Carfilzomib Cyclophosphamide and Dexamethasone CCd for Newly Diagnosed

Carfilzomib, Cyclophosphamide and Dexamethasone (CCd) for Newly Diagnosed Multiple Myeloma (MM) Patients: Initial Results of a Multicenter, Open Label Phase II Study Bringhen S et al. Proc EHA 2013; Abstract S 578.

Background VMP and MPT, which are standard therapies for elderly patients with newly diagnosed multiple myeloma (NDMM), induce about a 30% near-complete response/complete response rate, with a 35% discontinuation rate due to adverse events. l Carfilzomib, an irreversible proteasome inhibitor, has shown significant activity and favorable toxicity in MM. l Preliminary results with a combination of carfilzomib with cyclophosphamide and dexamethasone (CCd) showed encouraging activity in elderly patients with NDMM (Proc ASH 2012; Abstract 730). l Study objective: To present updated efficacy and safety results with the CCd regimen after 8 months of follow-up for patients with symptomatic NDMM who are ≥ 65 years old or ineligible for autologous stem cell transplantation. l Bringhen S et al. Proc EHA 2013; Abstract S 578.
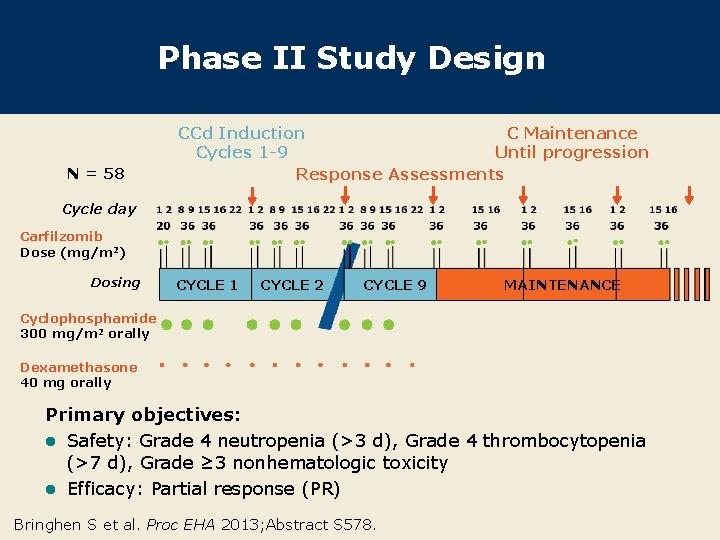
Phase II Study Design N = 58 CCd Induction C Maintenance Cycles 1 -9 Until progression Response Assessments Cycle day Carfilzomib Dose (mg/m 2) Dosing CYCLE 1 CYCLE 2 CYCLE 9 MAINTENANCE Cyclophosphamide 300 mg/m 2 orally Dexamethasone 40 mg orally Primary objectives: l Safety: Grade 4 neutropenia (>3 d), Grade 4 thrombocytopenia (>7 d), Grade ≥ 3 nonhematologic toxicity l Efficacy: Partial response (PR) Bringhen S et al. Proc EHA 2013; Abstract S 578.
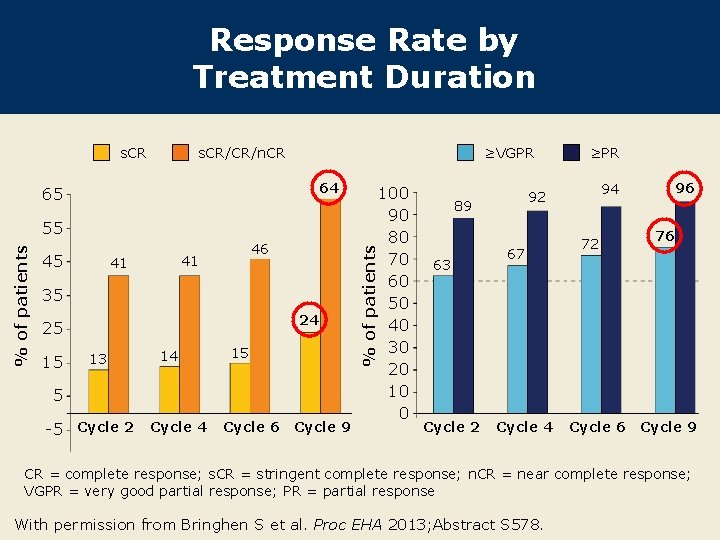
Response Rate by Treatment Duration s. CR ≥VGPR s. CR/CR/n. CR % of patients 55 45 46 41 41 35 24 25 15 13 14 15 5 -5 Cycle 2 Cycle 4 Cycle 6 Cycle 9 100 90 80 70 60 50 40 30 20 10 0 % of patients 64 65 Cycle 2 67 Cycle 4 96 94 92 89 63 ≥PR 72 Cycle 6 76 Cycle 9 CR = complete response; s. CR = stringent complete response; n. CR = near complete response; VGPR = very good partial response; PR = partial response With permission from Bringhen S et al. Proc EHA 2013; Abstract S 578.
