Cardiovascular Disease and Diabetes Tara Dall MD www

+ Cardiovascular Disease and Diabetes Tara Dall, MD www. advlip. com

Clinical Pearls + n n Screen all individuals aged 45 years or older for diabetes: n Screen younger individuals if BMI ≥ 25 kg/m 2 plus one additional risk factor n Diagnose diabetes when A 1 C is ≥ 6. 5% Check A 1 C every 3 months until A 1 C < 7. 0%, then at least every 6 months thereafter: n Intensify glucose-lowering regimen whenever A 1 C is ≥ 7. 0% n Hypoglycemia is a leading limiting factor in achieving and maintaining glycemic control n Intensive glycemic control exceeding an A 1 C goal of < 7. 0% yields no significant reduction in CV disease (CVD) outcomes compared to standard glycemic control

+ Case Study 1: Introduction Juanita comes in for a required physical examination prior to attending her daughter’s summer camp as a chaperone: n 42 -year-old Latina woman n Family history: type 2 diabetes in mother at age 56 years n 2 children; one was > 9 lbs at birth n BMI: 28. 2 kg/m 2 Should Juanita be screened for diabetes at today’s visit?

ADA Recommendations for Type 2 Diabetes Screening: Risk Factors + • Screen all individuals aged ≥ 45 years; if normal, repeat every 3 years • Consider earlier, more frequent screening if BMI ≥ 25 kg/m 2 plus any of these additional risk factors: n Habitual physical inactivity n First-degree relative with diabetes n Polycystic ovarian syndrome n History of CVD n n n Hypertension (≥ 140/90 mm Hg) History of impaired glucose tolerance (IGT) or impaired fasting glucose (IFG) Other conditions associated with insulin resistance: n n History of delivering a baby weighing > 9 lbs and/or gestational diabetes mellitus n High-density lipoprotein cholesterol (HDL-C) level < 35 mg/d. L and/or triglyceride level > 250 mg/d. L n Member of a high-risk ethnic population: n n n African American Latino Native American Asian American Pacific Islander Acanthosis nigricans American Diabetes Association. Diabetes Care. 2009; 32(suppl 1): S 13 -S 61.

2009 International Expert Committee Recommendation for Diagnosis + n Diagnosis of diabetes is made when A 1 C is ≥ 6. 5%: n Diagnosis should be confirmed with a repeat A 1 C test n No confirmation needed if symptomatic and plasma glucose levels are > 200 mg/d. L n If A 1 C testing is not available, fasting plasma glucose or 2 -hour oral glucose tolerance test is acceptable n Test A 1 C in children if diabetes is suspected but classic symptoms and casual plasma glucose level > 200 mg/d. L are not found n The categories of “prediabetes”, “IFG”, and “IGT” fail to capture the continuum of risk and will be phased out of use n Use prevention measures in individuals with A 1 C < 6. 5% but ≥ 6. 0%: n A 1 C levels below this range may indicate risk and, depending on the presence of other risk factors, prevention efforts may be beneficial International Expert Committee. Diabetes Care. 2009; 32: 1327 -1334.
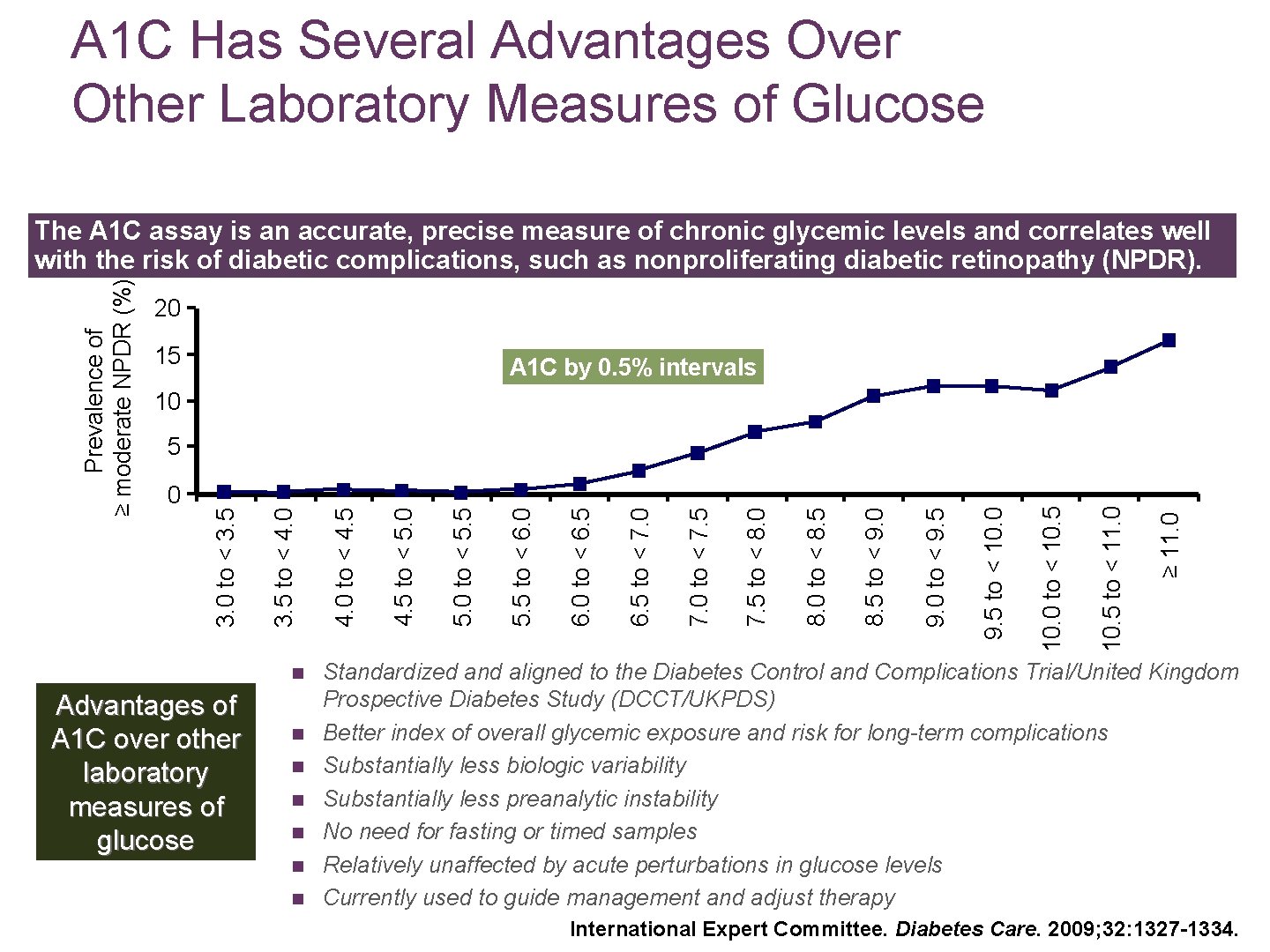
A 1 C Has Several Advantages Over Other Laboratory Measures of Glucose 20 15 A 1 C by 0. 5% intervals 10 n Advantages of A 1 C over other laboratory measures of glucose n n n ≥ 11. 0 10. 5 to < 11. 0 10. 0 to < 10. 5 9. 5 to < 10. 0 9. 0 to < 9. 5 8. 5 to < 9. 0 8. 0 to < 8. 5 7. 5 to < 8. 0 7. 0 to < 7. 5 6. 5 to < 7. 0 6. 0 to < 6. 5 5. 5 to < 6. 0 5. 0 to < 5. 5 4. 5 to < 5. 0 4. 0 to < 4. 5 0 3. 5 to < 4. 0 5 3. 0 to < 3. 5 Prevalence of ≥ moderate NPDR (%) The A 1 C assay is an accurate, precise measure of chronic glycemic levels and correlates well with the risk of diabetic complications, such as nonproliferating diabetic retinopathy (NPDR). Standardized and aligned to the Diabetes Control and Complications Trial/United Kingdom Prospective Diabetes Study (DCCT/UKPDS) Better index of overall glycemic exposure and risk for long-term complications Substantially less biologic variability Substantially less preanalytic instability No need for fasting or timed samples Relatively unaffected by acute perturbations in glucose levels Currently used to guide management and adjust therapy International Expert Committee. Diabetes Care. 2009; 32: 1327 -1334.
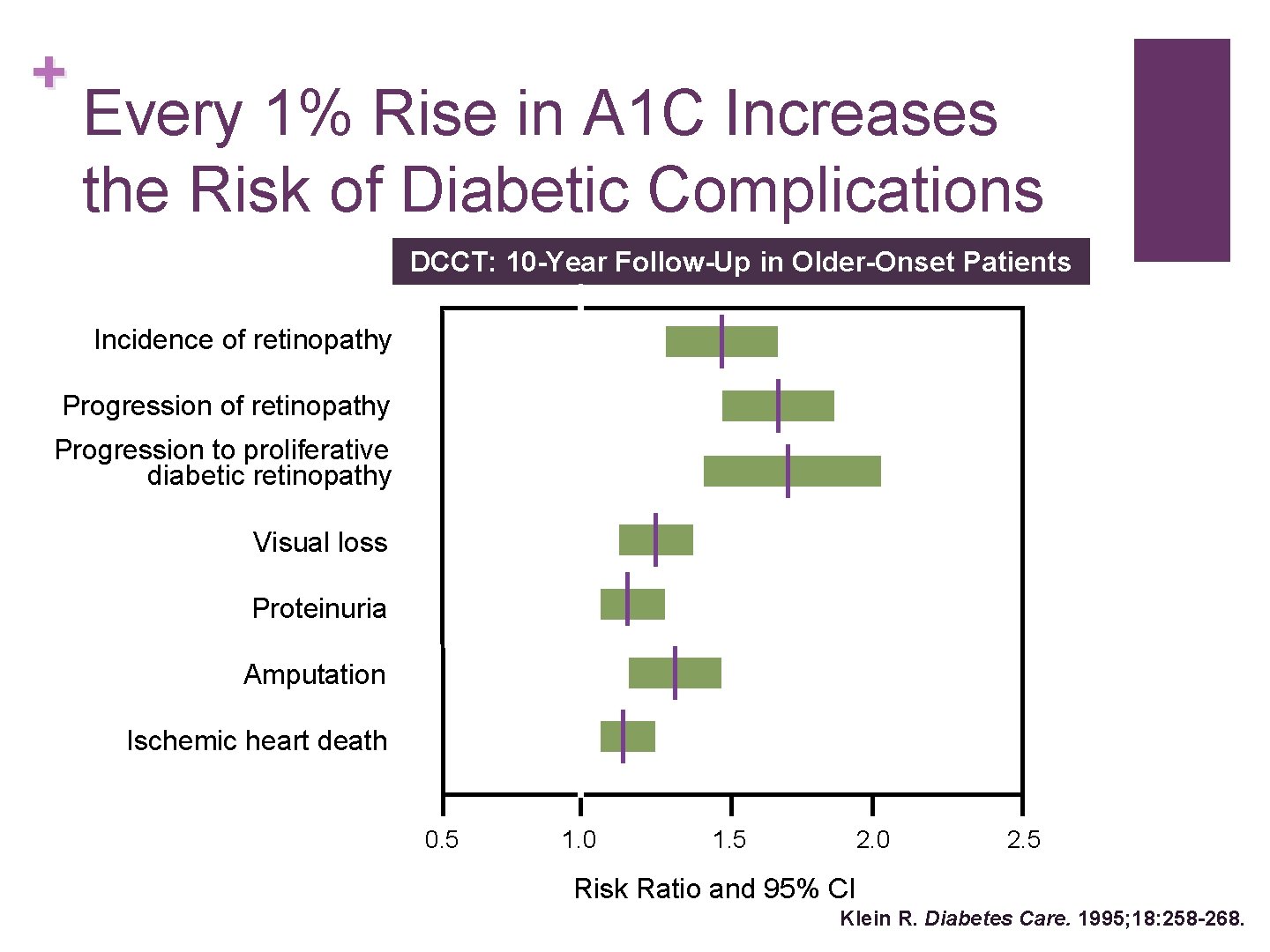
+ Every 1% Rise in A 1 C Increases the Risk of Diabetic Complications DCCT: 10 -Year Follow-Up in Older-Onset Patients Incidence of retinopathy Progression to proliferative diabetic retinopathy Visual loss Proteinuria Amputation Ischemic heart death 0. 5 1. 0 1. 5 2. 0 2. 5 Risk Ratio and 95% CI Klein R. Diabetes Care. 1995; 18: 258 -268.
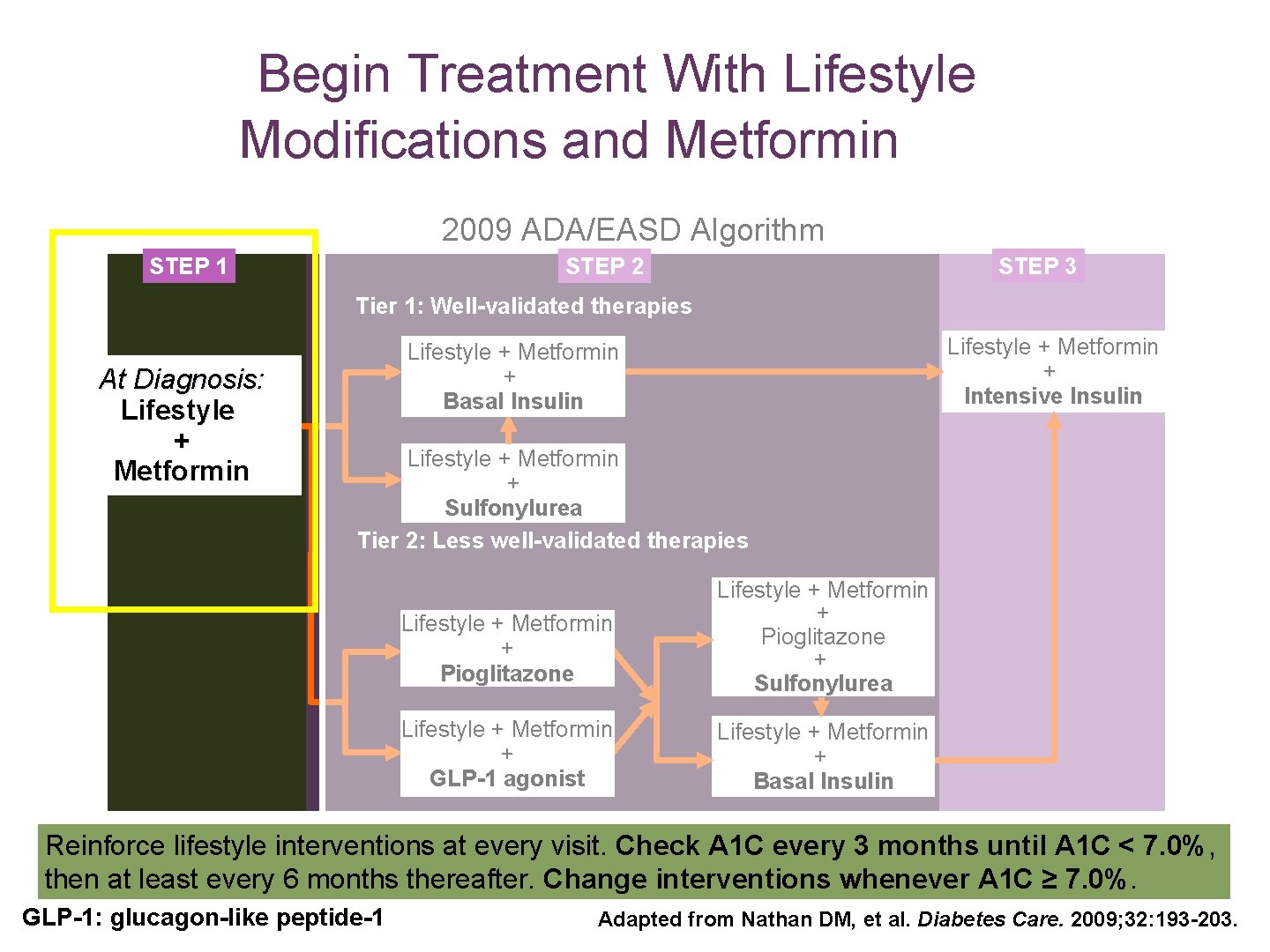
Begin Treatment With Lifestyle Modifications and Metformin 2009 ADA/EASD Algorithm STEP 1 STEP 2 STEP 3 Tier 1: Well-validated therapies At Diagnosis: Lifestyle + Metformin + Intensive Insulin Lifestyle + Metformin + Basal Insulin Lifestyle + Metformin + Sulfonylurea Tier 2: Less well-validated therapies Lifestyle + Metformin + Pioglitazone + Sulfonylurea Lifestyle + Metformin + GLP-1 agonist Lifestyle + Metformin + Basal Insulin Reinforce lifestyle interventions at every visit. Check A 1 C every 3 months until A 1 C < 7. 0%, then at least every 6 months thereafter. Change interventions whenever A 1 C ≥ 7. 0%. GLP-1: glucagon-like peptide-1 Adapted from Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203.
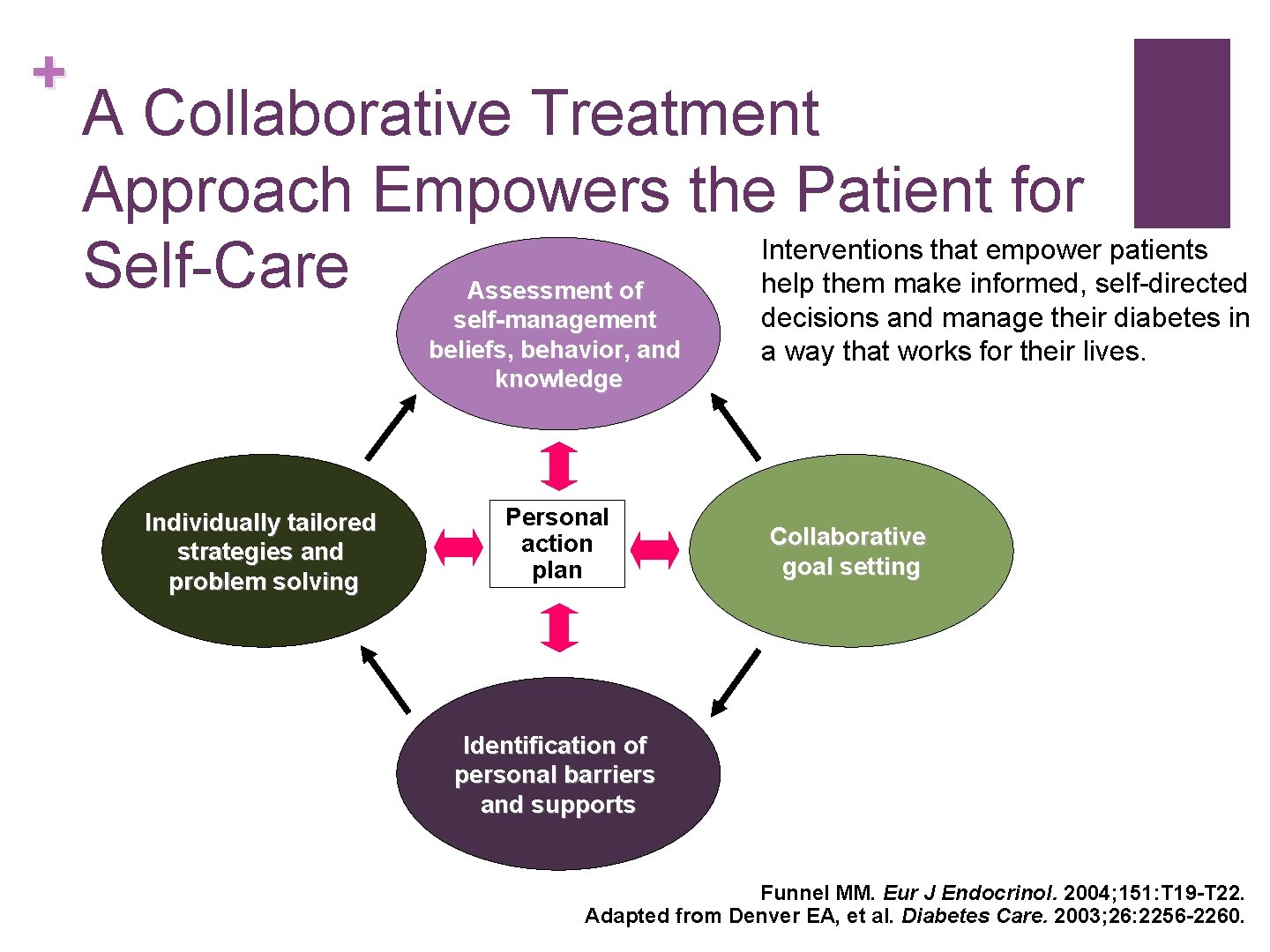
+ A Collaborative Treatment Approach Empowers the Patient for Interventions that empower patients Self-Care help them make informed, self-directed Assessment of self-management beliefs, behavior, and knowledge Individually tailored strategies and problem solving Personal action plan decisions and manage their diabetes in a way that works for their lives. Collaborative goal setting Identification of personal barriers and supports Funnel MM. Eur J Endocrinol. 2004; 151: T 19 -T 22. Adapted from Denver EA, et al. Diabetes Care. 2003; 26: 2256 -2260.

