Cardio Diabetes Master Class European chapter Munich Germany

Cardio Diabetes Master Class European chapter Munich, Germany May 6 -8, 2011 Presentation topic RAS blockade in the real world: Clinical lessons from recent trials Slide lecture prepared and held by: Peter Meredith Ph. D University of Glasgow, Department of Medicine and Therapeutics, Gardiner Institute Western Infirmary Glasgow, United Kingdom
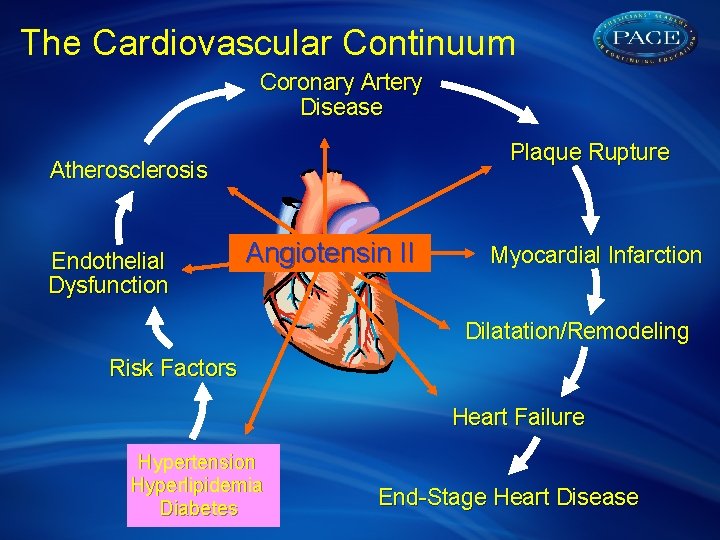
The Cardiovascular Continuum Coronary Artery Disease Plaque Rupture Atherosclerosis Endothelial Dysfunction Angiotensin II Myocardial Infarction Dilatation/Remodeling Risk Factors Heart Failure Hypertension Hyperlipidemia Diabetes End-Stage Heart Disease

RAS Blockade Across the CV Continuum Hypertension • LIFE • SCOPE • VALUE • KYOTO HEART • CAPPP • ANBP-2 • ALLHAT • CASE-J Heart Failure • ELITE II • Val-He. FT • CHARM • CONSENSUS I • SOLVD • PEP-CHF • I-PRESERVE Vascular • ELITE II • Val-He. FT • CHARM • CONSENSUS I • SOLVD • PEP-CHF • I-PRESERVE Diabetes - Renal • RENAAL • IDNT • ABCD-2 V • AASK • MARVAL • ADVANCE • DETAIL • DIRECT • ROADMAP Pre-Diabetes • NAVIGATOR • DREAM MI • OPTIMAAL • VALIANT • CONSENSUS II • ISIS-4 • GISSI-3 • SMILE • SAVE • AIRE • TRACE CAD • EUROPA • PEACE • IMAGINE 2 o Stroke Prevention • ACCESS • PROGRESS • PRo. FESS • SCAST

2007 ESH/ESC Guidelines: Choice of Antihypertensive Drugs § The main benefits of antihypertensive therapy are due to lowering of BP per se § Five major classes of antihypertensive agents – thiazide diuretics, CCBs, ACE inhibitors, angiotensin receptor blockers (ARBs) and b-blockers – are suitable for the initiation & maintenance of antihypertensive treatment, alone or in combination. b-blockers, especially in combination with a thiazide diuretic, should not be used in patients with the metabolic syndrome or at high risk of incident diabetes § Because in many patients more than one drug is needed, emphasis on identification of the first class of drugs to be used is often futile. Nevertheless, there are many conditions for which there is evidence in favour of some drugs versus others either as initial treatment or as part of a combination
