Cardiovascular Issues in Scleroderma James R Seibold M

Cardiovascular Issues in Scleroderma James R. Seibold, M. D. Professor and Chief. Division of Rheumatology University of Connecticut Health System Farmington, Connecticut, USA

Disclosures Funded Research/Consultancy Ø Ø Ø Ø Actelion (bosentan, macitentan, spouse) Bristol-Myers Squibb (tyrosine kinase inhibitor) Pfizer (PDGF 2α, sitaxsentan, VEGF) Gilead (ambrisentan) Roche (tyrosine kinase inhibitor) Centocor (IL-13, TH 17 biology) Fibro. Gen (anti. CTGF) United Therapeutics (oral treprostinil, tadalafil)

The Heart and Lungs in Scleroderma • OVERALL SURVIVAL IS IMPROVING • LUNG INVOLVEMENT IS NOW THE LEADING ISSUE • PULMONARY HYPERTENSION versus • INTERSTITIAL LUNG DISEASE
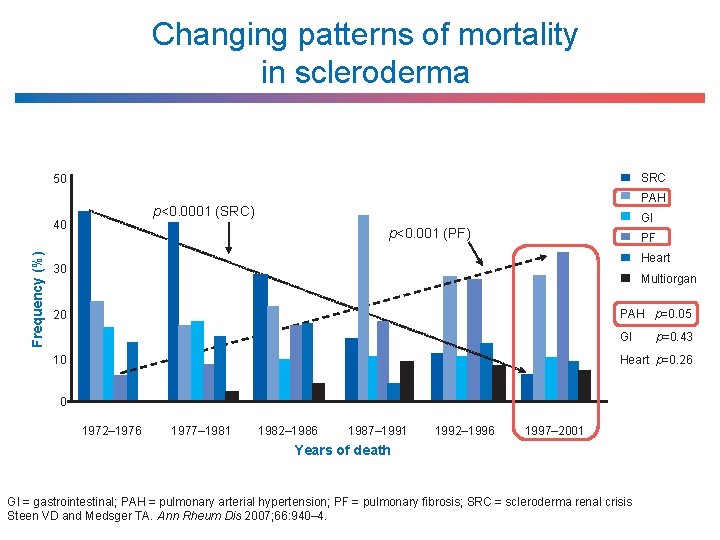
Changing patterns of mortality in scleroderma SRC 50 p<0. 0001 (SRC) 40 Frequency (%) PAH GI p<0. 001 (PF) PF Heart 30 Multiorgan PAH p=0. 05 20 GI 10 p=0. 43 Heart p=0. 26 0 1972– 1976 1977– 1981 1982– 1986 1987– 1991 1992– 1996 1997– 2001 Years of death GI = gastrointestinal; PAH = pulmonary arterial hypertension; PF = pulmonary fibrosis; SRC = scleroderma renal crisis Steen VD and Medsger TA. Ann Rheum Dis 2007; 66: 940– 4.
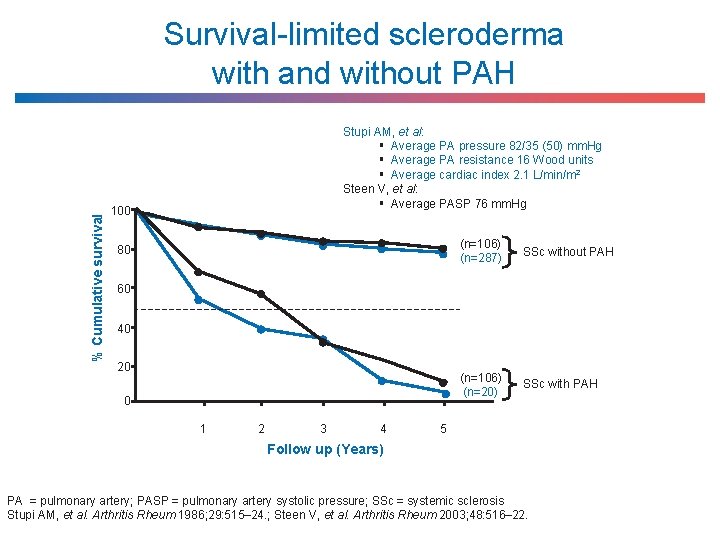
% Cumulative survival Survival-limited scleroderma with and without PAH Stupi AM, et al: § Average PA pressure 82/35 (50) mm. Hg § Average PA resistance 16 Wood units § Average cardiac index 2. 1 L/min/m 2 Steen V, et al: § Average PASP 76 mm. Hg 100 80 (n=106) (n=287) SSc without PAH (n=106) (n=20) SSc with PAH 60 40 20 0 1 2 3 4 5 Follow up (Years) PA = pulmonary artery; PASP = pulmonary artery systolic pressure; SSc = systemic sclerosis Stupi AM, et al. Arthritis Rheum 1986; 29: 515– 24. ; Steen V, et al. Arthritis Rheum 2003; 48: 516– 22.

Some Rash Statements!! • SSc-PAH is overdiagnosed in the community • SSc-PAH is undertreated in centers • Evidence-based medicine is lagging behind evolving standards of practice

Prevalence of PAH-CTD Disease Echo prevalence Catheter/clinical prevalence Systemic sclerosis 20– 50%1, 2 7. 85– 12%3, 4 Lupus 4– 43%5, 6 0. 9%3 20%7 <0. 01%8 Rheumatoid arthritis CTD = connective tissue disease; PAH = pulmonary arterial hypertension 1. Rheumatismo 2005; 57: 114. 2. Battle RW, et al. Chest 1996; 110: 1515‒ 9. 3. Eur J Respir Dis 2004; 59. 4. Mukerjee D, et al. Ann Rheum Dis 2003; 62: 1088‒ 93. 5. Pan TL, et al. Lupus 2000; 9: 338‒ 42. 6. Winslow TM, et al. Am Heart J 1995; 129: 510‒ 5. 7. Dawson JK, et al. Rheumatology 2000; 3: 1320‒ 5. 8. Post Marketing Surveillance Study (Data on File).

Differential diagnosis of “PH” CTD Pulmonary arterial hypertension (PAH) Pulmonary veno-occlusive disorder (PVOD) Myocardial involvement PH = pulmonary hypertension Interstitial lung disease (ILD) Chronic thromboembolic pulmonary hypertension (CTEPH) Others
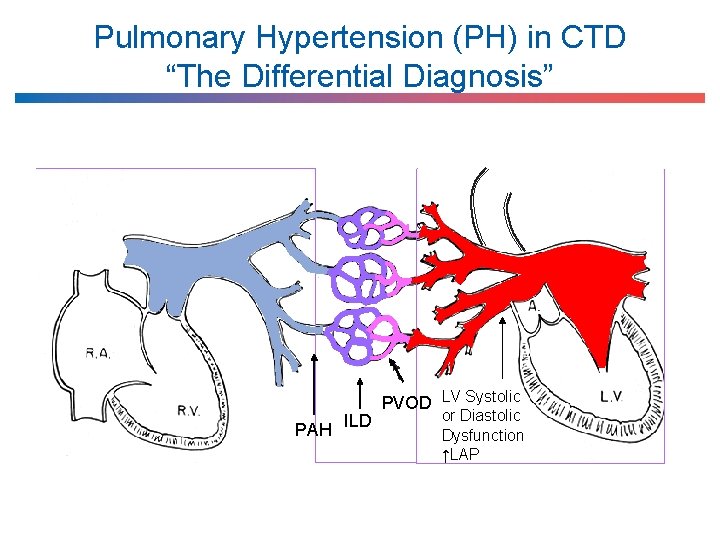
Pulmonary Hypertension (PH) in CTD “The Differential Diagnosis” PAH ILD PVOD LV Systolic or Diastolic Dysfunction ↑LAP
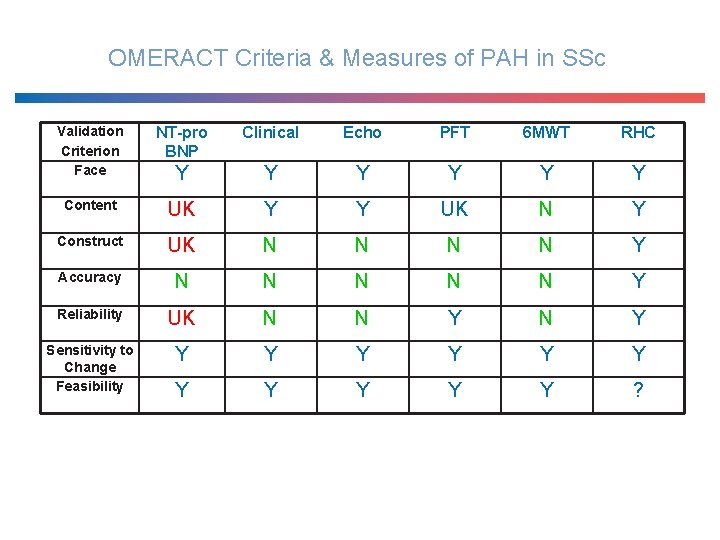
OMERACT Criteria & Measures of PAH in SSc Validation Criterion Face NT-pro BNP Clinical Echo PFT 6 MWT RHC Y Y Y Content UK Y Y UK N Y Construct UK N N Y Accuracy N N N Y Reliability UK N N Y Sensitivity to Change Feasibility Y Y Y ?