+ Principles Underlying Patient. Centered Diabetes Care n People with diabetes live multifaceted lives with competing demands that influence their diabetes self-care n People living with diabetes make complex self-care decisions every day n Family and other support systems strongly influence diabetes self-care n People with diabetes learn ideas and concepts that they perceive as important n Learning occurs when the individuals are engaged n Learning is a process that requires reinforcement and flexibility Joslin Diabetes Center Web site. http: //joslin. org/755. asp. Accessed August 11, 2009.
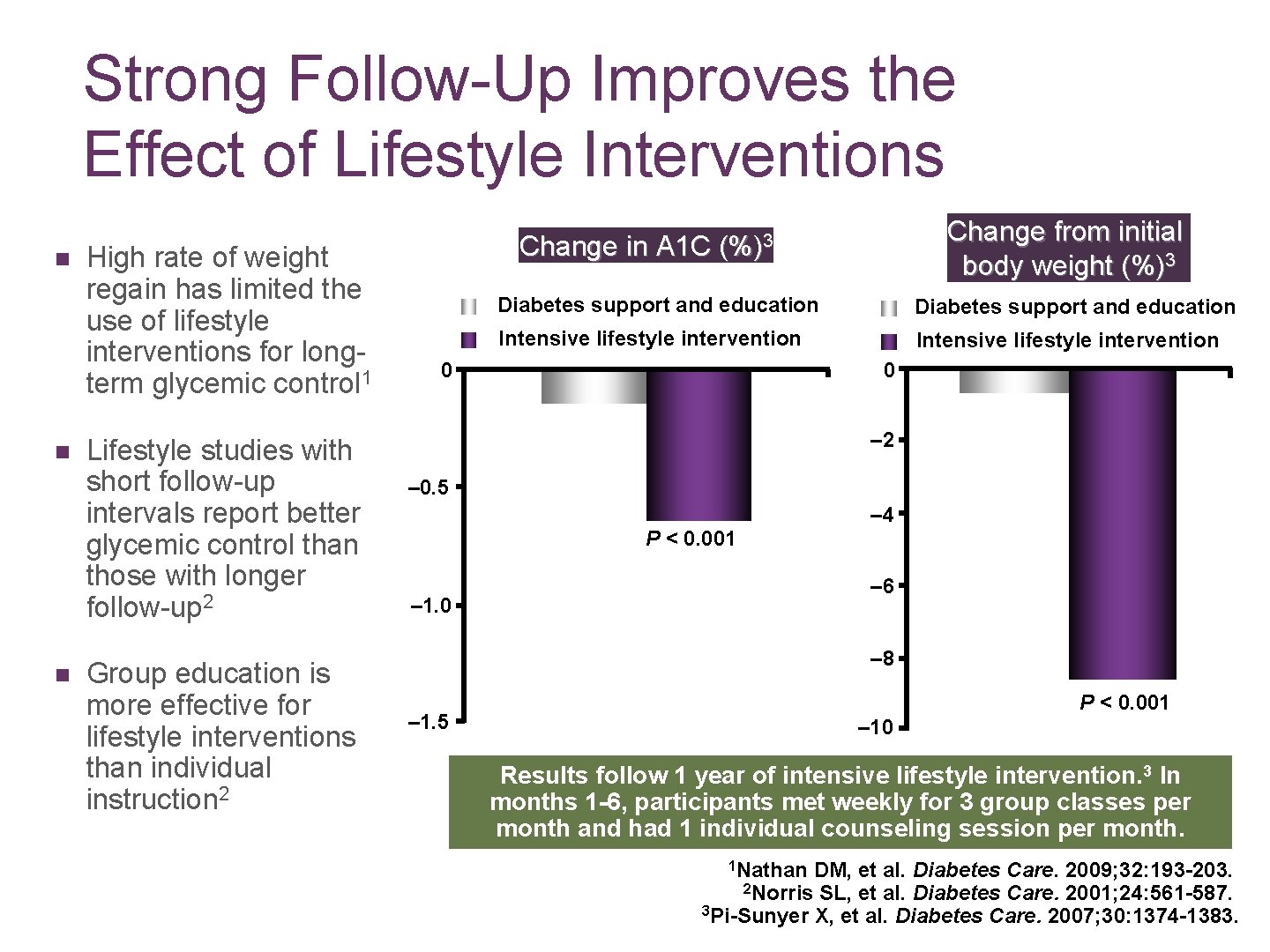
Strong Follow-Up Improves the Effect of Lifestyle Interventions n n n High rate of weight regain has limited the use of lifestyle interventions for longterm glycemic control 1 Lifestyle studies with short follow-up intervals report better glycemic control than those with longer follow-up 2 Group education is more effective for lifestyle interventions than individual instruction 2 Change in A 1 C Change from initial body weight (%)3 Diabetes support and education Intensive lifestyle intervention 0 0 – 2 – 0. 5 – 4 P < 0. 001 – 6 – 1. 0 – 8 P < 0. 001 – 1. 5 – 10 Results follow 1 year of intensive lifestyle intervention. 3 In months 1 -6, participants met weekly for 3 group classes per month and had 1 individual counseling session per month. 1 Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203. 2 Norris SL, et al. Diabetes Care. 2001; 24: 561 -587. 3 Pi-Sunyer X, et al. Diabetes Care. 2007; 30: 1374 -1383.

Case Study 1 Juanita comes in for a required physical examination prior to attending her daughter’s summer camp as a chaperone: n 42 -year-old Latina woman n Family history: type 2 diabetes in mother at age 56 years n 2 children; one was > 9 lbs at birth n BMI: 28. 2 kg/m 2 Should Juanita be screened for diabetes at today’s visit?