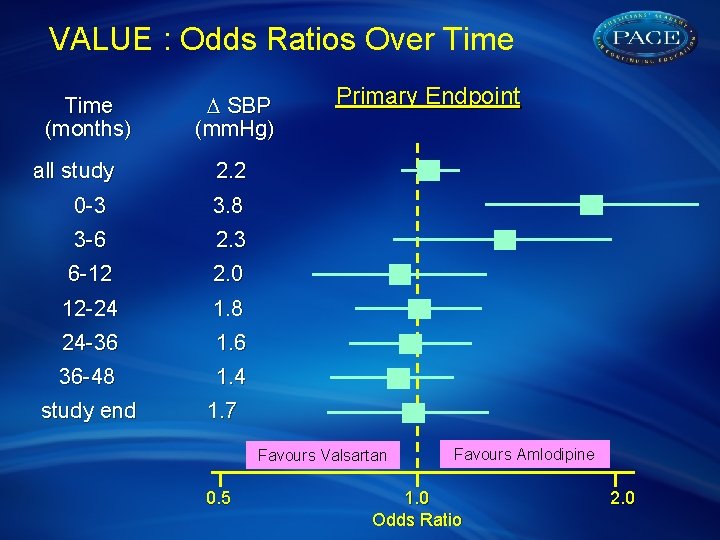
VALUE : Odds Ratios Over Time (months) D SBP (mm. Hg) all study 2. 2 0 -3 3. 8 3 -6 2. 3 6 -12 2. 0 12 -24 1. 8 24 -36 1. 6 36 -48 1. 4 study end 1. 7 Primary Endpoint Favours Valsartan 0. 5 Favours Amlodipine 1. 0 Odds Ratio 2. 0
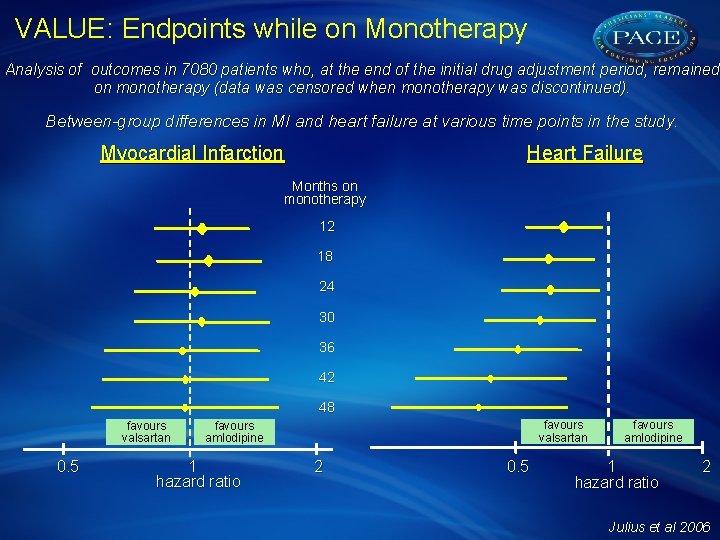
VALUE: Endpoints while on Monotherapy Analysis of outcomes in 7080 patients who, at the end of the initial drug adjustment period, remained on monotherapy (data was censored when monotherapy was discontinued). Between-group differences in MI and heart failure at various time points in the study. Myocardial Infarction Heart Failure Months on monotherapy 12 18 24 30 36 42 48 favours valsartan 0. 5 favours valsartan favours amlodipine 1 hazard ratio 2 0. 5 favours amlodipine 1 hazard ratio 2 Julius et al 2006
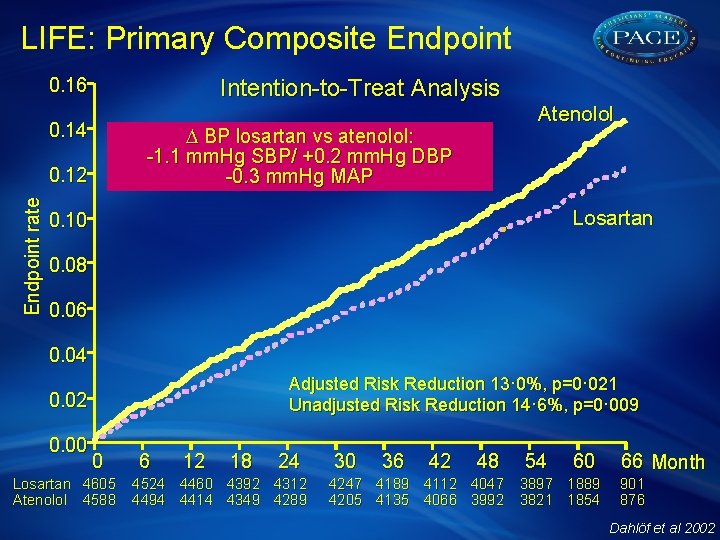
LIFE: Primary Composite Endpoint 0. 16 Intention-to-Treat Analysis 0. 14 D BP losartan vs atenolol: -1. 1 mm. Hg SBP/ +0. 2 mm. Hg DBP -0. 3 mm. Hg MAP Endpoint rate 0. 12 Atenolol Losartan 0. 10 0. 08 0. 06 0. 04 Adjusted Risk Reduction 13· 0%, p=0· 021 Unadjusted Risk Reduction 14· 6%, p=0· 009 0. 02 0. 00 0 Losartan 4605 Atenolol 4588 6 12 18 24 4524 4460 4392 4312 4494 4414 4349 4289 30 36 42 48 54 60 4247 4189 4112 4047 3897 1889 4205 4135 4066 3992 3821 1854 66 Month 901 876 Dahlöf et al 2002
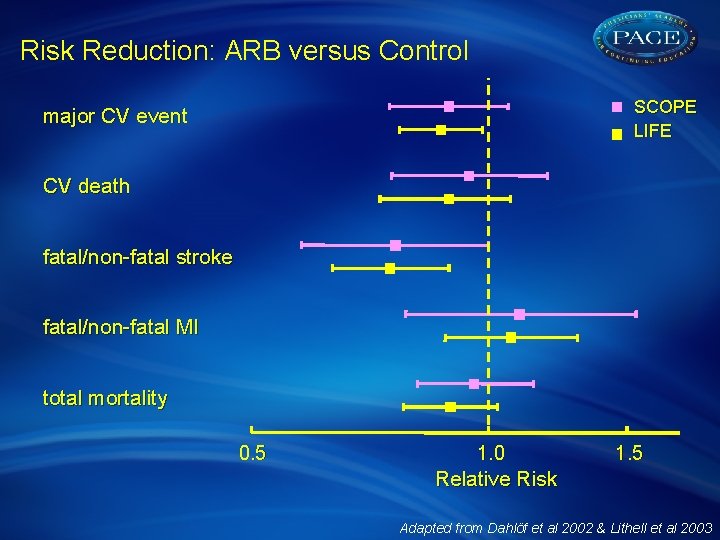
Risk Reduction: ARB versus Control SCOPE LIFE major CV event CV death fatal/non-fatal stroke fatal/non-fatal MI total mortality 0. 5 1. 0 1. 5 Relative Risk Adapted from Dahlöf et al 2002 & Lithell et al 2003
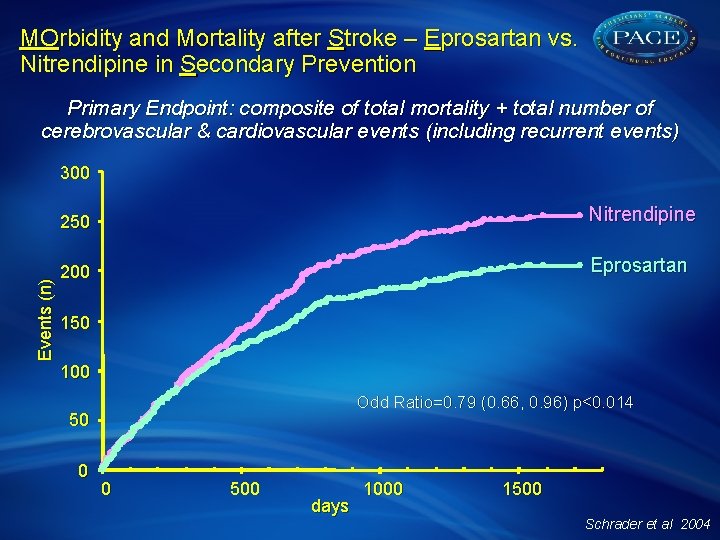
MOrbidity and Mortality after Stroke – Eprosartan vs. Nitrendipine in Secondary Prevention Primary Endpoint: composite of total mortality + total number of cerebrovascular & cardiovascular events (including recurrent events) Events (n) 300 250 Nitrendipine 200 Eprosartan 150 100 Odd Ratio=0. 79 (0. 66, 0. 96) p<0. 014 50 0 0 500 days 1000 1500 Schrader et al 2004