Causes of Shortness of Breath in Scleroderma Ø Interstitial Lung Disease Ø Pulmonary Arterial Hypertension Ø Left Heart Disease Ø Anemia Ø Physical deconditioning - Reduced V 02 Max - Reduced Metabolic Equivalent at V 02 Max What’s the most physical activity that you do and what symptom limits your capabilities?
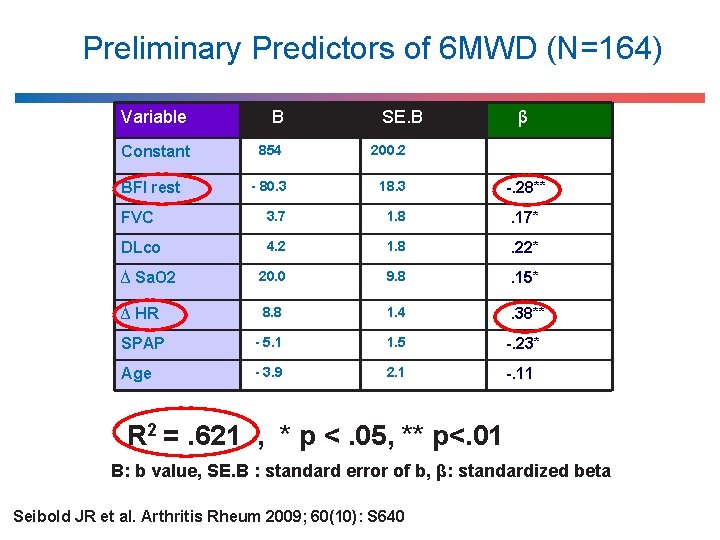
Preliminary Predictors of 6 MWD (N=164) Variable B Constant 854 200. 2 - 80. 3 18. 3 -. 28** FVC 3. 7 1. 8 . 17* DLco 4. 2 1. 8 . 22* ∆ Sa. O 2 20. 0 9. 8 . 15* ∆ HR 8. 8 1. 4 . 38** SPAP - 5. 1 1. 5 -. 23* Age - 3. 9 2. 1 -. 11 BFI rest SE. B β R 2 =. 621 , * p <. 05, ** p<. 01 B: b value, SE. B : standard error of b, β: standardized beta Seibold JR et al. Arthritis Rheum 2009; 60(10): S 640

Ratio of % FVC to % DLCO Influences Survival in SSc 1. 0 % FVC/% DLCO <1. 8 (n=337) 0. 9 Probability of survival 0. 8 0. 7 p=0. 007 0. 6 % FVC/% DLCO 1. 8 (n=169) 0. 5 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 Duration of disease (yr from onset) Disproportionate and/or isolated reduction in gas exchange (diffusing capacity) is dominant determinant of survival in all forms of SSc lung. Seibold JR et al. J Rheumatol (in press)
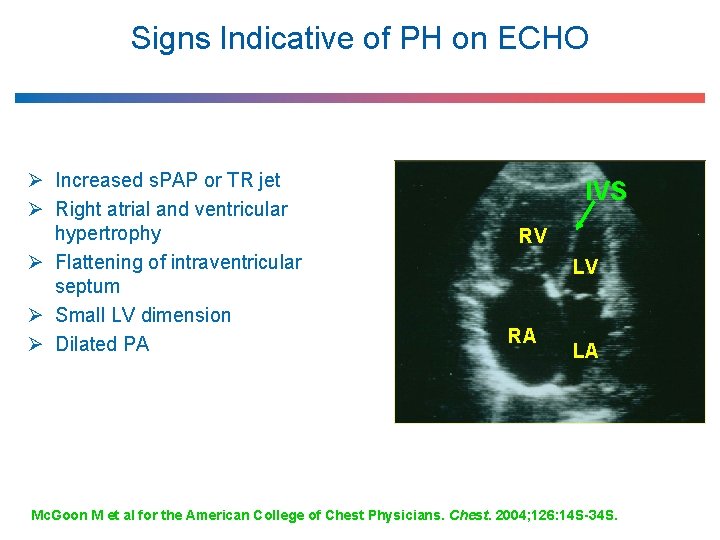
Signs Indicative of PH on ECHO Ø Increased s. PAP or TR jet Ø Right atrial and ventricular hypertrophy Ø Flattening of intraventricular septum Ø Small LV dimension Ø Dilated PA IVS RV LV RA LA Mc. Goon M et al for the American College of Chest Physicians. Chest. 2004; 126: 14 S-34 S.
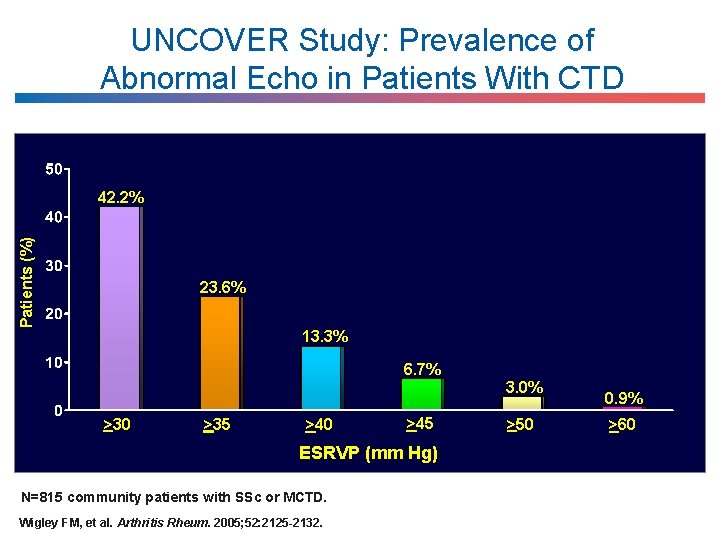
UNCOVER Study: Prevalence of Abnormal Echo in Patients With CTD Patients (%) 42. 2% 23. 6% 13. 3% 6. 7% 3. 0% >30 >35 >40 >45 ESRVP (mm Hg) N=815 community patients with SSc or MCTD. Wigley FM, et al. Arthritis Rheum. 2005; 52: 2125 -2132. >50 0. 9% >60
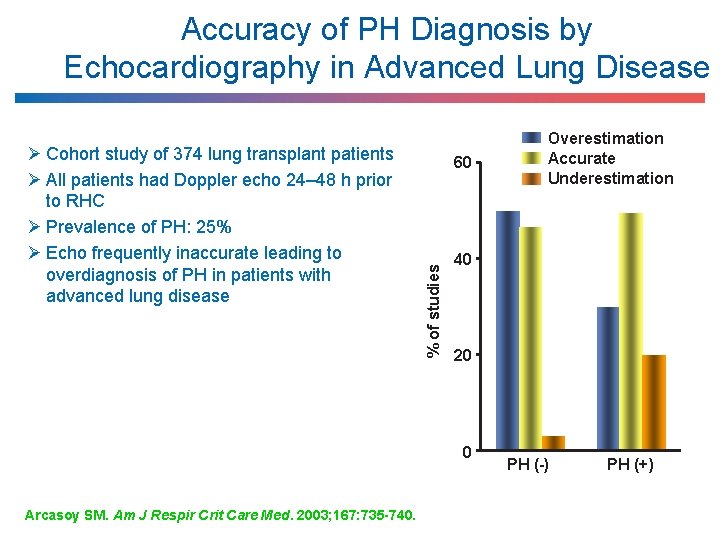
Accuracy of PH Diagnosis by Echocardiography in Advanced Lung Disease 60 % of studies Ø Cohort study of 374 lung transplant patients Ø All patients had Doppler echo 24– 48 h prior to RHC Ø Prevalence of PH: 25% Ø Echo frequently inaccurate leading to overdiagnosis of PH in patients with advanced lung disease 40 20 0 Arcasoy SM. Am J Respir Crit Care Med. 2003; 167: 735 -740. Overestimation Accurate Underestimation PH (-) PH (+)