+ n Case Study 2: Introduction James (aged 63 years) n Diagnosis: type 2 diabetes (September 2007); A 1 C 8. 5% n Initial regimen: metformin 850 mg BID; glimepiride 1 mg once daily n March 2008: A 1 C 7. 0% n October 2008: A 1 C 6. 9% n May 2009: A 1 C 8. 0%; increase glimepiride (2 mg) n August 2008: A 1 C 7. 6%; James now complains of 3 episodes of hypoglycemia in the past month

+ Type 2 Diabetes: Unmet Needs 20

21 Unmet therapeutic needs in diabetes Further improving the management of diabetes requires an approach that will: n Achieve current treatment targets (including Hb. A 1 c, fasting and postprandial glucose, weight, lipids, blood pressure) n Prevent / delay the decline in glycemic control over time n Improve patient compliance n Minimize adverse events, especially with respect to cardiovascular safety
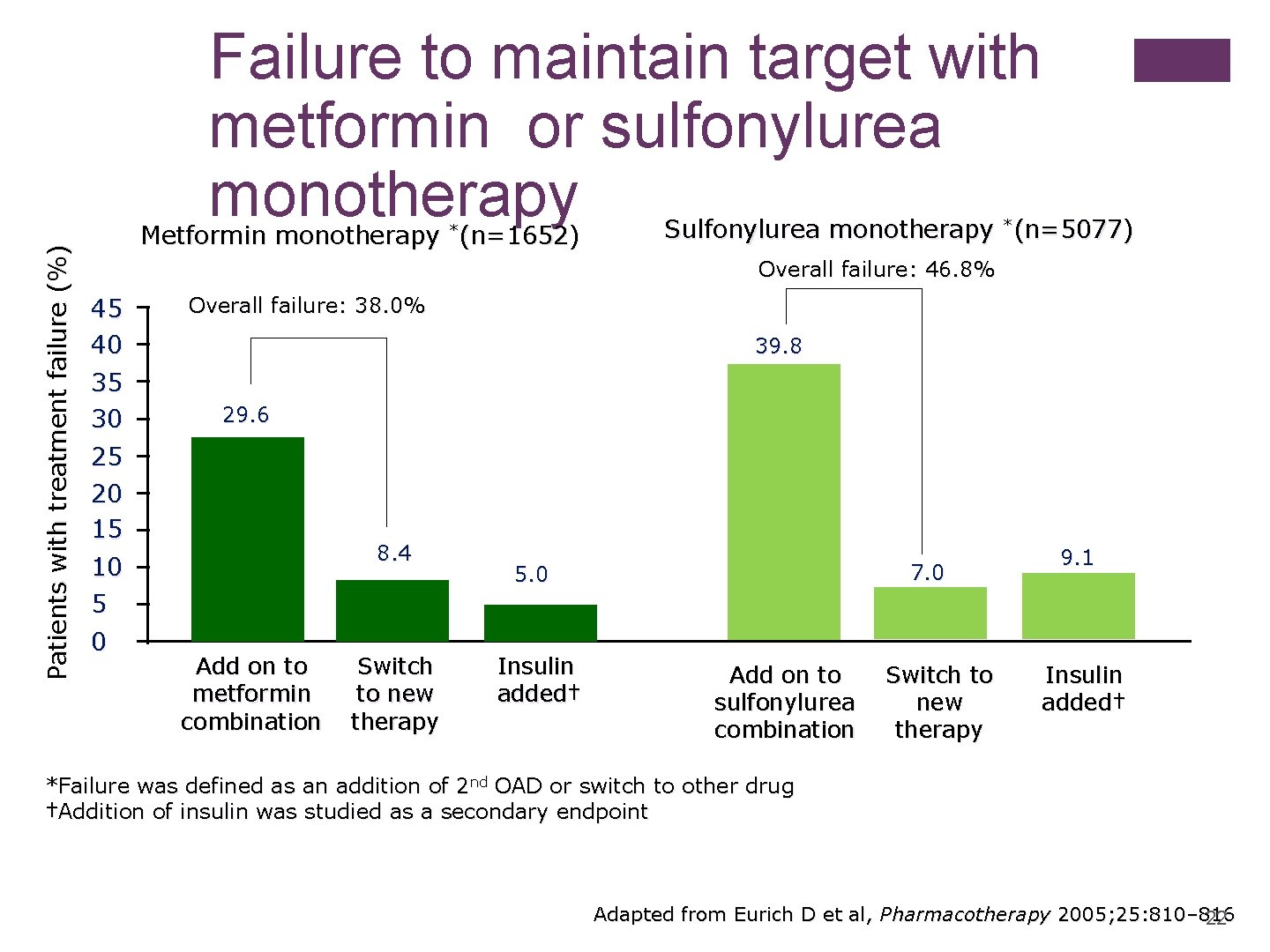
Patients with treatment failure (%) Failure to maintain target with metformin or sulfonylurea monotherapy Metformin monotherapy *(n=1652) Sulfonylurea monotherapy *(n=5077) Overall failure: 46. 8% 45 40 35 30 Overall failure: 38. 0% 39. 8 29. 6 25 20 15 10 5 0 8. 4 Add on to metformin combination Switch to new therapy 7. 0 5. 0 Insulin added† Add on to sulfonylurea combination Switch to new therapy 9. 1 Insulin added† *Failure was defined as an addition of 2 nd OAD or switch to other drug †Addition of insulin was studied as a secondary endpoint Adapted from Eurich D et al, Pharmacotherapy 2005; 25: 810– 816 22
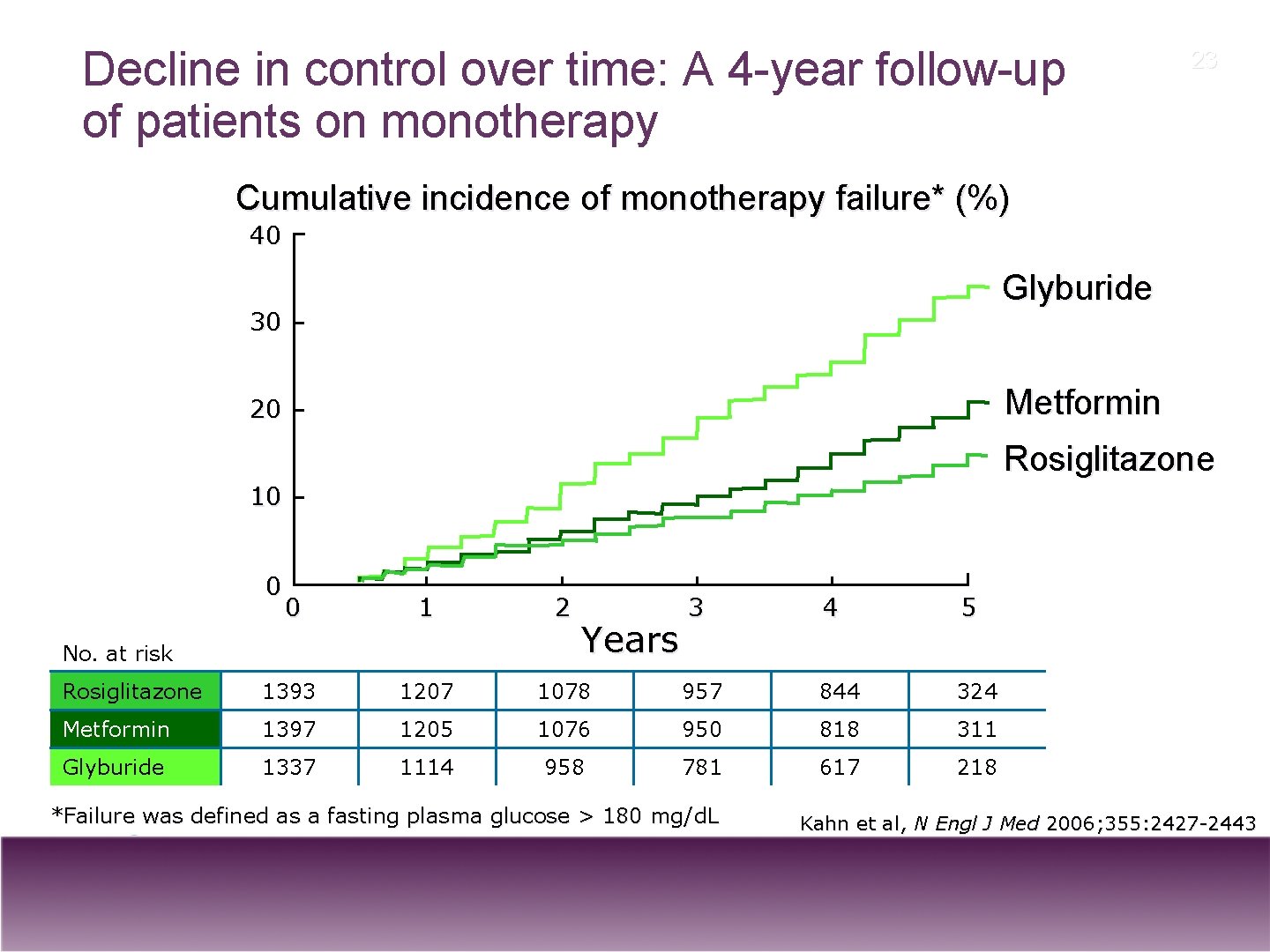
Decline in control over time: A 4 -year follow-up of patients on monotherapy 23 Cumulative incidence of monotherapy failure* (%) 40 Glyburide 30 Metformin 20 Rosiglitazone 10 0 0 1 2 3 4 5 Rosiglitazone 1393 1207 1078 957 844 324 Metformin 1397 1205 1076 950 818 311 Glyburide 1337 1114 958 781 617 218 No. at risk Years *Failure was defined as a fasting plasma glucose > 180 mg/ d. L Kahn et al, N Engl J Med 2006; 355: 2427 -2443

Non-compliance rates with antihyperglycemic medication, based on a patient survey 24 n Beliefs about Medication Questionnaire (BMQ) for antihyperglycemic medication: n Medication under use n Perceived necessity and harmfulness n Among 803 patients: n 46. 6% of participants reported non-compliance with antihyperglycemic medication n Many of these patients attributed this underuse to cost or concerns about medication harmfulness Aikens and Piette, Diabetes Care 2009; 32: 19 -24
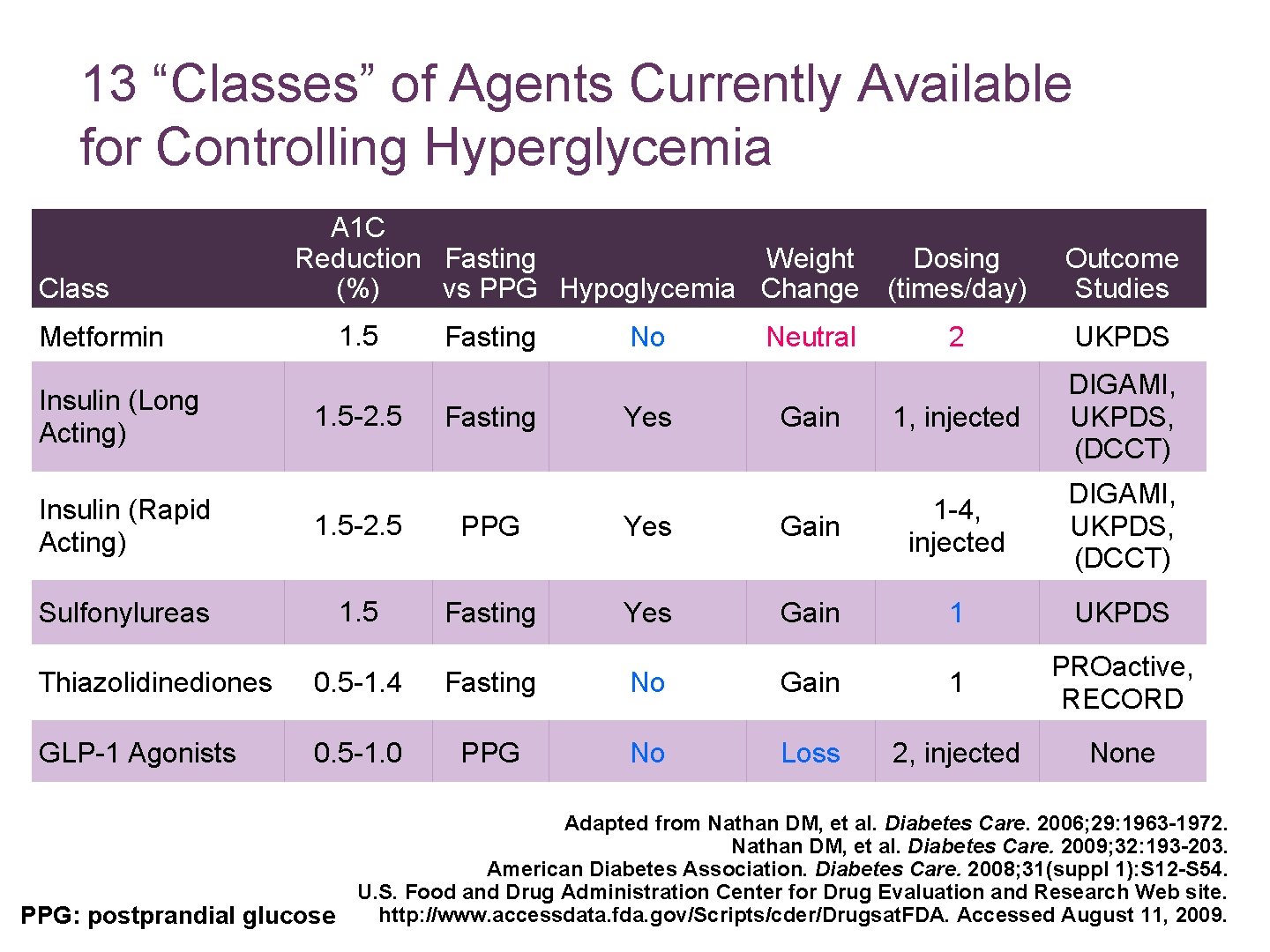
13 “Classes” of Agents Currently Available for Controlling Hyperglycemia Class A 1 C Reduction Fasting Weight Dosing (%) vs PPG Hypoglycemia Change (times/day) 1. 5 Metformin Insulin (Long Acting) 1. 5 -2. 5 Fasting No Yes Neutral Gain Outcome Studies 2 UKPDS 1, injected DIGAMI, UKPDS, (DCCT) Insulin (Rapid Acting) 1. 5 -2. 5 PPG Yes Gain 1 -4, injected Sulfonylureas 1. 5 Fasting Yes Gain 1 UKPDS Thiazolidinediones 0. 5 -1. 4 Fasting No Gain 1 PROactive, RECORD GLP-1 Agonists 0. 5 -1. 0 PPG No Loss 2, injected None PPG: postprandial glucose Adapted from Nathan DM, et al. Diabetes Care. 2006; 29: 1963 -1972. Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203. American Diabetes Association. Diabetes Care. 2008; 31(suppl 1): S 12 -S 54. U. S. Food and Drug Administration Center for Drug Evaluation and Research Web site. http: //www. accessdata. fda. gov/Scripts/cder/Drugsat. FDA. Accessed August 11, 2009.