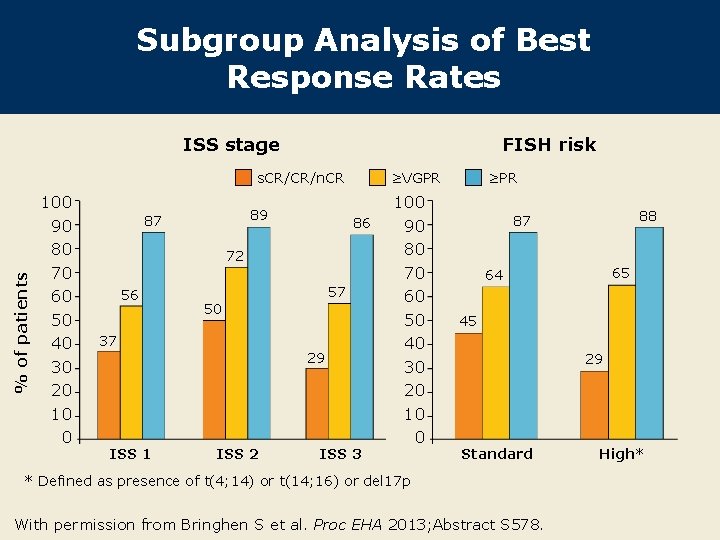
Subgroup Analysis of Best Response Rates ISS stage FISH risk % of patients s. CR/CR/n. CR 100 90 80 70 60 50 40 30 20 10 0 89 87 ≥VGPR 86 72 56 57 50 37 29 ISS 1 ISS 2 ISS 3 100 90 80 70 60 50 40 30 20 10 0 ≥PR 88 87 65 64 45 29 Standard * Defined as presence of t(4; 14) or t(14; 16) or del 17 p With permission from Bringhen S et al. Proc EHA 2013; Abstract S 578. High*
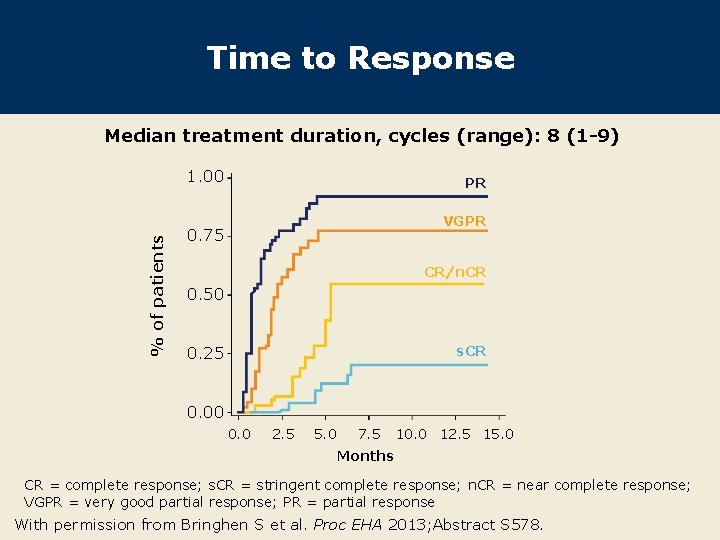
Time to Response Median treatment duration, cycles (range): 8 (1 -9) % of patients 1. 00 PR VGPR 0. 75 CR/n. CR 0. 50 s. CR 0. 25 0. 00 0. 0 2. 5 5. 0 7. 5 10. 0 12. 5 15. 0 Months CR = complete response; s. CR = stringent complete response; n. CR = near complete response; VGPR = very good partial response; PR = partial response With permission from Bringhen S et al. Proc EHA 2013; Abstract S 578.
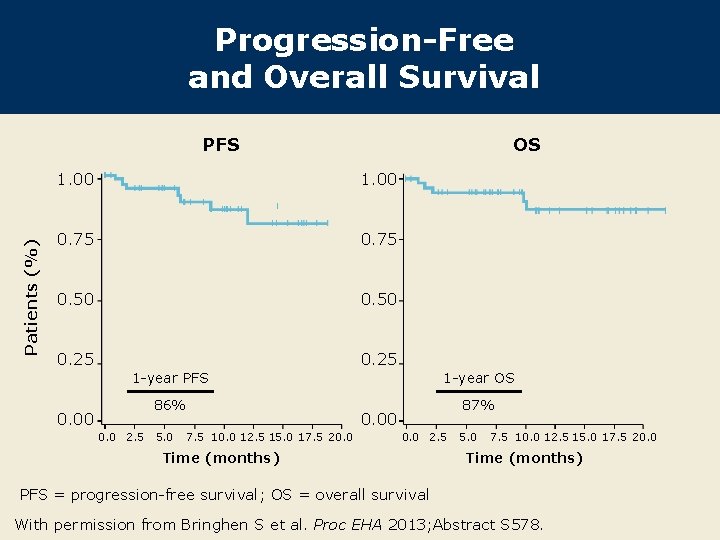
Progression-Free and Overall Survival Patients (%) PFS OS 1. 00 0. 75 0. 50 0. 25 1 -year PFS 1 -year OS 86% 87% 0. 00 0. 0 2. 5 5. 0 7. 5 10. 0 12. 5 15. 0 17. 5 20. 0 2. 5 Time (months) 5. 0 7. 5 10. 0 12. 5 15. 0 17. 5 20. 0 Time (months) PFS = progression-free survival; OS = overall survival With permission from Bringhen S et al. Proc EHA 2013; Abstract S 578.
