Cardiovascular outcomes trials with DPP4 inhibitors EDUCATIONAL SLIDE

Cardiovascular outcomes trials with DPP-4 inhibitors EDUCATIONAL SLIDE MODULE Date of preparation: March 2019 Version: 2. 0 Date of preparation: February 2016 SC-CRP-02183
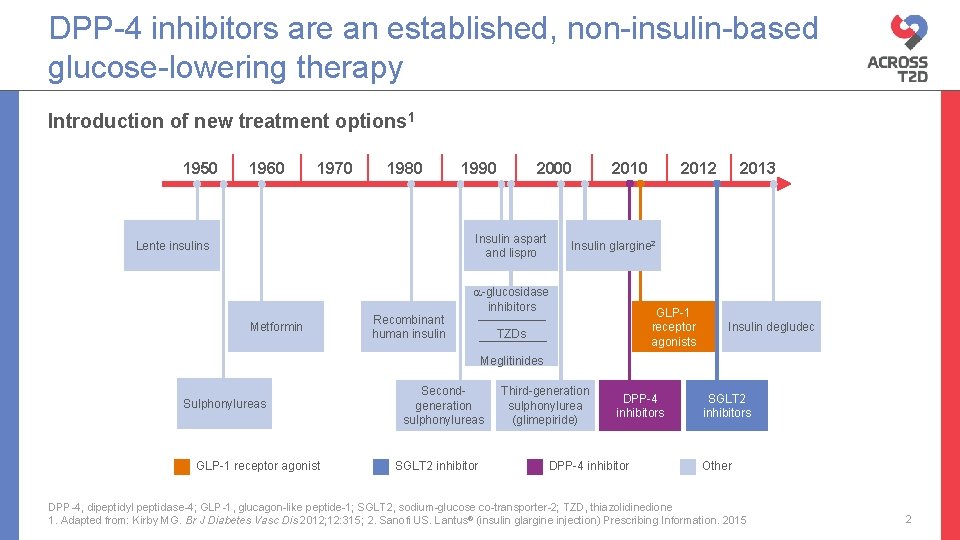
DPP-4 inhibitors are an established, non-insulin-based glucose-lowering therapy Introduction of new treatment options 1 1950 1960 1970 1980 1990 2000 Insulin aspart and lispro Lente insulins Metformin Recombinant human insulin 2010 2012 2013 Insulin glargine 2 -glucosidase inhibitors GLP-1 receptor agonists TZDs Insulin degludec Meglitinides Sulphonylureas GLP-1 receptor agonist Secondgeneration sulphonylureas SGLT 2 inhibitor Third-generation sulphonylurea (glimepiride) DPP-4 inhibitors DPP-4 inhibitor SGLT 2 inhibitors Other DPP-4, dipeptidyl peptidase-4; GLP-1, glucagon-like peptide-1; SGLT 2, sodium-glucose co-transporter-2; TZD, thiazolidinedione 1. Adapted from: Kirby MG. Br J Diabetes Vasc Dis 2012; 12: 315; 2. Sanofi US. Lantus® (insulin glargine injection) Prescribing Information. 2015 2
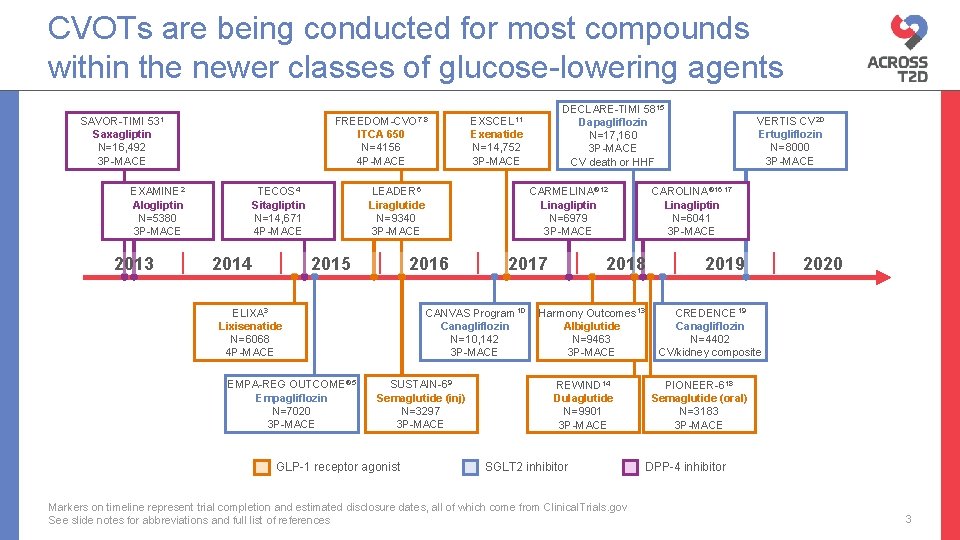
CVOTs are being conducted for most compounds within the newer classes of glucose-lowering agents 531 SAVOR-TIMI Saxagliptin N=16, 492 3 P-MACE EXAMINE 2 Alogliptin N=5380 3 P-MACE 2013 FREEDOM-CVO 7, 8 EXSCEL 11 ITCA 650 N=4156 4 P-MACE Exenatide N=14, 752 3 P-MACE TECOS 4 Sitagliptin N=14, 671 4 P-MACE 2014 LEADER 6 Liraglutide N=9340 3 P-MACE 2015 EMPA-REG OUTCOME® 5 Empagliflozin N=7020 3 P-MACE CARMELINA® 12 Linagliptin N=6979 3 P-MACE 2016 ELIXA 3 Lixisenatide N=6068 4 P-MACE 2017 CANVAS Program 10 Canagliflozin N=10, 142 3 P-MACE SUSTAIN-69 Semaglutide (inj) N=3297 3 P-MACE GLP-1 receptor agonist DECLARE-TIMI 5815 Dapagliflozin N=17, 160 3 P-MACE CV death or HHF CAROLINA® 16, 17 Linagliptin N=6041 3 P-MACE 2018 Harmony Outcomes 13 Albiglutide N=9463 3 P-MACE REWIND 14 Dulaglutide N=9901 3 P-MACE SGLT 2 inhibitor Markers on timeline represent trial completion and estimated disclosure dates, all of which come from Clinical. Trials. gov See slide notes for abbreviations and full list of references VERTIS CV 20 Ertugliflozin N=8000 3 P-MACE 2019 2020 CREDENCE 19 Canagliflozin N=4402 CV/kidney composite PIONEER-618 Semaglutide (oral) N=3183 3 P-MACE DPP-4 inhibitor 3
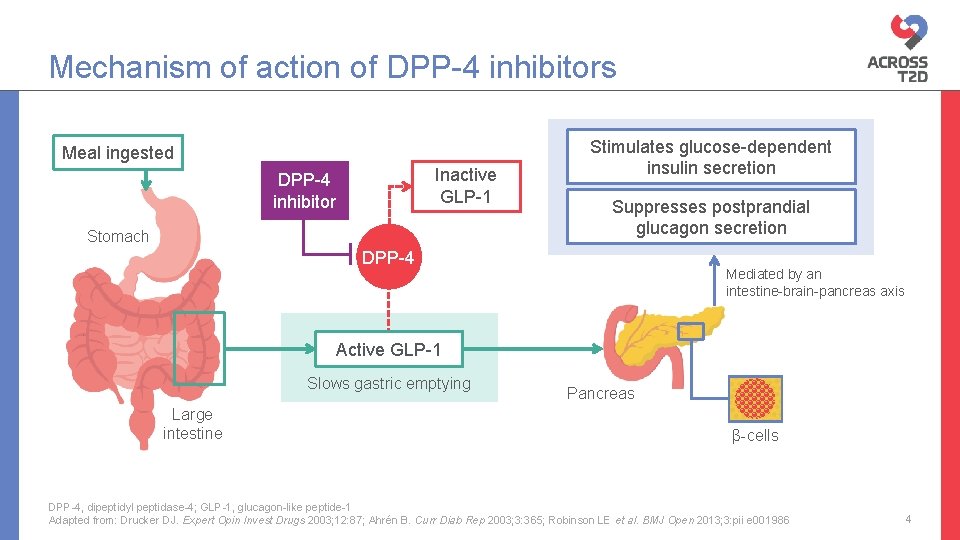
Mechanism of action of DPP-4 inhibitors Meal ingested Inactive GLP-1 DPP-4 inhibitor Stomach Stimulates glucose-dependent insulin secretion Suppresses postprandial glucagon secretion DPP-4 Mediated by an intestine-brain-pancreas axis Active GLP-1 Slows gastric emptying Large intestine Pancreas β-cells DPP-4, dipeptidyl peptidase-4; GLP-1, glucagon-like peptide-1 Adapted from: Drucker DJ. Expert Opin Invest Drugs 2003; 12: 87; Ahrén B. Curr Diab Rep 2003; 3: 365; Robinson LE et al. BMJ Open 2013; 3: pii e 001986 4
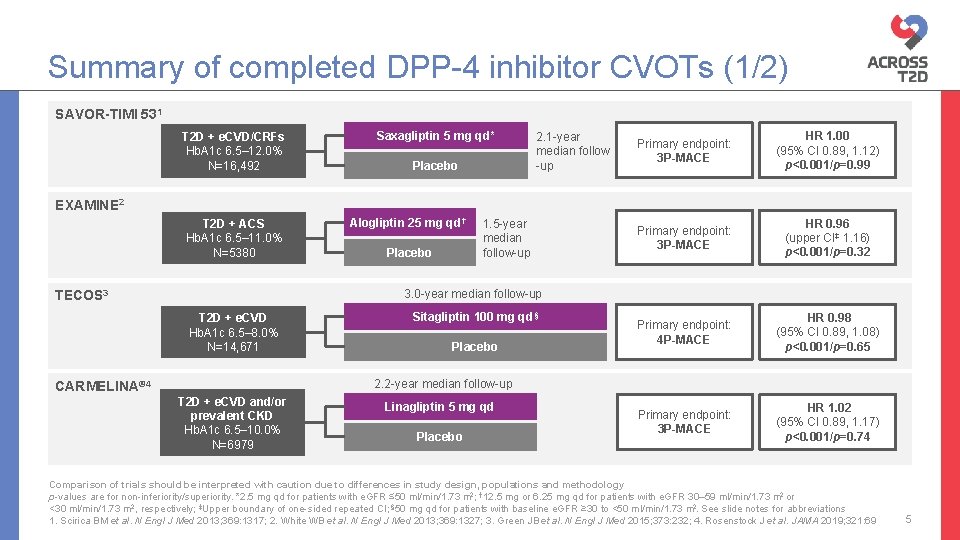
Summary of completed DPP-4 inhibitor CVOTs (1/2) SAVOR-TIMI 531 T 2 D + e. CVD/CRFs Hb. A 1 c 6. 5– 12. 0% N=16, 492 Saxagliptin 5 mg qd* Placebo 2. 1 -year median follow -up Primary endpoint: 3 P-MACE HR 1. 00 (95% CI 0. 89, 1. 12) p<0. 001/p=0. 99 Primary endpoint: 3 P-MACE HR 0. 96 (upper CI‡ 1. 16) p<0. 001/p=0. 32 Primary endpoint: 4 P-MACE HR 0. 98 (95% CI 0. 89, 1. 08) p<0. 001/p=0. 65 Primary endpoint: 3 P-MACE HR 1. 02 (95% CI 0. 89, 1. 17) p<0. 001/p=0. 74 EXAMINE 2 T 2 D + ACS Hb. A 1 c 6. 5– 11. 0% N=5380 Alogliptin 25 mg qd † Placebo 1. 5 -year median follow-up 3. 0 -year median follow-up TECOS 3 Sitagliptin 100 mg qd § T 2 D + e. CVD Hb. A 1 c 6. 5– 8. 0% N=14, 671 Placebo 2. 2 -year median follow-up CARMELINA® 4 T 2 D + e. CVD and/or prevalent CKD Hb. A 1 c 6. 5– 10. 0% N=6979 Linagliptin 5 mg qd Randomisation 3 1 2 Placebo Median follow-up (years) Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology p-values are for non-inferiority/superiority. *2. 5 mg qd for patients with e. GFR ≤ 50 ml/min/1. 73 m 2; † 12. 5 mg or 6. 25 mg qd for patients with e. GFR 30– 59 ml/min/1. 73 m 2 or <30 ml/min/1. 73 m 2, respectively; ‡Upper boundary of one-sided repeated CI; § 50 mg qd for patients with baseline e. GFR ≥ 30 to <50 ml/min/1. 73 m 2. See slide notes for abbreviations 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327; 3. Green JB et al. N Engl J Med 2015; 373: 232; 4. Rosenstock J et al. JAMA 2019; 321: 69 5
