Case study Chronic HBV infection Marion Peters University

Case study: Chronic HBV infection Marion Peters University of California San Francisco 2009

HBV case • 45 year old man admitted with fatigue, malaise and abdominal swelling in June 2003 • He was born in Greece, came to US age 14 • His brother had a liver transplant for HBV in 1998 • On Examination: jaundice, ascites, no muscle wasting, spider nevi

HBV Laboratory and Imaging • Bilirubin 3. 7, AST 129, ALT 106, albumin 2. 4, PT 1. 7, Ammonia 51, Creatinine 0. 9 • MELD 19 • HBs. Ag and HBe. Ag positive • HBV DNA 340, 000 IU/m. L • AFP 741 mcg/L • Acites: paracentesis WCC 183, albumin <1

How would you treat his HBV? Blue Green Red Yellow Pegylated interferon for 48 week Lamivudine 100 mg per day Entecavir 0. 5 mg per day Tenofovir 300 mg per day/ Combo

How would you treat his HBV? Blue Green Red Yellow Pegylated interferon for 48 week Lamivudine 100 mg per day Entecavir 0. 5 mg per day Tenofovir 300 mg per day/ Combo

HBV case-3 • June 2003 started lamivudine 100 mg daily – Well tolerated, lost ascites – Patient had improved liver function • Listed for liver transplantation • Ultrasound cirrhotic liver no masses • CT quadruple phase no masses
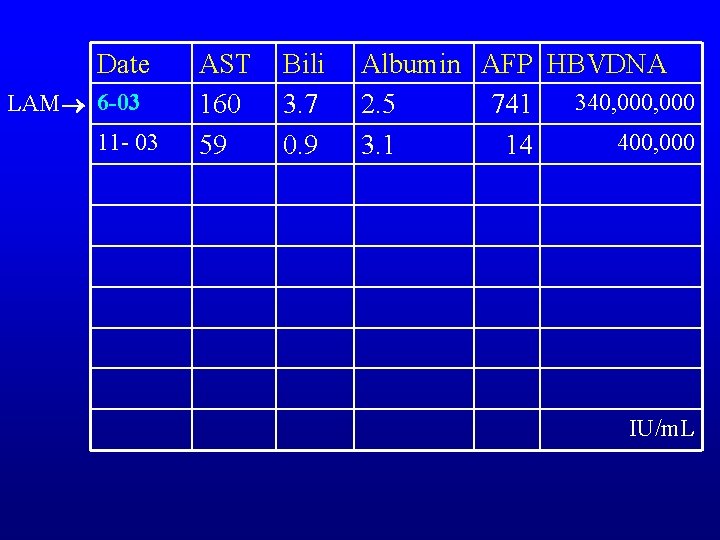
Date LAM 6 -03 11 - 03 AST 160 59 Bili 3. 7 0. 9 Albumin AFP HBVDNA 340, 000 2. 5 741 400, 000 3. 1 14 IU/m. L
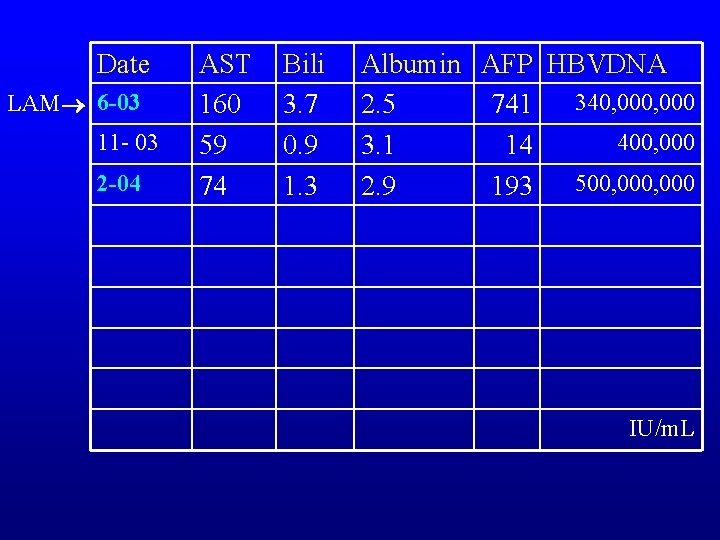
Date LAM 6 -03 11 - 03 2 -04 AST 160 59 74 Bili 3. 7 0. 9 1. 3 Albumin AFP HBVDNA 340, 000 2. 5 741 400, 000 3. 1 14 500, 000 2. 9 193 IU/m. L

What has occurred? Blue Red Green LAM non response LAM resistance Non compliance

What has occurred? Blue Red Green LAM non response LAM resistance Non compliance

100 Patients With YMDD Variants (%) Patients With Resistance (%) HBV DNA at Month 6 of LAM Predicts Later Risk of Resistance N = 159 HBe. Ag-positive patients Median follow-up: 29. 6 months 80 64 60 40 20 32 13 8 12 23 41 118 0 n= ≤ 2 ≤ 3 ≤ 4 >4 HBV DNA at 6 Months (log 10 copies/m. L) Yuen ME, et al. Hepatology. 2001; 34: 785 -791.

