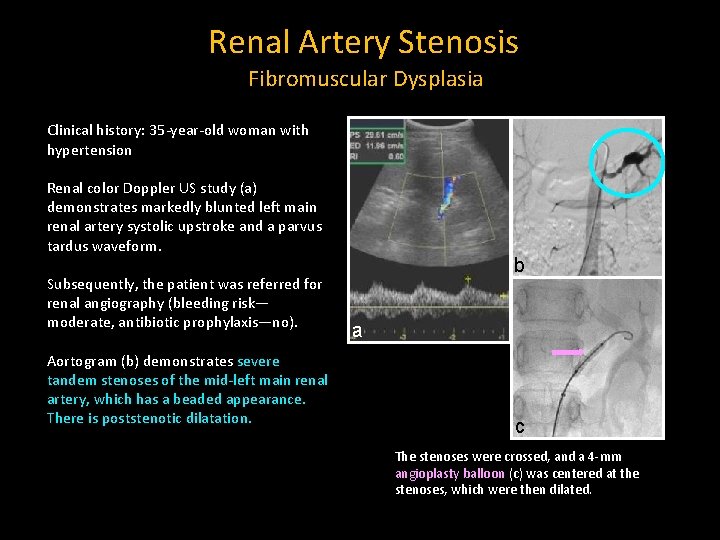
Renal Artery Stenosis Fibromuscular Dysplasia Clinical history: 35 -year-old woman with hypertension Renal color Doppler US study (a) demonstrates markedly blunted left main renal artery systolic upstroke and a parvus tardus waveform. Subsequently, the patient was referred for renal angiography (bleeding risk— moderate, antibiotic prophylaxis—no). Aortogram (b) demonstrates severe tandem stenoses of the mid-left main renal artery, which has a beaded appearance. There is poststenotic dilatation. b a c The stenoses were crossed, and a 4 -mm angioplasty balloon (c) was centered at the stenoses, which were then dilated.
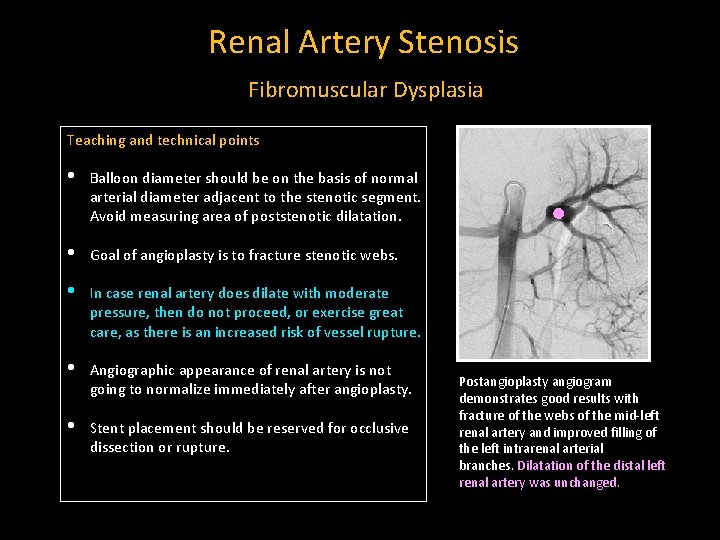
Renal Artery Stenosis Fibromuscular Dysplasia Teaching and technical points • Balloon diameter should be on the basis of normal arterial diameter adjacent to the stenotic segment. Avoid measuring area of poststenotic dilatation. • Goal of angioplasty is to fracture stenotic webs. • In case renal artery does dilate with moderate pressure, then do not proceed, or exercise great care, as there is an increased risk of vessel rupture. • Angiographic appearance of renal artery is not going to normalize immediately after angioplasty. • Stent placement should be reserved for occlusive dissection or rupture. Postangioplasty angiogram demonstrates good results with fracture of the webs of the mid-left renal artery and improved filling of the left intrarenal arterial branches. Dilatation of the distal left renal artery was unchanged.

Summary Endovascular renal interventions are commonly performed procedures at most institutions. Diagnostic and interventional radiologists and trainees should have a better understanding of these commonly performed procedures, including the technical aspects and associated imaging findings.