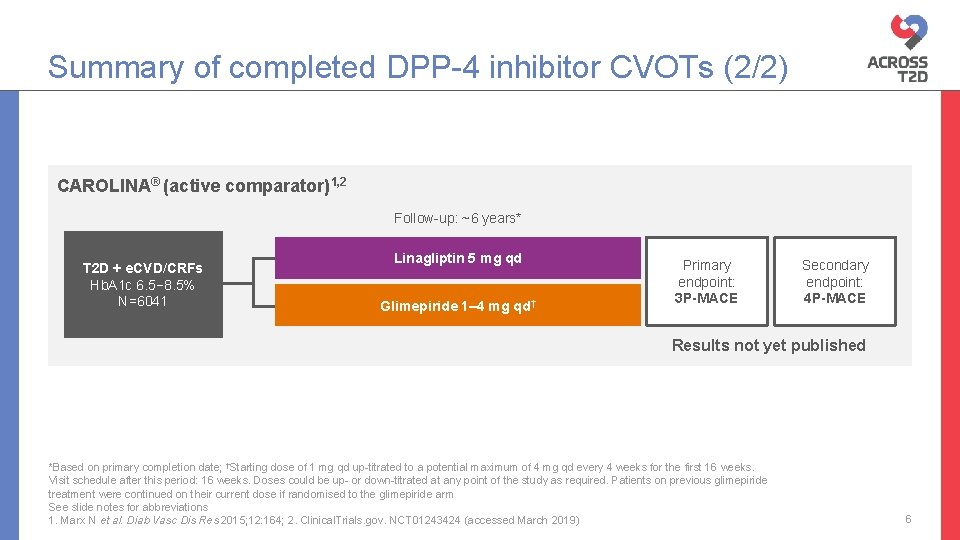
Summary of completed DPP-4 inhibitor CVOTs (2/2) CAROLINA® (active comparator)1, 2 Follow-up: ~6 years* T 2 D + e. CVD/CRFs Hb. A 1 c 6. 5− 8. 5% N=6041 Linagliptin 5 mg qd Glimepiride 1– 4 mg qd† Primary endpoint: 3 P-MACE Secondary endpoint: 4 P-MACE Results not yet published *Based on primary completion date; †Starting dose of 1 mg qd up-titrated to a potential maximum of 4 mg qd every 4 weeks for the first 16 weeks. Visit schedule after this period: 16 weeks. Doses could be up- or down-titrated at any point of the study as required. Patients on previous glimepiride treatment were continued on their current dose if randomised to the glimepiride arm See slide notes for abbreviations 1. Marx N et al. Diab Vasc Dis Res 2015; 12: 164; 2. Clinical. Trials. gov. NCT 01243424 (accessed March 2019) 6
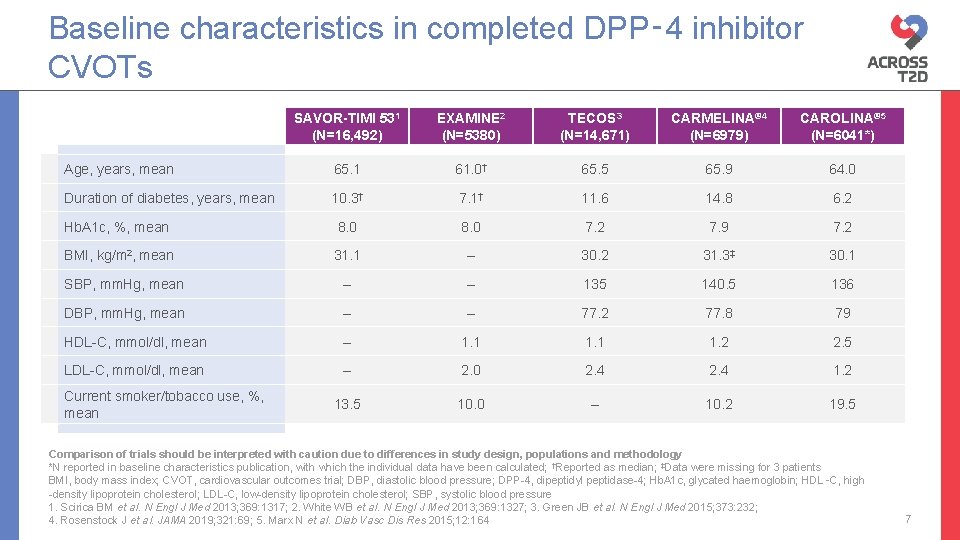
Baseline characteristics in completed DPP‑ 4 inhibitor CVOTs SAVOR-TIMI 531 (N=16, 492) EXAMINE 2 (N=5380) TECOS 3 (N=14, 671) CARMELINA® 4 (N=6979) CAROLINA® 5 (N=6041*) Age, years, mean 65. 1 61. 0† 65. 5 65. 9 64. 0 Duration of diabetes, years, mean 10. 3† 7. 1† 11. 6 14. 8 6. 2 Hb. A 1 c, %, mean 8. 0 7. 2 7. 9 7. 2 BMI, kg/m 2, mean 31. 1 – 30. 2 31. 3‡ 30. 1 SBP, mm. Hg, mean – – 135 140. 5 136 DBP, mm. Hg, mean – – 77. 2 77. 8 79 HDL-C, mmol/dl, mean – 1. 1 1. 2 2. 5 LDL-C, mmol/dl, mean – 2. 0 2. 4 1. 2 13. 5 10. 0 – 10. 2 19. 5 Current smoker/tobacco use, %, mean Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology *N reported in baseline characteristics publication, with which the individual data have been calculated; †Reported as median; ‡Data were missing for 3 patients BMI, body mass index; CVOT, cardiovascular outcomes trial; DBP, diastolic blood pressure; DPP-4, dipeptidyl peptidase-4; Hb. A 1 c, glycated haemoglobin; HDL‑C, high -density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SBP, systolic blood pressure 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327; 3. Green JB et al. N Engl J Med 2015; 373: 232; 4. Rosenstock J et al. JAMA 2019; 321: 69; 5. Marx N et al. Diab Vasc Dis Res 2015; 12: 164 7
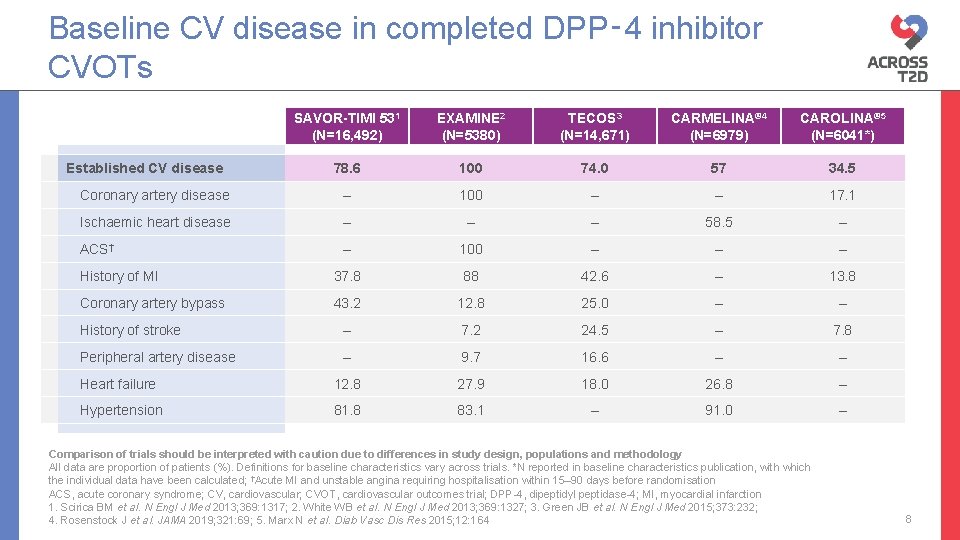
Baseline CV disease in completed DPP‑ 4 inhibitor CVOTs SAVOR-TIMI 531 (N=16, 492) EXAMINE 2 (N=5380) TECOS 3 (N=14, 671) CARMELINA® 4 (N=6979) CAROLINA® 5 (N=6041*) 78. 6 100 74. 0 57 34. 5 Coronary artery disease – 100 – – 17. 1 Ischaemic heart disease – – – 58. 5 – ACS† – 100 – – – History of MI 37. 8 88 42. 6 – 13. 8 Coronary artery bypass 43. 2 12. 8 25. 0 – – History of stroke – 7. 2 24. 5 – 7. 8 Peripheral artery disease – 9. 7 16. 6 – – Heart failure 12. 8 27. 9 18. 0 26. 8 – Hypertension 81. 8 83. 1 – 91. 0 – Established CV disease Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology All data are proportion of patients (%). Definitions for baseline characteristics vary across trials. *N reported in baseline characteristics publication, with which the individual data have been calculated; †Acute MI and unstable angina requiring hospitalisation within 15– 90 days before randomisation ACS, acute coronary syndrome; CV, cardiovascular; CVOT, cardiovascular outcomes trial; DPP-4, dipeptidyl peptidase-4; MI, myocardial infarction 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327; 3. Green JB et al. N Engl J Med 2015; 373: 232; 4. Rosenstock J et al. JAMA 2019; 321: 69; 5. Marx N et al. Diab Vasc Dis Res 2015; 12: 164 8
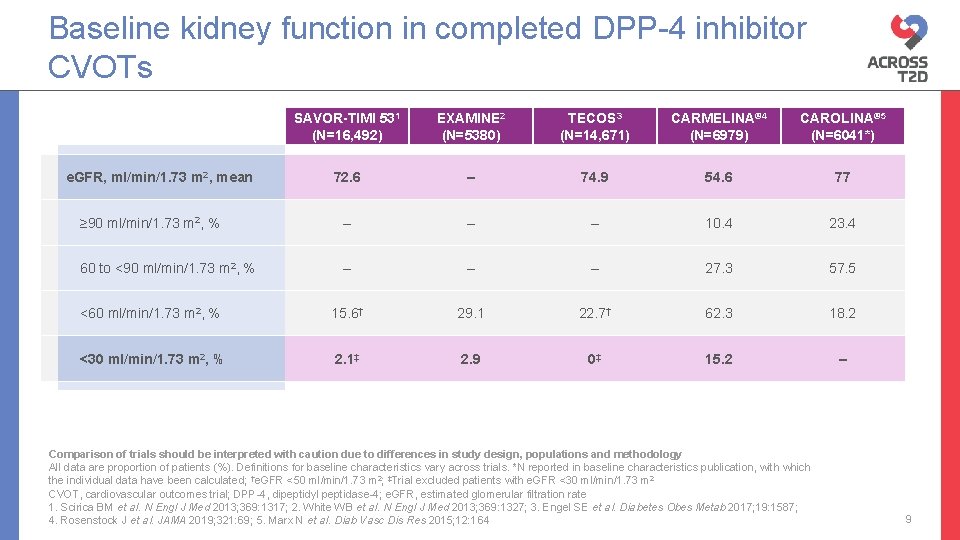