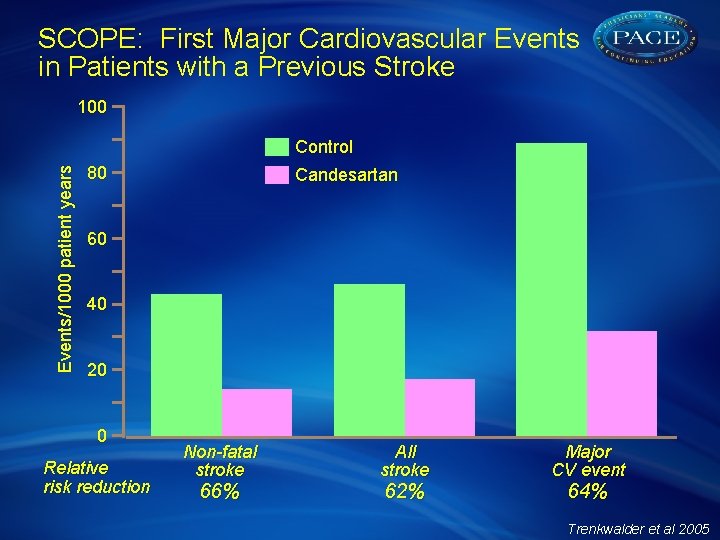
SCOPE: First Major Cardiovascular Events in Patients with a Previous Stroke 100 Events/1000 patient years Control 80 Candesartan 60 40 20 0 Relative risk reduction Non-fatal stroke 66% All stroke 62% Major CV event 64% Trenkwalder et al 2005
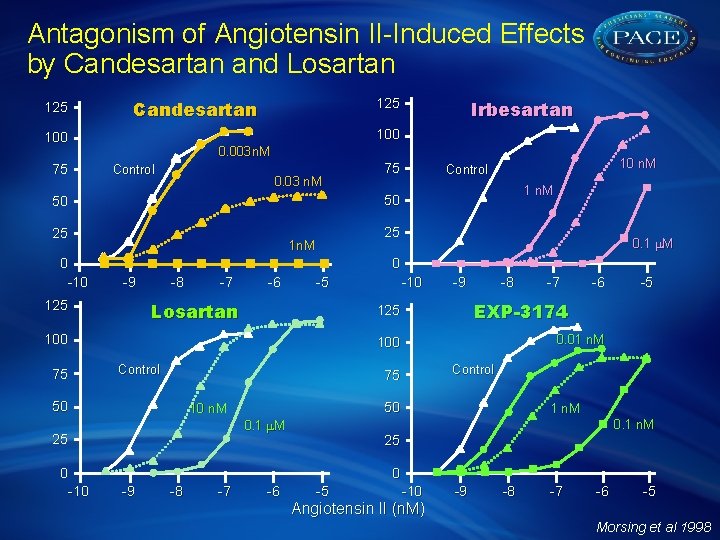
Antagonism of Angiotensin II-Induced Effects by Candesartan and Losartan 125 0. 003 n. M Control 0. 03 n. M 25 1 n. M 0. 1 M 0 -9 -8 -7 -6 -5 Losartan -10 -9 -8 50 10 n. M 1 n. M 0. 1 M 25 -5 Control 75 50 -6 0. 01 n. M 100 Control -7 EXP-3174 125 100 0 -10 10 n. M Control 25 1 n. M 125 75 75 50 50 0 -10 Irbesartan 100 75 125 Candesartan 25 0 -9 -8 -7 -6 -5 -10 -9 -8 -7 -6 -5 Angiotensin II (n. M) Morsing et al 1998
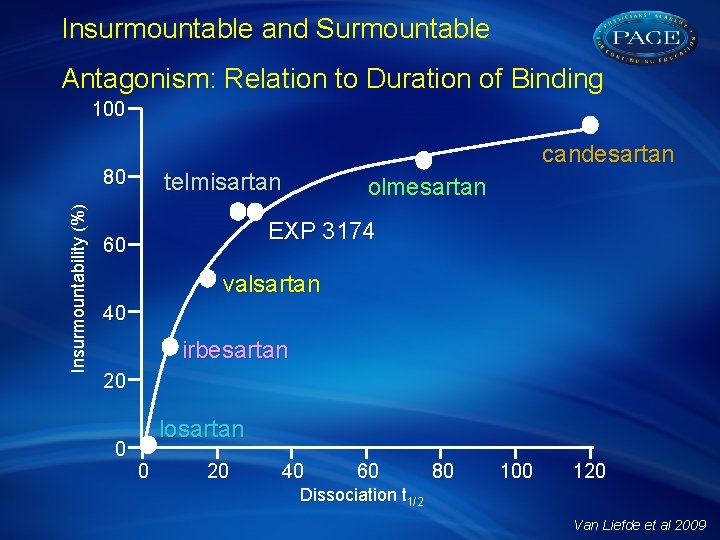
Insurmountable and Surmountable Antagonism: Relation to Duration of Binding 100 candesartan Insurmountability (%) 80 telmisartan olmesartan EXP 3174 60 valsartan 40 irbesartan 20 0 losartan 0 20 40 60 80 100 120 Dissociation t 1/2 Van Liefde et al 2009
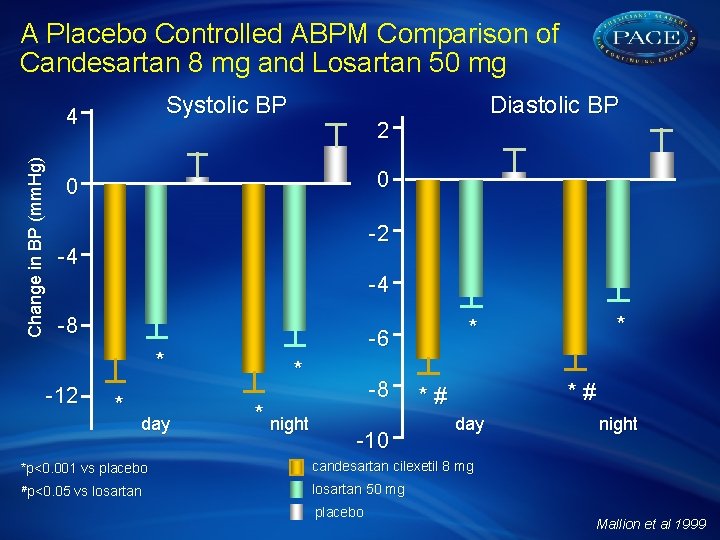
A Placebo Controlled ABPM Comparison of Candesartan 8 mg and Losartan 50 mg Systolic BP Change in BP (mm. Hg) 4 Diastolic BP 2 0 0 -2 -4 -4 -8 * -12 * day * * night -8 -10 * # day *p<0. 001 vs placebo candesartan cilexetil 8 mg #p<0. 05 vs losartan 50 mg placebo * * -6 night Mallion et al 1999
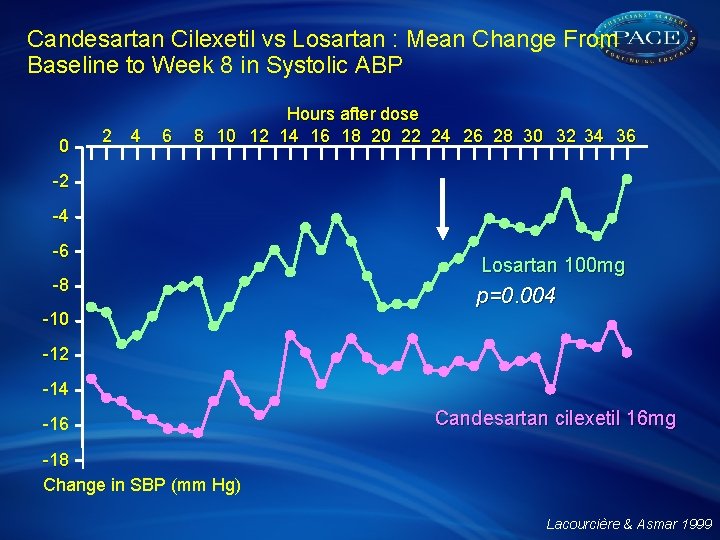
Candesartan Cilexetil vs Losartan : Mean Change From Baseline to Week 8 in Systolic ABP 0 2 4 6 Hours after dose 8 10 12 14 16 18 20 22 24 26 28 30 32 34 36 -2 -4 -6 -8 Losartan 100 mg p=0. 004 -10 -12 -14 -16 Candesartan cilexetil 16 mg -18 Change in SBP (mm Hg) Lacourcière & Asmar 1999

Comparison of the Efficacy of Candesartan & Losartan: Meta-Analysis of Trials in the Treatment of Hypertension A systematic literature search of databases from 1980 to 1 October 2008 identified 13 studies in which candesartan and losartan (as mono-therapy or in fixed combination with HCTZ) were compared in randomised trials in hypertensive patients. Data from 4066 patients were included in the statistical analysis which was performed using Rev. Man software (v 5), provided by the Cochrane Information Management System using a random effect model. Mean changes in SBP and DBP were compared for each drug alone and after stratification for dose and for combination with HCTZ. Meredith et al 2009