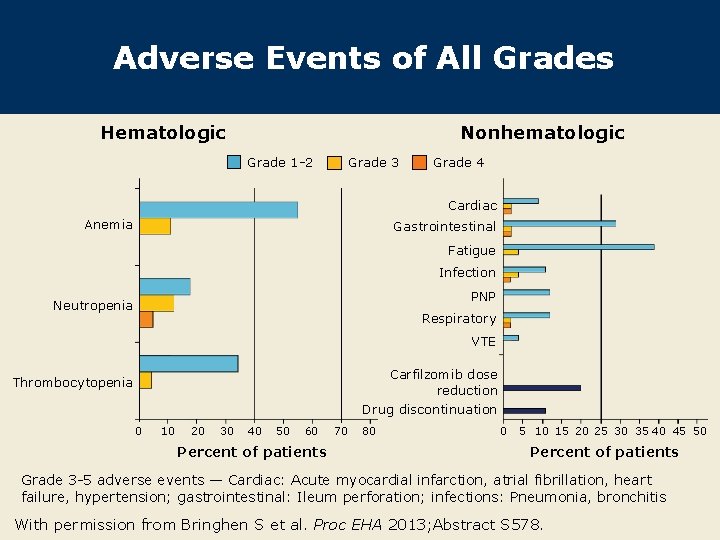
Adverse Events of All Grades Hematologic Nonhematologic Grade 1 -2 Grade 3 Grade 4 Cardiac Anemia Gastrointestinal Fatigue Infection PNP Neutropenia Respiratory VTE Carfilzomib dose reduction Drug discontinuation Thrombocytopenia 0 10 20 30 40 50 60 Percent of patients 70 80 0 5 10 15 20 25 30 35 40 45 50 Percent of patients Grade 3 -5 adverse events — Cardiac: Acute myocardial infarction, atrial fibrillation, heart failure, hypertension; gastrointestinal: Ileum perforation; infections: Pneumonia, bronchitis With permission from Bringhen S et al. Proc EHA 2013; Abstract S 578.

Author Conclusions CCd showed encouraging activity in elderly patients with NDMM in comparison to results with MPT and VMP from other studies. 1 -3 – ≥VGPR: 76% vs 36% (MPT) or 41% (VMP) – n. CR/CR/s. CR: 64% vs 27% (MPT) or 30%* (VMP) – s. CR: 24% (not reported for MPT or VMP) l The CCd combination was well tolerated. Grade 3 or 4 adverse events included – Thrombocytopenia: 4% vs 3% (MPT) or 37% (VMP) – Peripheral neuropathy: 0% vs 6% (MPT) or 14% (VMP) – Venous thromboembolic events: 0% vs 9% (MPT) or 1% l (VMP) – Treatment discontinuation: 11% vs 35% (MPT) or 33% (VMP) * CR only, n. CR not reported 1 Blood 2011; 118: 1239; 2 New Engl J Med 2008; 359: 906; Bringhen S et al. Proc EHA 2013; Abstract S 578. 3 Lancet 2006; 367: 825

Investigator Commentary: Initial Results of the Phase II Study of CCd for Patients with NDMM From this and several other studies, it is increasingly obvious that melphalan is much more toxic than dexamethasone and cyclophosphamide. Melphalan probably adds some benefit over cyclophosphamide and dexamethasone, but in continuous or maintenance therapy the advantages are limited and do not counterbalance the disadvantages of its toxicity. Cyclophosphamide is better tolerated than melphalan, including by patients older than 75 years. More importantly, this study showed that it is possible to double the rate of CR or n. CR with CCd (64%) in comparison to the VMP regimen (30%). Patients achieved a stringent CR of 24% with CCd. As other studies have demonstrated, carfilzomib is well tolerated. When it was used in doses up to 36 mg/m 2 in patients older than 75, no major side effects were observed. Interview with Antonio Palumbo, MD, August 12, 2013
- Slides: 10