Baseline kidney function in completed DPP-4 inhibitor CVOTs SAVOR-TIMI 531 (N=16, 492) EXAMINE 2 (N=5380) TECOS 3 (N=14, 671) CARMELINA® 4 (N=6979) CAROLINA® 5 (N=6041*) 72. 6 – 74. 9 54. 6 77 ≥ 90 ml/min/1. 73 m 2, % – – – 10. 4 23. 4 60 to <90 ml/min/1. 73 m 2, % – – – 27. 3 57. 5 <60 ml/min/1. 73 m 2, % 15. 6† 29. 1 22. 7† 62. 3 18. 2 <30 ml/min/1. 73 m 2, % 2. 1‡ 2. 9 0‡ 15. 2 – e. GFR, ml/min/1. 73 m 2, mean Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology All data are proportion of patients (%). Definitions for baseline characteristics vary across trials. *N reported in baseline characteristics publication, with which the individual data have been calculated; †e. GFR <50 ml/min/1. 73 m 2; ‡Trial excluded patients with e. GFR <30 ml/min/1. 73 m 2 CVOT, cardiovascular outcomes trial; DPP-4, dipeptidyl peptidase-4; e. GFR, estimated glomerular filtration rate 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327; 3. Engel SE et al. Diabetes Obes Metab 2017; 19: 1587; 4. Rosenstock J et al. JAMA 2019; 321: 69; 5. Marx N et al. Diab Vasc Dis Res 2015; 12: 164 9
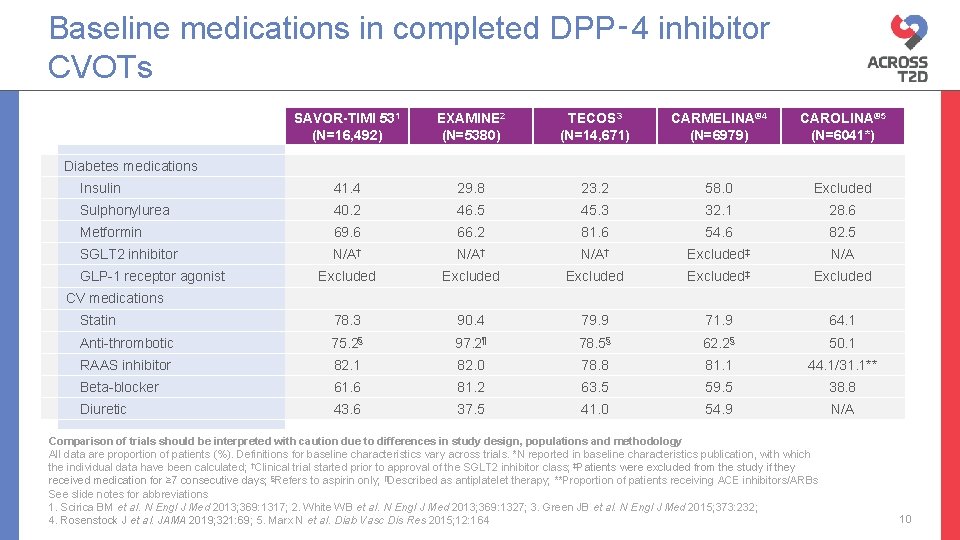
Baseline medications in completed DPP‑ 4 inhibitor CVOTs SAVOR-TIMI 531 (N=16, 492) EXAMINE 2 (N=5380) TECOS 3 (N=14, 671) CARMELINA® 4 (N=6979) CAROLINA® 5 (N=6041*) Insulin 41. 4 29. 8 23. 2 58. 0 Excluded Sulphonylurea 40. 2 46. 5 45. 3 32. 1 28. 6 Metformin 69. 6 66. 2 81. 6 54. 6 82. 5 SGLT 2 inhibitor N/A† Excluded‡ N/A Excluded‡ Excluded Statin 78. 3 90. 4 79. 9 71. 9 64. 1 Anti-thrombotic 75. 2§ 97. 2¶ 78. 5§ 62. 2§ 50. 1 RAAS inhibitor 82. 1 82. 0 78. 8 81. 1 44. 1/31. 1** Beta-blocker 61. 6 81. 2 63. 5 59. 5 38. 8 Diuretic 43. 6 37. 5 41. 0 54. 9 N/A Diabetes medications GLP-1 receptor agonist CV medications Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology All data are proportion of patients (%). Definitions for baseline characteristics vary across trials. *N reported in baseline characteristics publication, with which the individual data have been calculated; †Clinical trial started prior to approval of the SGLT 2 inhibitor class; ‡Patients were excluded from the study if they received medication for ≥ 7 consecutive days; §Refers to aspirin only; ¶Described as antiplatelet therapy; **Proportion of patients receiving ACE inhibitors/ARBs See slide notes for abbreviations 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327; 3. Green JB et al. N Engl J Med 2015; 373: 232; 4. Rosenstock J et al. JAMA 2019; 321: 69; 5. Marx N et al. Diab Vasc Dis Res 2015; 12: 164 10
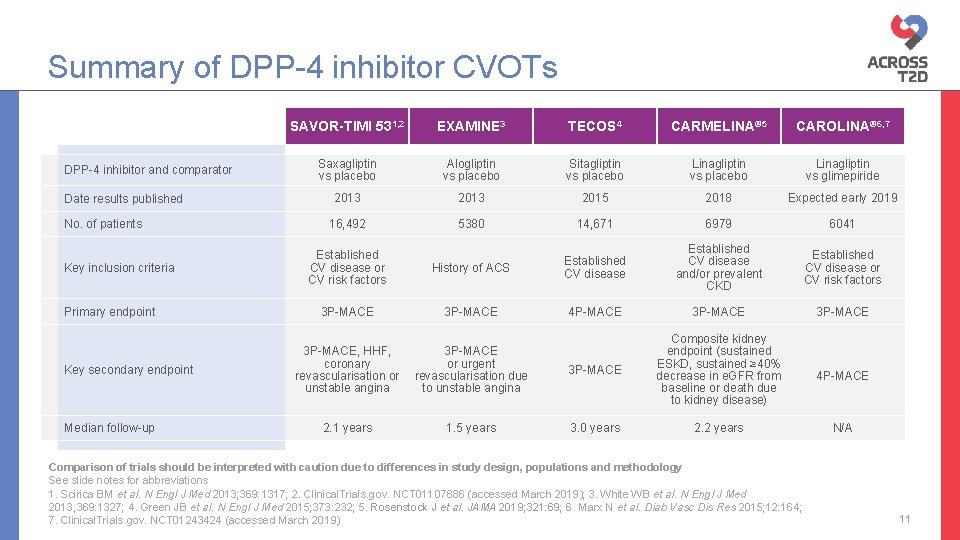
Summary of DPP-4 inhibitor CVOTs DPP-4 inhibitor and comparator Date results published No. of patients Key inclusion criteria Primary endpoint Key secondary endpoint Median follow-up SAVOR-TIMI 531, 2 EXAMINE 3 TECOS 4 CARMELINA® 5 CAROLINA® 6, 7 Saxagliptin vs placebo Alogliptin vs placebo Sitagliptin vs placebo Linagliptin vs glimepiride 2013 2015 2018 Expected early 2019 16, 492 5380 14, 671 6979 6041 Established CV disease or CV risk factors History of ACS Established CV disease and/or prevalent CKD Established CV disease or CV risk factors 3 P-MACE 4 P-MACE 3 P-MACE, HHF, coronary revascularisation or unstable angina 3 P-MACE or urgent revascularisation due to unstable angina 3 P-MACE Composite kidney endpoint (sustained ESKD, sustained ≥ 40% decrease in e. GFR from baseline or death due to kidney disease) 2. 1 years 1. 5 years 3. 0 years 2. 2 years Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology See slide notes for abbreviations 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. Clinical. Trials. gov. NCT 01107886 (accessed March 2019); 3. White WB et al. N Engl J Med 2013; 369: 1327; 4. Green JB et al. N Engl J Med 2015; 373: 232; 5. Rosenstock J et al. JAMA 2019; 321: 69; 6. Marx N et al. Diab Vasc Dis Res 2015; 12: 164; 7. Clinical. Trials. gov. NCT 01243424 (accessed March 2019) 4 P-MACE N/A 11
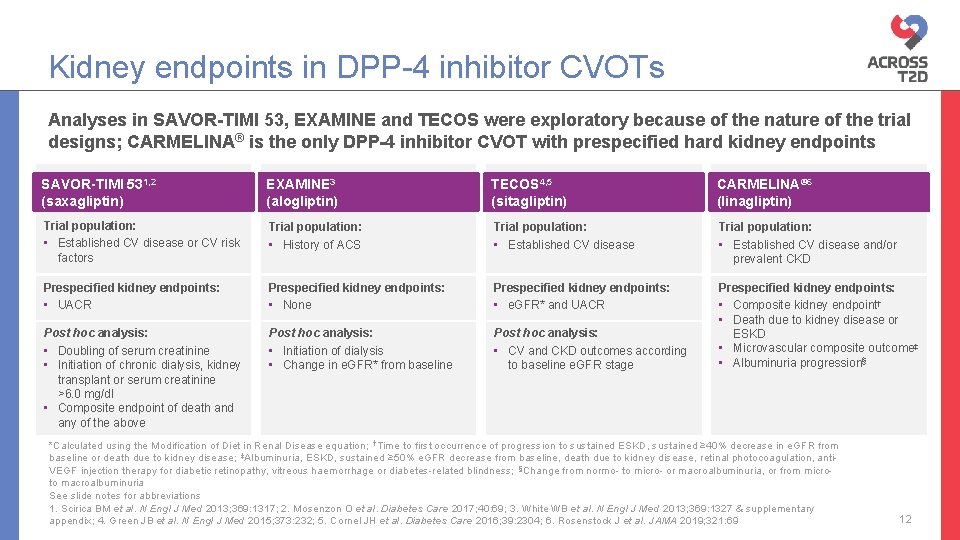