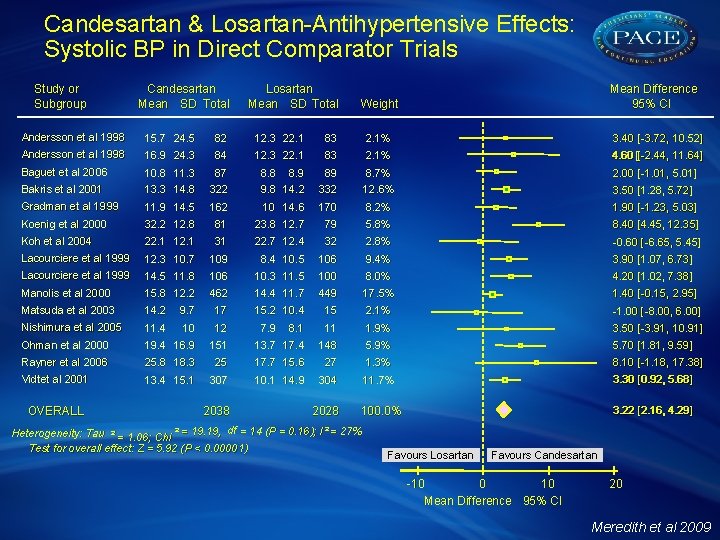
Candesartan & Losartan-Antihypertensive Effects: Systolic BP in Direct Comparator Trials Study or Subgroup Candesartan Mean SD Total Losartan Mean SD Total Mean Difference 95% CI Weight Andersson et al 1998 15. 7 24. 5 82 12. 3 22. 1 83 2. 1% 3. 40 [-3. 72, 10. 52] Andersson et al 1998 16. 9 24. 3 84 12. 3 22. 1 83 2. 1% 4. 60 [4. 60 [-2. 44, 11. 64] Baguet et al 2006 87 322 8. 8 8. 9 9. 8 14. 2 89 332 8. 7% 12. 6% 2. 00 [-1. 01, 5. 01] Bakris et al 2001 10. 8 11. 3 13. 3 14. 8 Gradman et al 1999 11. 9 14. 5 162 10 14. 6 170 8. 2% 1. 90 [-1. 23, 5. 03] Koenig et al 2000 32. 2 12. 8 81 23. 8 12. 7 79 5. 8% 8. 40 [4. 45, 12. 35] Koh et al 2004 22. 1 12. 1 31 22. 7 12. 4 32 2. 8% -0. 60 [ -6. 65, 5. 45] Lacourciere et al 1999 12. 3 10. 7 Lacourciere et al 1999 14. 5 11. 8 Manolis et al 2000 15. 8 12. 2 109 8. 4 10. 5 106 9. 4% 3. 90 [1. 07, 6. 73] 106 10. 3 11. 5 100 8. 0% 4. 20 [1. 02, 7. 38] 462 14. 4 11. 7 449 17. 5% 1. 40 [-0. 15, 2. 95] 15. 2 10. 4 15 2. 1% -1. 00 [ -8. 00, 6. 00] 8. 1 11 1. 9% 3. 50 [-3. 91, 10. 91] 3. 50 [1. 28, 5. 72] Matsuda et al 2003 14. 2 9. 7 17 Nishimura et al 2005 11. 4 10 12 Ohman et al 2000 19. 4 16. 9 151 13. 7 17. 4 148 5. 9% 5. 70 [1. 81, 9. 59] Rayner et al 2006 25. 8 18. 3 25 17. 7 15. 6 27 1. 3% 8. 10 [-1. 18, 17. 38] Vidt et al 2001 13. 4 15. 1 307 10. 1 14. 9 304 11. 7% 3. 30 [0. 92, 5. 68] 2028 100. 0% 3. 22 [2. 16, 4. 29] OVERALL 2038 7. 9 Heterogeneity: Tau ² = 1. 06; Chi ² = 19. 19, df = 14 (P = 0. 16); I ² = 27% Test for overall effect: Z = 5. 92 (P < 0. 00001) Favours Losartan Favours Candesartan -10 0 10 Mean Difference 95% CI 20 Meredith et al 2009
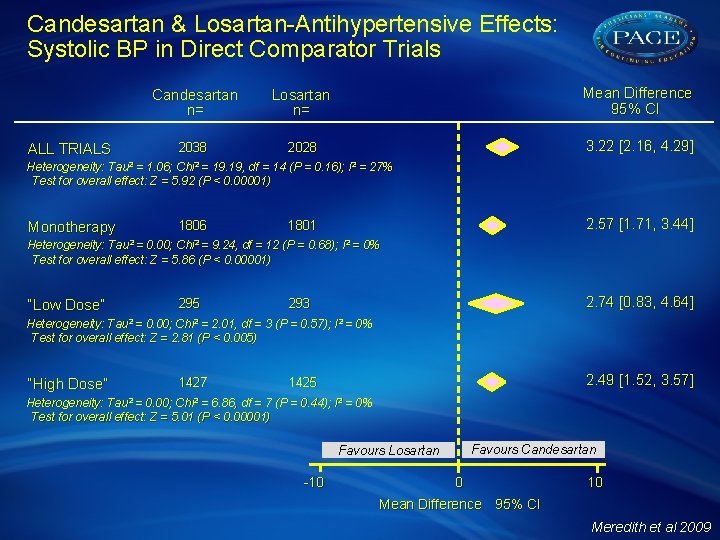