HBV status • HBV Genotype A, HBe. Ag positive • Polymerase mutations – L 180 M, +M 204 V – no precore mutations detected – No ADV mutations detected • HIV negative • HDV negative

HBV: How would you treat his HBV now with LAM resistance? Blue Red Green Yellow Switch to Adefovir/TDF Switch to Entecavir 0. 5 mg per day Add Adefovir/TDF

HBV: How would you treat his HBV now? Blue Red Green Yellow Switch to Adefovir/TDF Switch to Entecavir 0. 5 mg per day Add Adefovir/TDF
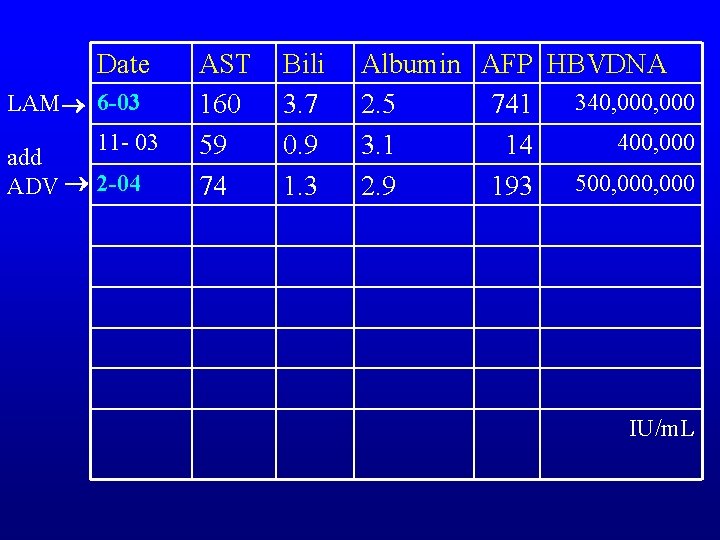
Date LAM 6 -03 11 - 03 add ADV 2 -04 AST 160 59 74 Bili 3. 7 0. 9 1. 3 Albumin AFP HBVDNA 340, 000 2. 5 741 400, 000 3. 1 14 500, 000 2. 9 193 IU/m. L
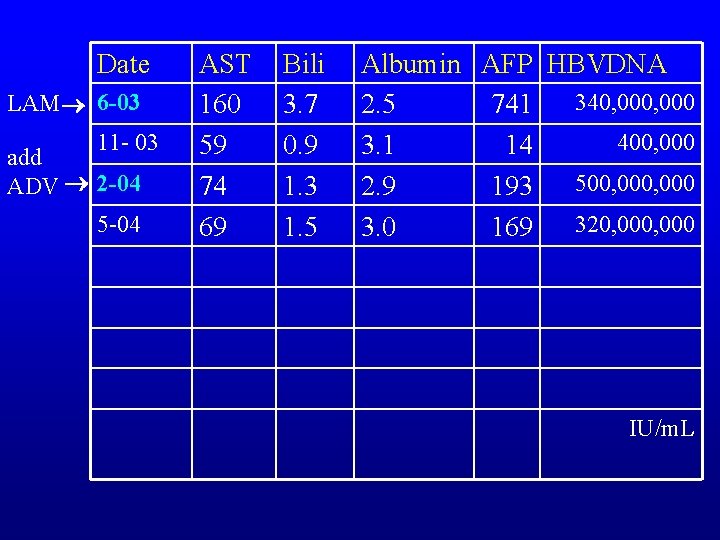
Date LAM 6 -03 11 - 03 add ADV 2 -04 5 -04 AST 160 59 74 69 Bili 3. 7 0. 9 1. 3 1. 5 Albumin AFP HBVDNA 340, 000 2. 5 741 400, 000 3. 1 14 500, 000 2. 9 193 320, 000 3. 0 169 IU/m. L

What has occurred? Blue Red Green Yellow ADV resistance ADV primary non response ADV suboptimal response Non compliance