Kidney endpoints in DPP-4 inhibitor CVOTs Analyses in SAVOR-TIMI 53, EXAMINE and TECOS were exploratory because of the nature of the trial designs; CARMELINA® is the only DPP-4 inhibitor CVOT with prespecified hard kidney endpoints SAVOR-TIMI 531, 2 (saxagliptin) EXAMINE 3 (alogliptin) TECOS 4, 5 (sitagliptin) CARMELINA® 6 (linagliptin) Trial population: • Established CV disease or CV risk factors Trial population: • History of ACS Trial population: • Established CV disease and/or prevalent CKD Prespecified kidney endpoints: • UACR Prespecified kidney endpoints: • None Prespecified kidney endpoints: • e. GFR* and UACR Post hoc analysis: • Doubling of serum creatinine • Initiation of chronic dialysis, kidney transplant or serum creatinine >6. 0 mg/dl • Composite endpoint of death and any of the above Post hoc analysis: • Initiation of dialysis • Change in e. GFR* from baseline Post hoc analysis: • CV and CKD outcomes according to baseline e. GFR stage Prespecified kidney endpoints: • Composite kidney endpoint† • Death due to kidney disease or ESKD • Microvascular composite outcome‡ • Albuminuria progression§ *Calculated using the Modification of Diet in Renal Disease equation; †Time to first occurrence of progression to sustained ESKD, sustained ≥ 40% decrease in e. GFR from baseline or death due to kidney disease; ‡Albuminuria, ESKD, sustained ≥ 50% e. GFR decrease from baseline, death due to kidney disease, retinal photocoagulation, anti. VEGF injection therapy for diabetic retinopathy, vitreous haemorrhage or diabetes-related blindness; §Change from normo- to micro- or macroalbuminuria, or from microto macroalbuminuria See slide notes for abbreviations 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. Mosenzon O et al. Diabetes Care 2017; 40: 69; 3. White WB et al. N Engl J Med 2013; 369: 1327 & supplementary appendix; 4. Green JB et al. N Engl J Med 2015; 373: 232; 5. Cornel JH et al. Diabetes Care 2016; 39: 2304; 6. Rosenstock J et al. JAMA 2019; 321: 69 12
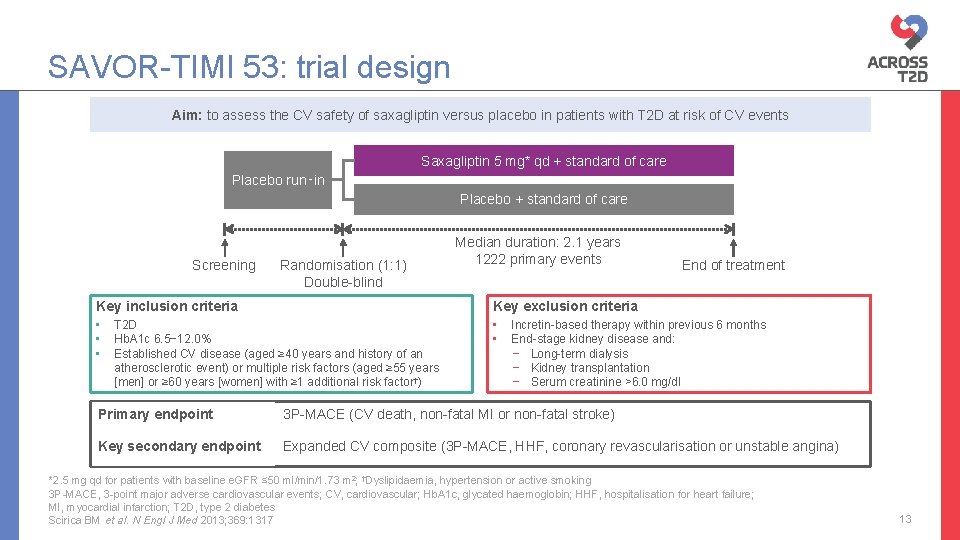
SAVOR-TIMI 53: trial design Aim: to assess the CV safety of saxagliptin versus placebo in patients with T 2 D at risk of CV events Saxagliptin 5 mg* qd + standard of care Placebo run‑in Placebo + standard of care Screening Randomisation (1: 1) Double-blind Median duration: 2. 1 years 1222 primary events Key inclusion criteria Key exclusion criteria • • • T 2 D Hb. A 1 c 6. 5− 12. 0% Established CV disease (aged ≥ 40 years and history of an atherosclerotic event) or multiple risk factors (aged ≥ 55 years [men] or ≥ 60 years [women] with ≥ 1 additional risk factor†) End of treatment Incretin-based therapy within previous 6 months End-stage kidney disease and: − Long-term dialysis − Kidney transplantation − Serum creatinine >6. 0 mg/dl Primary endpoint 3 P-MACE (CV death, non-fatal MI or non-fatal stroke) Key secondary endpoint Expanded CV composite (3 P-MACE, HHF, coronary revascularisation or unstable angina) *2. 5 mg qd for patients with baseline e. GFR ≤ 50 ml/min/1. 73 m 2; †Dyslipidaemia, hypertension or active smoking 3 P-MACE, 3 -point major adverse cardiovascular events; CV, cardiovascular; Hb. A 1 c, glycated haemoglobin; HHF, hospitalisation for heart failure; MI, myocardial infarction; T 2 D, type 2 diabetes Scirica BM et al. N Engl J Med 2013; 369: 1317 13
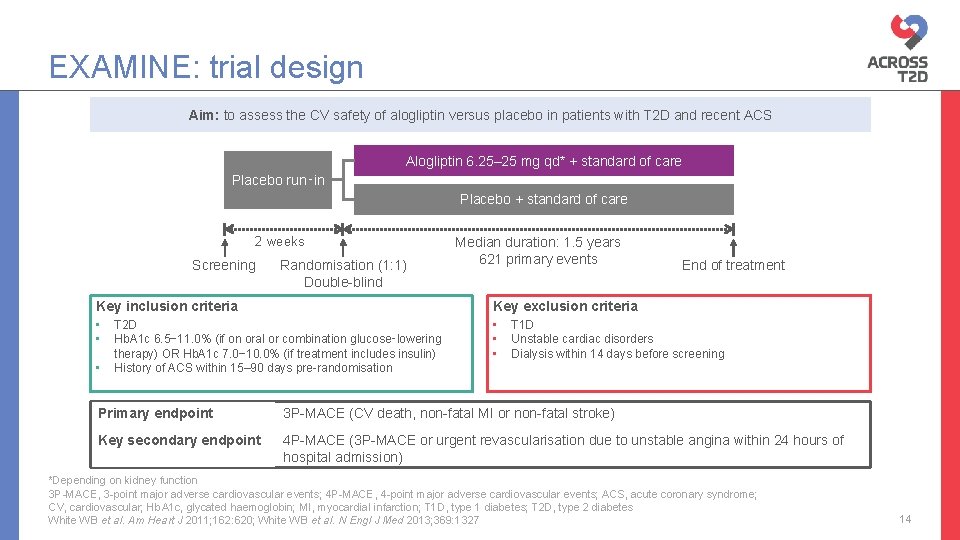
EXAMINE: trial design Aim: to assess the CV safety of alogliptin versus placebo in patients with T 2 D and recent ACS Alogliptin 6. 25– 25 mg qd* + standard of care Placebo run‑in Placebo + standard of care 2 weeks Screening Randomisation (1: 1) Double-blind Median duration: 1. 5 years 621 primary events Key inclusion criteria Key exclusion criteria • • • T 2 D Hb. A 1 c 6. 5− 11. 0% (if on oral or combination glucose‑lowering therapy) OR Hb. A 1 c 7. 0− 10. 0% (if treatment includes insulin) History of ACS within 15– 90 days pre-randomisation End of treatment T 1 D Unstable cardiac disorders Dialysis within 14 days before screening Primary endpoint 3 P-MACE (CV death, non-fatal MI or non-fatal stroke) Key secondary endpoint 4 P-MACE (3 P-MACE or urgent revascularisation due to unstable angina within 24 hours of hospital admission) *Depending on kidney function 3 P-MACE, 3 -point major adverse cardiovascular events; 4 P-MACE, 4 -point major adverse cardiovascular events; ACS, acute coronary syndrome; CV, cardiovascular; Hb. A 1 c, glycated haemoglobin; MI, myocardial infarction; T 1 D, type 1 diabetes; T 2 D, type 2 diabetes White WB et al. Am Heart J 2011; 162: 620; White WB et al. N Engl J Med 2013; 369: 1327 14
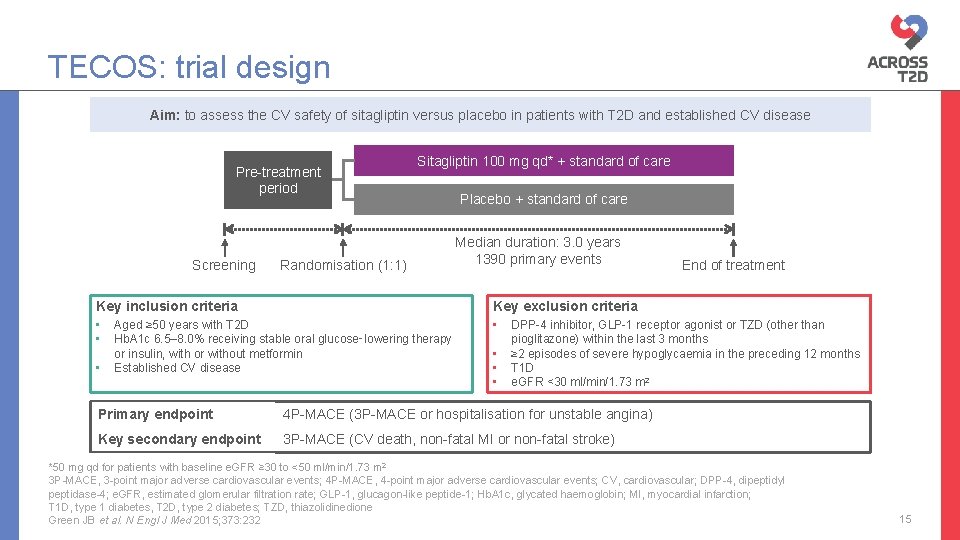