Adverse events from current oral agents for type 2 diabetes 26 n 216 controlled trials and cohort studies and 2 systematic reviews that addressed benefits and harms of oral diabetes drugs were reviewed n Commonly reported adverse events included: n Increases in body weight by 1 to 5 kg (except with metformin) n Hypoglycemia with sulfonylureas (minor 17. 5%, major 2. 5% with glyburide in 10 -year UKPDS follow-up) n Gastrointestinal problems with metformin (reported incidence 2% – 63%) n Congestive heart failure (reported incidence 0. 8% to 3. 6% with thiazolidinediones) n Peripheral edema (reported incidence 0% to 26% with thiazolidinediones) Bolen et al, Ann Intern Med 2007; 147: 386 -399
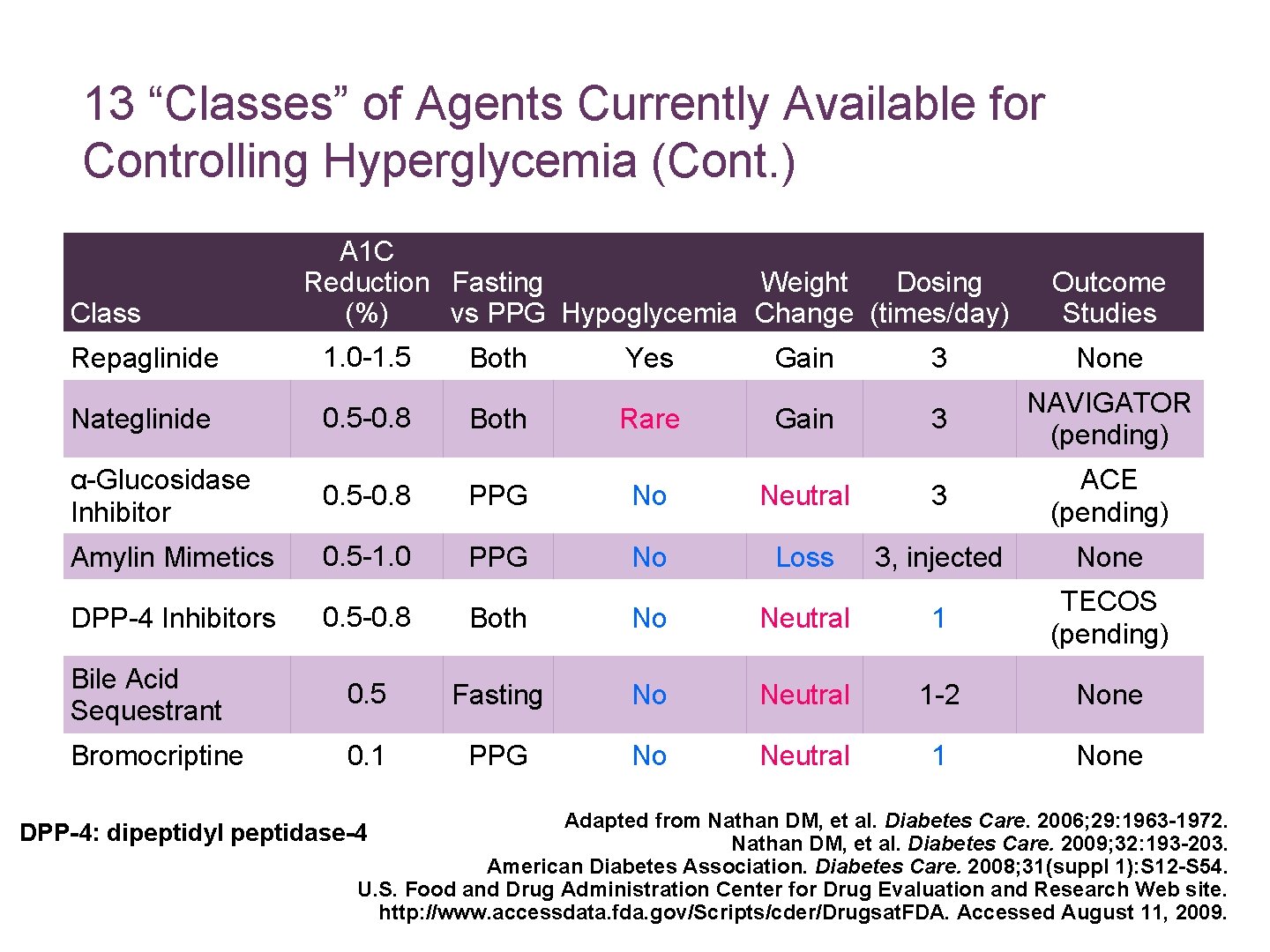
13 “Classes” of Agents Currently Available for Controlling Hyperglycemia (Cont. ) Class Repaglinide Nateglinide A 1 C Reduction Fasting Weight Dosing (%) vs PPG Hypoglycemia Change (times/day) 1. 0 -1. 5 0. 5 -0. 8 Both Yes Rare Gain Outcome Studies 3 None 3 NAVIGATOR (pending) α-Glucosidase Inhibitor 0. 5 -0. 8 PPG No Neutral 3 ACE (pending) Amylin Mimetics 0. 5 -1. 0 PPG No Loss 3, injected None 0. 5 -0. 8 Both No Neutral 1 TECOS (pending) Bile Acid Sequestrant 0. 5 Fasting No Neutral 1 -2 None Bromocriptine 0. 1 PPG No Neutral 1 None DPP-4 Inhibitors Adapted from Nathan DM, et al. Diabetes Care. 2006; 29: 1963 -1972. DPP-4: dipeptidyl peptidase-4 Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203. American Diabetes Association. Diabetes Care. 2008; 31(suppl 1): S 12 -S 54. U. S. Food and Drug Administration Center for Drug Evaluation and Research Web site. http: //www. accessdata. fda. gov/Scripts/cder/Drugsat. FDA. Accessed August 11, 2009.
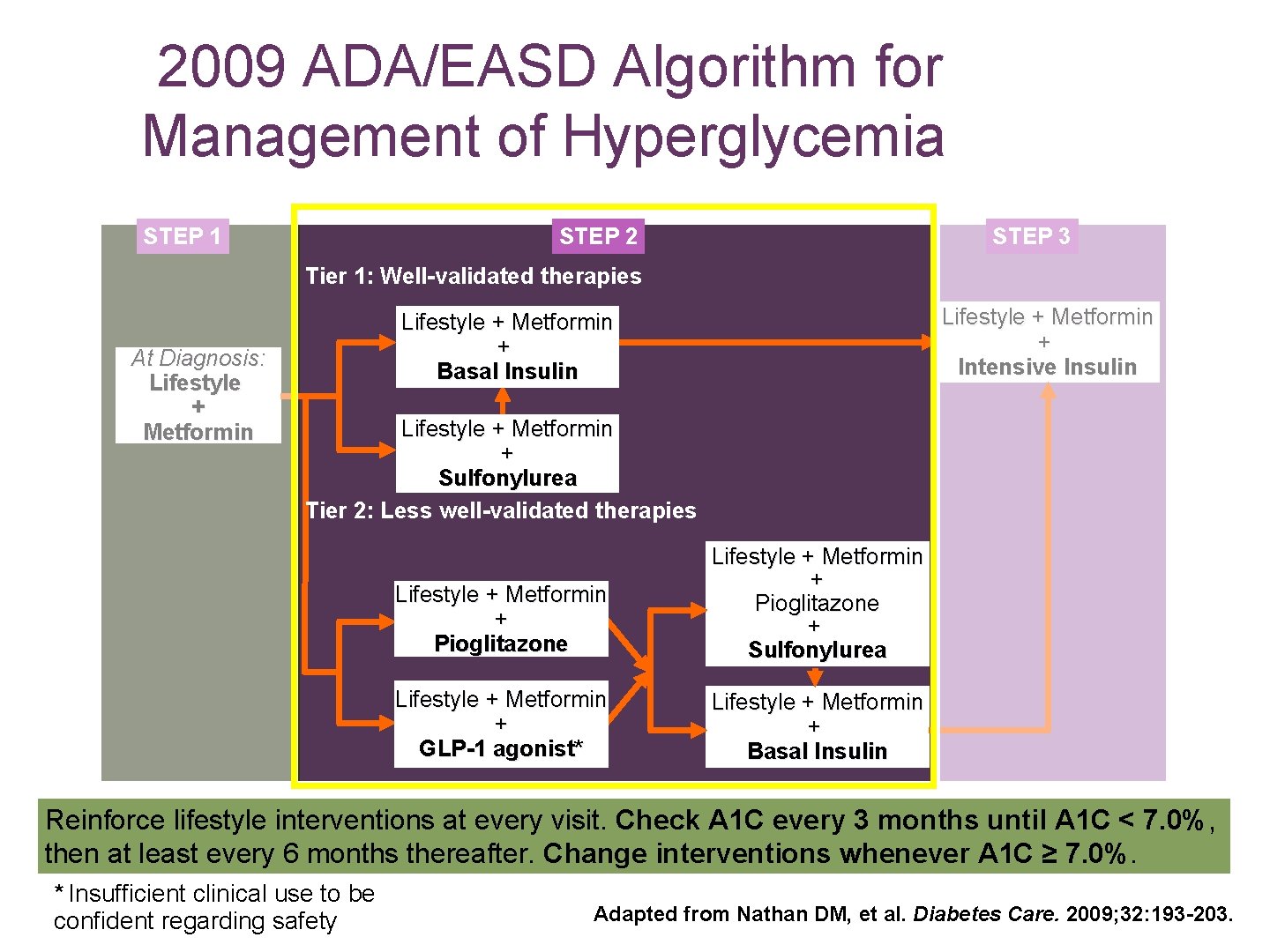
2009 ADA/EASD Algorithm for Management of Hyperglycemia STEP 1 STEP 2 STEP 3 Tier 1: Well-validated therapies At Diagnosis: Lifestyle + Metformin + Intensive Insulin Lifestyle + Metformin + Basal Insulin Lifestyle + Metformin + Sulfonylurea Tier 2: Less well-validated therapies Lifestyle + Metformin + Pioglitazone + Sulfonylurea Lifestyle + Metformin + GLP-1 agonist* Lifestyle + Metformin + Basal Insulin Reinforce lifestyle interventions at every visit. Check A 1 C every 3 months until A 1 C < 7. 0%, then at least every 6 months thereafter. Change interventions whenever A 1 C ≥ 7. 0%. * Insufficient clinical use to be confident regarding safety Adapted from Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203.

ADA/EASD Algorithm: Recent Revisions Similarities with previous algorithm n Test A 1 C at least every 3 months: n n Differences from previous algorithm n Intensify the glycemic regimen whenever A 1 C is ≥ 7. 0% In patients newly diagnosed with type 2 diabetes: n n Institute lifestyle interventions that promote weight loss and increase physical activity Metformin is the initial recommended glycemic agent in newly diagnosed patients n Recommend insulin to patients whose A 1 C is not controlled by oral agents n Tier 1: basal insulin or sulfonylurea n Tier 2: pioglitazone or exenatide n The sulfonylureas glibenclamide (glyburide) and chlorpropamide are no longer recommended n Rosiglitazone is no longer recommended, but pioglitazone is still recommended n The incretin mimetic exenatide is now a recommended agent Encourage smoking cessation n Glucose-lowering agents now fall into 2 tiers that are recommended in different circumstances: Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203.

Selecting an Individualized Glycemic Regimen From Tier 1 or Tier 2 Tier 1 Agents Tier 2 Agents n When metformin is not tolerated or is contraindicated: n Glomerular filtration rate < 30 m. L/min n When a large reduction in A 1 C is needed: n A 1 C n When reduction ≥ 1. 5% medication cost is a hardship n When avoidance of hypoglycemia is important: n Certain occupations (eg, drivers, heavy equipment operators, using power tools, public safety jobs) n Certain populations (eg, history of hypoglycemia, alcohol use, elderly patients, renal insufficiency) n When promotion of weight loss is important Adapted from Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203. Lassmann-Vague V. Diabetes Metab. 2005; 31: 5 S 53 -5 S 57.
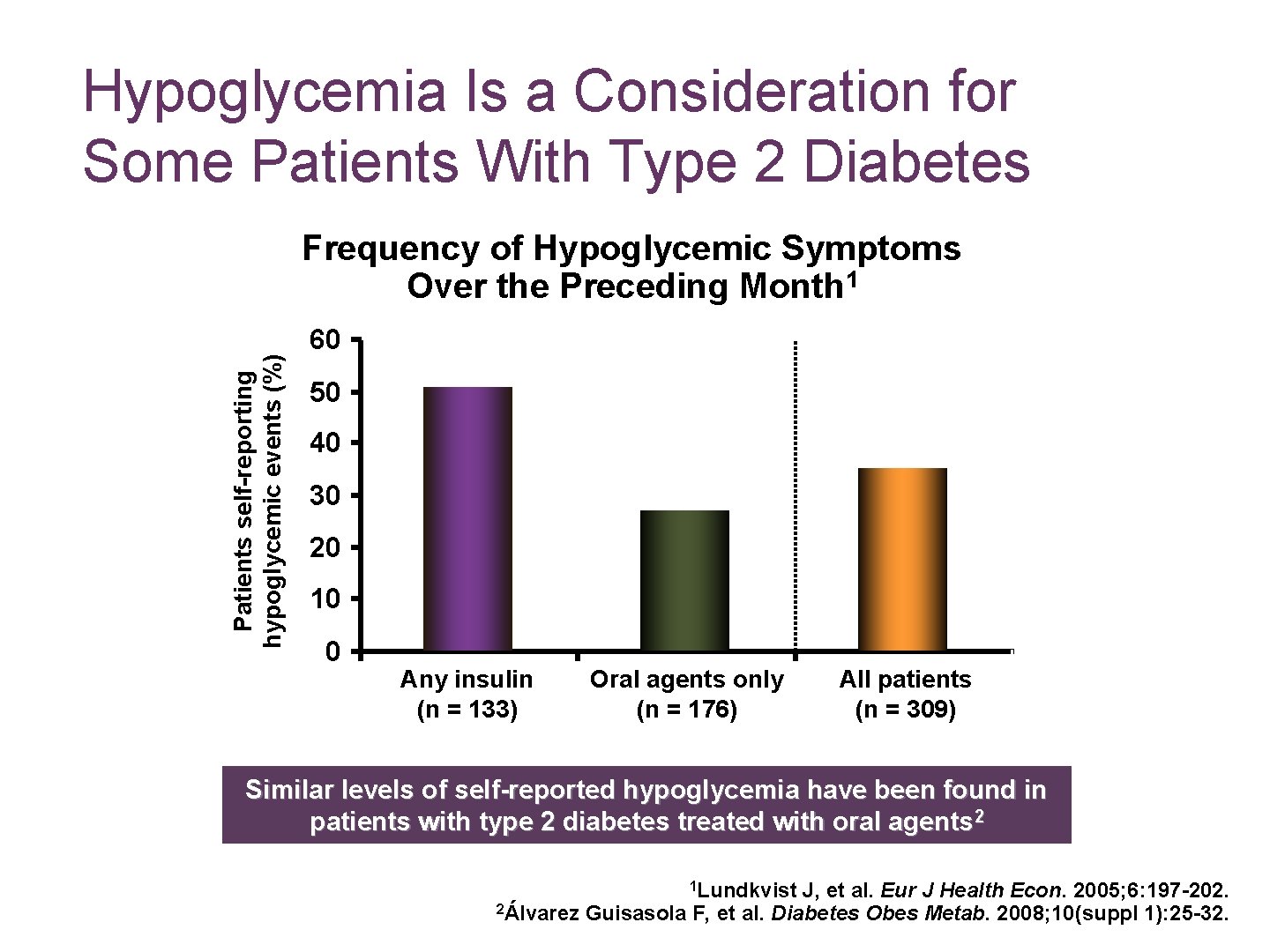
Hypoglycemia Is a Consideration for Some Patients With Type 2 Diabetes Patients self-reporting hypoglycemic events (%) Frequency of Hypoglycemic Symptoms Over the Preceding Month 1 60 50 40 30 20 10 0 Any insulin (n = 133) Oral agents only (n = 176) All patients (n = 309) Similar levels of self-reported hypoglycemia have been found in patients with type 2 diabetes treated with oral agents 2 1 Lundkvist J, et al. Eur J Health Econ. 2005; 6: 197 -202. 2Álvarez Guisasola F, et al. Diabetes Obes Metab. 2008; 10(suppl 1): 25 -32.