Should we do annual echocardiography? • • • Estimated Event Rate 0. 6 per 100 patient years 167 baseline echocardiograms 167 follow up echocardiograms University of Michigan charges $2200 per test $734, 800 per case • This assumes 100% sensitivity/specificity
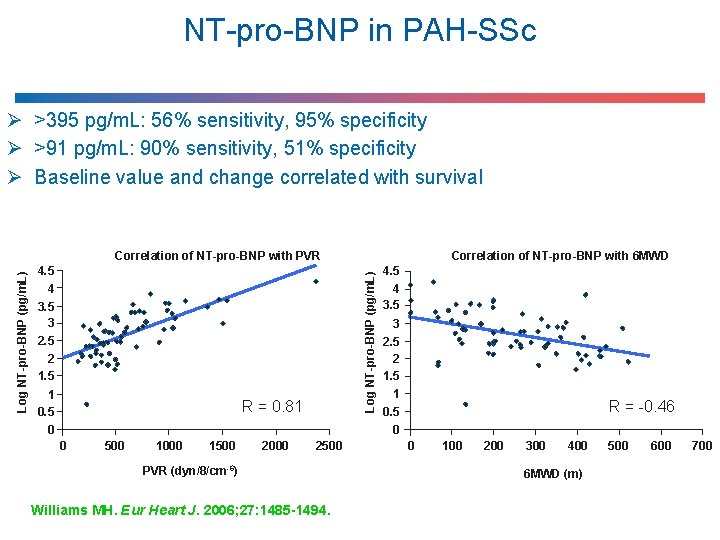
NT-pro-BNP in PAH-SSc Ø >395 pg/m. L: 56% sensitivity, 95% specificity Ø >91 pg/m. L: 90% sensitivity, 51% specificity Ø Baseline value and change correlated with survival Correlation of NT-pro-BNP with 6 MWD 4. 5 4 3. 5 3 2. 5 2 1. 5 1 0. 5 0 Log NT-pro-BNP (pg/m. L) Correlation of NT-pro-BNP with PVR R = 0. 81 0 500 1000 1500 2000 2500 PVR (dyn/8/cm-6) Williams MH. Eur Heart J. 2006; 27: 1485 -1494. 4. 5 4 3. 5 3 2. 5 2 1. 5 1 0. 5 0 R = -0. 46 0 100 200 300 400 6 MWD (m) 500 600 700
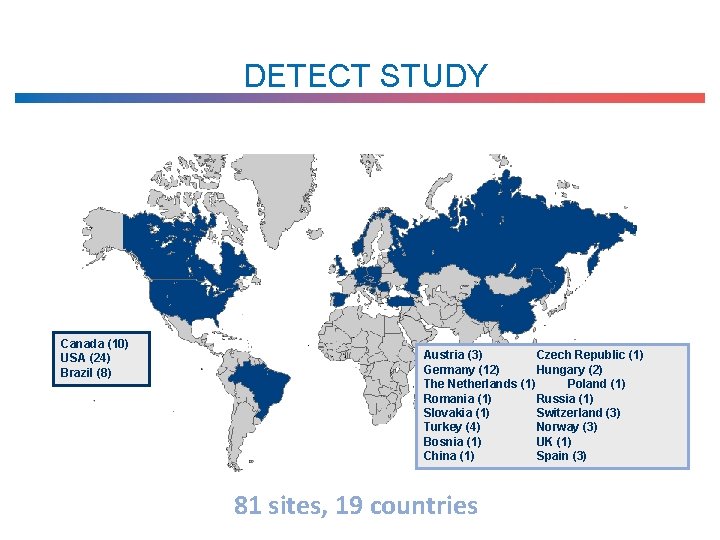
DETECT STUDY Canada (10) USA (24) Brazil (8) Austria (3) Czech Republic (1) Germany (12) Hungary (2) The Netherlands (1) Poland (1) Romania (1) Russia (1) Slovakia (1) Switzerland (3) Turkey (4) Norway (3) Bosnia (1) UK (1) China (1) Spain (3) 81 sites, 19 countries
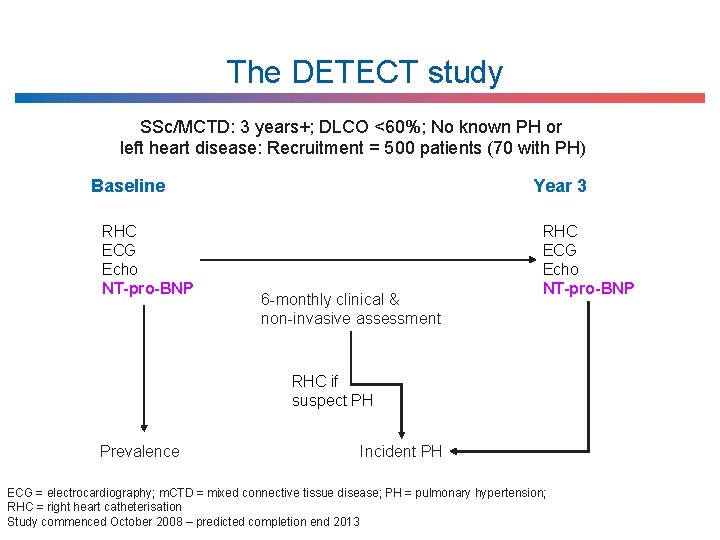
The DETECT study SSc/MCTD: 3 years+; DLCO <60%; No known PH or left heart disease: Recruitment = 500 patients (70 with PH) Baseline RHC ECG Echo NT-pro-BNP Year 3 6 -monthly clinical & non-invasive assessment RHC ECG Echo NT-pro-BNP RHC if suspect PH Prevalence Incident PH ECG = electrocardiography; m. CTD = mixed connective tissue disease; PH = pulmonary hypertension; RHC = right heart catheterisation Study commenced October 2008 – predicted completion end 2013
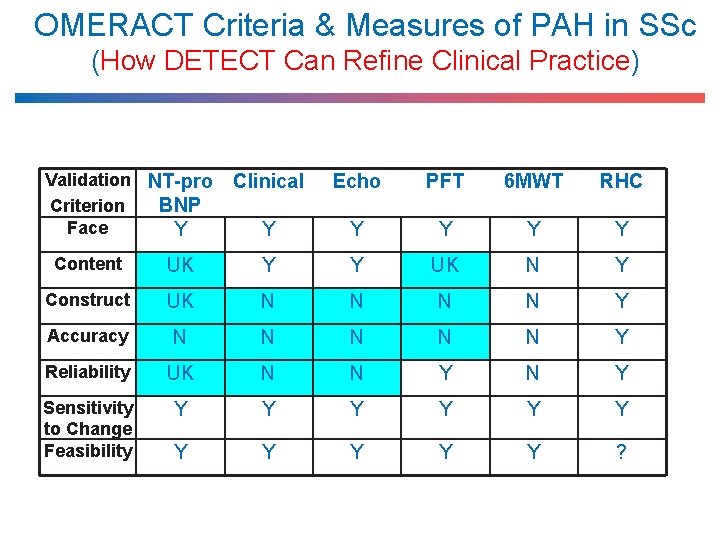
OMERACT Criteria & Measures of PAH in SSc (How DETECT Can Refine Clinical Practice) Validation NT-pro Criterion BNP Face Y Clinical Echo PFT 6 MWT RHC Y Y Y Content UK Y Y UK N Y Construct UK N N Y Accuracy N N N Y Reliability UK N N Y Sensitivity to Change Feasibility Y Y Y ?

Early and Reliable DETECTion of PAH in SSc • Earlier diagnosis • Precise diagnosis • Validation of screening measures • Definition of role of right heart catheterization • Earlier intervention for PAH • Earlier intervention for other forms of PH • Event rate and risk profile for “prevention” study • Event rate and risk profile for “vascular deremodeling” study