TECOS: trial design Aim: to assess the CV safety of sitagliptin versus placebo in patients with T 2 D and established CV disease Pre-treatment period Screening Sitagliptin 100 mg qd* + standard of care Randomisation (1: 1) Placebo + standard of care Median duration: 3. 0 years 1390 primary events Key inclusion criteria Key exclusion criteria • • Aged ≥ 50 years with T 2 D Hb. A 1 c 6. 5– 8. 0% receiving stable oral glucose‑lowering therapy or insulin, with or without metformin Established CV disease • • • End of treatment DPP-4 inhibitor, GLP-1 receptor agonist or TZD (other than pioglitazone) within the last 3 months ≥ 2 episodes of severe hypoglycaemia in the preceding 12 months T 1 D e. GFR <30 ml/min/1. 73 m 2 Primary endpoint 4 P-MACE (3 P-MACE or hospitalisation for unstable angina) Key secondary endpoint 3 P-MACE (CV death, non-fatal MI or non-fatal stroke) *50 mg qd for patients with baseline e. GFR ≥ 30 to <50 ml/min/1. 73 m 2 3 P-MACE, 3 -point major adverse cardiovascular events; 4 P-MACE, 4 -point major adverse cardiovascular events; CV, cardiovascular; DPP-4, dipeptidyl peptidase-4; e. GFR, estimated glomerular filtration rate; GLP-1, glucagon-like peptide-1; Hb. A 1 c, glycated haemoglobin; MI, myocardial infarction; T 1 D, type 1 diabetes, T 2 D, type 2 diabetes; TZD, thiazolidinedione Green JB et al. N Engl J Med 2015; 373: 232 15
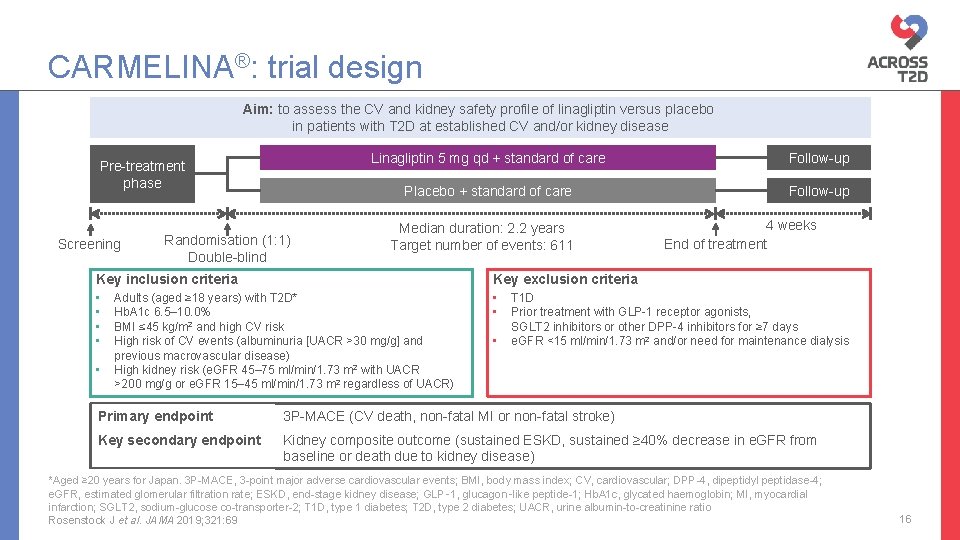
CARMELINA®: trial design Aim: to assess the CV and kidney safety profile of linagliptin versus placebo in patients with T 2 D at established CV and/or kidney disease Pre-treatment phase Screening Randomisation (1: 1) Double-blind Linagliptin 5 mg qd + standard of care Follow-up Placebo + standard of care Follow-up Median duration: 2. 2 years Target number of events: 611 Key inclusion criteria Key exclusion criteria • • Adults (aged ≥ 18 years) with T 2 D* Hb. A 1 c 6. 5– 10. 0% BMI ≤ 45 kg/m 2 and high CV risk High risk of CV events (albuminuria [UACR >30 mg/g] and previous macrovascular disease) High kidney risk (e. GFR 45– 75 ml/min/1. 73 m 2 with UACR >200 mg/g or e. GFR 15– 45 ml/min/1. 73 m 2 regardless of UACR) • 4 weeks End of treatment T 1 D Prior treatment with GLP-1 receptor agonists, SGLT 2 inhibitors or other DPP-4 inhibitors for ≥ 7 days e. GFR <15 ml/min/1. 73 m 2 and/or need for maintenance dialysis Primary endpoint 3 P-MACE (CV death, non-fatal MI or non-fatal stroke) Key secondary endpoint Kidney composite outcome (sustained ESKD, sustained ≥ 40% decrease in e. GFR from baseline or death due to kidney disease) *Aged ≥ 20 years for Japan. 3 P-MACE, 3 -point major adverse cardiovascular events; BMI, body mass index; CV, cardiovascular; DPP-4, dipeptidyl peptidase-4; e. GFR, estimated glomerular filtration rate; ESKD, end-stage kidney disease; GLP‑ 1, glucagon‑like peptide-1; Hb. A 1 c, glycated haemoglobin; MI, myocardial infarction; SGLT 2, sodium-glucose co-transporter-2; T 1 D, type 1 diabetes; T 2 D, type 2 diabetes; UACR, urine albumin-to-creatinine ratio Rosenstock J et al. JAMA 2019; 321: 69 16
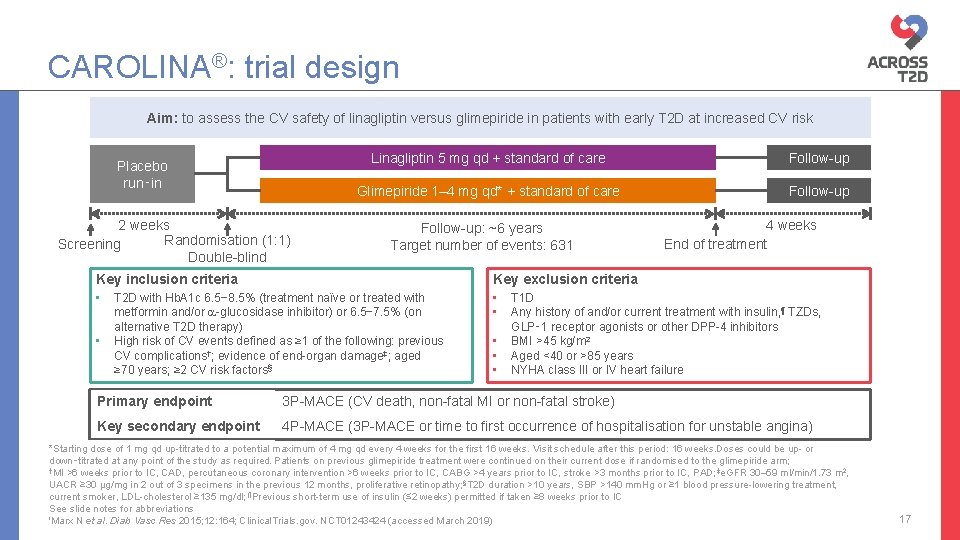
CAROLINA®: trial design Aim: to assess the CV safety of linagliptin versus glimepiride in patients with early T 2 D at increased CV risk Placebo run‑in 2 weeks Randomisation (1: 1) Screening Double-blind Linagliptin 5 mg qd + standard of care Follow-up Glimepiride 1– 4 mg qd* + standard of care Follow-up: ~6 years Target number of events: 631 Key inclusion criteria Key exclusion criteria • • T 2 D with Hb. A 1 c 6. 5− 8. 5% (treatment naïve or treated with metformin and/or -glucosidase inhibitor) or 6. 5− 7. 5% (on alternative T 2 D therapy) High risk of CV events defined as ≥ 1 of the following: previous CV complications†; evidence of end-organ damage‡; aged ≥ 70 years; ≥ 2 CV risk factors§ • • • 4 weeks End of treatment T 1 D Any history of and/or current treatment with insulin, ¶ TZDs, GLP‑ 1 receptor agonists or other DPP-4 inhibitors BMI >45 kg/m 2 Aged <40 or >85 years NYHA class III or IV heart failure Primary endpoint 3 P-MACE (CV death, non-fatal MI or non-fatal stroke) Key secondary endpoint 4 P-MACE (3 P-MACE or time to first occurrence of hospitalisation for unstable angina) *Starting dose of 1 mg qd up-titrated to a potential maximum of 4 mg qd every 4 weeks for the first 16 weeks. Visit schedule after this period: 16 weeks. Doses could be up- or down‑titrated at any point of the study as required. Patients on previous glimepiride treatment were continued on their current dose if randomised to the glimepiride arm; †MI >6 weeks prior to IC, CAD, percutaneous coronary intervention >6 weeks prior to IC, CABG >4 years prior to IC, stroke >3 months prior to IC, PAD; ‡e. GFR 30– 59 ml/min/1. 