Candesartan & Losartan-Antihypertensive Effects: Systolic BP in Direct Comparator Trials ALL TRIALS Candesartan n= Losartan n= Mean Difference 95% CI 2038 2028 3. 22 [2. 16, 4. 29] Heterogeneity: Tau² = 1. 06; Chi² = 19. 19, df = 14 (P = 0. 16); I² = 27% Test for overall effect: Z = 5. 92 (P < 0. 00001) Monotherapy 1806 2. 57 [1. 71, 3. 44] 1801 Heterogeneity: Tau² = 0. 00; Chi² = 9. 24, df = 12 (P = 0. 68); I² = 0% Test for overall effect: Z = 5. 86 (P < 0. 00001) “Low Dose” 295 2. 74 [0. 83, 4. 64] 293 Heterogeneity: Tau² = 0. 00; Chi² = 2. 01, df = 3 (P = 0. 57); I² = 0% Test for overall effect: Z = 2. 81 (P < 0. 005) “High Dose” 1427 2. 49 [1. 52, 3. 57] 1425 Heterogeneity: Tau² = 0. 00; Chi² = 6. 86, df = 7 (P = 0. 44); I² = 0% Test for overall effect: Z = 5. 01 (P < 0. 00001) Favours Candesartan Favours Losartan -10 0 10 Mean Difference 95% CI Meredith et al 2009
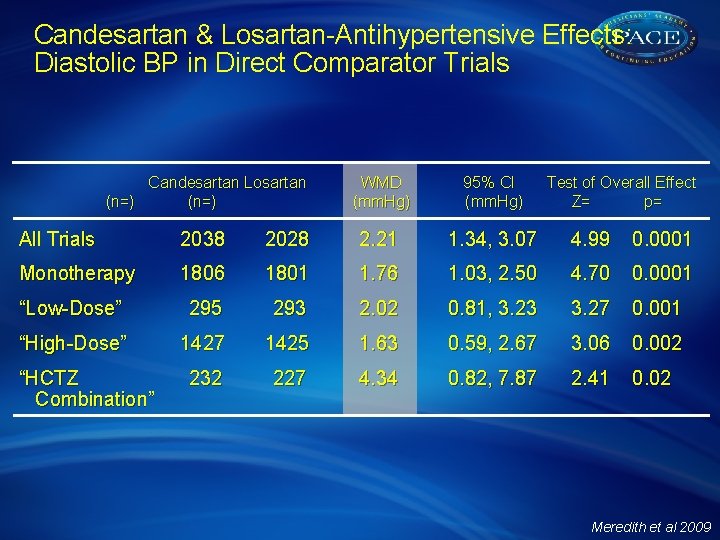
Candesartan & Losartan-Antihypertensive Effects: Diastolic BP in Direct Comparator Trials Candesartan Losartan (n=) WMD 95% CI Test of Overall Effect (mm. Hg) Z= p= All Trials 2038 2028 2. 21 1. 34, 3. 07 4. 99 0. 0001 Monotherapy 1806 1801 1. 76 1. 03, 2. 50 4. 70 0. 0001 “Low-Dose” 295 293 2. 02 0. 81, 3. 23 3. 27 0. 001 “High-Dose” 1427 1425 1. 63 0. 59, 2. 67 3. 06 0. 002 “HCTZ Combination” 232 227 4. 34 0. 82, 7. 87 2. 41 0. 02 Meredith et al 2009
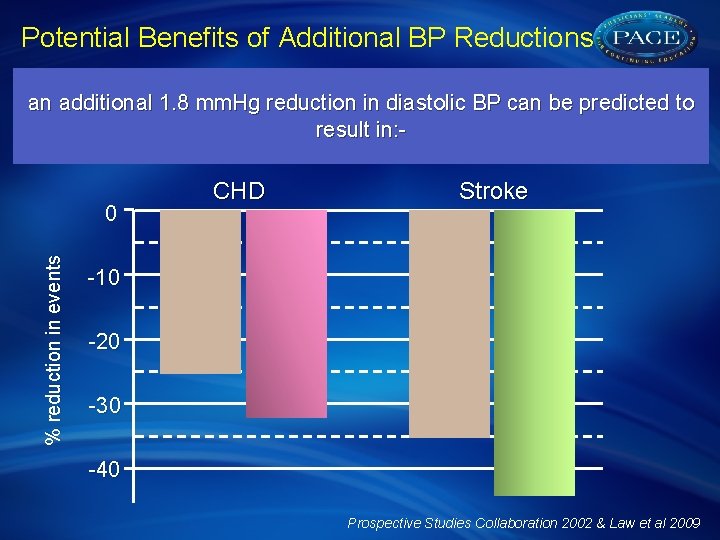
Potential Benefits of Additional BP Reductions Meta-analysis of 61 cohort studies and 147 randomised trials suggests that in a 65 year old, monotherapy with a standard dose of an an additional 1. 8 mm. Hg reduction in diastolic BP can be predicted to antihypertensive, reduces diastolic BP by approximately 5 mm. Hg result in: resulting in: - % reduction in events 0 CHD Stroke -10 -20 -30 -40 Prospective Studies Collaboration 2002 & Law et al 2009

Hypothesis and Aim § Losartan and candesartan have different pharmacological properties and blood pressure lowering abilities § The aim of the Real Life study was to test the hypothesis that losartan and candesartan have different primary preventive effects on CVD risk § The hypothesis was tested by setting up a large retrospective observation study in 72 Health Care Centres in Sweden REAL-LIFE