PAH–A Distinct Entity PAH Idiopathic/Familial Intimal Proliferation Associated With • Connective tissue disease • Congenital systemic-pulmonary shunts • Portal hypertension • HIV infection • Drugs and toxins • Other Medial Hypertrophy Plexiform lesion
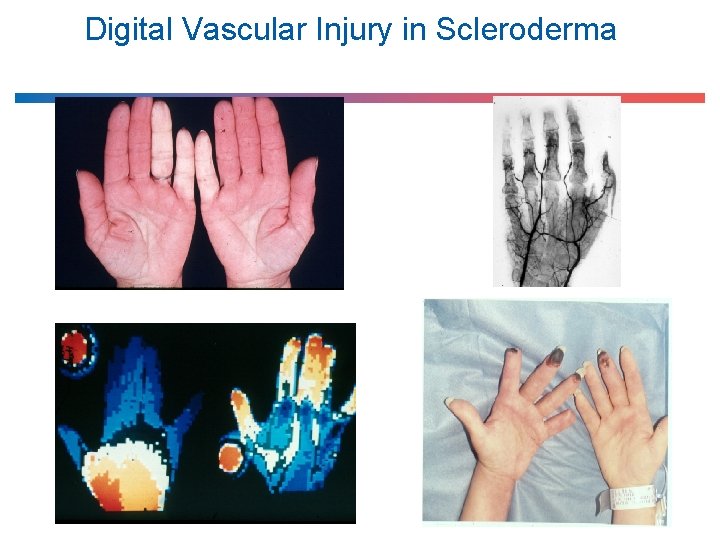
Digital Vascular Injury in Scleroderma
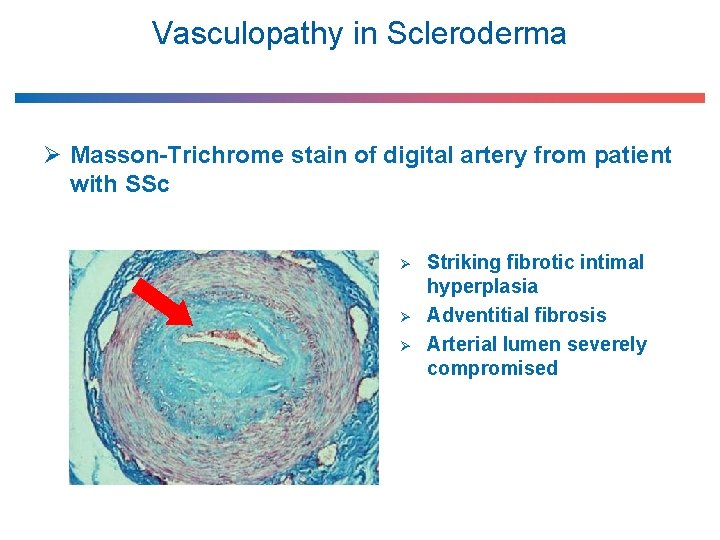
Vasculopathy in Scleroderma Ø Masson-Trichrome stain of digital artery from patient with SSc Ø Ø Ø Striking fibrotic intimal hyperplasia Adventitial fibrosis Arterial lumen severely compromised

Vascular Features and Outcome in SSc Ø Scleroderma renal crisis Ø Pulmonary arterial hypertension Ø Myocardial involvement Ø Sudden death Ø ? ? Role in gastrointestinal involvement
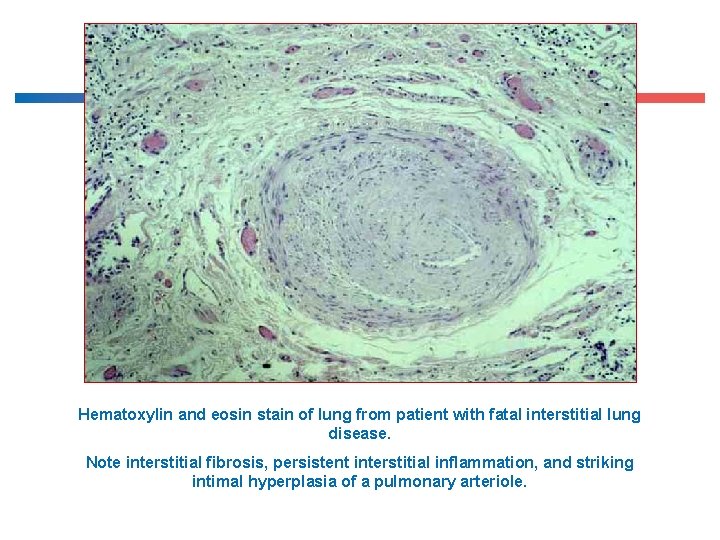
Hematoxylin and eosin stain of lung from patient with fatal interstitial lung disease. Note interstitial fibrosis, persistent interstitial inflammation, and striking intimal hyperplasia of a pulmonary arteriole.

Why is SSc-PH/PAH so Difficult to Treat? Ø Older Patients Ø Interstitial Lung Disease Ø LV Diastolic Dysfunction Ø RV Diastolic Dysfunction Ø More severe structural vasculopathy Ø Outcome measures may be inappropriate Ø Poor recognition in the community
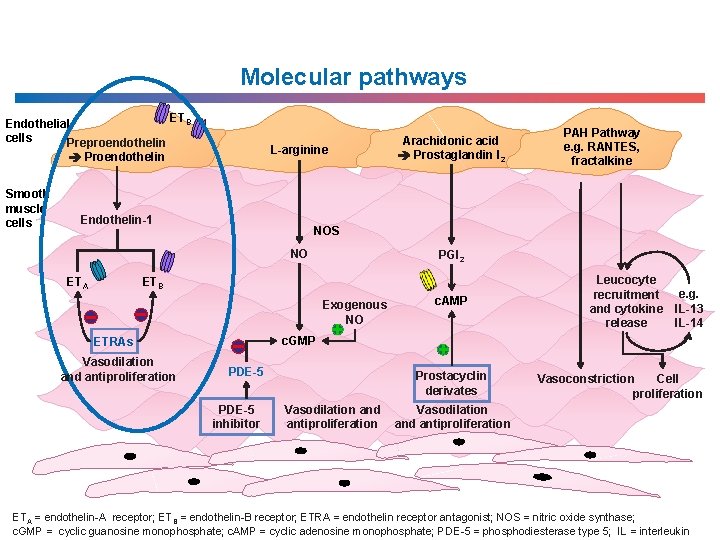
Molecular pathways Endothelial cells Preproendothelin ETB L-arginine Proendothelin Smooth muscle cells Endothelin-1 PGI 2 ETB Exogenous NO c. AMP Leucocyte e. g. recruitment and cytokine IL-13 IL-14 release c. GMP ETRAs Vasodilation and antiproliferation PAH Pathway e. g. RANTES, fractalkine NOS NO ETA Arachidonic acid Prostaglandin I 2 PDE-5 inhibitor Prostacyclin derivates Vasodilation and antiproliferation Cell Vasoconstriction proliferation Vasodilation and antiproliferation ETA = endothelin-A receptor; ETB = endothelin-B receptor; ETRA = endothelin receptor antagonist; NOS = nitric oxide synthase; c. GMP = cyclic guanosine monophosphate; c. AMP = cyclic adenosine monophosphate; PDE-5 = phosphodiesterase type 5; IL = interleukin