73 m 2, UACR ≥ 30 µg/mg in 2 out of 3 specimens in the previous 12 months, proliferative retinopathy; §T 2 D duration >10 years, SBP >140 mm. Hg or ≥ 1 blood pressure-lowering treatment, current smoker, LDL-cholesterol ≥ 135 mg/dl; ¶Previous short-term use of insulin (≤ 2 weeks) permitted if taken ≥ 8 weeks prior to IC See slide notes for abbreviations 'Marx N et al. Diab Vasc Res 2015; 12: 164; Clinical. Trials. gov. NCT 01243424 (accessed March 2019) 17
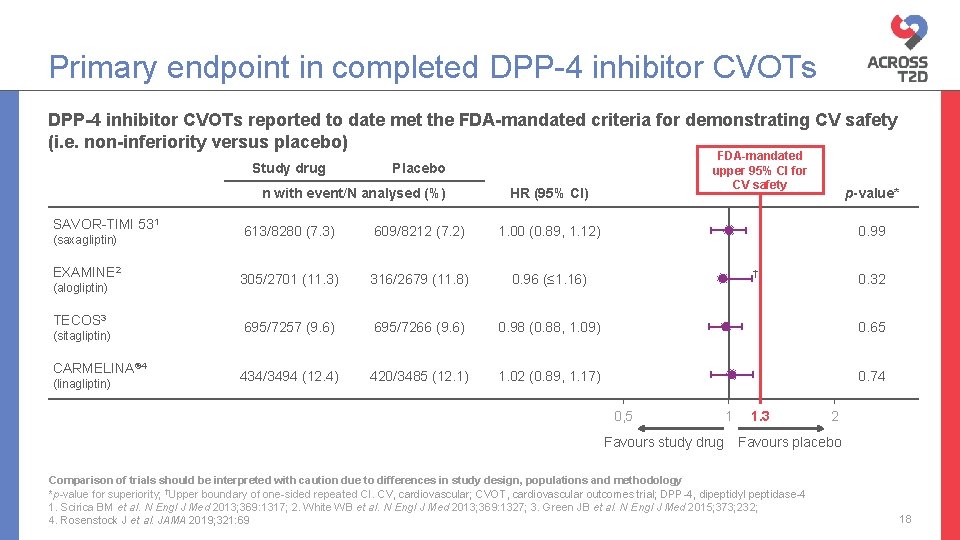
Primary endpoint in completed DPP-4 inhibitor CVOTs reported to date met the FDA-mandated criteria for demonstrating CV safety (i. e. non-inferiority versus placebo) Study drug n with event/N analysed (%) SAVOR-TIMI 531 (saxagliptin) EXAMINE 2 (alogliptin) TECOS 3 (sitagliptin) CARMELINA® 4 (linagliptin) FDA-mandated upper 95% CI for CV safety Placebo HR (95% CI) p-value* 613/8280 (7. 3) 609/8212 (7. 2) 1. 00 (0. 89, 1. 12) 0. 99 305/2701 (11. 3) 316/2679 (11. 8) 0. 96 (≤ 1. 16) 695/7257 (9. 6) 695/7266 (9. 6) 0. 98 (0. 88, 1. 09) 0. 65 434/3494 (12. 4) 420/3485 (12. 1) 1. 02 (0. 89, 1. 17) 0. 74 † 0, 5 Favours study drug 1 1. 3 0. 32 2 Favours placebo Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology *p-value for superiority; †Upper boundary of one-sided repeated CI. CV, cardiovascular; CVOT, cardiovascular outcomes trial; DPP-4, dipeptidyl peptidase-4 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327; 3. Green JB et al. N Engl J Med 2015; 373; 232; 4. Rosenstock J et al. JAMA 2019; 321: 69 18
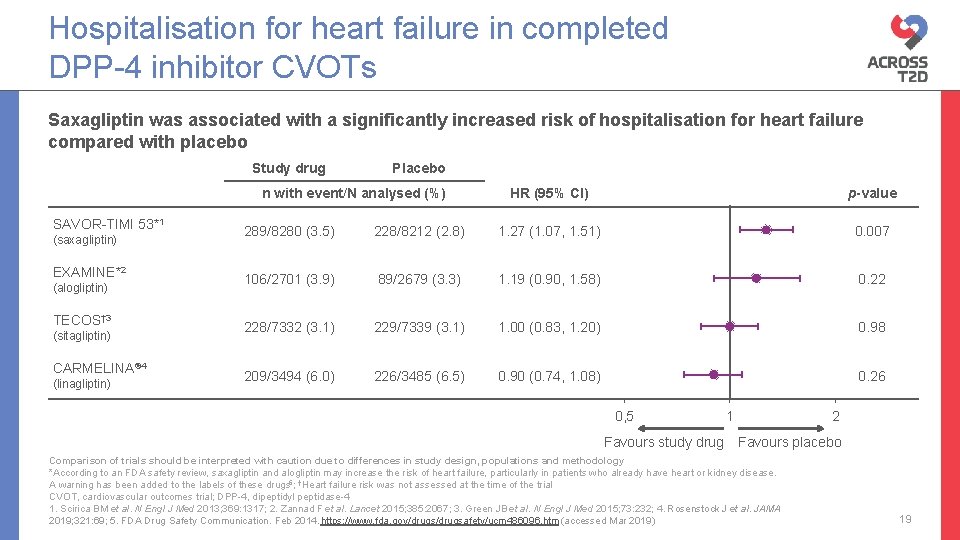
Hospitalisation for heart failure in completed DPP-4 inhibitor CVOTs Saxagliptin was associated with a significantly increased risk of hospitalisation for heart failure compared with placebo Study drug Placebo n with event/N analysed (%) SAVOR-TIMI 53*1 (saxagliptin) EXAMINE*2 (alogliptin) TECOS† 3 (sitagliptin) CARMELINA® 4 (linagliptin) HR (95% CI) p-value 289/8280 (3. 5) 228/8212 (2. 8) 1. 27 (1. 07, 1. 51) 0. 007 106/2701 (3. 9) 89/2679 (3. 3) 1. 19 (0. 90, 1. 58) 0. 22 228/7332 (3. 1) 229/7339 (3. 1) 1. 00 (0. 83, 1. 20) 0. 98 209/3494 (6. 0) 226/3485 (6. 5) 0. 90 (0. 74, 1. 08) 0. 26 0, 5 Favours study drug 1 2 Favours placebo Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology *According to an FDA safety review, saxagliptin and alogliptin may increase the risk of heart failure, particularly in patients who already have heart or kidney disease. A warning has been added to the labels of these drugs 5; †Heart failure risk was not assessed at the time of the trial CVOT, cardiovascular outcomes trial; DPP-4, dipeptidyl peptidase-4 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. Zannad F et al. Lancet 2015; 385: 2067; 3. Green JB et al. N Engl J Med 2015; 73: 232; 4. Rosenstock J et al. JAMA 2019; 321: 69; 5. FDA Drug Safety Communication. Feb 2014. https: //www. fda. gov/drugsafety/ucm 486096. htm (accessed Mar 2019) 19
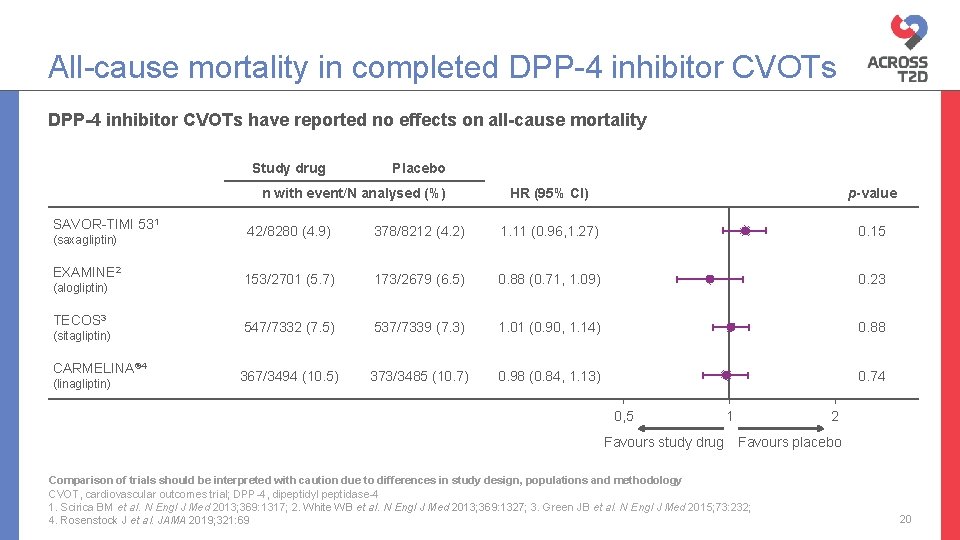
All-cause mortality in completed DPP-4 inhibitor CVOTs have reported no effects on all-cause mortality Study drug Placebo n with event/N analysed (%) SAVOR-TIMI 531 (saxagliptin) EXAMINE 2 (alogliptin) TECOS 3 (sitagliptin) CARMELINA® 4 (linagliptin) HR (95% CI) p-value 42/8280 (4. 9) 378/8212 (4. 2) 1. 11 (0. 96, 1. 27) 0. 15 153/2701 (5. 7) 173/2679 (6. 5) 0. 88 (0. 71, 1. 09) 0. 23 547/7332 (7. 5) 537/7339 (7. 3) 1. 01 (0. 90, 1. 14) 0. 88 367/3494 (10. 5) 373/3485 (10. 7) 0. 98 (0. 84, 1. 13) 0. 74 0, 5 Favours study drug 1 2 Favours placebo Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology CVOT, cardiovascular outcomes trial; DPP-4, dipeptidyl peptidase-4 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327; 3. Green JB et al. N Engl J Med 2015; 73: 232; 4. Rosenstock J et al. JAMA 2019; 321: 69 20
