Case two Outbreak of a PVL producing MRSA

Case two: Outbreak of a PVL producing MRSA Dr Mark Garvey, Principal Clinical Scientist in Microbiology Associate Director of Infection Prevention and Control mark. garvey@uhb. nhs. uk or m. i. garvey@bham. ac. uk @drmarkgarvey @uhbipc

• • • Overview MRSA Panton Valentine leukocidin (PVL) Case studies PVL Outbreak Conclusions
Staphylococcus aureus • S. aureus: – Gram positive cocci – Frequently coloniser in humans – found in nose of up to 30% of healthy individuals • Opportunistic pathogen: – Wide range of infections including skin and soft tissue infections (SSTI’s), bloodstream infection, pneumonia, endocarditis • Toxins – mediated infection such as Toxic shock syndrome

Meticillin-resistant (‘flucloxacillinresistant’) Staphylococcus aureus (MRSA) • Contains resistance island called Staphylococcal chromosome cassette (SCC) • mec is the genetic element confers resistance to meticillin • mec. A encodes for PBP 2 a; low affinity for meticillin and confers meticillin resistance on S. aureus (ie MRSA) • Six types of SCC harbouring different resistance determinants

Problem – route of transmission • The bugs themselves aren’t the problem • Its where they can get to that is • Healthcare is increasingly invasive and we are working with even more vulnerable patients as medical science progresses • MRSA seek and destroy i. e. screen and decolonise Kiernan M IPS 2018

Panton Valentine Leukocidin (PVL)
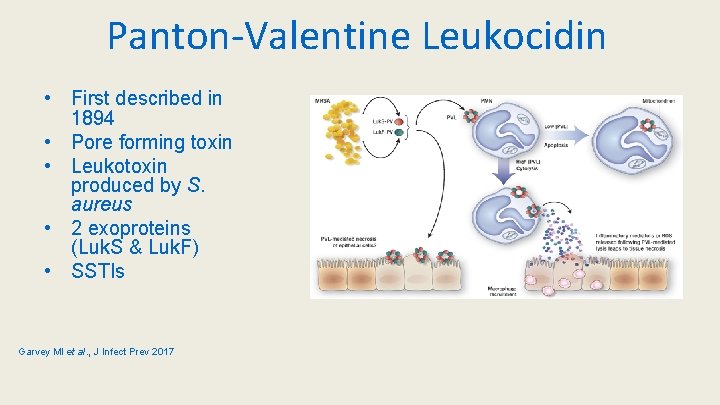
Panton-Valentine Leukocidin • First described in 1894 • Pore forming toxin • Leukotoxin produced by S. aureus • 2 exoproteins (Luk. S & Luk. F) • SSTIs Garvey MI et al. , J Infect Prev 2017

PVL: epidemiology & risk factors • 2 -10% clinical S. aureus isolates are PVL positive • Predominantly associated with community S. aureus (MSSA & MRSA) • Modest disease burden in UK & rest of Europe currently compared with USA • Highly transmissible especially in community – Close contacts e. g. families, social groups, military, gyms. Garvey MI et al. , J Infect Prev 2017

UK epidemiology of PVL • UK PVL have been estimated to be carried in less than 2% of clinical isolates of MSSA. • Reference laboratories only receive a small number of selected isolates so this is likely to under estimate the true burden of the disease • In England, the majority of PVL-positive strains have been MSSA Garvey MI et al. , J Infect Prev 2017

Questions • When to suspect a PVL? • How many do laboratory inhouse PVL testing? • How many send away for PVL testing?

When to suspect PVL? S. aureus (MSSA/MRSA) from a patient with: • Recurrent/multiple boils/abscess if • Necrotising skin and soft tissue infection Especially <40 yrs • Necrotising pneumonia Antimicrobial susceptibility patterns are highly variable • Detection of PVL S. aureus infection depends on clinical suspicion of PVL-related syndrome Garvey MI et al. , J Infect Prev 2017