Median Change in 6 MWD (m) Epoprostenol Effect on Exercise Capacity Scleroderma with PAH: Treated versus Control 70 60 50 40 30 20 10 0 -10 -20 -30 -40 63. 5 Conventional (55) Flolan (56) 48. 5 13. 25 p=0. 003 -7 p=0. 001 -14 -36 Week 1 Week 6 Badesch et al: Ann Int Med 2000; 132: 425 Week 12 PPH: Barst et al: NEJM 1996; 334: 296
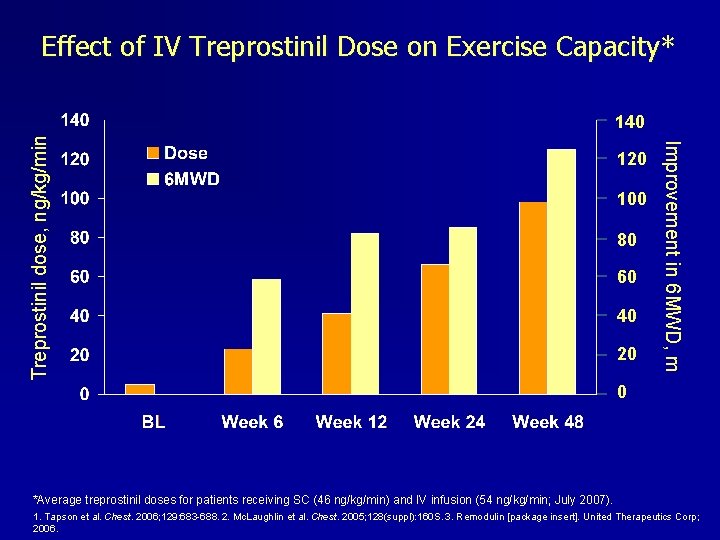
Effect of IV Treprostinil Dose on Exercise Capacity* 120 100 80 60 40 20 Improvement in 6 MWD, m Treprostinil dose, ng/kg/min 140 0 *Average treprostinil doses for patients receiving SC (46 ng/kg/min) and IV infusion (54 ng/kg/min; July 2007). 1. Tapson et al. Chest. 2006; 129: 683 -688. 2. Mc. Laughlin et al. Chest. 2005; 128(suppl): 160 S. 3. Remodulin [package insert]. United Therapeutics Corp; 2006.
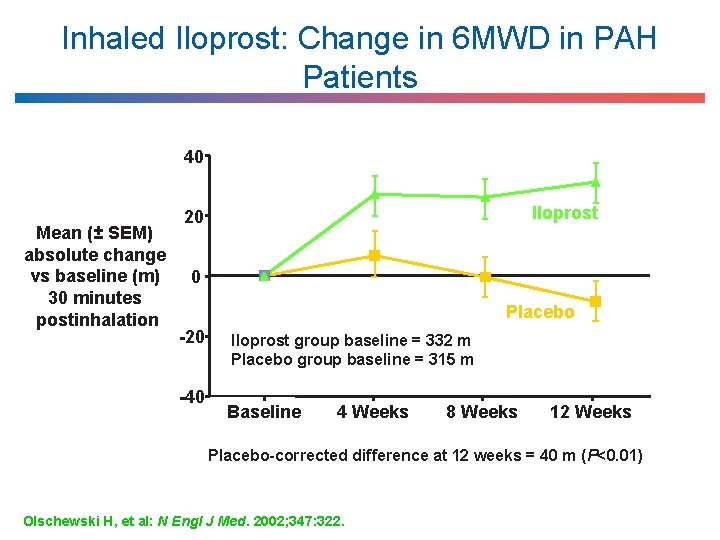
Inhaled Iloprost: Change in 6 MWD in PAH Patients 40 Mean (± SEM) absolute change vs baseline (m) 30 minutes postinhalation Iloprost 20 0 Placebo -20 -40 Iloprost group baseline = 332 m Placebo group baseline = 315 m Baseline 4 Weeks 8 Weeks 12 Weeks Placebo-corrected difference at 12 weeks = 40 m (P<0. 01) Olschewski H, et al: N Engl J Med. 2002; 347: 322.
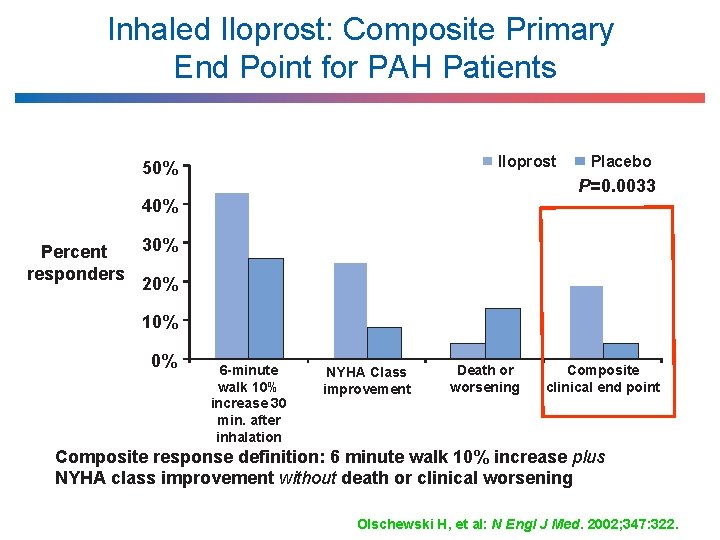
Inhaled Iloprost: Composite Primary End Point for PAH Patients 50% Iloprost 43% P=0. 0033 40% 30% Percent responders 20% 26% 25% 19% 13% 8% 10% 0% Placebo 4% 6 -minute walk 10% increase 30 min. after inhalation NYHA Class improvement Death or worsening 4% Composite clinical end point Composite response definition: 6 minute walk 10% increase plus NYHA class improvement without death or clinical worsening Olschewski H, et al: N Engl J Med. 2002; 347: 322.

% of patients with a greater 6 MWD change Inhaled Treprostinil Distribution of Changes in 6 MWD Treprostinil (n=115) Placebo (n=120) 52% 32% ≤ 6 MWD change from baseline (m) 6 MWD, 6 -minute walk distance. 34
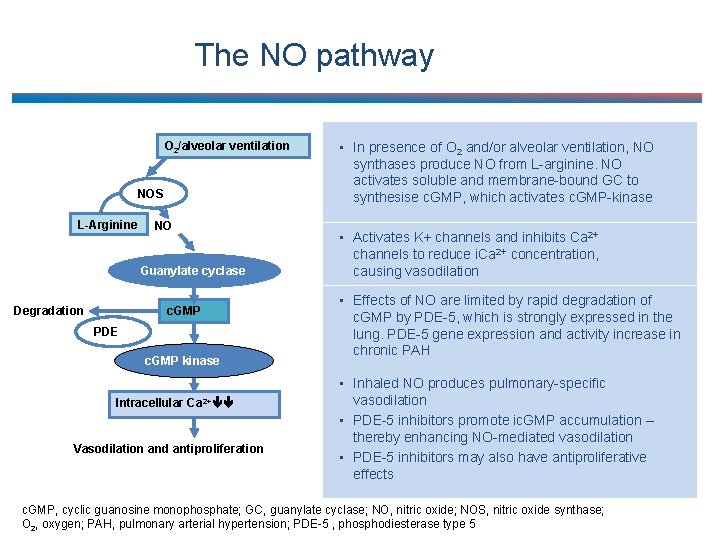
The NO pathway O 2/alveolar ventilation NOS L-Arginine NO Guanylate cyclase Degradation c. GMP PDE c. GMP kinase Intracellular Ca 2+ Vasodilation and antiproliferation • In presence of O 2 and/or alveolar ventilation, NO synthases produce NO from L-arginine. NO activates soluble and membrane-bound GC to synthesise c. GMP, which activates c. GMP-kinase • Activates K+ channels and inhibits Ca 2+ channels to reduce i. Ca 2+ concentration, causing vasodilation • Effects of NO are limited by rapid degradation of c. GMP by PDE-5, which is strongly expressed in the lung. PDE-5 gene expression and activity increase in chronic PAH • Inhaled NO produces pulmonary-specific vasodilation • PDE-5 inhibitors promote ic. GMP accumulation – thereby enhancing NO-mediated vasodilation • PDE-5 inhibitors may also have antiproliferative effects c. GMP, cyclic guanosine monophosphate; GC, guanylate cyclase; NO, nitric oxide; NOS, nitric oxide synthase; O 2, oxygen; PAH, pulmonary arterial hypertension; PDE-5 , phosphodiesterase type 5
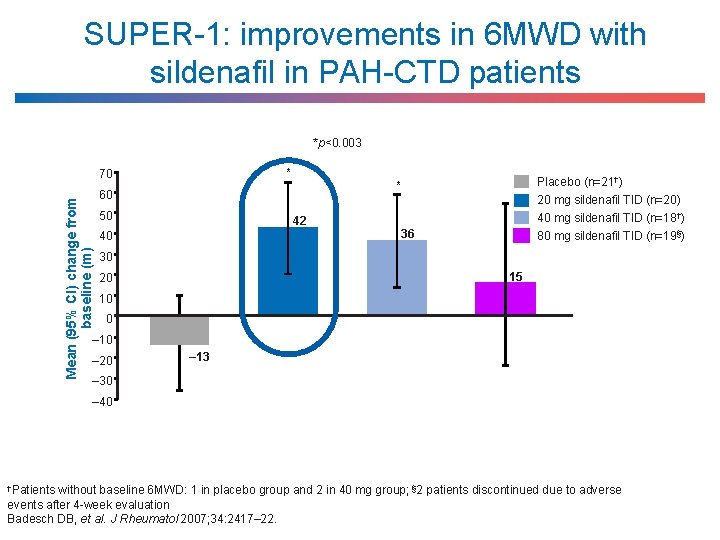
SUPER-1: improvements in 6 MWD with sildenafil in PAH-CTD patients *p<0. 003 * Mean (95% CI) change from baseline (m) 70 60 50 42 40 Placebo (n=21†) * 20 mg sildenafil TID (n=20) 40 mg sildenafil TID (n=18†) 80 mg sildenafil TID (n=19§) 36 30 15 20 10 0 – 10 – 20 – 13 – 30 – 40 †Patients without baseline 6 MWD: 1 in placebo group and 2 in 40 mg group; § 2 patients discontinued due to adverse events after 4 -week evaluation Badesch DB, et al. J Rheumatol 2007; 34: 2417– 22.