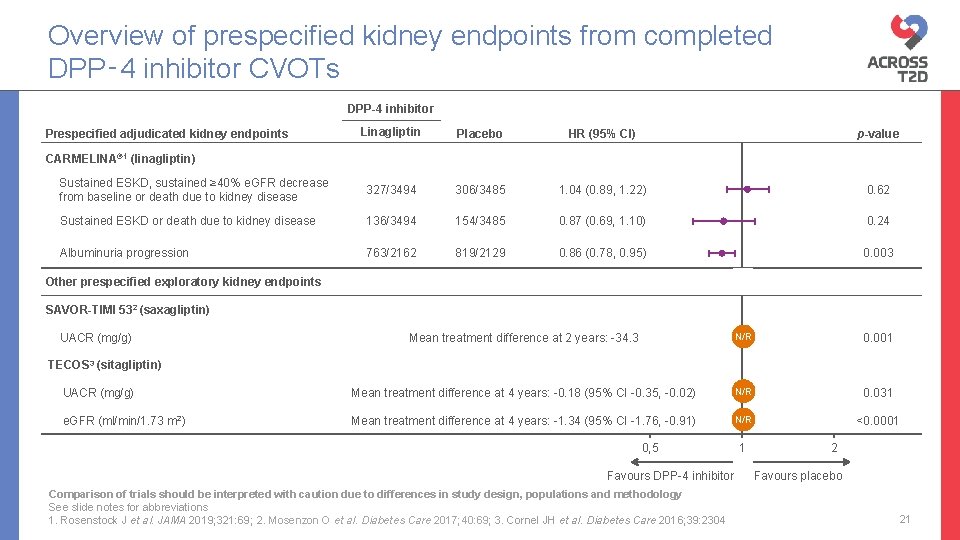
DPP-4 inhibitor Linagliptin Placebo HR (95% CI) p-value Sustained ESKD, sustained ≥ 40% e. GFR decrease from baseline or death due to kidney disease 327/3494 306/3485 1. 04 (0. 89, 1. 22) 0. 62 Sustained ESKD or death due to kidney disease 136/3494 154/3485 0. 87 (0. 69, 1. 10) 0. 24 Albuminuria progression 763/2162 819/2129 0. 86 (0. 78, 0. 95) 0. 003 Prespecified adjudicated kidney endpoints CARMELINA® 1 (linagliptin) Other prespecified exploratory kidney endpoints SAVOR-TIMI 532 (saxagliptin) UACR (mg/g) Mean treatment difference at 2 years: -34. 3 0. 001 UACR (mg/g) Mean treatment difference at 4 years: -0. 18 (95% CI -0. 35, -0. 02) 0. 031 e. GFR (ml/min/1. 73 m 2) Mean treatment difference at 4 years: -1. 34 (95% CI -1. 76, -0. 91) <0. 0001 TECOS 3 (sitagliptin)
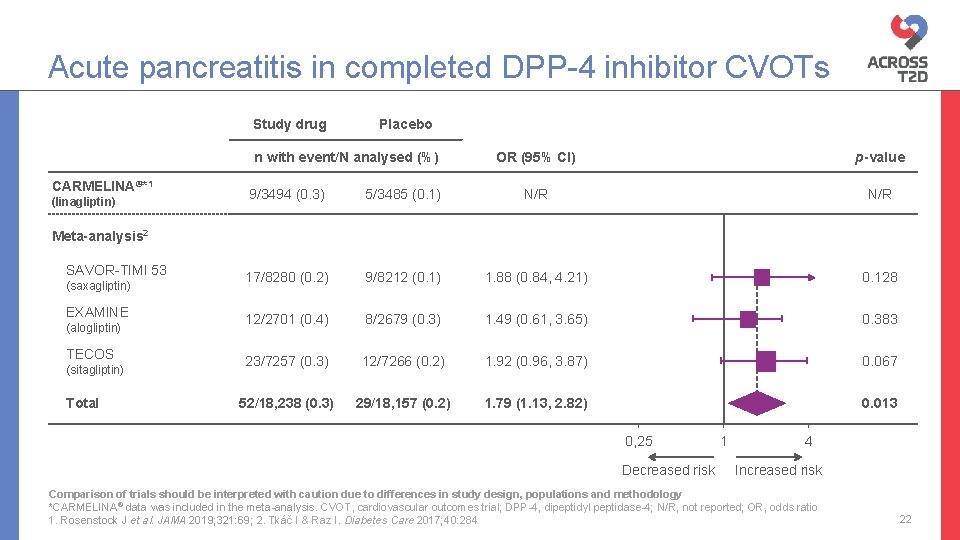
Acute pancreatitis in completed DPP-4 inhibitor CVOTs Study drug Placebo n with event/N analysed (%) CARMELINA®*1 (linagliptin) OR (95% CI) p-value 9/3494 (0. 3) 5/3485 (0. 1) N/R 17/8280 (0. 2) 9/8212 (0. 1) 1. 88 (0. 84, 4. 21) 0. 128 12/2701 (0. 4) 8/2679 (0. 3) 1. 49 (0. 61, 3. 65) 0. 383 23/7257 (0. 3) 12/7266 (0. 2) 1. 92 (0. 96, 3. 87) 0. 067 52/18, 238 (0. 3) 29/18, 157 (0. 2) 1. 79 (1. 13, 2. 82) 0. 013 Meta-analysis 2 SAVOR-TIMI 53 (saxagliptin) EXAMINE (alogliptin) TECOS (sitagliptin) Total 0, 25 Decreased risk 1 4 Increased risk Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology *CARMELINA® data was included in the meta-analysis. CVOT, cardiovascular outcomes trial; DPP-4, dipeptidyl peptidase-4; N/R, not reported; OR, odds ratio 1. Rosenstock J et al. JAMA 2019; 321: 69; 2. Tkáč I & Raz I. Diabetes Care 2017; 40: 284 22
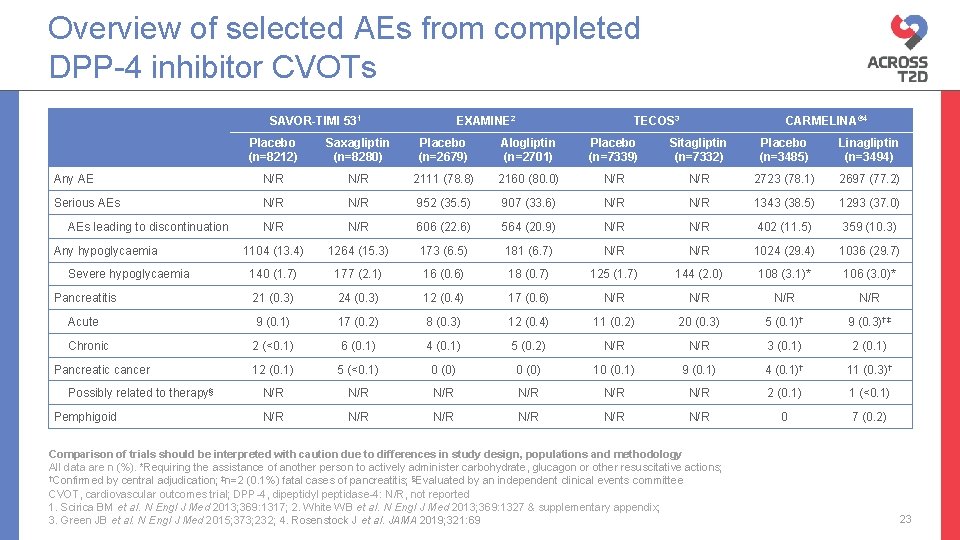
Overview of selected AEs from completed DPP-4 inhibitor CVOTs SAVOR-TIMI 531 EXAMINE 2 TECOS 3 CARMELINA® 4 Placebo (n=8212) Saxagliptin (n=8280) Placebo (n=2679) Alogliptin (n=2701) Placebo (n=7339) Sitagliptin (n=7332) Placebo (n=3485) Linagliptin (n=3494) Any AE N/R 2111 (78. 8) 2160 (80. 0) N/R 2723 (78. 1) 2697 (77. 2) Serious AEs N/R 952 (35. 5) 907 (33. 6) N/R 1343 (38. 5) 1293 (37. 0) N/R 606 (22. 6) 564 (20. 9) N/R 402 (11. 5) 359 (10. 3) 1104 (13. 4) 1264 (15. 3) 173 (6. 5) 181 (6. 7) N/R 1024 (29. 4) 1036 (29. 7) 140 (1. 7) 177 (2. 1) 16 (0. 6) 18 (0. 7) 125 (1. 7) 144 (2. 0) 108 (3. 1)* 106 (3. 0)* Pancreatitis 21 (0. 3) 24 (0. 3) 12 (0. 4) 17 (0. 6) N/R N/R Acute 9 (0. 1) 17 (0. 2) 8 (0. 3) 12 (0. 4) 11 (0. 2) 20 (0. 3) 5 (0. 1)† 9 (0. 3)†‡ Chronic 2 (<0. 1) 6 (0. 1) 4 (0. 1) 5 (0. 2) N/R 3 (0. 1) 2 (0. 1) 12 (0. 1) 5 (<0. 1) 0 (0) 10 (0. 1) 9 (0. 1) 4 (0. 1)† 11 (0. 3)† N/R N/R N/R 2 (0. 1) 1 (<0. 1) N/R N/R N/R 0 7 (0. 2) AEs leading to discontinuation Any hypoglycaemia Severe hypoglycaemia Pancreatic cancer Possibly related to therapy§ Pemphigoid Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology All data are n (%). *Requiring the assistance of another person to actively administer carbohydrate, glucagon or other resuscitative actions; †Confirmed by central adjudication; ‡n=2 (0. 1%) fatal cases of pancreatitis; §Evaluated by an independent clinical events committee CVOT, cardiovascular outcomes trial; DPP-4, dipeptidyl peptidase-4: N/R, not reported 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327 & supplementary appendix; 3. Green JB et al. N Engl J Med 2015; 373; 232; 4. Rosenstock J et al. JAMA 2019; 321: 69 23