Help Patients Gain Confidence in Controlling the Risk of Hypoglycemia n n n Patients can fight fear with knowledge: n Encourage patient questions about hypoglycemia n Suggest enrollment in a diabetes education program n Suggest subscribing to a popular diabetes magazine Patients should not simply tolerate frequent low blood sugars: n Avoid overreactions that could lead to chronically high blood sugars n Avoid an overly restrictive lifestyle; discuss changes in the medication schedule or changing medications n Consider meal plans, daily activity, alcohol consumption, and overall diabetes management Help patients harness the fear: n It is poorly controlled diabetes that typically causes serious problems, not diabetes itself Behavioral Diabetes Institute Web site. http: //www. behavioraldiabetes. org/downloads/ brochure-Diabetes-10 -Things-To-Know. pdf#page=1&zoom=100&view=Fit. Accessed August 11, 2009.

Intensifying the Regimen: Guidelines for Combining Glycemic Agents ADA Recommendations for Medication Combinations n If glycemic control needs intensification, agents that have different mechanisms of action produce greater synergy of effect n Also consider combinations of agents that have complementary extraglycemic effects n The “glinides”, α-glucosidase inhibitors, amylin agonists, DPP-4 inhibitors, bile acid sequestrants, and bromocriptine are not included in the 2 tiers of preferred agents in the ADA/EASD algorithm. However, they may be appropriate choices in selected patients. Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203.
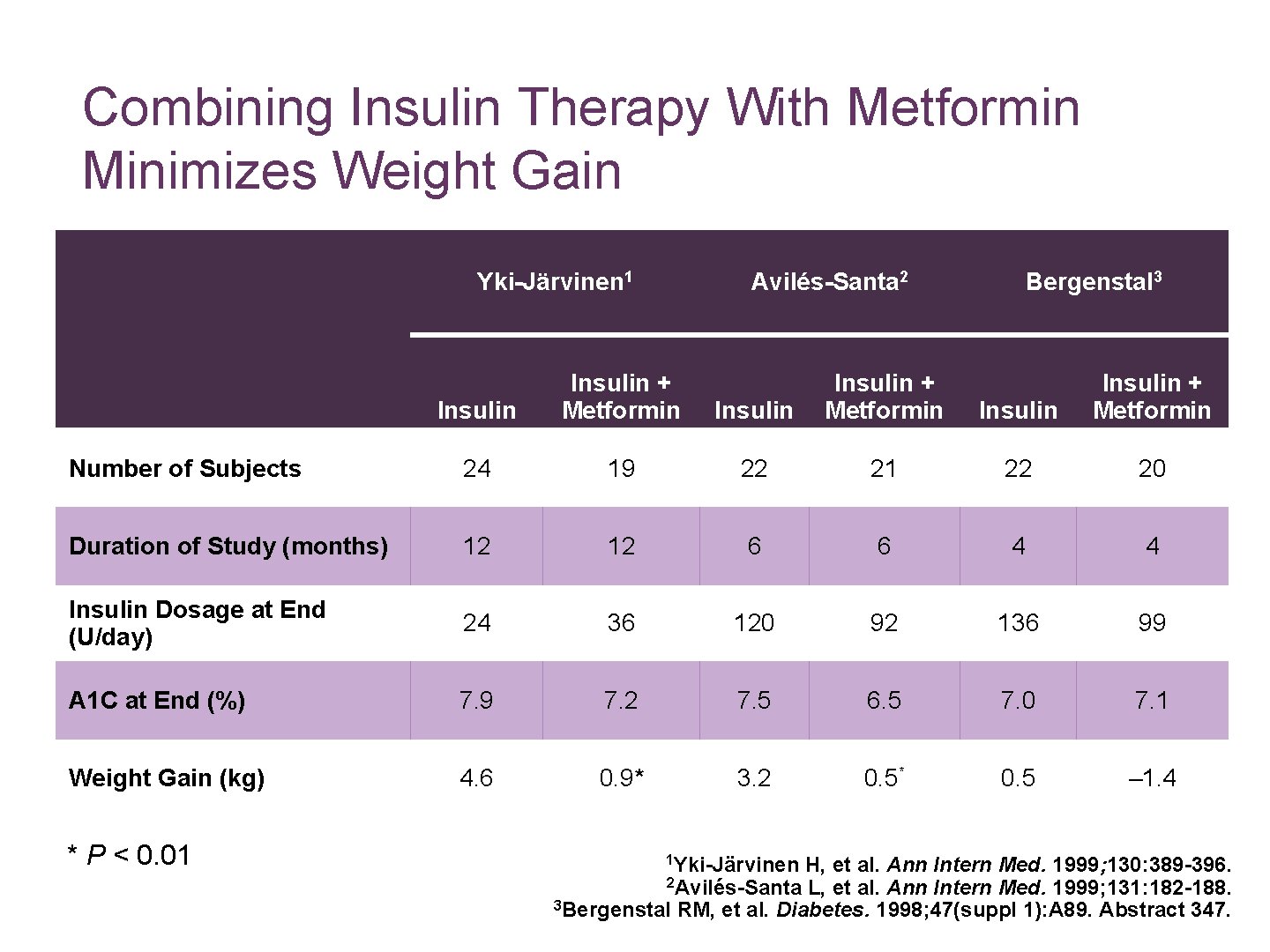
Combining Insulin Therapy With Metformin Minimizes Weight Gain Yki-Järvinen 1 Avilés-Santa 2 Insulin + Metformin Number of Subjects 24 Duration of Study (months) Bergenstal 3 Insulin + Metformin 19 22 21 22 20 12 12 6 6 4 4 Insulin Dosage at End (U/day) 24 36 120 92 136 99 A 1 C at End (%) 7. 9 7. 2 7. 5 6. 5 7. 0 7. 1 Weight Gain (kg) 4. 6 0. 9* 3. 2 0. 5* 0. 5 – 1. 4 * P < 0. 01 1 Yki-Järvinen H, et al. Ann Intern Med. 1999; 130: 389 -396. 2 Avilés-Santa L, et al. Ann Intern Med. 1999; 131: 182 -188. 3 Bergenstal RM, et al. Diabetes. 1998; 47(suppl 1): A 89. Abstract 347.
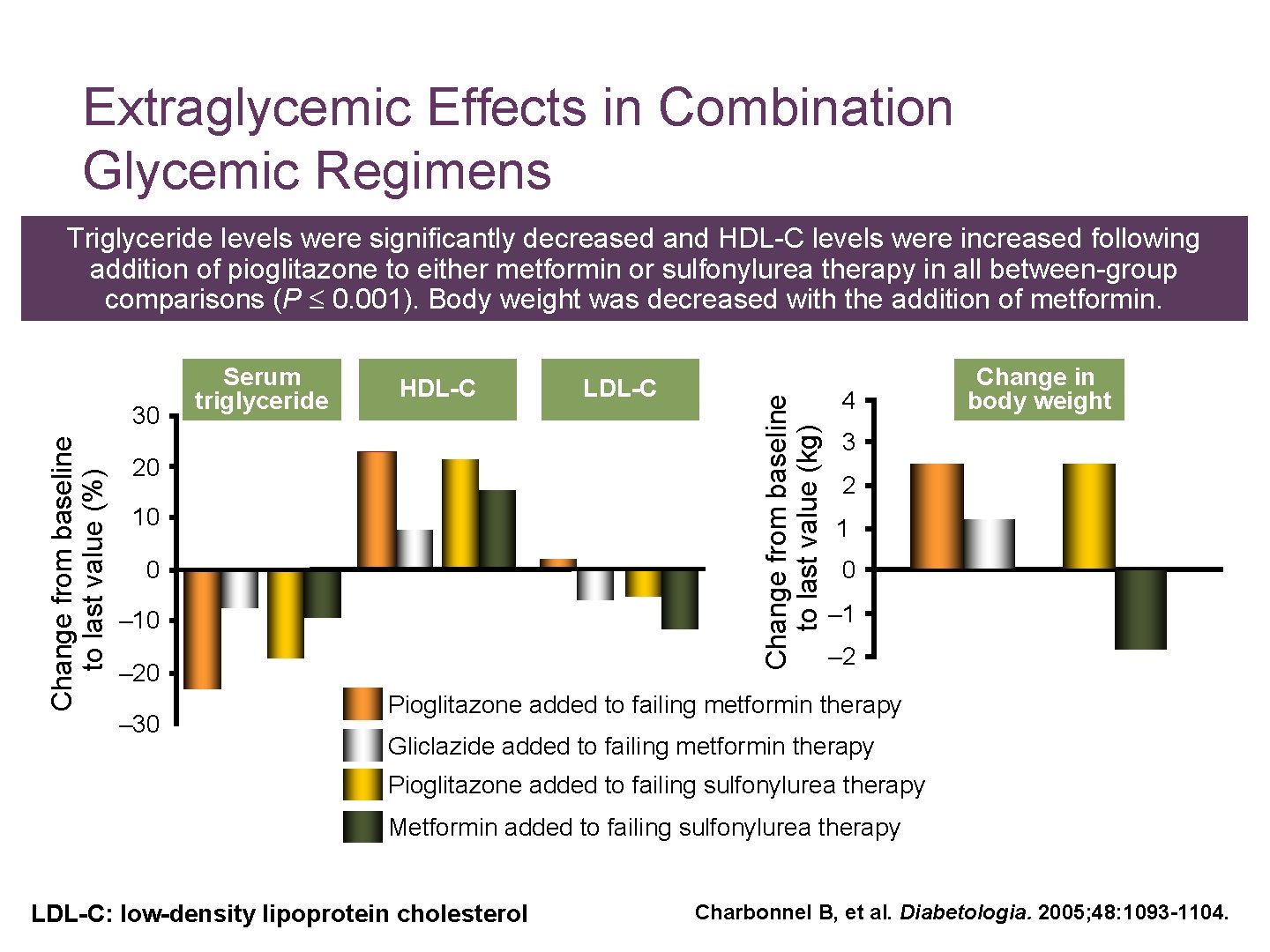
Extraglycemic Effects in Combination Glycemic Regimens Change from baseline to last value (%) 30 Serum triglyceride HDL-C 20 10 0 – 10 – 20 – 30 LDL-C Change from baseline to last value (kg) Triglyceride levels were significantly decreased and HDL-C levels were increased following addition of pioglitazone to either metformin or sulfonylurea therapy in all between-group comparisons (P 0. 001). Body weight was decreased with the addition of metformin. 4 Change in body weight 3 2 1 0 – 1 – 2 Pioglitazone added to failing metformin therapy Gliclazide added to failing metformin therapy Pioglitazone added to failing sulfonylurea therapy Metformin added to failing sulfonylurea therapy LDL-C: low-density lipoprotein cholesterol Charbonnel B, et al. Diabetologia. 2005; 48: 1093 -1104.

Weight Change Following Combination Therapies With Exenatide added to metformin (30 weeks) Exenatide added to sulfonylurea and metformin (30 weeks) Exenatide added to glitazone ± metformin (16 weeks) A 1 C reduction in each trial ≈ 1% versus placebo * (n = 336) * P ≤ 0. 001 vs placebo † P < 0. 05 vs placebo ‡ P ≤ 0. 01 vs placebo (n = 377) (n = 733) (n = 233) De. Fronzo R, et al. Diabetes Care. 2005; 28: 1092 -1100. Buse J, et al. Diabetes Care. 2004; 27: 2628 -2635. Kendall D, et al. Diabetes Care. 2005; 28: 1083 -1091. Zinman B, et al. Ann Intern Med. 2007; 146: 477 -485.
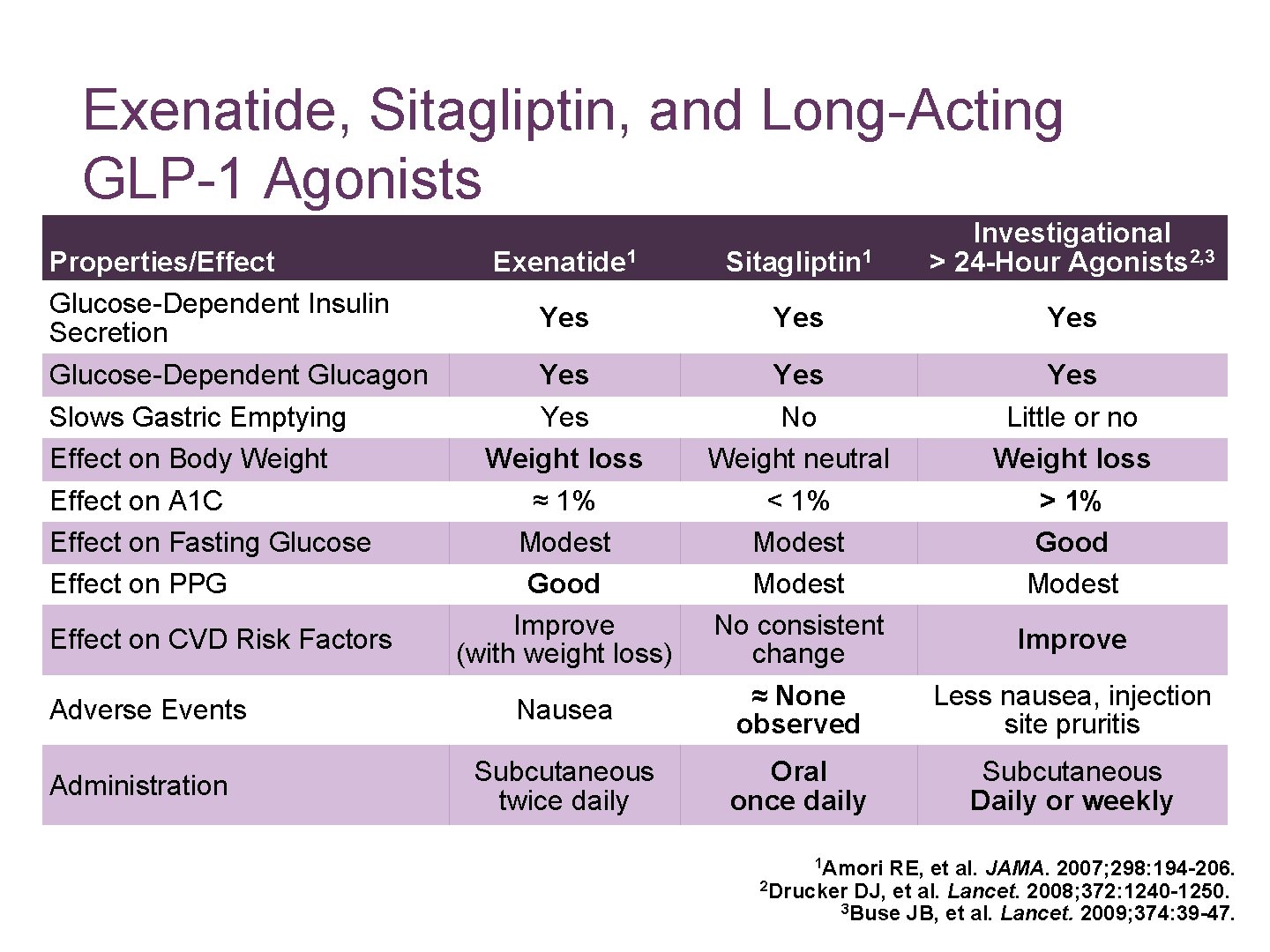
Exenatide, Sitagliptin, and Long-Acting GLP-1 Agonists Properties/Effect Glucose-Dependent Insulin Secretion Glucose-Dependent Glucagon Slows Gastric Emptying Effect on Body Weight Effect on A 1 C Effect on Fasting Glucose Effect on PPG Effect on CVD Risk Factors Adverse Events Administration Exenatide 1 Sitagliptin 1 Investigational > 24 -Hour Agonists 2, 3 Yes Yes Weight loss ≈ 1% Modest Good Improve (with weight loss) No Weight neutral < 1% Modest No consistent change ≈ None observed Little or no Weight loss > 1% Good Modest Less nausea, injection site pruritis Oral once daily Subcutaneous Daily or weekly Nausea Subcutaneous twice daily 1 Amori Improve RE, et al. JAMA. 2007; 298: 194 -206. 2 Drucker DJ, et al. Lancet. 2008; 372: 1240 -1250. 3 Buse JB, et al. Lancet. 2009; 374: 39 -47.