What has occurred? Blue Red Green Yellow ADV resistance ADV primary non response ADV suboptimal response Non compliance
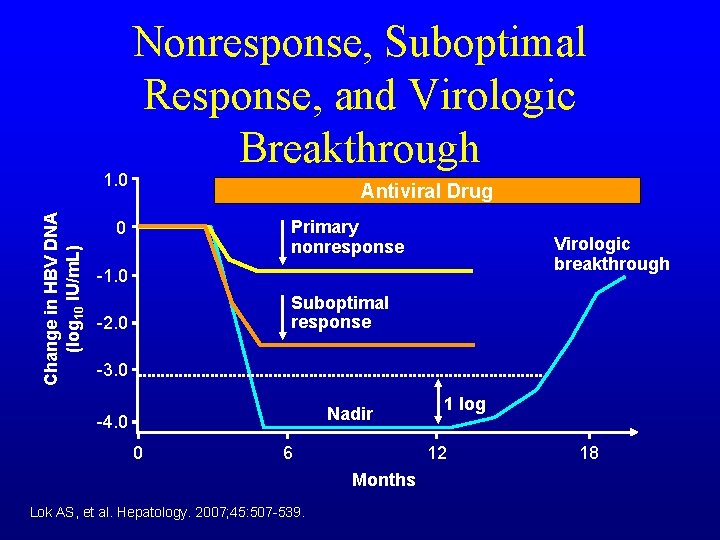
Change in HBV DNA (log 10 IU/m. L) 1. 0 Nonresponse, Suboptimal Response, and Virologic Breakthrough Antiviral Drug Primary nonresponse 0 Virologic breakthrough -1. 0 Suboptimal response -2. 0 -3. 0 Nadir -4. 0 0 6 12 Months Lok AS, et al. Hepatology. 2007; 45: 507 -539. 1 log 18
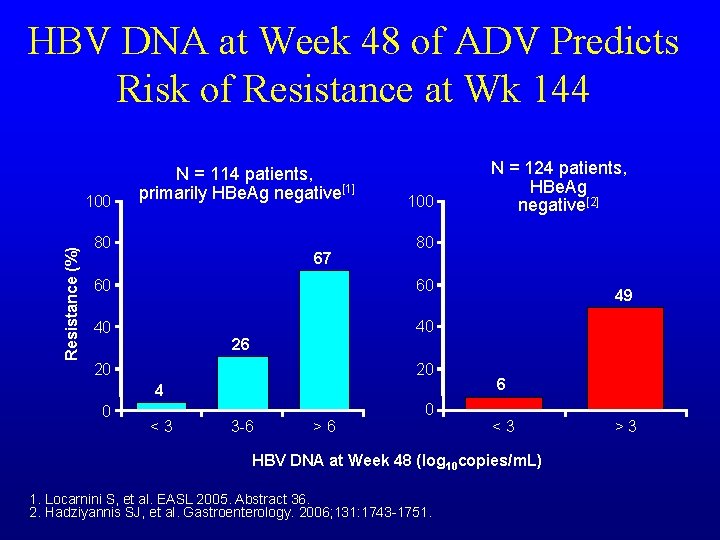
HBV DNA at Week 48 of ADV Predicts Risk of Resistance at Wk 144 Resistance (%) 100 N = 114 patients, primarily HBe. Ag negative[1] 80 67 100 80 60 60 40 40 26 20 20 4 0 <3 3 -6 >6 N = 124 patients, HBe. Ag negative[2] 0 49 6 <3 HBV DNA at Week 48 (log 10 copies/m. L) 1. Locarnini S, et al. EASL 2005. Abstract 36. 2. Hadziyannis SJ, et al. Gastroenterology. 2006; 131: 1743 -1751. >3

HBV-case: What would you do? Blue Red Green Yellow Continue ADV Add Tenofovir 300 mg Change to TDF and ETV Change to TDF and Lam/FTC