DPP-4 inhibitor CVOTs: summary Primary outcome All DPP-4 inhibitor CVOTs reported to date were non-inferior to placebo for the primary endpoint (3 P-MACE or 4 P-MACE)1– 4 Saxagliptin was associated with an increased risk of HHF 1 Microvascular outcomes Other CV endpoints CARMELINA® is the only DPP-4 inhibitor CVOT with prespecified hard kidney outcomes; no increased risk in the composite kidney endpoints was demonstrated 1– 7 CVOTs demonstrated no significant difference in the incidence of severe hypoglycaemia between DPP-4 inhibitors and placebo 1– 4 One further DPP-4 inhibitor CVOT with an active comparator will add to existing clinical data on the CV safety of DPP-4 inhibitors 8, 9 Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology 3 P-MACE, 3 -point major adverse cardiovascular events; 4 P-MACE, 4 -point major adverse cardiovascular events; CV, cardiovascular; CVOT, cardiovascular outcomes trial; DPP-4, dipeptidyl peptidase-4; HHF, hospitalisation for heart failure 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327; 3. Green JB et al. N Engl J Med 2015; 373: 232; 4. Rosenstock J et al. JAMA 2019; 321: 69; 5. Zannad F et al. Lancet 2015; 385: 2067; 6. Mosenzon O et al. Diabetes Care 2017; 40: 69; 7. Cornel JH et al. Diabetes Care 2016; 39: 2304; 8. Marx N et al. Diab Vasc Dis Res 2015; 12: 164; 9. Clinical. Trials. gov. NCT 01243424 (accessed March 2019) Safety 24

Back-up slides
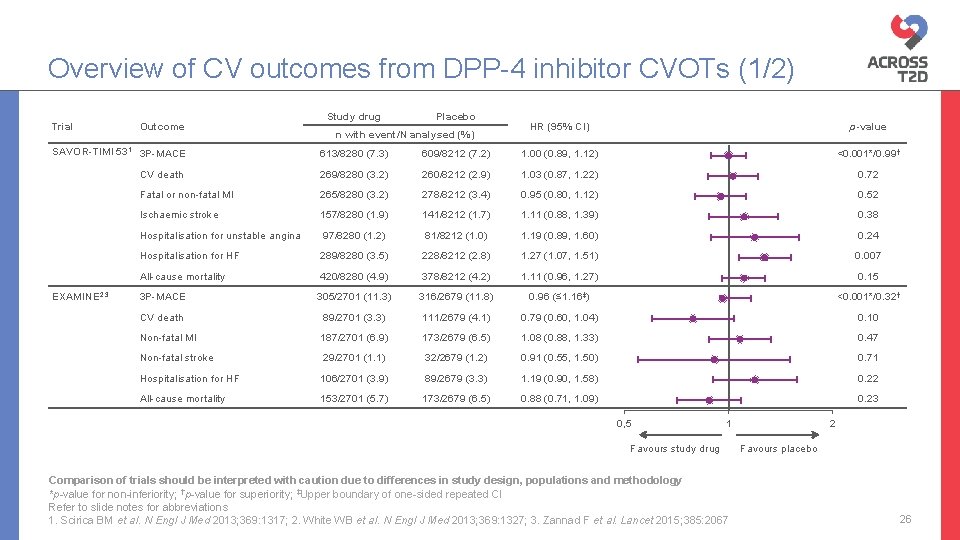
Overview of CV outcomes from DPP-4 inhibitor CVOTs (1/2) Trial Outcome Study drug Placebo n with event/N analysed (%) HR (95% CI) p-value SAVOR-TIMI 531 3 P-MACE 613/8280 (7. 3) 609/8212 (7. 2) 1. 00 (0. 89, 1. 12) <0. 001*/0. 99† CV death 269/8280 (3. 2) 260/8212 (2. 9) 1. 03 (0. 87, 1. 22) 0. 72 Fatal or non-fatal MI 265/8280 (3. 2) 278/8212 (3. 4) 0. 95 (0. 80, 1. 12) 0. 52 Ischaemic stroke 157/8280 (1. 9) 141/8212 (1. 7) 1. 11 (0. 88, 1. 39) 0. 38 Hospitalisation for unstable angina 97/8280 (1. 2) 81/8212 (1. 0) 1. 19 (0. 89, 1. 60) 0. 24 Hospitalisation for HF 289/8280 (3. 5) 228/8212 (2. 8) 1. 27 (1. 07, 1. 51) 0. 007 All-cause mortality 420/8280 (4. 9) 378/8212 (4. 2) 1. 11 (0. 96, 1. 27) 0. 15 3 P-MACE 305/2701 (11. 3) 316/2679 (11. 8) 0. 96 (≤ 1. 16‡) <0. 001*/0. 32† CV death 89/2701 (3. 3) 111/2679 (4. 1) 0. 79 (0. 60, 1. 04) 0. 10 Non-fatal MI 187/2701 (6. 9) 173/2679 (6. 5) 1. 08 (0. 88, 1. 33) 0. 47 Non-fatal stroke 29/2701 (1. 1) 32/2679 (1. 2) 0. 91 (0. 55, 1. 50) 0. 71 Hospitalisation for HF 106/2701 (3. 9) 89/2679 (3. 3) 1. 19 (0. 90, 1. 58) 0. 22 All-cause mortality 153/2701 (5. 7) 173/2679 (6. 5) 0. 88 (0. 71, 1. 09) 0. 23 EXAMINE 2, 3 0, 5 1 Favours study drug Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology *p-value for non-inferiority; †p-value for superiority; ‡Upper boundary of one-sided repeated CI Refer to slide notes for abbreviations 1. Scirica BM et al. N Engl J Med 2013; 369: 1317; 2. White WB et al. N Engl J Med 2013; 369: 1327; 3. Zannad F et al. Lancet 2015; 385: 2067 2 Favours placebo 26
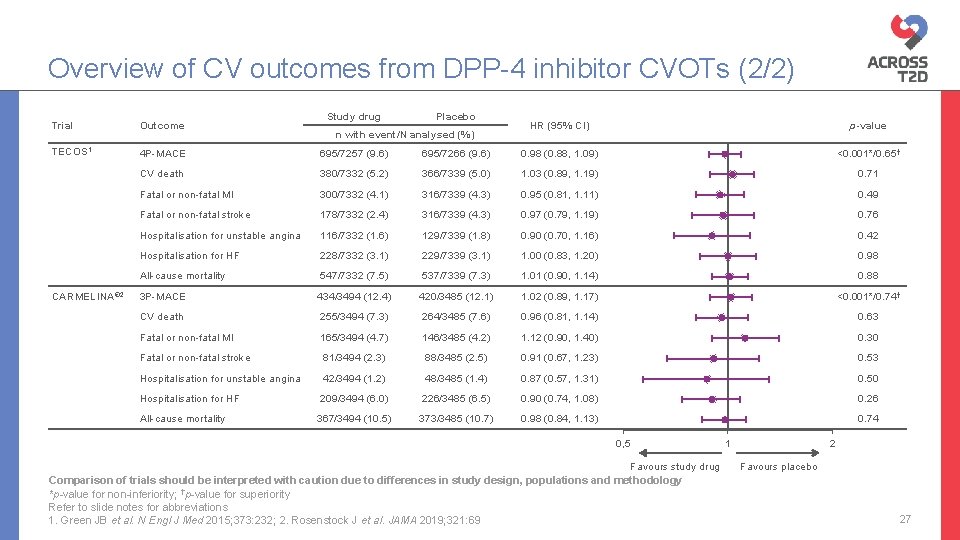
Overview of CV outcomes from DPP-4 inhibitor CVOTs (2/2) Study drug Trial Outcome TECOS 1 4 P-MACE 695/7257 (9. 6) CV death CARMELINA® 2 Placebo HR (95% CI) p-value 695/7266 (9. 6) 0. 98 (0. 88, 1. 09) <0. 001*/0. 65† 380/7332 (5. 2) 366/7339 (5. 0) 1. 03 (0. 89, 1. 19) 0. 71 Fatal or non-fatal MI 300/7332 (4. 1) 316/7339 (4. 3) 0. 95 (0. 81, 1. 11) 0. 49 Fatal or non-fatal stroke 178/7332 (2. 4) 316/7339 (4. 3) 0. 97 (0. 79, 1. 19) 0. 76 Hospitalisation for unstable angina 116/7332 (1. 6) 129/7339 (1. 8) 0. 90 (0. 70, 1. 16) 0. 42 Hospitalisation for HF 228/7332 (3. 1) 229/7339 (3. 1) 1. 00 (0. 83, 1. 20) 0. 98 All-cause mortality 547/7332 (7. 5) 537/7339 (7. 3) 1. 01 (0. 90, 1. 14) 0. 88 3 P-MACE 434/3494 (12. 4) 420/3485 (12. 1) 1. 02 (0. 89, 1. 17) <0. 001*/0. 74† CV death 255/3494 (7. 3) 264/3485 (7. 6) 0. 96 (0. 81, 1. 14) 0. 63 Fatal or non-fatal MI 165/3494 (4. 7) 146/3485 (4. 2) 1. 12 (0. 90, 1. 40) 0. 30 Fatal or non-fatal stroke 81/3494 (2. 3) 88/3485 (2. 5) 0. 91 (0. 67, 1. 23) 0. 53 Hospitalisation for unstable angina 42/3494 (1. 2) 48/3485 (1. 4) 0. 87 (0. 57, 1. 31) 0. 50 Hospitalisation for HF 209/3494 (6. 0) 226/3485 (6. 5) 0. 90 (0. 74, 1. 08) 0. 26 All-cause mortality 367/3494 (10. 5) 373/3485 (10. 7) 0. 98 (0. 84, 1. 13) 0. 74 n with event/N analysed (%) 0, 5 Favours study drug Comparison of trials should be interpreted with caution due to differences in study design, populations and methodology *p-value for non-inferiority; †p-value for superiority Refer to slide notes for abbreviations 1. Green JB et al. N Engl J Med 2015; 373: 232; 2. Rosenstock J et al. JAMA 2019; 321: 69 1 2 Favours placebo 27
- Slides: 27