+ ® CYCLOSET (quick release bromocriptine): Proposed mechanism of action Morning administration (within 2 hours of waking) of CYCLOSET Corrects Low dopaminergic tone in hypothalamus in early morning in diabetes Restoration of morning peak in dopaminergic activity (via D 2 receptormediated activity) ÇSympathetic tone ÇHPA axis tone Hepatic gluconeogenesis FFA and TG Insulin resistance Inflammation/hypercoagulation Impaired glucose metabolism, hyperglycemia and insulin resistance Adverse cardiovascular pathology È Sympathetic tone È HPA axis tone Hepatic gluconeogenesis FFA and TG Insulin resistance Inflammation/hypercoagulation Decreased postprandial glucose levels Reduction in insulin resistance Day-long reduction in plasma glucose, TGs and FFAs Fonseca. Use of Dopamine agonists in Type-2 -Diabetes. Oxford American Pocket Cards. OUP, 2010 Cincotta. Hypothalamic role in Insulin Resistance and insulin Resistance Syndrome. Frontiers in Animal Diabetes Research Series. Taylor and Francis, Eds Hansen, B Shafrir, E London, pp 271 -312, 2002 38

Case Study 2 n James (aged 63 years) n n Diagnosis: type 2 diabetes (September 2007); A 1 C 8. 5% Initial regimen: metformin 850 mg BID; glimepiride 1 mg once daily n March 2008: A 1 C 7. 0% n October 2008: A 1 C 6. 9% n May 2009: A 1 C 8. 0%; increase glimepiride (2 mg) n August 2008: A 1 C 7. 6%; James now complains of 3 episodes of hypoglycemia in the past month Since concerns about hypoglycemia may be causing inconsistency in his medication use and eating patterns, what approach would you take to discuss regimen adherence with James? What therapeutic options would you offer him at this point?

+ Case Study 3: Introduction Lyle is a 68 -year-old male diagnosed with type 2 diabetes 1 year ago. Three months ago, Lyle increased his metformin dose to 500 mg BID (from once daily). He now complains of gastrointestinal discomfort. kg/m 2 n BMI: 29. 7 n A 1 C 7. 4% n Hypertension is well-controlled (lisinopril/hydrochlorothiazide) n Simvastatin (10 mg once daily) n Metformin (500 mg BID) Lyle has some concerns about the fact that he is treating his diabetes in the context of elevated CV risk. How would you address his concerns about CV risk?
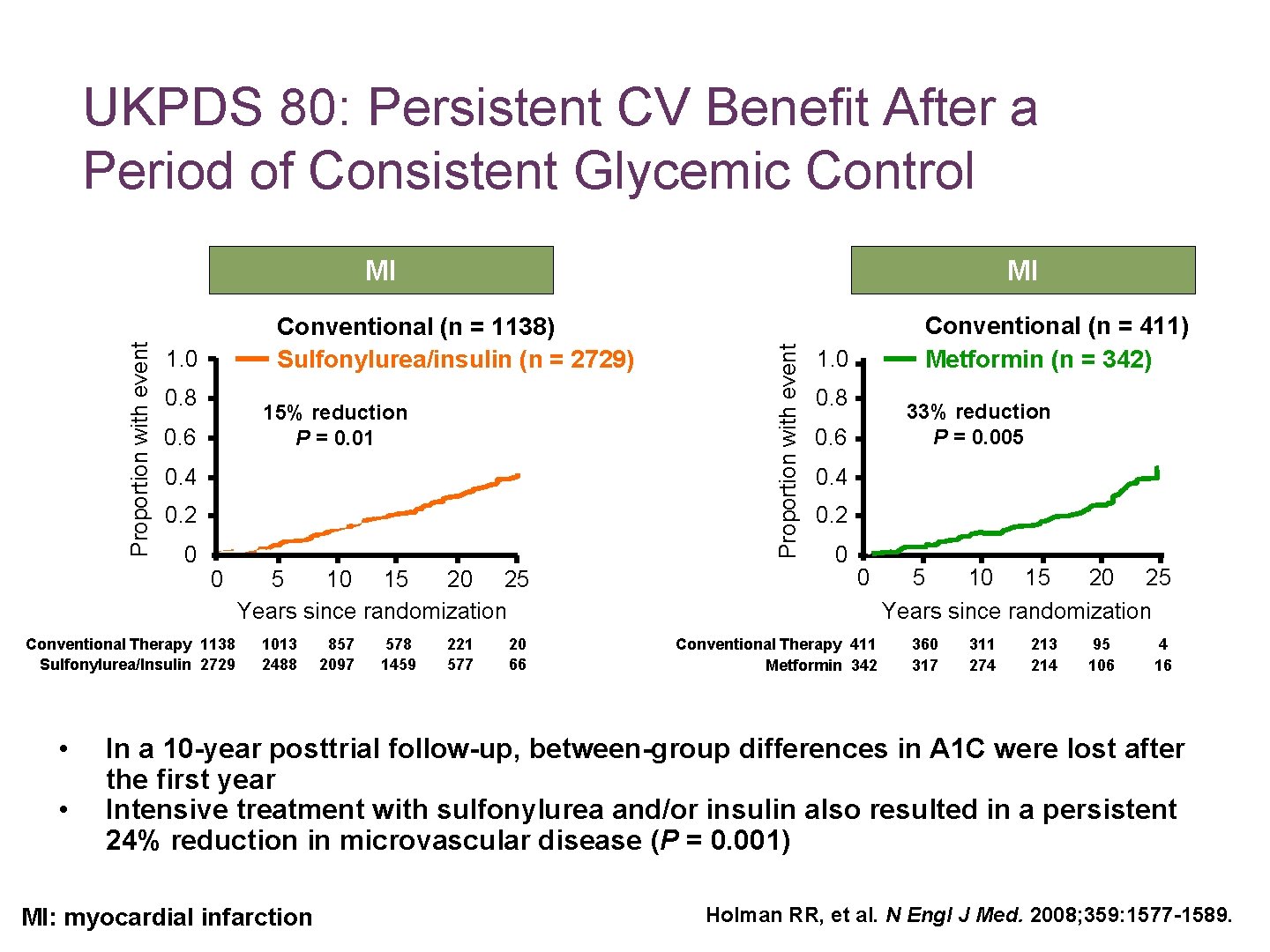
UKPDS 80: Persistent CV Benefit After a Period of Consistent Glycemic Control Conventional (n = 1138) Sulfonylurea/insulin (n = 2729) 1. 0 0. 8 15% reduction P = 0. 01 0. 6 0. 4 0. 2 0 0 Conventional Therapy 1138 Sulfonylurea/Insulin 2729 • • MI 5 10 15 20 25 Years since randomization 1013 2488 857 2097 578 1459 221 577 20 66 Proportion with event MI Conventional (n = 411) Metformin (n = 342) 1. 0 0. 8 33% reduction P = 0. 005 0. 6 0. 4 0. 2 0 0 Conventional Therapy 411 Metformin 342 5 10 15 20 25 Years since randomization 360 317 311 274 213 214 95 106 4 16 In a 10 -year posttrial follow-up, between-group differences in A 1 C were lost after the first year Intensive treatment with sulfonylurea and/or insulin also resulted in a persistent 24% reduction in microvascular disease (P = 0. 001) MI: myocardial infarction Holman RR, et al. N Engl J Med. 2008; 359: 1577 -1589.
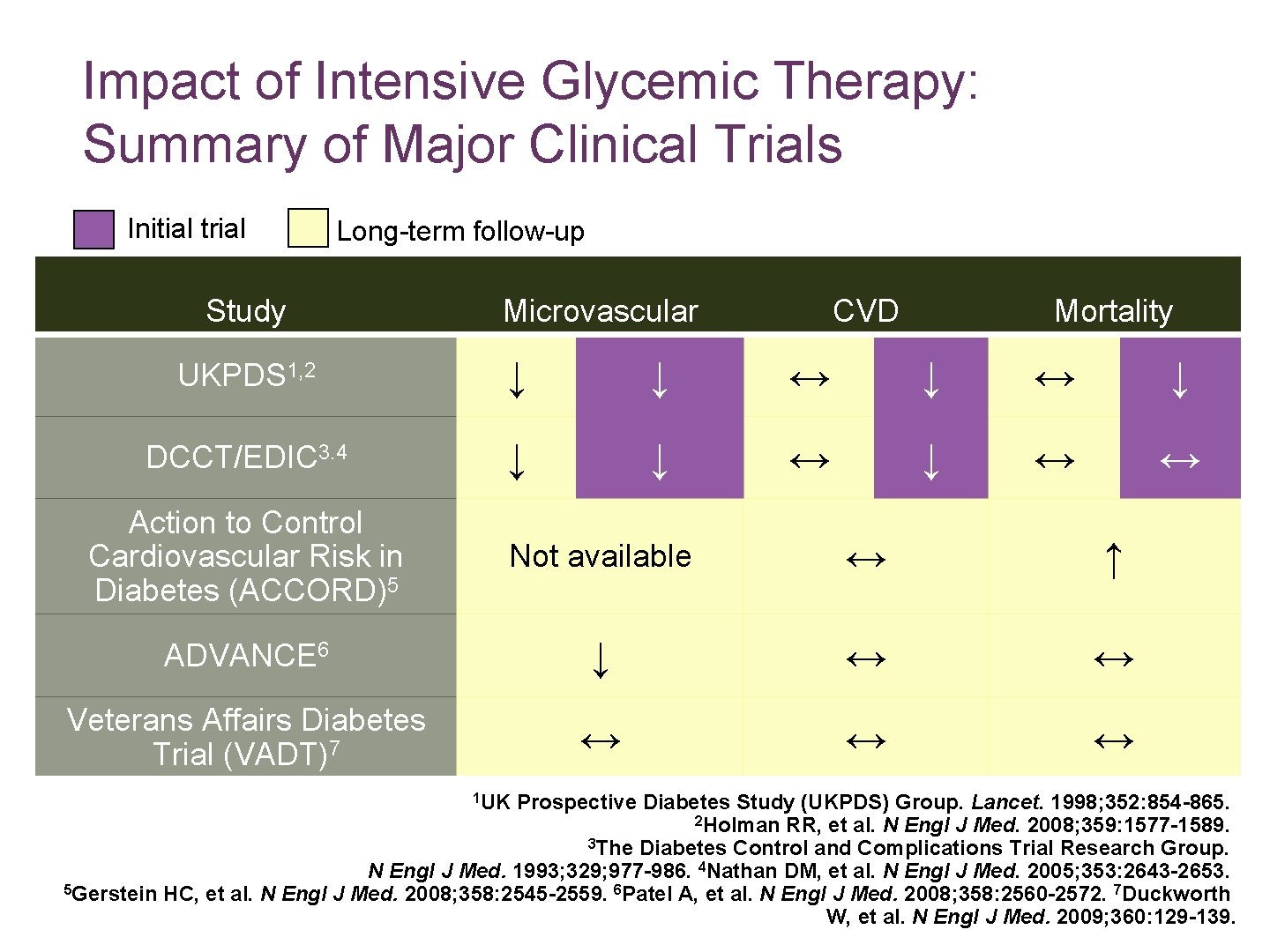
Impact of Intensive Glycemic Therapy: Summary of Major Clinical Trials Initial trial Long-term follow-up Study Microvascular CVD Mortality UKPDS 1, 2 ↓ ↓ ↔ ↓ DCCT/EDIC 3. 4 ↓ ↓ ↔ ↔ Action to Control Cardiovascular Risk in Diabetes (ACCORD)5 Not available ↔ ↑ ADVANCE 6 ↓ ↔ ↔ Veterans Affairs Diabetes Trial (VADT)7 ↔ ↔ ↔ 1 UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998; 352: 854 -865. 2 Holman RR, et al. N Engl J Med. 2008; 359: 1577 -1589. 3 The Diabetes Control and Complications Trial Research Group. N Engl J Med. 1993; 329; 977 -986. 4 Nathan DM, et al. N Engl J Med. 2005; 353: 2643 -2653. 5 Gerstein HC, et al. N Engl J Med. 2008; 358: 2545 -2559. 6 Patel A, et al. N Engl J Med. 2008; 358: 2560 -2572. 7 Duckworth W, et al. N Engl J Med. 2009; 360: 129 -139.
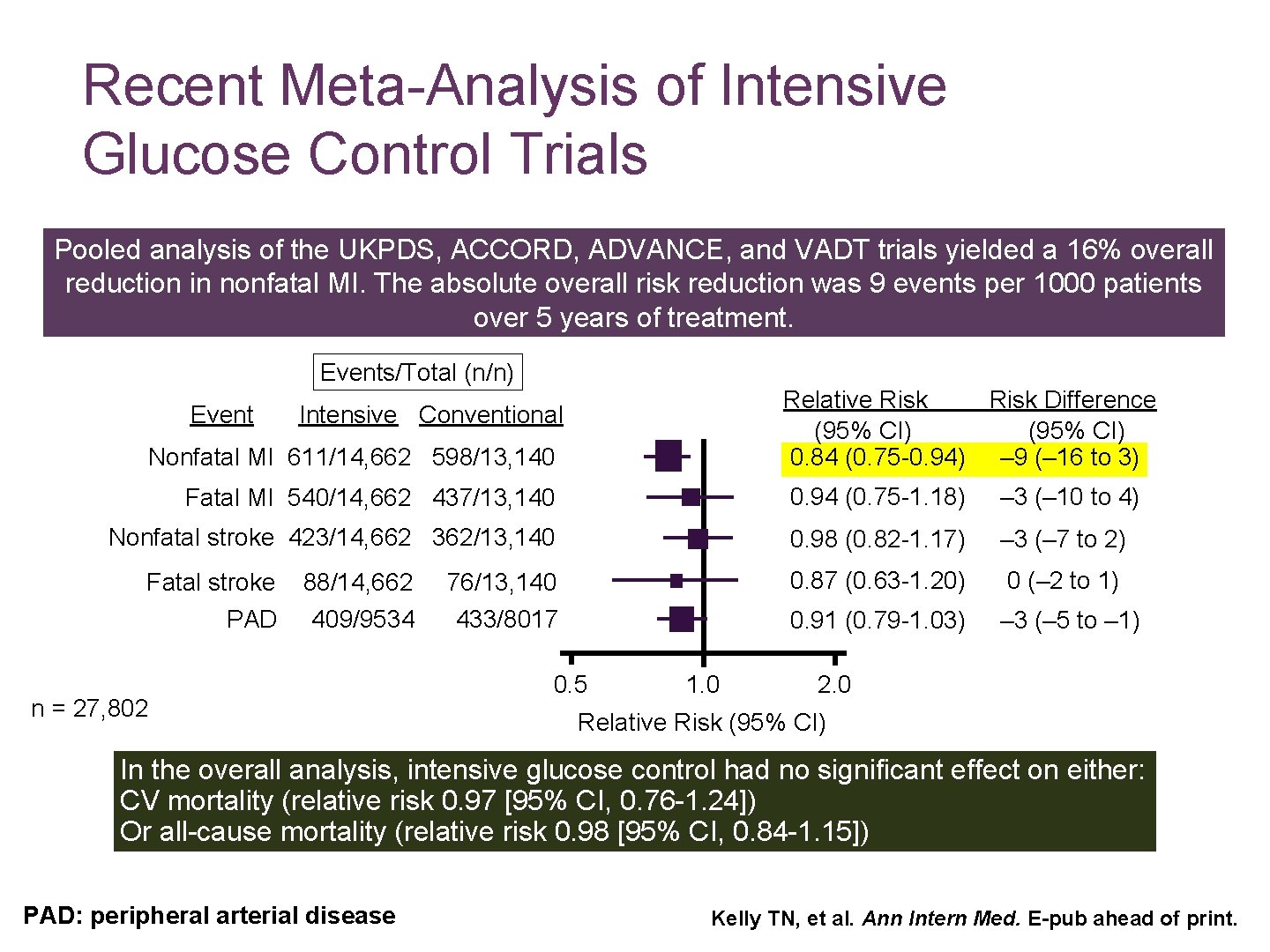
Recent Meta-Analysis of Intensive Glucose Control Trials Pooled analysis of the UKPDS, ACCORD, ADVANCE, and VADT trials yielded a 16% overall reduction in nonfatal MI. The absolute overall risk reduction was 9 events per 1000 patients over 5 years of treatment. Events/Total (n/n) Event Relative Risk Difference (95% CI) 0. 84 (0. 75 -0. 94) – 9 (– 16 to 3) Intensive Conventional Nonfatal MI 611/14, 662 598/13, 140 Fatal MI 540/14, 662 437/13, 140 0. 94 (0. 75 -1. 18) – 3 (– 10 to 4) Nonfatal stroke 423/14, 662 362/13, 140 0. 98 (0. 82 -1. 17) – 3 (– 7 to 2) 0. 87 (0. 63 -1. 20) 0 (– 2 to 1) 0. 91 (0. 79 -1. 03) – 3 (– 5 to – 1) Fatal stroke PAD 88/14, 662 409/9534 n = 27, 802 76/13, 140 433/8017 0. 5 1. 0 2. 0 Relative Risk (95% CI) In the overall analysis, intensive glucose control had no significant effect on either: CV mortality (relative risk 0. 97 [95% CI, 0. 76 -1. 24]) Or all-cause mortality (relative risk 0. 98 [95% CI, 0. 84 -1. 15]) PAD: peripheral arterial disease Kelly TN, et al. Ann Intern Med. E-pub ahead of print.