Suggested Readings • Angle JF, Siddiqi NH, Wallace MJ, et al. Quality improvement guidelines for percutaneous transcatheter embolization: Society of Interventional Radiology Standards of Practice Committee. J Vasc Interv Radiol 2010; 21(10): 1479– 1486. • Chimpiri AR, Natarajan B. Renal vascular lesions: diagnosis and endovascular management. Semin Intervent Radiol 2009; 26(3): 253– 261. • Davis C, Boyett T, Caridi J. Renal artery embolization: application and success in patients with renal cell carcinoma and angiomyolipoma. Semin Intervent Radiol 2007; 24(1): 111– 116. • Greaves NS, Katsogridakis E, Faris B, Murray D. Prophylactic antibiotics for percutaneous endovascular procedures. Eur J Clinical Microbiol Infect Dis 2017; 36(4): 597– 601. • Haas NB, Nathanson KL. Hereditary kidney cancer syndromes. Adv Chronic Kidney Dis 2014; 21(1): 81– 90. • Khoury MH, Gornik HL. Fibromuscular dysplasia (FMD). Vasc Med 2017; 22(3): 248– 252. • Kohi MP, Fidelman N, Behr S, et al. Periprocedural patient care. Radio. Graphics 2015; 35(6): 1766– 1778. • Laberge JM. Interventional management of renal transplant arteriovenous fistula. Semin Intervent Radiol 2004; 21(4): 239– 246. • Leesar MA, Varma J, Shapira A, et al. Prediction of hypertension improvement after stenting of renal artery stenosis: comparative accuracy of translesional pressure gradients, intravascular ultrasound, and angiography. J Am Coll Cardiol 2009; 53(25): 2363– 2371.

Suggested Readings • Leesar MA, Varma J, Shapira A, et al. Prediction of hypertension improvement after stenting of renal artery stenosis: comparative accuracy of translesional pressure gradients, intravascular ultrasound, and angiography. J Am Coll Cardiol 2009; 53(25): 2363 -2371. • Leyon JJ, Littlehales T, Rangarajan B, Hoey ET, Ganeshan A. Endovascular embolization: review of currently available embolization agents. Curr Probl Diagn Radiol 2014; 43(1): 35– 53. • Li D, Pua BB, Madoff DC. Role of embolization in the treatment of renal masses. Semin Intervent Radiol 2014; 31(1): 70– 81. • Lubarsky M, Ray CE, Funaki B. Embolization agents-which one should be used when? part 1: large-vessel embolization. Semin Intervent Radiol 2009; 26(4): 352– 357. • Maruno M, Kiyosue H, Tanoue S, et al. Renal arteriovenous shunts: clinical features, imaging appearance, and transcatheter embolization based on angioarchitecture. Radio. Graphics 2016; 36(2): 580– 595. • Medsinge A, Zajko A, Orons P, Amesur N, Santos E. A case-based approach to common embolization agents used in vascular interventional radiology. AJR Am J Roentgenol 2014; 203(4): 699– 708. • Mousa AY, Abu. Rahma AF, Bozzay J, Broce M, Bates M. Update on intervention versus medical therapy for atherosclerotic renal artery stenosis. J Vasc Surg 2015; 61(6): 1613– 1623.

Suggested Readings • Saba R, Davis J, Balavenkataraman A, et al. Pulmonary arterial hypertension secondary to ethanol sclerotherapy for renal artery embolization. Case Rep Critical Care 2014; 10. 1155/2014/452452. • Tabei T, Tajirika H, Yoshigi J, Kobayashi K. Extensive renal arteriovenous malformations treated by transcatheter arterial embolization. Case reports in Urology. 2017; 10. 1155/2017/2376034. • Vaidya S, Tozer KR, Chen J. An overview of embolic agents. Semin Intervent Radiol 2008; 25(3): 204– 215. • van Brussel PM, van de Hoef TP, de Winter RJ, Vogt L, van den Born BJ. Hemodynamic measurements for the selection of patients with renal artery stenosis: a systematic review. JACC Cardiovasc Interv 2017; 10(10): 973– 985. • Varennes L, Tahon F, Kastler A, et al. Fibromuscular dysplasia: what the radiologist should know: a pictorial review. Insights Imaging. 2015; 6(3): 295– 307. • Venkatesan AM, Kundu S, Sacks D, et al. Practice guidelines for adult antibiotic prophylaxis during vascular and interventional radiology procedures. Written by the Standards of Practice Committee for the Society of Interventional Radiology and Endorsed by the Cardiovascular Interventional Radiological Society of Europe and Canadian Interventional Radiology Association [corrected]: J Vasc Interv Radiol 2010; 21(11): 1611– 1630.
- Slides: 46