24, 943 patients started on either losartan or candesartan from 1999– 2007 Included 14, 100 Selection Method Losartan 6, 771 (48. 0%) Follow-up average 2. 0 years (36, 339 patient years) Candesartan 7, 329 (52. 0%) 10, 843 (44%) patients were excluded: 1. CV-disease or prescription of warfarin/digitalis/nitrates 2. Malignancy 3. Another RAS-inhibitor in the first week after inclusion REAL-LIFE
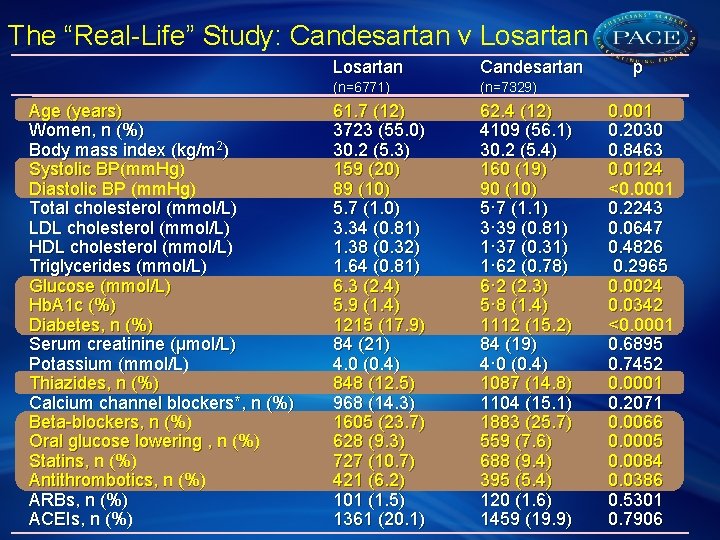
The “Real-Life” Study: Candesartan v Losartan Age (years) Women, n (%) Body mass index (kg/m 2) Systolic BP(mm. Hg) Diastolic BP (mm. Hg) Total cholesterol (mmol/L) LDL cholesterol (mmol/L) HDL cholesterol (mmol/L) Triglycerides (mmol/L) Glucose (mmol/L) Hb. A 1 c (%) Diabetes, n (%) Serum creatinine (μmol/L) Potassium (mmol/L) Thiazides, n (%) Calcium channel blockers*, n (%) Beta-blockers, n (%) Oral glucose lowering , n (%) Statins, n (%) Antithrombotics, n (%) ARBs, n (%) ACEIs, n (%) Losartan Candesartan (n=6771) (n=7329) 61. 7 (12) 3723 (55. 0) 30. 2 (5. 3) 159 (20) 89 (10) 5. 7 (1. 0) 3. 34 (0. 81) 1. 38 (0. 32) 1. 64 (0. 81) 6. 3 (2. 4) 5. 9 (1. 4) 1215 (17. 9) 84 (21) 4. 0 (0. 4) 848 (12. 5) 968 (14. 3) 1605 (23. 7) 628 (9. 3) 727 (10. 7) 421 (6. 2) 101 (1. 5) 1361 (20. 1) 62. 4 (12) 4109 (56. 1) 30. 2 (5. 4) 160 (19) 90 (10) 5· 7 (1. 1) 3· 39 (0. 81) 1· 37 (0. 31) 1· 62 (0. 78) 6· 2 (2. 3) 5· 8 (1. 4) 1112 (15. 2) 84 (19) 4· 0 (0. 4) 1087 (14. 8) 1104 (15. 1) 1883 (25. 7) 559 (7. 6) 688 (9. 4) 395 (5. 4) 120 (1. 6) 1459 (19. 9) p 0. 001 0. 2030 0. 8463 0. 0124 <0. 0001 0. 2243 0. 0647 0. 4826 0. 2965 0. 0024 0. 0342 <0. 0001 0. 6895 0. 7452 0. 0001 0. 2071 0. 0066 0. 0005 0. 0084 0. 0386 0. 5301 0. 7906
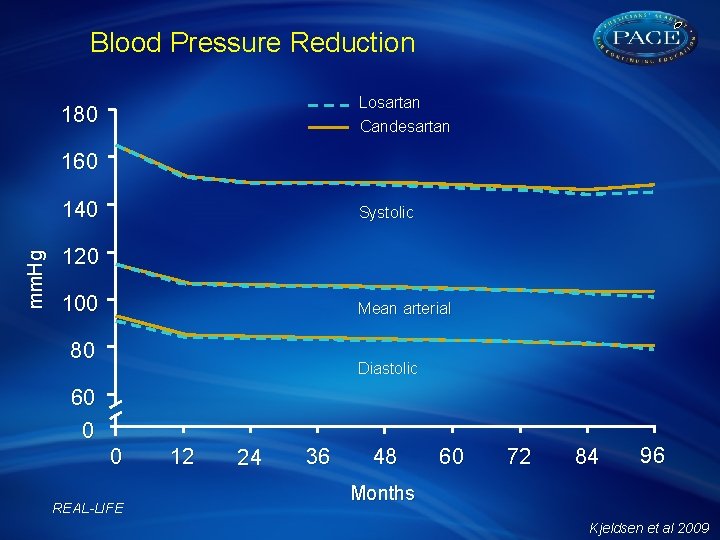
Blood Pressure Reduction Losartan Candesartan 180 160 mm. Hg 140 Systolic 120 100 Mean arterial 80 Diastolic 60 0 0 REAL-LIFE 12 24 36 48 60 72 84 96 Months Kjeldsen et al 2009
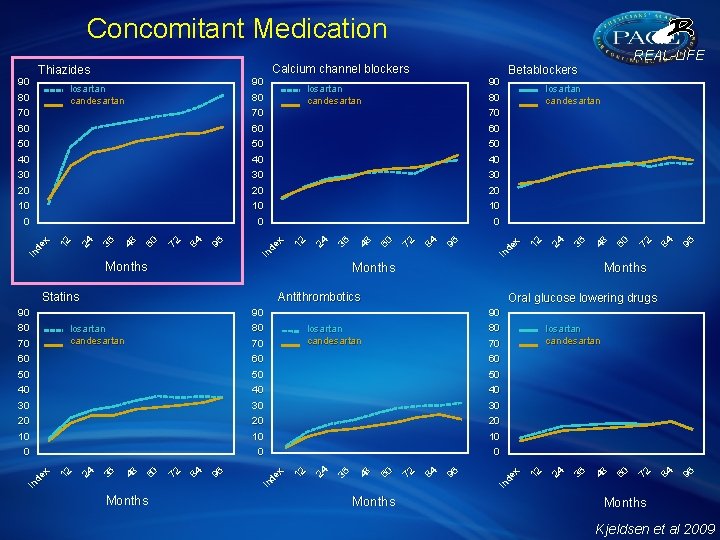