Implications of ACCORD, ADVANCE, and VADT for Microvascular Risk Microvascular disease: n Lowering A 1 C to ≤ 7. 0% reduces microvascular and neuropathic complications in type 2 diabetes n If achievable without causing significant hypoglycemia or other adverse events, even lower A 1 C goals may be suggested for selected individuals having: n Short duration of diabetes n Long life expectancy n No significant CVD Skyler JS, et al. Diabetes Care. 2009; 32: 187 -192.

Implications of ACCORD, ADVANCE, and VADT for Macrovascular Risk + Macrovascular disease: n Intensive glycemic control that exceeds an A 1 C goal of < 7. 0% yields no significant reduction in CVD outcomes compared to standard glycemic control n Lowering A 1 C to a goal of ≤ 7. 0% is a reasonable glycemic goal until more evidence becomes available n Long-term follow-up of the DCCT and UKPDS cohorts suggests that treating to an A 1 C goal below or near 7. 0% yields longterm reductions in the risk of macrovascular disease if it is instituted in the years soon after diagnosis of diabetes Skyler JS, et al. Diabetes Care. 2009; 32: 187 -192.
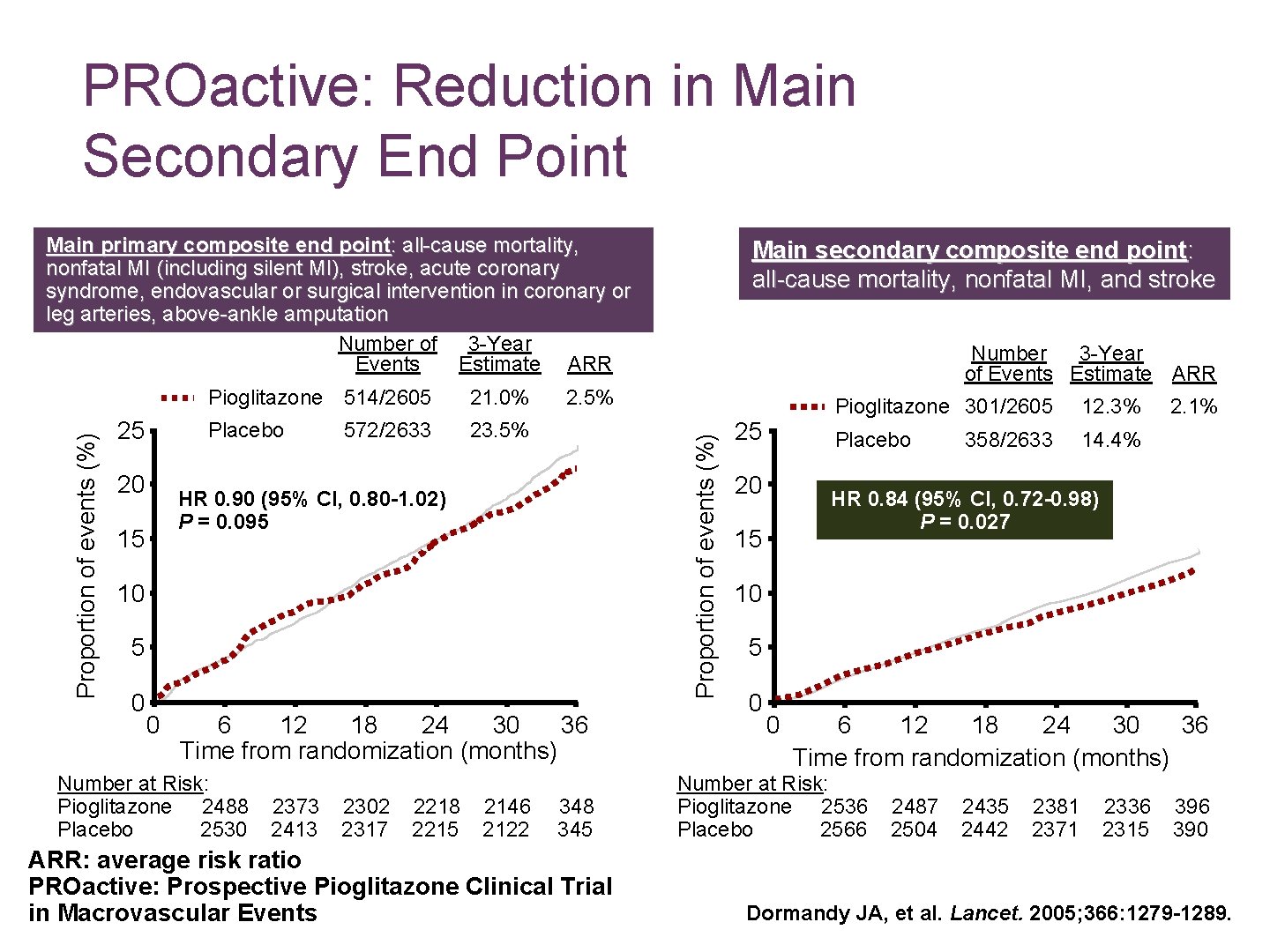
PROactive: Reduction in Main Secondary End Point 25 20 Pioglitazone 514/2605 21. 0% Placebo 23. 5% 572/2633 10 5 0 0 6 12 18 24 30 36 Time from randomization (months) Number at Risk: Pioglitazone 2488 Placebo 2530 2373 2413 2302 2317 2218 2215 2146 2122 Number 3 -Year of Events Estimate ARR 2. 5% HR 0. 90 (95% CI, 0. 80 -1. 02) P = 0. 095 15 Main secondary composite end point: all-cause mortality, nonfatal MI, and stroke Proportion of events (%) Main primary composite end point: all-cause mortality, nonfatal MI (including silent MI), stroke, acute coronary syndrome, endovascular or surgical intervention in coronary or leg arteries, above-ankle amputation Number of 3 -Year Events Estimate ARR 348 345 ARR: average risk ratio PROactive: Prospective Pioglitazone Clinical Trial in Macrovascular Events 25 20 Pioglitazone 301/2605 12. 3% Placebo 14. 4% 358/2633 2. 1% HR 0. 84 (95% CI, 0. 72 -0. 98) P = 0. 027 15 10 5 0 0 6 12 18 24 30 36 Time from randomization (months) Number at Risk: Pioglitazone 2536 Placebo 2566 2487 2504 2435 2442 2381 2371 2336 2315 396 390 Dormandy JA, et al. Lancet. 2005; 366: 1279 -1289.
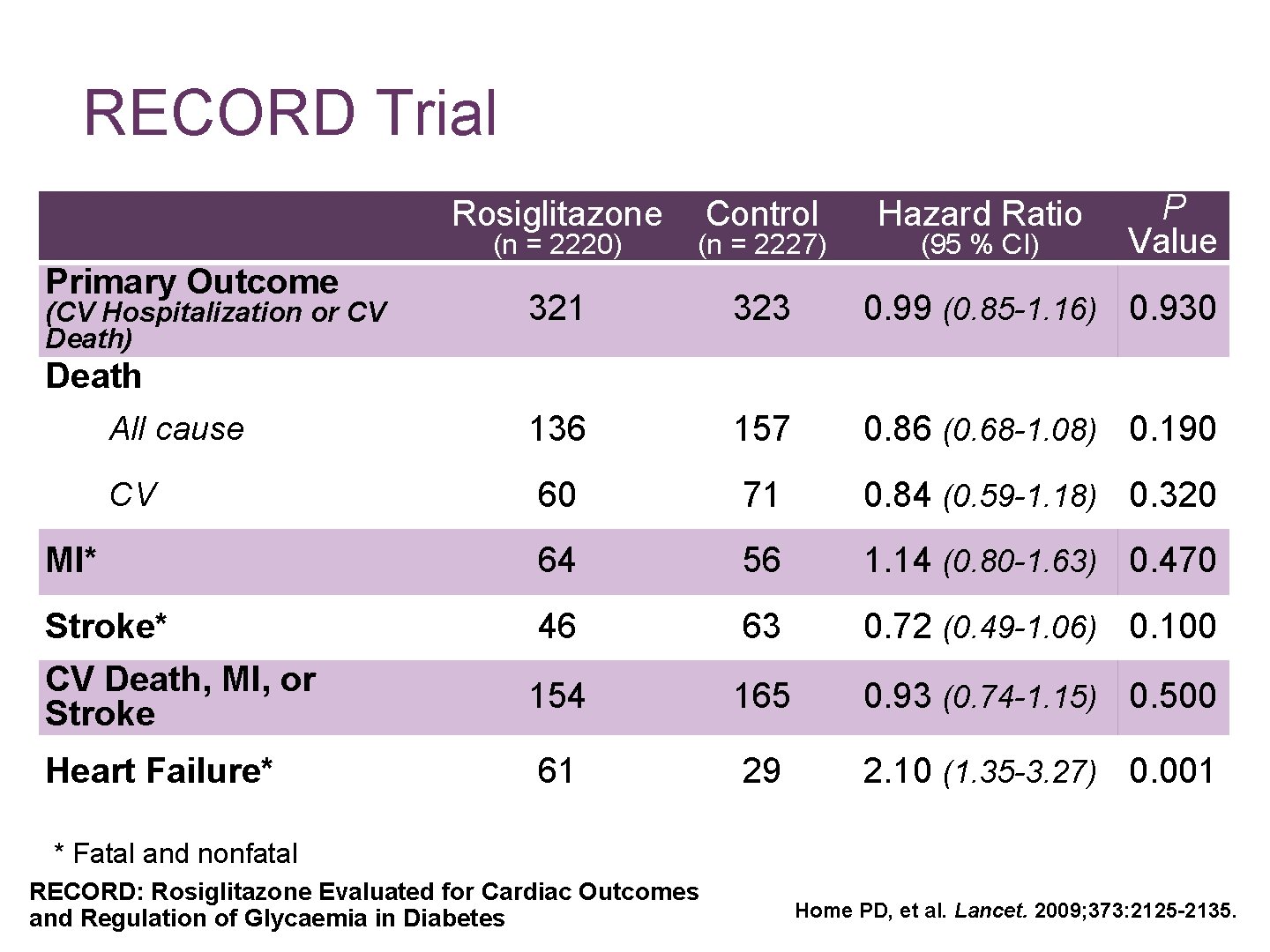
RECORD Trial Rosiglitazone Control Hazard Ratio (n = 2220) (n = 2227) 321 323 0. 99 (0. 85 -1. 16) 0. 930 All cause 136 157 0. 86 (0. 68 -1. 08) 0. 190 CV 60 71 0. 84 (0. 59 -1. 18) 0. 320 MI* 64 56 1. 14 (0. 80 -1. 63) 0. 470 Stroke* 46 63 0. 72 (0. 49 -1. 06) 0. 100 CV Death, MI, or Stroke 154 165 0. 93 (0. 74 -1. 15) 0. 500 Heart Failure* 61 29 2. 10 (1. 35 -3. 27) 0. 001 Primary Outcome (CV Hospitalization or CV Death) (95 % CI) P Value Death * Fatal and nonfatal RECORD: Rosiglitazone Evaluated for Cardiac Outcomes and Regulation of Glycaemia in Diabetes Home PD, et al. Lancet. 2009; 373: 2125 -2135.
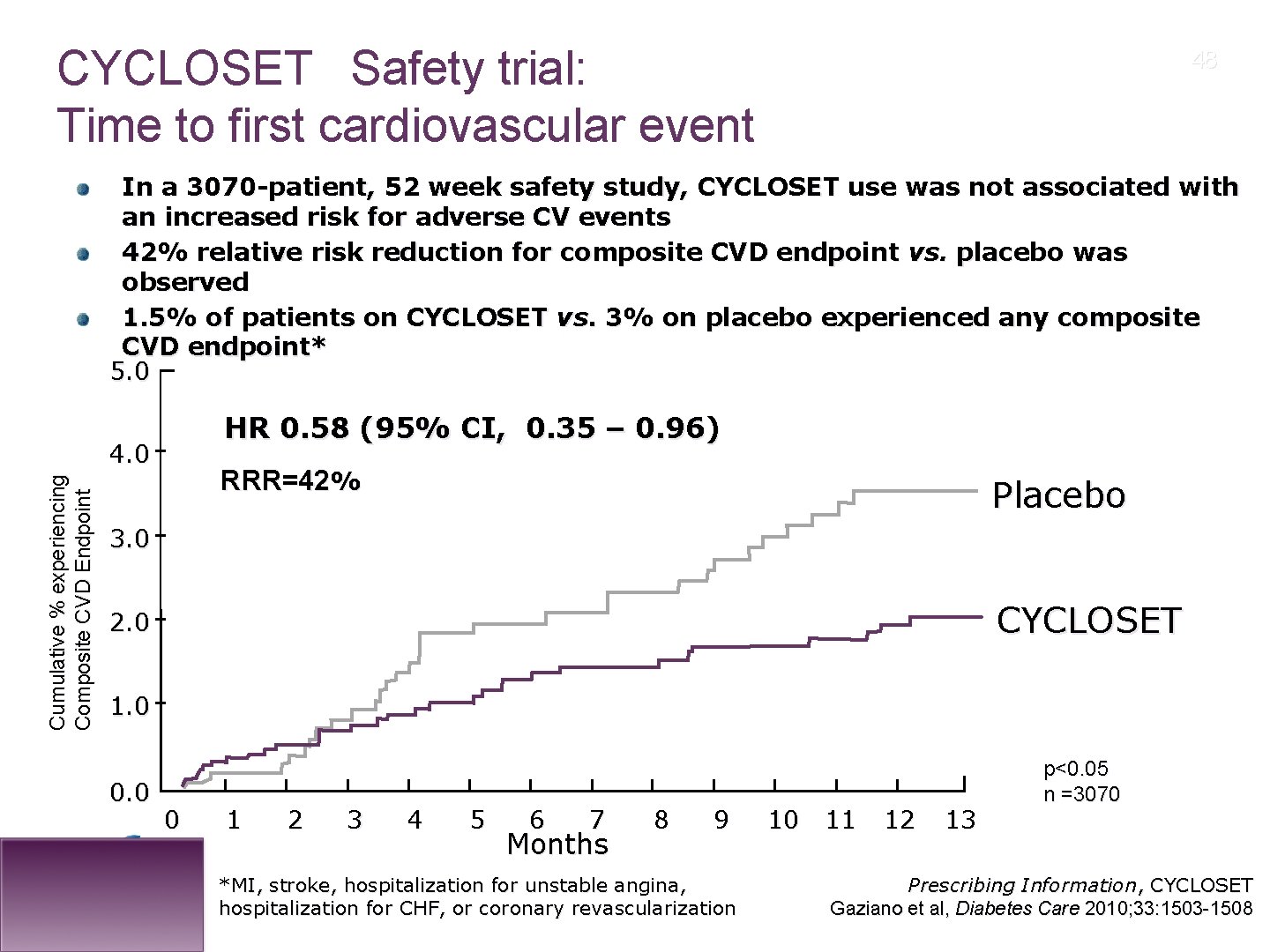
® CYCLOSET Safety trial: Time to first cardiovascular event 48 In a 3070 -patient, 52 week safety study, CYCLOSET use was not associated with an increased risk for adverse CV events 42% relative risk reduction for composite CVD endpoint vs. placebo was observed 1. 5% of patients on CYCLOSET vs. 3% on placebo experienced any composite CVD endpoint* 5. 0 HR 0. 58 (95% CI, 0. 35 – 0. 96) Cumulative % experiencing Composite CVD Endpoint 4. 0 RRR=42% Placebo 3. 0 CYCLOSET 2. 0 1. 0 0 1 2 3 4 5 6 7 Months 8 9 *MI, stroke, hospitalization for unstable angina, hospitalization for CHF, or coronary revascularization 10 11 12 13 p<0. 05 n =3070 Prescribing Information, CYCLOSET Gaziano et al, Diabetes Care 2010; 33: 1503 -1508