PVL Outbreak

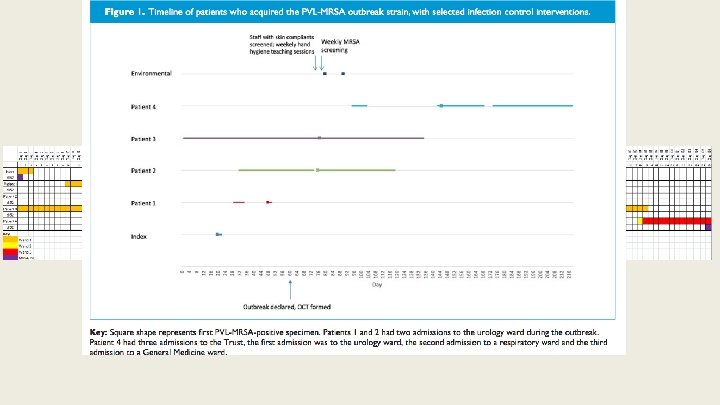
Description of cases Sept 14 – Jan 15: 4 patients acquired PVL MRSA, spa type t 852, associated with an index case. Index case • Admitted to a bay • Admission screen negative for MRSA Garvey MI et al. , J Infect Prev 2017

Patient 1 • Patient 1 was admitted on 2 separate occasions • First admission was in the same bay as index case • Patient 1 subsequently acquired MRSA in October 2014 (acquisition) Garvey MI et al. , J Infect Prev 2017

Patient 2 • Patient 2 admitted October 2014 for elective surgery • Acquired MRSA during second admission to Trust • During second admission was in same bay as patient 1 Garvey MI et al. , J Infect Prev 2017

Patient 3 • Patient 3 complex surgical case admitted in Sept 2014 • Found to be MRSA positive in December 2014 (acquisition) • MRSA bacteraemia January 2015 Garvey MI et al. , J Infect Prev 2017

Patient 4 • • Patient 4 admitted and discharged in December 2014 Patient 4 readmitted to Trust with community acquired pneumonia in February 2015 and was subsequently found to be MRSA positive that month (acquisition), however was on a different ward at the time Typing revealed same strain as Urology outbreak MRSA bacteraemia March 2015 Garvey MI et al. , J Infect Prev 2017

Outbreak control • What would you do if had this situation? • Would you screen staff? • How many undertake staff screening in outbreaks? • What would you do with wondering patients?

What was done? • After the second case an outbreak control team was formed – Included DIPC, Infection Control Nurses, ward medical and nursing staff, domestics, PHE, local commissioners, managerial nurses and Trust communications • Screening of all patients in ward to identify other possible cases of transmission • Staff screening with skin complaints e. g. eczema & boils – undertaken after 4 th case no staff positives • Improving hand hygiene compliance Garvey MI et al. , J Infect Prev 2017

Other key interventions • Adherence to Trust MRSA screening procedures • Local infection prevention control training sessions • Improvement in antimicrobial prescribing – Warning when flucloxacillin is prescribed in MRSA positive patients via electronic prescribing system • Enhanced daily cleaning of bed spaces Garvey MI et al. , J Infect Prev 2017

Cleaning & environmental screening • Do you undertake environmental screening? • What do you screen? • When do you undertake? • What do you do with results?

What we did? • Before enhanced cleaning 16/ 40 sites were positive for MRSA • After enhanced cleaning 6/ 40 sites were positive for MRSA

Summary of PVL MRSA outbreak • Molecular typing & whole genome sequencing revealed all five strains were the same spa type t 852, MLST CC 22, EMRSA 15 (ST 22 -IV) • Strain recognised in Indian sub continent as well as UK • Epidemiology suggests person to person transmission occurred Garvey MI et al. , J Infect Prev 2017

Final thoughts • Risk of spread of this clone in a healthcare setting is high • Potential pathogenicity of strain seemed to be high – associated in this outbreak with 2 MRSA bacteraemias • Molecular typing of MRSA acquisitions was essential to identify and help control outbreak • Stringent adherence to infection prevention and control practice in outbreaks is key to prevent further spread of healthcare associated infections

Thank you Questions?