Pathophysiological effects of ET-1 Cell proliferation Migration Vasodilation Vasoconstriction Vascular remodelling Antiproliferation Fibrosis Hypertrophy ET-1 = endothelin-1 Apoptosis

Evidence of a Role for Endothelin-1 in Connective Tissue Disease Pathogenesis Ø Elevated plasma levels of ET-1 in SSc Ø Increased levels of ET-1 in bronchoalveolar lavage fluid in SSc Ø Increased ET-1 and ET-B receptors in early diffuse skin lesions Ø Increased ET-1 binding sites in SSc interstitial lung disease Control Abraham DJ, et al. Am J Pathol. 1997; 151: 831 -841. SSc
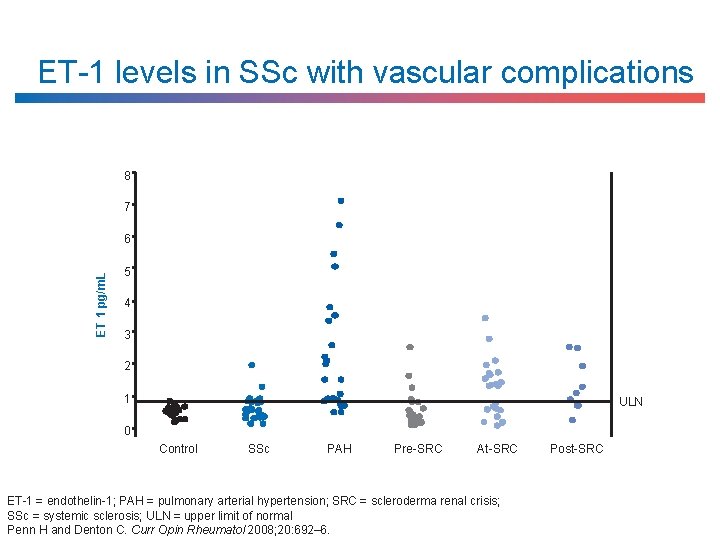
ET-1 levels in SSc with vascular complications 8 7 ET 1 pg/m. L 6 5 4 3 2 1 ULN 0 Control SSc PAH Pre-SRC At-SRC ET-1 = endothelin-1; PAH = pulmonary arterial hypertension; SRC = scleroderma renal crisis; SSc = systemic sclerosis; ULN = upper limit of normal Penn H and Denton C. Curr Opin Rheumatol 2008; 20: 692‒ 6. Post-SRC
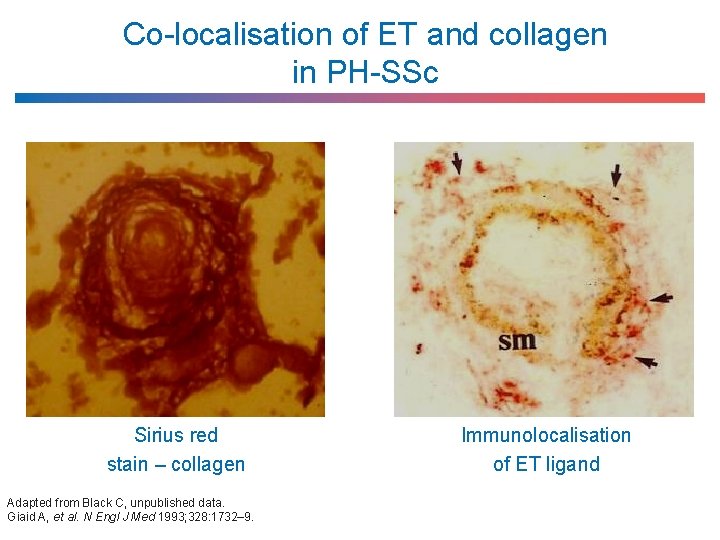
Co-localisation of ET and collagen in PH-SSc Sirius red stain – collagen Adapted from Black C, unpublished data. Giaid A, et al. N Engl J Med 1993; 328: 1732– 9. Immunolocalisation of ET ligand
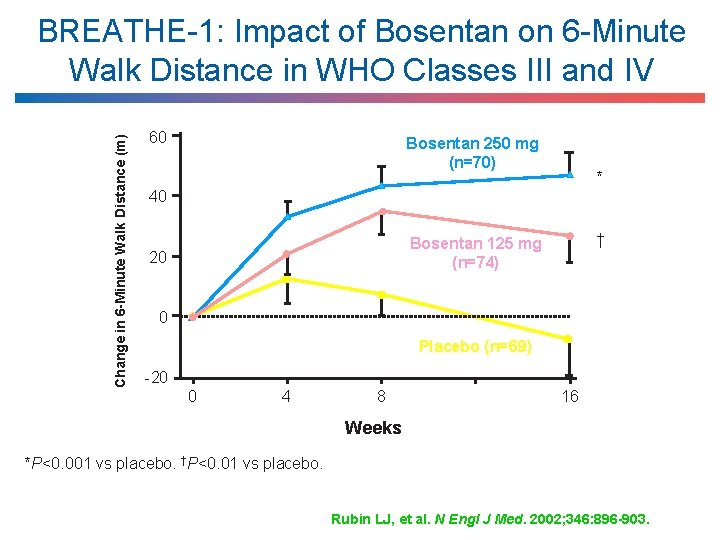
Change in 6 -Minute Walk Distance (m) BREATHE-1: Impact of Bosentan on 6 -Minute Walk Distance in WHO Classes III and IV 60 Bosentan 250 mg (n=70) * 40 † Bosentan 125 mg (n=74) 20 0 Placebo (n=69) -20 0 4 8 16 Weeks *P<0. 001 vs placebo. †P<0. 01 vs placebo. Rubin LJ, et al. N Engl J Med. 2002; 346: 896 -903.

Bosentan for PAH-CTD: Time to Clinical Worsening Event Free (%) 100 90. 3% 86. 4% 80 Difference not significant 60 Bosentan 40 20 0 0 Patients at risk 44 22 4 8 12 16 Weeks From Treatment Start 44 20 29 Bosentan 16 Placebo N=66. Retrospective subgroup analysis from placebo-controlled clinical trials and their extension. Event: death, premature withdrawal or hospitalization due to PAH worsening, or initiation of epoprostenol therapy. Denton S, et al. Ann Rheum Dis. 2006; 65: 1336 -1340.
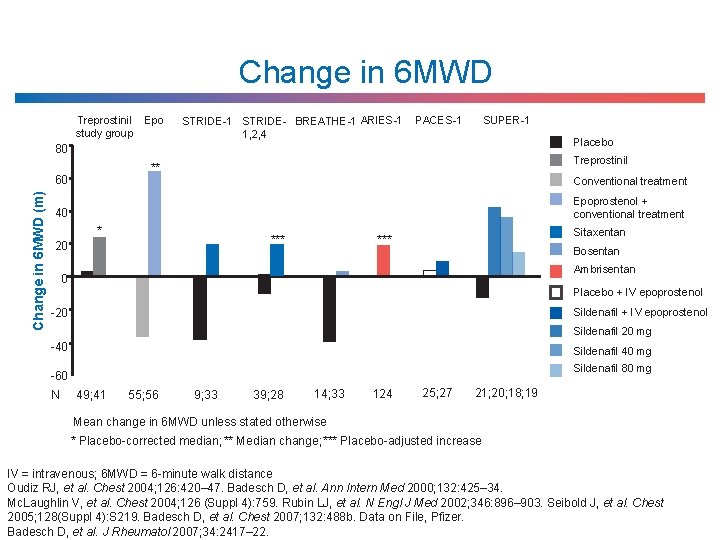
Change in 6 MWD Treprostinil Epo study group STRIDE-1 STRIDE- BREATHE-1 ARIES-1 1, 2, 4 PACES-1 SUPER-1 Placebo 80 60 Change in 6 MWD (m) Treprostinil ** Conventional treatment Epoprostenol + conventional treatment 40 * Sitaxentan *** 20 Bosentan Ambrisentan 0 Placebo + IV epoprostenol Sildenafil + IV epoprostenol -20 Sildenafil 20 mg -40 Sildenafil 40 mg Sildenafil 80 mg -60 N 49; 41 55; 56 9; 33 39; 28 14; 33 124 25; 27 21; 20; 18; 19 Mean change in 6 MWD unless stated otherwise * Placebo-corrected median; ** Median change; *** Placebo-adjusted increase IV = intravenous; 6 MWD = 6 -minute walk distance Oudiz RJ, et al. Chest 2004; 126: 420– 47. Badesch D, et al. Ann Intern Med 2000; 132: 425– 34. Mc. Laughlin V, et al. Chest 2004; 126 (Suppl 4): 759. Rubin LJ, et al. N Engl J Med 2002; 346: 896– 903. Seibold J, et al. Chest 2005; 128(Suppl 4): S 219. Badesch D, et al. Chest 2007; 132: 488 b. Data on File, Pfizer. Badesch D, et al. J Rheumatol 2007; 34: 2417– 22.
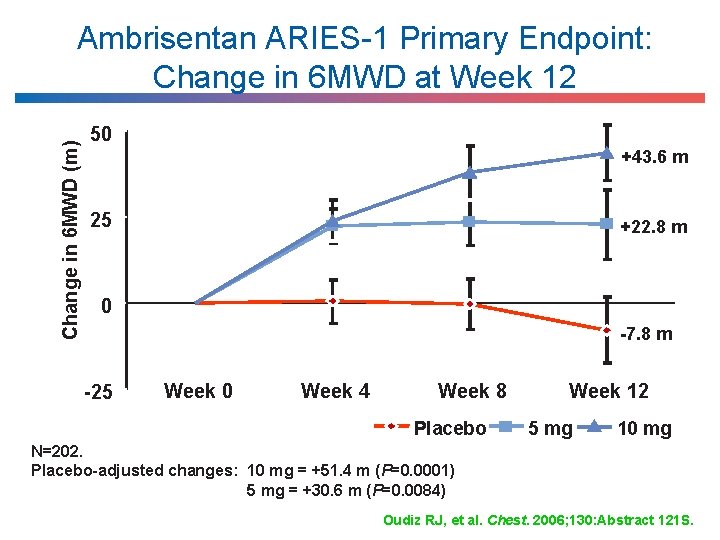
Change in 6 MWD (m) Ambrisentan ARIES-1 Primary Endpoint: Change in 6 MWD at Week 12 50 +43. 6 m 25 +22. 8 m 0 -7. 8 m -25 Week 0 Week 4 Week 8 Placebo Week 12 5 mg 10 mg N=202. Placebo-adjusted changes: 10 mg = +51. 4 m (P=0. 0001) 5 mg = +30. 6 m (P=0. 0084) Oudiz RJ, et al. Chest. 2006; 130: Abstract 121 S.