HBV-case: What would you do? Blue Red Green Yellow Continue ADV Add Tenofovir 300 mg Change to TDF and ETV Change to TDF and Lam/FTC
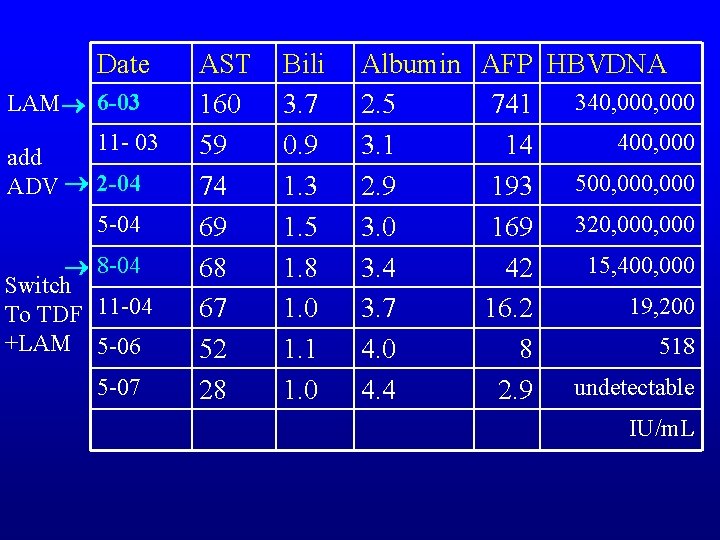
Date LAM 6 -03 11 - 03 add ADV 2 -04 5 -04 8 -04 Switch To TDF 11 -04 +LAM 5 -06 5 -07 AST 160 59 74 69 68 67 52 28 Bili 3. 7 0. 9 1. 3 1. 5 1. 8 1. 0 1. 1 1. 0 Albumin AFP HBVDNA 340, 000 2. 5 741 400, 000 3. 1 14 500, 000 2. 9 193 320, 000 3. 0 169 15, 400, 000 3. 4 42 19, 200 3. 7 16. 2 518 4. 0 8 4. 4 2. 9 undetectable IU/m. L
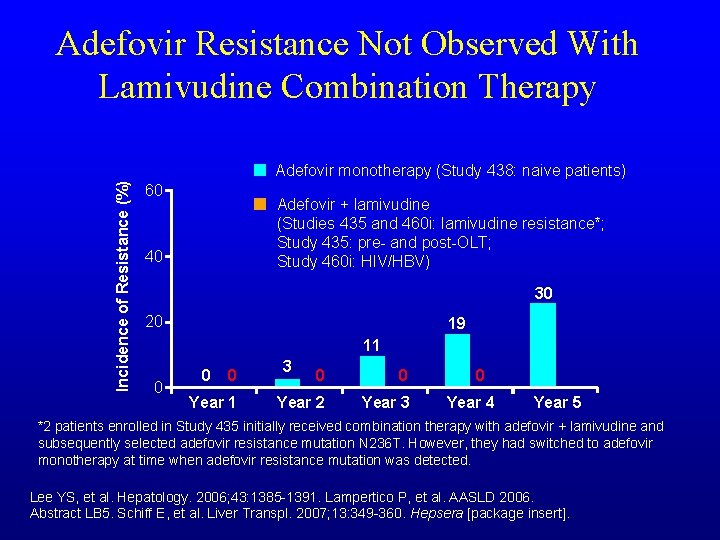
Adefovir Resistance Not Observed With Lamivudine Combination Therapy Incidence of Resistance (%) Adefovir monotherapy (Study 438: naive patients) 60 Adefovir + lamivudine (Studies 435 and 460 i: lamivudine resistance*; Study 435: pre- and post-OLT; Study 460 i: HIV/HBV) 40 30 20 19 11 0 0 0 Year 1 3 0 0 Year 2 Year 3 0 Year 4 Year 5 *2 patients enrolled in Study 435 initially received combination therapy with adefovir + lamivudine and subsequently selected adefovir resistance mutation N 236 T. However, they had switched to adefovir monotherapy at time when adefovir resistance mutation was detected. Lee YS, et al. Hepatology. 2006; 43: 1385 -1391. Lampertico P, et al. AASLD 2006. Abstract LB 5. Schiff E, et al. Liver Transpl. 2007; 13: 349 -360. Hepsera [package insert].

Management of HBV • Check response at 12 and 24 weeks • If no response switch • When virologic breakthrough occurs – “Switch to” another drug – “Add on” another drug – “Switch to” and “add on” another drug • Choice of second drug generally dictated by lack of cross-resistance

Combination therapy • Sequential monotherapy with nucleos(t)ide analogues has led to HBV resistance • Resistance has been low with combination therapy • Peg IFN and LAM showed more HBV DNA suppression while on therapy, but lost after end of therapy, no increased HBe. Ag serconversion • ADV and LAM/FTC less resistance but no increase in efficacy Lampertico Gastro 2007; Yim HJ, et al. Hepatology. 2006: 43: S 173 -181; Shaw T, et al. AASLD 2007. Abstract 986; Schildgen O, et al. N Engl J Med. 2006; 354: 1807 -1812; Reijnders JG, AASLD 2007. Abstract 951; Colonno R, et al. Hepatology. 2006; 44: 1656 -1665.
- Slides: 26