® CYCLOSET Safety trial: Summary n Demonstrated cardiovascular safety n In a 3070 -patient, 52 week safety study, CYCLOSET use was not associated with an increased risk for adverse CV events n 42% relative risk reduction for composite CVD endpoint vs. placebo was observed; Hazard ratio=0. 58 (95% CI, 0. 35 -0. 96) n 1. 5% of patients on CYCLOSET vs. 3% on placebo experienced any composite CVD endpoint n Demonstrated overall safety n Rate of SAEs vs. placebo 8. 5% vs. 9. 6%, respectively n No significant weight gain or severe hypoglycemia compared with placebo observed in clinical trials 50

51 CYCLOSET dosing n CYCLOSET is taken once daily within 2 hours of waking in the morning n The initial dose is one 0. 8 mg tablet daily, increased weekly by 1 tablet until therapeutic dose (1. 6 to 4. 8 mg, or between 2 and 6 tablets per day) is achieved n In clinical trials, most patients reached a dose between 3. 2 mg and 4. 8 mg per day Prescribing Information, CYCLOSET

CV Considerations in Diabetes Treatment + n In middle aged and older adults with long-standing type 2 diabetes and known CVD or who are at high risk for CVD, lower glycemic targets (ie, < 6. 0% or < 6. 5%) yielded no significant reduction in CVD outcomes compared to standard control targets (ie, < 7. 0%) and may be associated with adverse events (eg, hypoglycemia, weight gain, and death): n n ACCORD, ADVANCE, VADT Improved glycemic control is associated with a reduction in CVD end points: n n n UKPDS and DCCT long-term follow-ups Meta-analysis: UKPDS 33, UKPDS 34, ACCORD, ADVANCE, VADT PROactive

+ Case Study 3 Lyle is a 68 -year-old male diagnosed with type 2 diabetes 1 year ago. Three months ago, Lyle increased his metformin dose to 500 mg BID (from once daily). He now complains of gastrointestinal discomfort. n BMI: 29. 7 kg/m 2 n A 1 C 7. 4% n Hypertension is well-controlled (lisinopril/hydrochlorothiazide) n Simvastatin (10 mg once daily) n Metformin (500 mg BID) Is a goal of A 1 C < 7. 0% appropriate for Lyle? Lyle wants to discontinue metformin. What are his best alternatives for glycemic therapy?

Summary Slide + n Screen for diabetes in everyone aged 45 years and older: n n n Use an individualized glucose-lowering regimen to achieve the appropriate A 1 C goal: n n Lowering A 1 C to < 7. 0% is appropriate and well-supported by clinical evidence Check A 1 C at least every 3 months; intensify treatment until < 7. 0% A lower A 1 C goal than 7. 0% does not further reduce risk of CV death Consider adverse events associated with glucose-lowering therapy: n n If normal, repeat every 3 years Consider earlier, more frequent screenings if overweight with additional risk factors Hypoglycemia is a leading limiting factor in glycemic management Patient/clinician communication is crucial in diabetes self-care: n n Internally motivated patients learn and practice effective diabetes self-care At presentation, refer for diabetes education or nutrition counseling; assess anxiety/depression; suggest psychosocial support
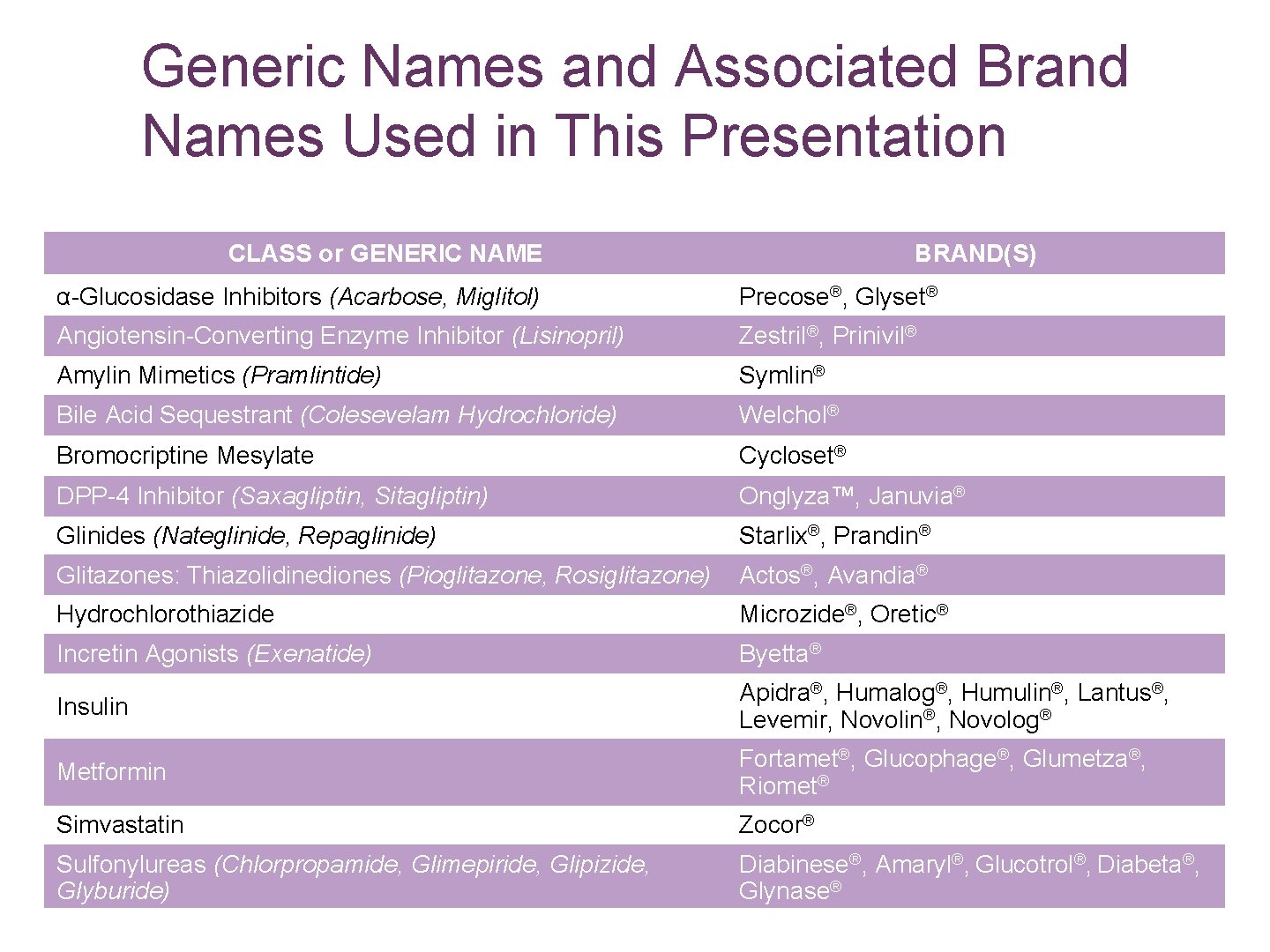
Generic Names and Associated Brand Names Used in This Presentation CLASS or GENERIC NAME BRAND(S) α-Glucosidase Inhibitors (Acarbose, Miglitol) Precose®, Glyset® Angiotensin-Converting Enzyme Inhibitor (Lisinopril) Zestril®, Prinivil® Amylin Mimetics (Pramlintide) Symlin® Bile Acid Sequestrant (Colesevelam Hydrochloride) Welchol® Bromocriptine Mesylate Cycloset® DPP-4 Inhibitor (Saxagliptin, Sitagliptin) Onglyza™, Januvia® Glinides (Nateglinide, Repaglinide) Starlix®, Prandin® Glitazones: Thiazolidinediones (Pioglitazone, Rosiglitazone) Actos®, Avandia® Hydrochlorothiazide Microzide®, Oretic® Incretin Agonists (Exenatide) Byetta® Insulin Apidra®, Humalog®, Humulin®, Lantus®, Levemir, Novolin®, Novolog® Metformin Fortamet®, Glucophage®, Glumetza®, Riomet® Simvastatin Zocor® Sulfonylureas (Chlorpropamide, Glimepiride, Glipizide, Glyburide) Diabinese®, Amaryl®, Glucotrol®, Diabeta®, Glynase®
- Slides: 48