Concomitant Medication Months Antithrombotics 80 80 losartan candesartan 70 30 30 20 20 10 0 Months 96 84 72 In de x 96 84 72 60 48 36 24 10 12 In de x 30 96 40 84 40 72 40 60 50 48 50 36 50 24 60 12 60 In de x 60 Months losartan candesartan 70 60 losartan candesartan 96 90 48 70 Oral glucose lowering drugs 90 36 80 Months 24 Statins 90 84 0 72 0 60 0 In de x 10 96 10 84 10 72 20 60 20 48 20 36 30 24 30 12 30 In de x 40 96 40 84 40 72 50 60 50 48 50 36 60 24 70 60 12 70 60 In de x 70 Months losartan candesartan 80 48 losartan candesartan 80 Betablockers 36 losartan candesartan 80 90 12 90 24 Calcium channel blockers Thiazides 12 90 REAL-LIFE Months Kjeldsen et al 2009
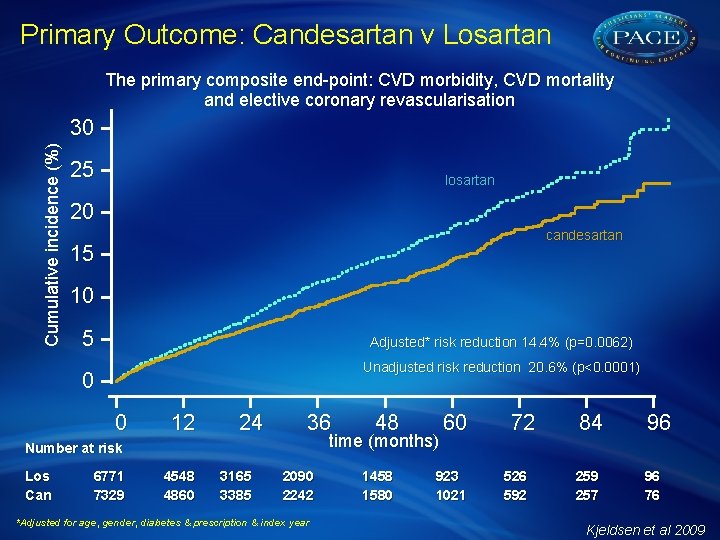
Primary Outcome: Candesartan v Losartan The primary composite end-point: CVD morbidity, CVD mortality and elective coronary revascularisation Cumulative incidence (%) 30 25 losartan 20 candesartan 15 10 5 Adjusted* risk reduction 14. 4% (p=0. 0062) Unadjusted risk reduction 20. 6% (p<0. 0001) 0 0 12 24 36 time (months) Number at risk Los Can 6771 7329 4548 4860 3165 3385 48 2090 2242 *Adjusted for age, gender, diabetes & prescription & index year 1458 1580 60 923 1021 72 526 592 84 96 259 257 96 76 Kjeldsen et al 2009
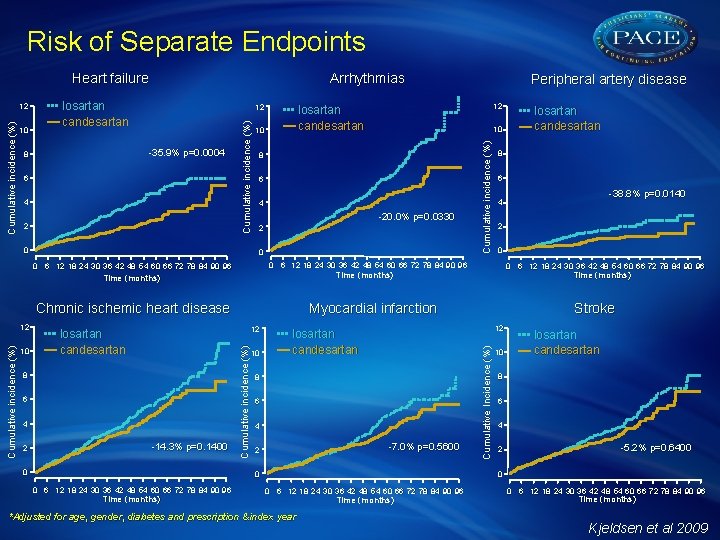
Risk of Separate Endpoints Heart failure 12 6 4 2 0 10 6 4 -20. 0% p=0. 0330 2 0 6 4 2 -14. 3% p=0. 1400 0 Cumulative incidence (%) 8 10 8 6 4 -7. 0% p=0. 5600 2 Time (months) 0 0 6 12 18 24 30 36 42 48 54 60 66 72 78 84 90 96 Time (months) Stroke 10 losartan candesartan 8 6 4 2 -5. 2% p=0. 6400 0 6 12 18 24 30 36 42 48 54 60 66 72 78 84 90 96 2 12 losartan candesartan -38. 8% p=0. 0140 4 Myocardial infarction 12 losartan candesartan 6 Time (months) Chronic ischemic heart disease losartan candesartan 8 0 6 12 18 24 30 36 42 48 54 60 66 72 78 84 90 96 Time (months) 10 10 8 0 6 12 18 24 30 36 42 48 54 60 66 72 78 84 90 96 12 12 losartan candesartan Cumulative incidence (%) -35. 9% p=0. 0004 8 Peripheral artery disease Cumulative Incidence (%) 10 losartan candesartan Cumulative incidence (%) 12 Arrhythmias 0 6 12 18 24 30 36 42 48 54 60 66 72 78 84 90 96 *Adjusted for age, gender, diabetes and prescription & index year Time (months) 0 6 12 18 24 30 36 42 48 54 60 66 72 78 84 90 96 Time (months) Kjeldsen et al 2009