Change From Baseline (meters) Ambrisentan for CTD-PAH: Change in 6 -Minute Walk Distance at 12 Weeks PAH (n=166) PAH-CTD (n=82) 64. 0 59. 0 49. 0 24. 0 -6. 4 29. 0 -13. 4 2. 5 mg 10 mg Ambrisentan Patients with PAH-CTD, post-hoc subgroup analysis from integrated analysis of double-blind, placebo-controlled trials. Coghlan JG, et al. Lupus. 2006; 15: 138 -142.
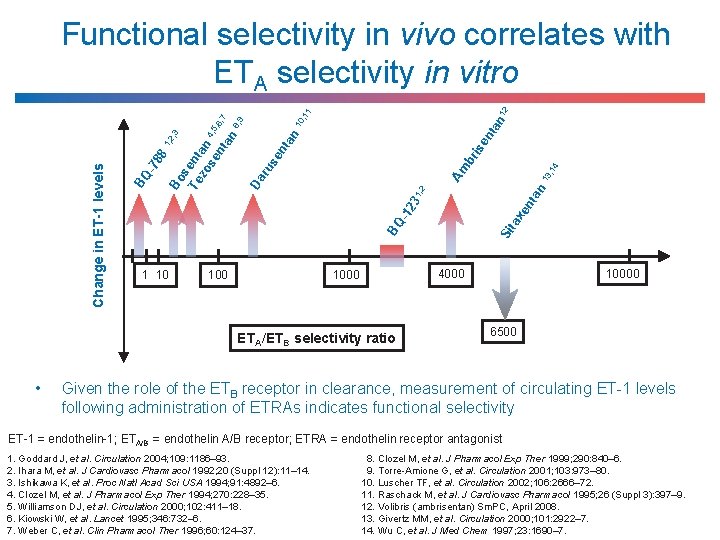
n 12 , 1 1 ta 10 nt an 13 Am , 2 xe 23 1 100 Si ta BQ -1 1 10 10000 4000 1000 ETA/ETB selectivity ratio • , 1 4 br is en an nt se Da ru 88 BQ -7 Change in ET-1 levels 1 8, 9 , 2 , 3 Bo se Te nt zo an se 4, 5, 6 nt an , 7 Functional selectivity in vivo correlates with ETA selectivity in vitro 6500 Given the role of the ETB receptor in clearance, measurement of circulating ET-1 levels following administration of ETRAs indicates functional selectivity ET-1 = endothelin-1; ETA/B = endothelin A/B receptor; ETRA = endothelin receptor antagonist 1. Goddard J, et al. Circulation 2004; 109: 1186‒ 93. 2. Ihara M, et al. J Cardiovasc Pharmacol 1992; 20 (Suppl 12): 11‒ 14. 3. Ishikawa K, et al. Proc Natl Acad Sci USA 1994; 91: 4892‒ 6. 4. Clozel M, et al. J Pharmacol Exp Ther 1994; 270: 228‒ 35. 5. Williamson DJ, et al. Circulation 2000; 102: 411‒ 18. 6. Kiowski W, et al. Lancet 1995; 346: 732‒ 6. 7. Weber C, et al. Clin Pharmacol Ther 1996; 60: 124‒ 37. 8. Clozel M, et al. J Pharmacol Exp Ther 1999; 290: 840‒ 6. 9. Torre-Amione G, et al. Circulation 2001; 103: 973‒ 80. 10. Luscher TF, et al. Circulation 2002; 106: 2666‒ 72. 11. Raschack M, et al. J Cardiovasc Pharmacol 1995; 26 (Suppl 3): 397‒ 9. 12. Volibris (ambrisentan) Sm. PC, April 2008. 13. Givertz MM, et al. Circulation 2000; 101: 2922‒ 7. 14. Wu C, et al. J Med Chem 1997; 23: 1690‒ 7.
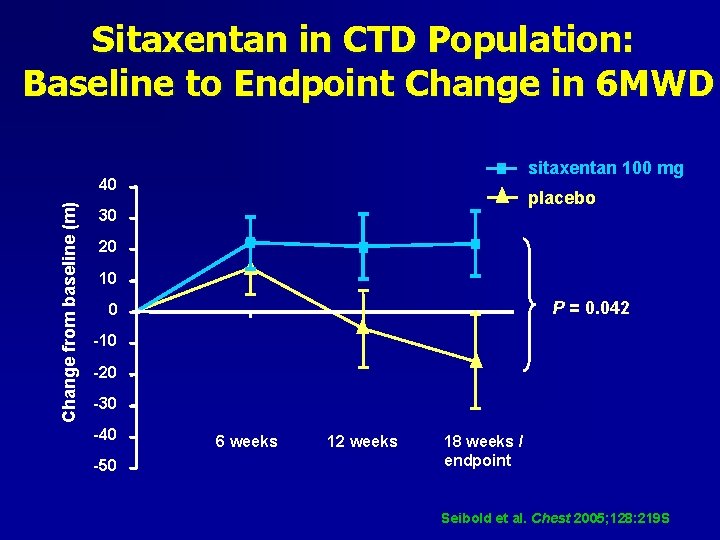
Sitaxentan in CTD Population: Baseline to Endpoint Change in 6 MWD sitaxentan 100 mg Change from baseline (m) 40 placebo 30 20 10 P = 0. 042 0 -10 -20 -30 -40 -50 6 weeks 12 weeks 18 weeks / endpoint Seibold et al. Chest 2005; 128: 219 S
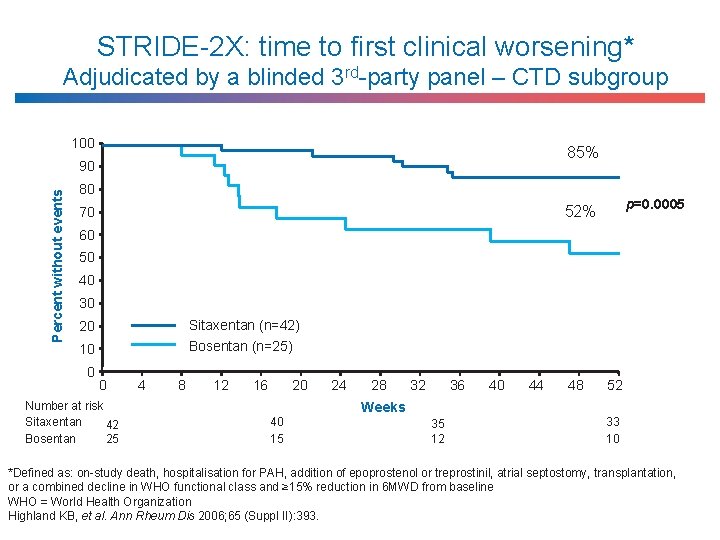
STRIDE-2 X: time to first clinical worsening* Adjudicated by a blinded 3 rd-party panel – CTD subgroup 100 85% Percent without events 90 80 p=0. 0005 52% 70 60 50 40 30 Sitaxentan (n=42) Bosentan (n=25) 20 10 0 0 Number at risk Sitaxentan 42 Bosentan 25 4 8 12 16 20 40 15 24 28 32 36 Weeks 35 12 40 44 48 52 33 10 *Defined as: on-study death, hospitalisation for PAH, addition of epoprostenol or treprostinil, atrial septostomy, transplantation, or a combined decline in WHO functional class and ≥ 15% reduction in 6 MWD from baseline WHO = World Health Organization Highland KB, et al. Ann Rheum Dis 2006; 65 (Suppl II): 393.
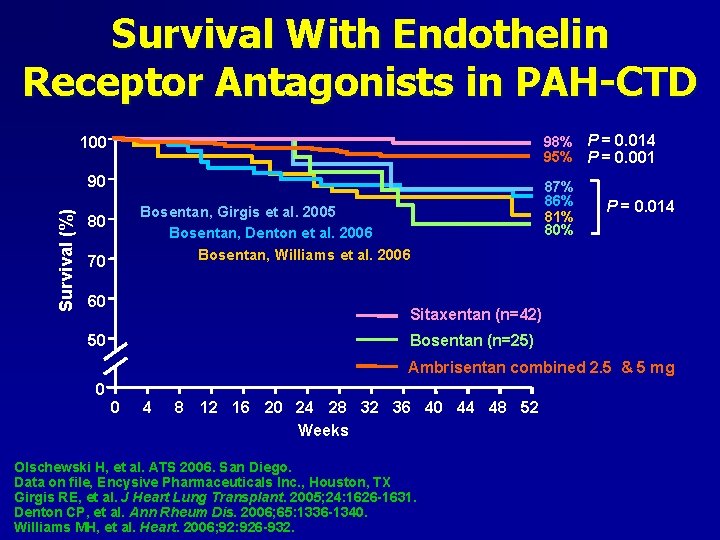
Survival With Endothelin Receptor Antagonists in PAH-CTD 98% P = 0. 014 95% P = 0. 001 100 Survival (%) 90 Bosentan, Girgis et al. 2005 Bosentan, Denton et al. 2006 Bosentan, Williams et al. 2006 80 70 60 87% 86% 81% 80% P = 0. 014 Sitaxentan (n=42) Bosentan (n=25) 50 Ambrisentan combined 2. 5 & 5 mg 0 0 4 8 12 16 20 24 28 32 36 40 44 48 52 Weeks Olschewski H, et al. ATS 2006. San Diego. Data on file, Encysive Pharmaceuticals Inc. , Houston, TX Girgis RE, et al. J Heart Lung Transplant. 2005; 24: 1626 -1631. Denton CP, et al. Ann Rheum Dis. 2006; 65: 1336 -1340. Williams MH, et al. Heart. 2006; 92: 926 -932.
Survival by PAH Etiology
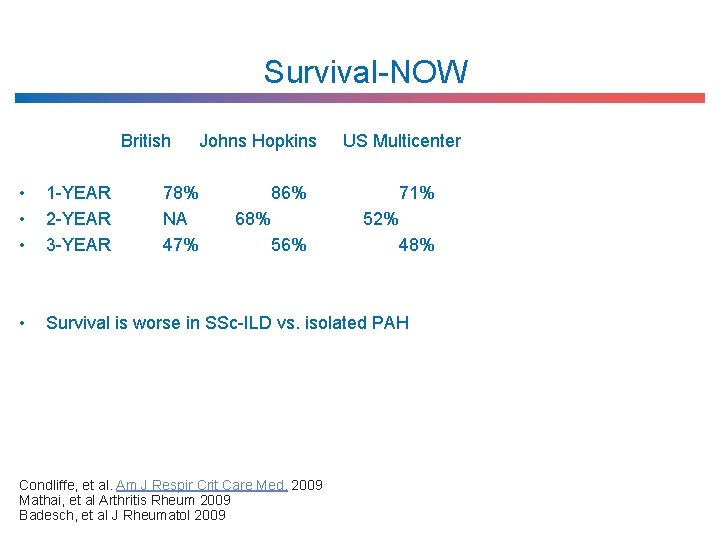
Survival-NOW British Johns Hopkins US Multicenter • • • 1 -YEAR 2 -YEAR 3 -YEAR • Survival is worse in SSc-ILD vs. isolated PAH 78% NA 47% 86% 68% 56% Condliffe, et al. Am J Respir Crit Care Med. 2009 Mathai, et al Arthritis Rheum 2009 Badesch, et al J Rheumatol 2009 71% 52% 48%