Supplementary - cleaning

Environmental survival of key pathogens on hospital surfaces Pathogens Survival times S. aureus (including MRSA) 7 days to >12 months Enterococcus spp. (including VRE) 5 days to >48 months Acinetobacter spp. 3 days to 11 months Clostridium difficile (spore form) >5 months Norovirus 8 hours to 28 days (temperature dependent) Pseudomonas aeruginosa 6 hours to 16 months Klebsiella spp. 2 hours to >30 months Neisseria gonorrhoae 20 seconds Hota B et al. , Clin Infect Dis 2011 Kramer A et al. , BMC Infect Dis 2007 Dancer SJ et al. Clin Micro Rev 2014

Vickery K et al. , J Hosp Infect 2012
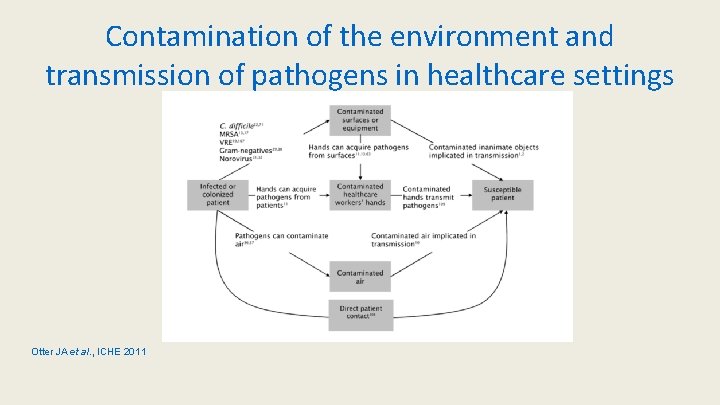
Contamination of the environment and transmission of pathogens in healthcare settings Otter JA et al. , ICHE 2011

Face touching S. aureus • Adults touch their face 23 times per hour – 44% mucous membrane • • 36% mouth 31% nose 27% eyes 6% all three – Mouth 4 x – Nose 3 x – Eye 3 x Kwok et al. , AJIC 2015

Evidence for organism transfer in clinical environments • Inoculation of cauliflower mosaic virus DNA onto phone in an neonatal unit ICU cubicle – Virus spread to 58% of ward sampling sites within 7 days of inoculation – Spread to all five other cubicles – Door handles in other cubicles became positive first • Oelberg DG et al. , Detection of pathogen transmission on neonatal nurseries using DNA markers as surrogate indicators pediatrics (2000)

Risk of transmission from previous room occupant • Meta analysis of all studies with evidence of transmission – Pooled acquisition odds for MRSA – 1. 89 for Gram positives (95% CI: 1. 62 -2. 21) Mitchell B et al. , J Hosp Infect 2015

Transmission of organisms • Prospective cohort study in ICU – Successive occupiers of a room at risk from organisms from previous occupants • Quality audits showed 56% of rooms were not cleaned correctly – Audits were visual only, failure in room door handles (45%), monitor screens (27%) and bedside tables (16%) Nseir et al. , CMI 2010

Who does the cleaning of bed spaces in your hospital?

MRSA and environment • Doorknobs, bed rails, curtains, touchscreens, keyboards contaminated by hands which transmit to other surfaces – MRSA on door handles of 19% of rooms housing MRSA & 7% of door handles of non-MRSA rooms • Oie S et al. , J Hosp Infect 2002 • Staff often say ‘but I did not touch the patient’ – 42% of nurses contaminated gloves with MRSA with no direct contact but by touching objects in rooms of MRSA patients • Boyce JM et al. , ICHE 1997

How many gloves? • Do you know the at our hospital 23 millions pairs of gloves are used • 40% of gloves used in NHS is inappropriate! • Cost savings huge

Audit of equipment • Many items of clinical equipment do not receive appropriate cleaning attention – ATP score showed surfaces cleaned by professional cleaning staff was 64% lower than those by other staff (P=0. 019) • Do we clean well? – Of 27 items cleaned by clinical staff, 89% failed – This is failure • Training • Allocation of responsibility Anderson RE et al. , J Hosp Infect 2011

Questions • • • Do we clean well? Do Doctors clean well? Or know about importance of cleaning? What do you think are the most clean areas? What about stethoscopes? How do you measure cleaning?

Supplementary - final thoughts • Cleaning is a Science – Time to recognize it as such – New RCT-level research on the use of UV-C showed clinically significant reductions in infections • Anderson DJ et al. , Lancet 2017
- Slides: 40