Candesartan v Losartan: Mortality in Heart Failure Patients § Data from 30, 254 unique patients registered from 62 hospitals and 60 outpatient clinics was extracted from the Swedish Heart Failure Registry between 2000 and 2009 § A total of 5139 patients (mean [SD] age, 74 [11] years; 39% women) were treated with candesartan (n=2639) or losartan (n=2500) § Survival as of 14 December, 2009, by ARB agent was analyzed by Kaplan-Meier method § Predictors of survival determined by univariate and multivariate proportional hazard regression models, with and without adjustment for propensity scores and interactions § Stratified analyses and quantification of residual confounding were also performed Eklind-Cervenka et al 2011
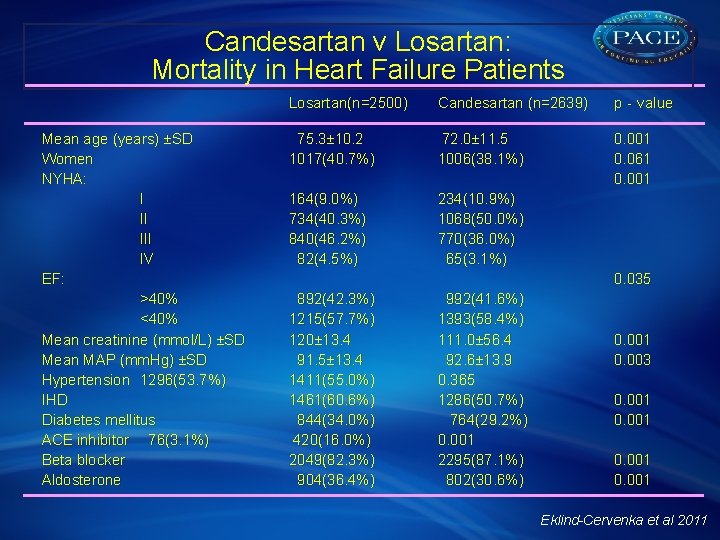
Candesartan v Losartan: Mortality in Heart Failure Patients Mean age (years) ±SD Women NYHA: I II IV EF: >40% <40% Mean creatinine (mmol/L) ±SD Mean MAP (mm. Hg) ±SD Hypertension 1296(53. 7%) IHD Diabetes mellitus ACE inhibitor 76(3. 1%) Beta blocker Aldosterone Losartan(n=2500) Candesartan (n=2639) p‐value 75. 3± 10. 2 1017(40. 7%) 72. 0± 11. 5 1006(38. 1%) 0. 001 0. 061 0. 001 164(9. 0%) 734(40. 3%) 840(46. 2%) 82(4. 5%) 234(10. 9%) 1068(50. 0%) 770(36. 0%) 65(3. 1%) 0. 035 892(42. 3%) 1215(57. 7%) 120± 13. 4 91. 5± 13. 4 1411(55. 0%) 1461(60. 6%) 844(34. 0%) 420(16. 0%) 2049(82. 3%) 904(36. 4%) 992(41. 6%) 1393(58. 4%) 111. 0± 56. 4 92. 6± 13. 9 0. 365 1286(50. 7%) 764(29. 2%) 0. 001 2295(87. 1%) 802(30. 6%) 0. 001 0. 003 0. 001 Eklind-Cervenka et al 2011
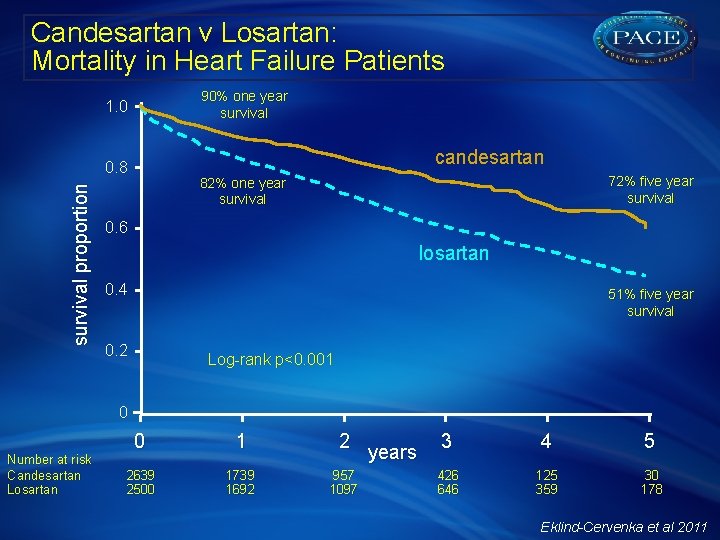
Candesartan v Losartan: Mortality in Heart Failure Patients 90% one year survival proportion 1. 0 candesartan 0. 8 72% five year survival 82% one year survival 0. 6 losartan 0. 4 51% five year survival 0. 2 Log-rank p<0. 001 0 Number at risk Candesartan Losartan 0 1 2639 2500 1739 1692 2 3 years 957 1097 426 646 4 5 125 359 30 178 Eklind-Cervenka et al 2011
- Slides: 29