Recommended stepwise approach to combination therapy Treatment goals not met Initial treatment with an ETRA + + Addition of a PDE-5 inhibitor Addition of inhaled prostanoid Transition from inhaled to IV prostanoid + Highly urgent lung transplantation
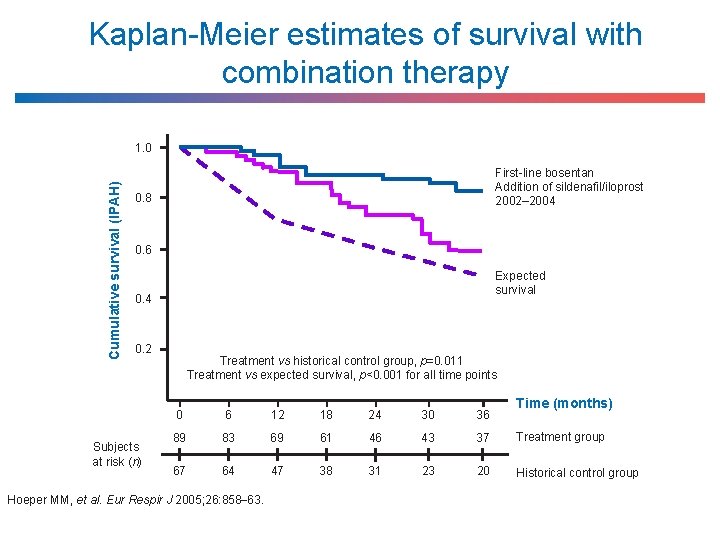
Kaplan-Meier estimates of survival with combination therapy Cumulative survival (IPAH) 1. 0 First-line bosentan Addition of sildenafil/iloprost 2002– 2004 0. 8 0. 6 Expected survival 0. 4 0. 2 Subjects at risk (n) Treatment vs historical control group, p=0. 011 Treatment vs expected survival, p<0. 001 for all time points Time (months) 0 6 12 18 24 30 36 89 83 69 61 46 43 37 Treatment group 67 64 47 38 31 23 20 Historical control group Hoeper MM, et al. Eur Respir J 2005; 26: 858– 63.
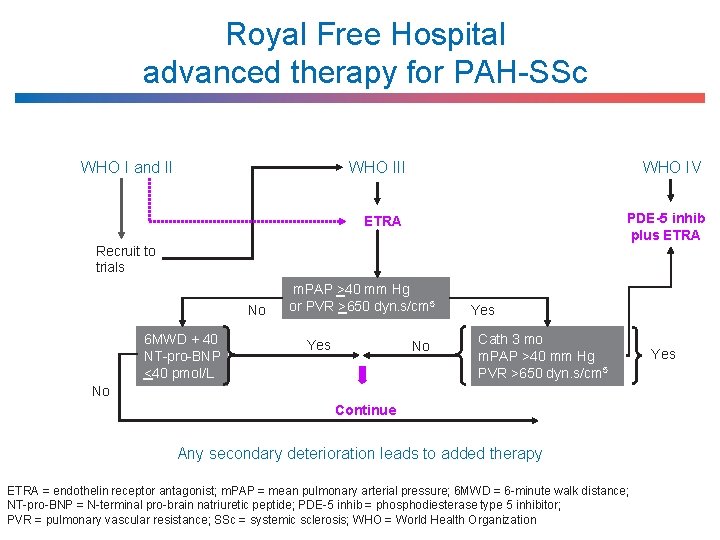
Royal Free Hospital advanced therapy for PAH-SSc WHO I and II WHO IV ETRA PDE-5 inhib plus ETRA Recruit to trials No 6 MWD + 40 NT-pro-BNP <40 pmol/L m. PAP >40 mm Hg or PVR >650 dyn. s/cm 5 Yes No Yes Cath 3 mo m. PAP >40 mm Hg PVR >650 dyn. s/cm 5 No Continue Any secondary deterioration leads to added therapy ETRA = endothelin receptor antagonist; m. PAP = mean pulmonary arterial pressure; 6 MWD = 6 -minute walk distance; NT-pro-BNP = N-terminal pro-brain natriuretic peptide; PDE-5 inhib = phosphodiesterase type 5 inhibitor; PVR = pulmonary vascular resistance; SSc = systemic sclerosis; WHO = World Health Organization Yes

Food for Thought? ? • PAH-CTD is overdiagnosed in community – primarily due to excessive reliance on echocardiography • PAH-CTD is under managed in centers – primarily due to avoidance of parenteral prostacyclin • Available data support strategies for EARLIER diagnosis and intervention • Available data support use of “step up” combination therapy in “goal directed approach” • Earlier use of right heart catheterization may permit true “prevention” studies
- Slides: 56